BGD Tutorial - Applied Embryology and Teratology
| Embryology - 9 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This Medicine Phase 2 tutorial introduces the topics of Applied Embryology and Teratology. This one and a half hour presentation uses your existing knowledge of normal human development in an applied clinical manner in relation to our existing knowledge of teratogens. In addition, you should begin considering the variables that will not change and those that will in future medical practice. Due to time limitations, only a brief coverage can be given of any one topic.
Self-Directed Learning boxes on this page will not be discussed within the tutorial. You should also return here and later work through the linked online resources for more detailed descriptions and an understanding of these issues. This current page appears in the lefthand menu under Medicine as BGD 2 Tutorial.
- 2014 Print Version PDF (10 pages, 670kb)
Similar content was covered in the previous online tutorials in 2012 | 2012 PDF | 2011 PDF and 2010.
| Whats in the News? | ||
|---|---|---|
|
Objectives
Applied Embryology: birth statistics, unintended pregnancies, ART, abnormalities statistics, timeline of development, trophoblastic disease, embryonic development, placenta, fetal development, maternal diet, multiple pregnancies.
Teratology: definitions, critical periods, medications, chromosomal abnormalities, environmental factors and infections.
Textbooks
Applied Embryology
|
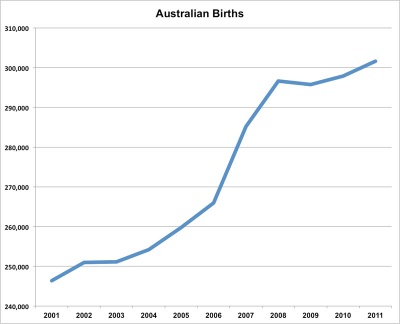
This recent data summarised below from Australia's mothers and babies 2011[1], 2009[2], 2008[3] and 2007[4]. This data should help you as a clinician and researcher to understand the current trends in reproductive medicine within Australia. Also see recent general population data in Australian Statistics.
|
|
| Mothers | ||||
|---|---|---|---|---|
Mothers
Smoking during pregnancy
Preterm Birth
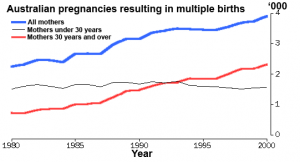
Multiple pregnancy
Presentation at birth2009 data
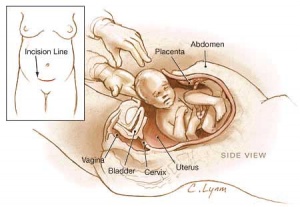
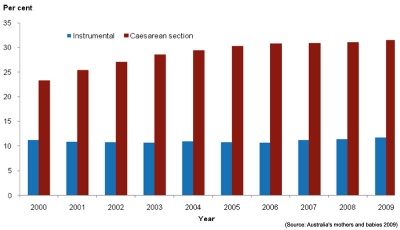
Method of birth2009 data
Postnatal length of stay2009 data
|
| Babies |
|---|
Babies2009 data
Gestational age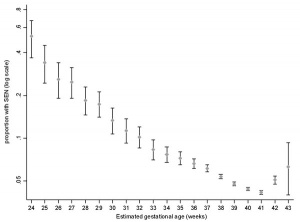 Special educational need by gestational age (UK data)[5] 2009 data
Birthweight2009 data
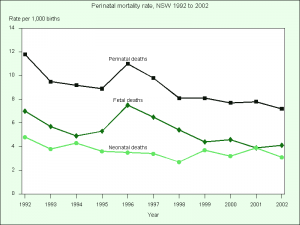
Apgar scores - 1.5% of liveborn babies had a low Apgar score (between 0 and 6) at 5 minutes (More? Apgar test) Special care nurseries (SCN) or neonatal intensive care units (NICU) - 14.2% of liveborn babies admitted (2008 - 14.5%) Perinatal mortality
|
| 2013 National core maternity indicators |
|---|
|
2013 National core maternity indicators[6] This report provides a baseline for monitoring changes in the quality of maternity services across Australia using 10 national core maternity indicators.
|
Unintended Pregnancy
| Approximately one-half of pregnancies in the United States (2001) were unintended (Finer 2006, Perspectives on Sexual and Reproductive Health).
An earlier 1995 USA National Survey of Family Growth (NSFG) found:
Unintended pregnancy is either mistimed (woman wanted to be pregnant later) or unwanted (did not want to ever be pregnant). |
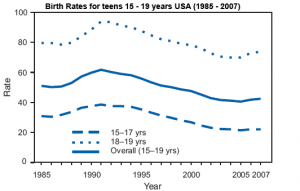
Teen pregnancy (USA) |
| Teen Pregnancy |
|---|
A recent Canadian retrospective study[7] of half a million births comparing pregnant women < 20 years of age (adolescent) were compared with those of women 20 to 35 years old.
|
Links: CDC Unintended Pregnancy Prevention | Pregnancy Risk Assessment Monitoring System USA | The Measurement and Meaning of Unintended Pregnancy
Assisted Reproduction Technology
Assisted Reproduction Technology (ART) is also sometimes also used to identify In vitro fertilization (IVF) but now includes many new techniques.
Assisted reproductive technology in Australia and New Zealand 2010[8] 26 Oct 2012
2010 ART treatment cycles
- 61,774 assisted reproductive technology (ART) treatment cycles performed in Australia and New Zealand.
- 23.9% resulted in a clinical pregnancy
- 18.1% in a live delivery (the birth of at least one liveborn baby).
- 12,056 liveborn babies following ART treatments in 2010.
Trends in ART procedures
- In the last 5 years there has been a shift from day 2-3 embryo (cleavage stage) transfers to day 5-6 embryo (blastocyst) transfers.
- The proportion of blastocyst transfers has increased from 27.1% in 2006 to 52.1% in 2010.
- Increase in the transfer of vitrified (ultra-rapid frozen) embryos. Compared with 2009, the proportion has more than doubled from 18.3% to 38.2%.
- reduction in the rate of multiple birth deliveries, with a decrease from 12% in 2006 to 7.9% in 2010.
- shifting to single embryo transfer, the proportion of which increased from 56.9% in 2006 to almost 70% in 2009 and 2010.
- decrease in the multiple delivery rate was achieved while clinical pregnancy rates remained stable at about 23% per cycle.
| 2009 Data |
|---|
|
The following data from Assisted reproductive technology in Australia and New Zealand 2009.[9] 9 Nov 2011 2009 ART treatment cycles
single embryo transfer
cryopreserved embryos
|
Early Development Issues
Abnormal Implantation
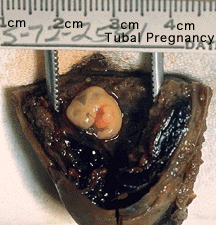
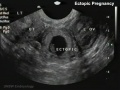
Ectopic Implantation (Pregnancy)
Abnormal implantation sites or Ectopic Pregnancy occurs if implantation is in uterine tube or outside the uterus.
Tubal pregnancy - 94% of ectopic pregnancies
|
|
This is also the most common cause of pregnancy-related deaths in the first trimester. A United Kingdom enquiry into maternal deaths[10], identified ectopic pregnancy as the fourth most common cause of maternal death (73% of early pregnancy deaths).
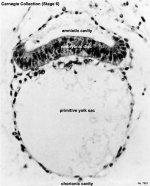
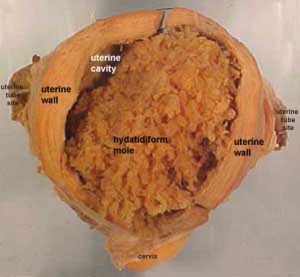
Hydatidiform Mole
Another type of abnormality is when only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole", which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus. The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo.
There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
This topic is also covered in Placenta - Abnormalities
Twinning
- Twin deliveries and place of birth in NSW 2001-2005[11] "Both infant and maternal morbidity increase from 39 weeks gestation. Delivery of twins before 36 weeks at smaller hospitals (< 500 deliveries per annum) should be avoided. A twin pregnancy where there is a greater or equal to 20% difference in estimated fetal weights should be considered for referral to a tertiary obstetric unit."
Dizygotic Twinning
Dizygotic twins (fraternal, non-identical) arise from separate fertilization events involving two separate oocyte (egg, ova) and spermatozoa (sperm). Dizygotic twinning can be increased by Assisted Reproductive Technologies (ART) that use double embryo transfer techniques.
Monoygotic Twinning
Monoygotic twins (identical) produced from a single fertilization event (one fertilised egg and a single spermatazoa, form a single zygote), these twins therefore share the same genetic makeup. Occurs in approximately 3-5 per 1000 pregnancies, more commonly with aged mothers. The later the twinning event, the less common are initially separate placental membranes and finally resulting in conjoined twins.
| Week | Week 1 | Week 2 | |||||||||||||
| Day | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
| Cell Number | 1 | 1 | 2 | 16 | 32 | 128 | bilaminar | ||||||||
| Event | Ovulation | fertilization | First cell division | Morula | Early blastocyst | Late blastocyst
Hatching |
Implantation starts | X inactivation | |||||||
|
|
|
|||||||||||||
| Monoygotic
Twin Type |
Diamniotic
Dichorionic |
Diamniotic
Monochorionic |
Monoamniotic
Monochorionic |
Conjoined | |||||||||||
Table based upon recent Twinning Review.[12]
- Links: Twinning
Abnormal Development
Embryological development is a robust biological system able to cope with many stresses without long-term consequences. When development does go wrong there are generally 3 major types groups: Genetic (inherited), Environmental (maternal) derived and Unknown (not determined or known) abnormalities. Also often not considered, is that pregnancy itself can also expose abnormalities in the mother (congenital heart disease, diabetes, reproductive disorders) that until the pregnancy had gone undetected.
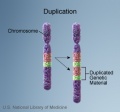
Genetic abnormalities in medicine are still mainly about determining a family history and good prenatal/neonatal diagnosis. Realise that there exists in all of us genetic variations and some variations which eventually expand be expressed as a genetic disorder (CAG expansions).
| Abnormality Links | ||||
|---|---|---|---|---|
|
Prenatal diagnosis are the clinical tools used to determine both normal and abnormal development. There are a growing number of new diagnostic techniques that are being applied to human embryonic development.
| Prenatal Diagnosis Links | |
|---|---|
|
While genetic abnormalities will have well-defined impacts upon development, environmentally derived effects can be harder to define and often variable depending on many different factors (timing, exposure level, and the combination effects with other factors). This combination effect can also be seen between genetic and environmental interacting to give an even broader spectrum of both major and minor abnormalities.
| Environmental Links | ||||||
|---|---|---|---|---|---|---|
|
International Classification of Diseases
The International Classification of Diseases (ICD) World Health Organization's classification used worldwide as the standard diagnostic tool for epidemiology, health management and clinical purposes. This includes the analysis of the general health situation of population groups. It is used to monitor the incidence and prevalence of diseases and other health problems. Within this classification "congenital malformations, deformations and chromosomal abnormalities" are (Q00-Q99) but excludes "inborn errors of metabolism" (E70-E90).
Australian Birth Anomalies System
- "The national collation and reporting of birth anomalies data has been suspended in recent years due to concerns about data quality and comparability."
- Variability among states and territories in scope of birth anomalies data collections: sources of birth anomalies notifications and definitions and classifications used; method of data collection and available resources.
- Variability among the states and territories in the timing and method of the provision of birth anomalies data to the AIHW National Perinatal Statistics Unit (NPSU) for national collation and reporting.
- New Australian Birth Anomalies System should be data for birth anomalies detected up to 1 year of age
- including data on terminations of pregnancies with birth anomalies and regardless of gestational age (i.e. including less than 20 weeks gestation)
- System will initially be based on data from the states able to detect birth anomalies at least up to 1 year of age (NSW, VIC, WA and SA), further extending the period of detection in the future.
- Congenital anomalies are coded using the British Paediatric Association Classification of Diseases (ICD-9-BPA), based on the International Classification of Diseases, 9th Revision (ICD-9).
- The Australian Congenital Anomalies Monitoring System (ACAMS) supersedes the National Congenital Malformations and Birth Defects Data Collection (NCM&BD).
Links: Australian Congenital Anomalies Monitoring System | Congenital Anomalies in Australia 2002-2003
NSW Data
Congenital Conditions Register
Scheduled congenital conditions (section 2) detected during pregnancy or in infants up to one year of age in NSW are required to be reported under the NSW Public Health Act 1991.
Scheduled congenital conditions include:
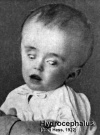
- All structural malformations. Examples include spina bifida, microcephaly, transposition of the great vessels, ventricular septal defects, pulmonary agenesis, polycystic lungs, duodenal atresia, exomphalos, hypospadias, cleft lip/palate, microphthalmia, limb reductions, polydactyly, birthmarks greater than 4 cms diameter, cystic hygroma and multisystem syndromes including at least one structural malformation.
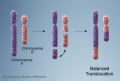
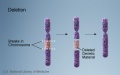
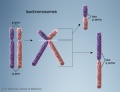
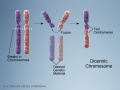
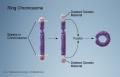
- Chromosomal abnormalities. Examples include Down syndrome and unbalanced translocations.
- Four medical conditions: cystic fibrosis, phenylketonuria, congenital hypothyroidism and thalassaemia major.
Congenital conditions that are not notifiable include:
- Minor anomalies occurring in isolation (Examples of minor anomalies include skin tags, deviated nasal septum, tongue tie, benign heart murmurs, clicky non-dislocating hips, sacral dimples, positional talipes, abnormal palmar creases, dysmorphic features).
- Birth injuries.
- Congenital infections which do not result in a structural malformation.
- Tumours and cysts.
- Conditions arising from prematurity or asphyxiation.
Links: NSW Health - Congenital Conditions Register - Reporting Requirements 2012 | PDF
Mothers and Babies Report 2010
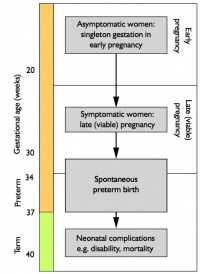
- preterm birth (less than 37 weeks gestation) was 7.4%.
- rate of low birth weight (less than 2,500 grams) was 6.1%
- in Aboriginal or Torres Strait Islander babies was 11.2%.
- About 2% of infants are born with congenital conditions each year in NSW.
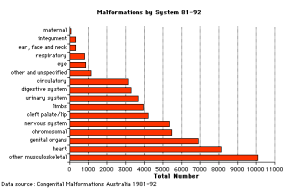
- In 2004–2010, anomalies of the cardiovascular system were most commonly reported, followed by anomalies of the musculoskeletal system and the genito-urinary system.
- Congenital conditions were more common among premature infants compared to full term infants, and among male infants compared to female infants.
- rate of congenital conditions increases with increasing maternal age, especially after age 35.
- However, as most babies are born to mothers aged less than 35 years, the majority of babies with congenital conditions were born to younger mothers.
- perinatal deaths 755, 134 (17.7%) of these deaths were unexplained stillbirths.
- neonatal death was extreme prematurity (41.3%), followed by congenital abnormalities (21.5%).
Data<refCentre for Epidemiology and Evidence. New South Wales Mothers and Babies 2010. Sydney: NSW Ministry of Health, 2012.</ref>
Links: New South Wales Mothers and Babies Report 2010
| Victoria - 10 most reported birth anomalies | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Based upon statistics from the Victorian Perinatal Data Collection Unit in Victoria between 2003-2004. | ||||||||||||||||||||
|
| USA Statistics | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| European Statistics | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Genetic
Teratology
Prenatal Screening
How and why do things go wrong in development?
These notes cover abnormalities that can occur during development often described as congenital defects or birth defects. There are many different ways that developmental abnormalities can occur the 3 major types are Genetic (inherited), Environmental (maternal) and Unknown (not determined) derived abnormalities. The environmental factors that cause or lead to any of these abnormalities are described as Teratogens.
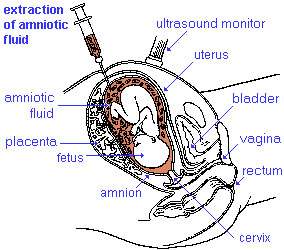
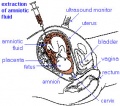
| Diagnosis Links: Prenatal Diagnosis | pregnancy test | amniocentesis | chorionic villus sampling | ultrasound | Alpha-Fetoprotein | Pregnancy-associated plasma protein-A | Fetal Blood Sampling | Magnetic Resonance Imaging | Computed Tomography | Non-Invasive Prenatal Testing | Fetal Cells in Maternal Blood | Preimplantation Genetic Screening | Comparative Genomic Hybridization | Genome Sequencing | Neonatal Diagnosis | Category:Prenatal Diagnosis | Fetal Surgery | Classification of Diseases | Category:Neonatal Diagnosis |
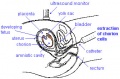
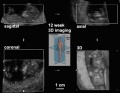
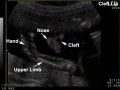
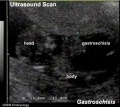
| Ultrasound | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Ultrasound
|
Now consider the terms used to describe the different environmental effects that can occur during pregnancy that may influence outcomes.
- Teratogen (Greek, teraton = monster) any agent that causes a structural abnormality (congenital abnormalities) following fetal exposure during pregnancy. The overall effect depends on dosage and time of exposure.
- Absolute risk the rate of occurrence of an abnormal phenotype among individuals exposed to the agent. (e.g. fetal alcohol syndrome)
- Relative risk the ratio of the rate of the condition among the exposed and the nonexposed. (e.g. smokers risk of having a low birth weight baby compared to non-smokers) A high relative risk may indicate a low absolute risk if the condition is rare.
- Mutagen a chemical or agent that can cause permanent damage to the deoxyribonucleic acid (DNA) in a cell. DNA damage in the human egg or sperm may lead to reduced fertility, spontaneous abortion (miscarriage), birth defects and heritable diseases.
- Fetotoxicant is a chemical that adversely affects the developing fetus, resulting in low birth weight, symptoms of poisoning at birth or stillbirth (fetus dies before it is born).
- Synergism when the combined effect of exposure to more than one chemical at one time, or to a chemical in combination with other hazards (heat, radiation, infection) results in effects of such exposure to be greater than the sum of the individual effects of each hazard by itself.
- Toxicogenomics the interaction between the genome, chemicals in the environment, and disease. Cells exposed to a stress, drug or toxicant respond by altering the pattern of expression of genes within their chromosomes. Based on new genetic and microarray technologies.
Teratogens
- Infections, collectively grouped under the acronym TORCH for Toxoplasmosis, Other organisms (parvovirus, HIV, Epstein-Barr, herpes 6 and 8, varicella, syphilis, enterovirus) , Rubella, Cytomegalovirus and Hepatitis. See also the related topics on maternal hyperthermia and bacterial infections. (More? Postnatal Immunisation)
- Maternal diet the best characterised is the role of low folic acid and Neural Tube Defects (NTDs) see also abnormal neural development and Neural Tube Defects (NTDs). More recently the focus has been on dietary iodine levels and the role they also play on neural development.
- Maternal drugs effects either prescription drugs (therapeutic chemicals/agents, thalidomide limb development), non-prescription drugs (smoking), and illegal drugs (Cannabis/Marijuana, Methamphetamine/Amphetamine, Cocaine, Heroin, Lysergic Acid Diethylamide)
- Environment (smoking, chemicals, heavy metals, radiation) and maternal endocrine function (maternal diabetes, thyroid development) and maternal stress.
- Teratogen synergism, different environmental effects can act individually or in combination on the same developing system. For example, neural development can be impacted upon by alcohol (fetal alcohol syndrome), viral infection (rubella) and/or inadequate dietry folate intake (neural tube defects). These effects may also not be seen as a direct effect on a system or systems but result in a reduced birth weight and the potential postnatal developmental effects. Consider also this in relation to the increasing support to the fetal origins hypothesis.
Links:
| Abnormality Links: abnormal development | abnormal genetic | abnormal environmental | Unknown | teratogens | ectopic pregnancy | cardiovascular abnormalities | coelom abnormalities | endocrine abnormalities | gastrointestinal abnormalities | genital abnormalities | head abnormalities | integumentary abnormalities | musculoskeletal abnormalities | limb abnormalities | neural abnormalities | neural crest abnormalities | placenta abnormalities | renal abnormalities | respiratory abnormalities | hearing abnormalities | vision abnormalities | twinning | Developmental Origins of Health and Disease | ICD-11 | ||
|
| Environmental Links: Introduction | low folic acid | iodine deficiency | Nutrition | Drugs | Australian Drug Categories | USA Drug Categories | thalidomide | herbal drugs | Illegal Drugs | smoking | Fetal Alcohol Syndrome | TORCH | viral infection | bacterial infection | fungal infection | zoonotic infection | toxoplasmosis | Malaria | maternal diabetes | maternal hypertension | maternal hyperthermia | Maternal Inflammation | Maternal Obesity | hypoxia | biological toxins | chemicals | heavy metals | air pollution | radiation | Prenatal Diagnosis | Neonatal Diagnosis | International Classification of Diseases | Fetal Origins Hypothesis |
| Genetic Links: genetic abnormalities | maternal age | Trisomy 21 | Trisomy 18 | Trisomy 13 | Trisomy X | trisomy mosaicism | Monosomy | Fragile X | Williams | Alagille | Philadelphia chromosome | mitochondria | VACTERL | hydatidiform mole | epigenetics | Prenatal Diagnosis | Neonatal Diagnosis | meiosis | mitosis | International Classification of Diseases | genetics |
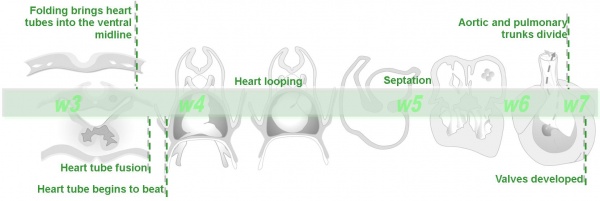
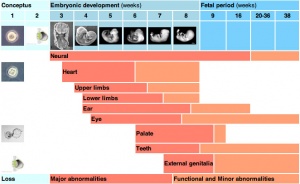
Critical Periods of Development
- Finally, when studying this topic remember the concept of critical periods of development that will affect the overall impact of the above listed factors. This can be extended to the potential differences between prenatal and postnatal effects, for example with infections and outcomes.
| Conceptus | Embryonic development (weeks) | Fetal period (weeks) | |||||||||||||||||
|
|||||||||||||||||||
| Neural | |||||||||||||||||||
| Heart | |||||||||||||||||||
| Upper limbs | |||||||||||||||||||
| Lower limbs | |||||||||||||||||||
| Ear | |||||||||||||||||||
| Eye | |||||||||||||||||||
| Palate | |||||||||||||||||||
| Teeth | |||||||||||||||||||
| External genitalia | |||||||||||||||||||
| Loss | Major abnormalities | Functional and Minor abnormalities | |||||||||||||||||
Links: Embryonic Development | Timeline human development | Movie - Human Development annotated cartoon | Human - critical periods
Australian Drug Categories
Legal drugs are classified, usually by each country's appropriate regulatory body, on the safety of drugs during pregnancy. In Australia, the Therapeutic Goods Authority has classes (A, B1, B2, B3, C, D and X) to define their safety. In the USA, drugs are classified by the Food and Drug Administration (FDA) into classes (A, B, C, D, and X) to define their safety. (More? Australian Drug Categories)
- Pregnancy Category A - Have been taken by a large number of pregnant women and women of childbearing age without an increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed.
- Pregnancy Category B1 - Drugs which have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals have not shown evidence of an increased occurrence of fetal damage.
- Pregnancy Category B2 - Have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals are inadequate or may be lacking, but available data show no evidence of an increased occurrence of fetal damage.
- Pregnancy Category B3 - Have been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human fetus having been observed. Studies in animals have shown evidence of an increased occurrence of fetal damage, the significance of which is considered uncertain in humans.
- Pregnancy Category C - Have caused or may be suspected of causing, harmful effects on the human fetus or neonate without causing malformations. These effects may be reversible.
- Pregnancy Category D - Have caused, are suspected to have caused or may be expected to cause, an increased incidence of human fetal malformations or irreversible damage. These drugs may also have adverse pharmacological effects.
- Pregnancy Category X - Have such a high risk of causing permanent damage to the fetus that they should NOT be used in pregnancy or when there is a possibility of pregnancy.
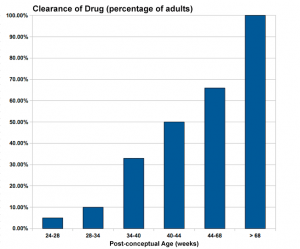
Infant Drug Clearance
The drug clearance data below are only approximate calculated rates for the fetus and infant from NZ Drug Safety in Lactation
| Post-conceptual Age (weeks) | Clearance of Drug (percentage of adults) |
| 24-28 | 5% |
| 28-34 | 10% |
| 34-40 | 33% |
| 40-44 | 50% |
| 44-68 | 66% |
| > 68 | 100% |
Links: Abnormal Development - Drugs | Australian Fetal Risk Categories | USA FDA Fetal Risk Categories | Therapeutic Goods Authority | Australian Drug Evaluation Committee (ADEC) | TGA - Medicines Pregnancy Database | Appendix A: Therapeutic goods exempted from pregnancy classification | NSW Poisons Information Centre
References
- ↑ Li Z, Zeki R, Hilder L & Sullivan EA 2013. Australia’s mothers and babies 2011. Perinatal statistics series no. 28. Cat. no. PER 59. Canberra: AIHW.
- ↑ Li Z, McNally L, Hilder L & Sullivan EA 2011. Australia's mothers and babies 2009 AIHW Perinatal statistics series no. 25 Cat. no. PER 52. Sydney: AIHW National Perinatal Epidemiology and Statistics Unit.
- ↑ Laws P & Sullivan EA 2010 Australia's mothers and babies 2008 AIHW Perinatal statistics series no. 24 Cat. no. PER 48. Sydney: AIHW National Perinatal Statistics Unit.
- ↑ Laws P & Sullivan EA 2009. Australia's mothers and babies 2007 AIHW Perinatal statistics series no. 23 Cat. no. PER 48. Sydney: AIHW National Perinatal Statistics Unit.
- ↑ <pubmed>20543995</pubmed>| PLoS Medicine
- ↑ AIHW National Perinatal Epidemiology and Statistics Unit and AIHW 2013. National core maternity indicators. Cat. no. PER 58. Canberra: AIHW.
- ↑ <pubmed>23470111</pubmed>
- ↑ AIHW, Macaldowie A, Wang YA, Chambers GM & Sullivan EA 2012. Assisted reproductive technology in Australia and New Zealand 2010. Assisted reproduction technology series. Cat. no. PER 55. Canberra: AIHW. Online Summary | PDF
- ↑ Wang YA, Macaldowie A, Hayward I, Chambers GM, & Sullivan EA 2011. Assisted reproductive technology in Australia and New Zealand 2009. Assisted reproduction technology series no. 15. Cat. no. PER 51. Canberra: AIHW. Online Summary | PDF
- ↑ Confidential Enquiry into Maternal Deaths (CEMD) Why Mothers Die 2000–2002 PDFPDF2
- ↑ <pubmed>19780726</pubmed>
- ↑ <pubmed>12957099</pubmed>
Links
The following are links to relevant notes pages that cover the key embryology concepts in this tutorial. These pages and their links will provide further detailed information.
Applied Embryology
Timeline human development | Fetal Development | Birth | Apgar test | Neonatal Development | Week 2 Abnormalities - Trophoblastic Disease | Placenta Development | Neural Abnormalities | Abnormal Development - Folic Acid and Neural Tube Defects | Week 3 | Cardiovascular Abnormalities | Twinning | Blastocyst | Molecular Development
Teratology Links
Human Abnormal Development | Genetic Abnormalities | Environmental Factors | Drugs | Trisomy 21 (Down Syndrome) | Fetal Alcohol Syndrome | Viral Infection | Rubella Virus | Hyperthermia
Self-Directed Learning
| Self-Directed Learning 1 - Australian Statistics |
Once you have thought about the Australian statistics, now look at the latest report summary Australia’s mothers and babies 2010 and Australian Statistics.
|
| Self-Directed Learning 2 - Pregnancy |
|
| Self-Directed Learning 3 - Assisted Reproductive Technologies |
|
| Self-Directed Learning 4 - The First Few Weeks |
|
| Self-Directed Learning 5 - Abnormal Development |
|
| Self-Directed Learning 6 - Prenatal Diagnosis |
|
| Self-Directed Learning 7 - Medications in Pregnancy |
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Department of Health and Ageing The National Maternity Services Plan 2010 | National Maternity Services Plan: 2010 -2011 Annual Report
- Australia AIHW National Perinatal Statistics Unit | Victorian Birth Defects Register (VBDR) | Victorian Birth Defects Register brochure
- National Perinatal Statistics Unit Congenital Anomalies Neural tube defects in Australia - An epidemiological report | Congenital Anomalies in Australia 2002-2003 | Congenital Anomalies in Australia 1998-2001 | Congenital Malformations Australia 1981-1997 | Congenital Malformations Australia 1995 and 1996 | Congenital Malformations Australia 1993 and 1994 | Congenital Malformations Australia 1981-1992
- Neonatal Networks
- Australian & New Zealand Neonatal Network (ANZNN) Neonatal Intensive Care Units
- Canada Canadian Neonatal Network
- European Neonatal Network EuroNeoNet
- USA and Other International Vermont Oxford Network
- Therapeutic Goods Authority TGA | Australian Drug Evaluation Committee (ADEC) | Prescribing Medicines in Pregnancy | Appendix A: Therapeutic goods exempted from pregnancy classification
- NSW Poisons Information Centre Poisons Information Centre
- USA Food and Drug Administration Evaluating the Risks of Drug Exposure in Human Pregnancies | Centers for Disease Control and Prevention (CDC, USA) Pregnancy Risk Assessment Monitoring System (PRAMS) collects state-specific, population-based data on maternal attitudes and experiences before, during, and shortly after pregnancy.
- Other Motherisk (Canada) Drugs, chemicals, radiation and herbal products in pregnancy | International Society for the Study of Trophoblastic Diseases Trophoblastic Diseases
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 9) Embryology BGD Tutorial - Applied Embryology and Teratology. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/BGD_Tutorial_-_Applied_Embryology_and_Teratology
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G