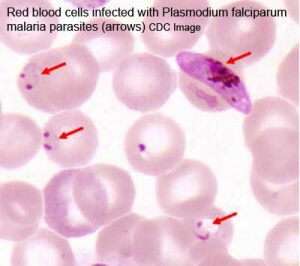
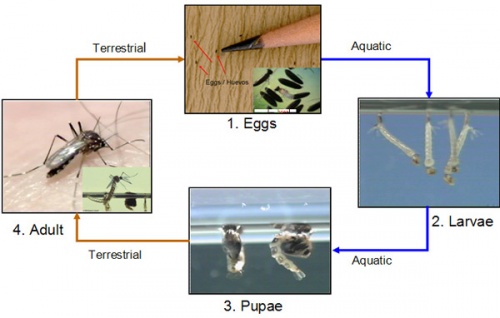
Abnormal Development - Malaria
| Embryology - 25 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
About 10,000 women and 200,000 babies die annually because of malaria in pregnancy, which can cause miscarriages, preterm births, and low-birth-weight births.[1] There are about 156 species of Plasmodium which infect different vertebrate species. In humans there are four types of malaria caused by the protozoan parasite Plasmodium falciparum (main), Plasmodium vivax, Plasmodium ovale, Plasmodium malariae.
These malarial parasites are carried by the female mosquito (anopheles species) and about 100 different species can transmit human malaria. When an infected mosquito bites a human, the parasites can enter the bloodstream and travel for about an hour before entering the liver and then multiplying. Between 6 to 16 days (depending on the species), the parasites reenter the bloodstream to invade and multiply inside red blood cells (RBCs). These infected RBCs rupture and released more parasites that infect and destroy further RBCs.
Placental infection is common in regions where malaria is endemic with women carrying their first pregnancy (primigravida). (More? Placenta - Abnormalities)
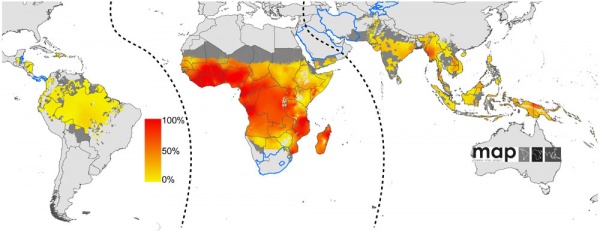
|
| Global limits and endemicity of P. falciparum in 2007 |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Maternal Malaria | Placental Malaria | Fetal Malaria |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Mosquito Lifecycle
Placental Malaria
Pregnant women have an increased susceptibility to malaria infection. Malarial infection of the placenta by sequestration of the infected red blood cells leading to low birth weight and other effects.
- Several infective agents may cross into the placenta from the maternal circulation, as well as enter the embryo/fetal circulation.
- Pregnant women have an increased susceptibility to malaria infection.
- Malarial infection of the placenta by sequestration of the infected red blood cells leading to low birth weight and other effects.
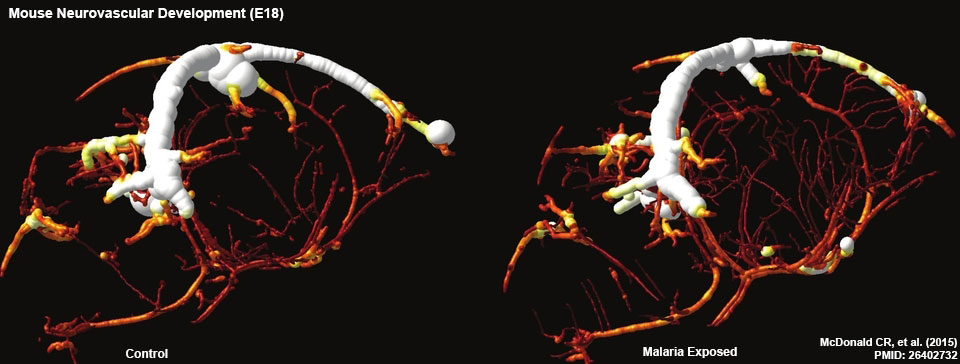
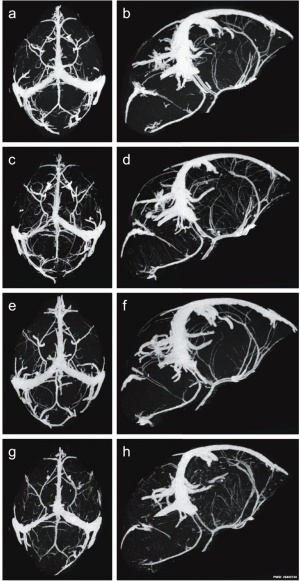
Mouse Model
Mouse E18 neurovasculature MicroCT[2]
References
- ↑ Dellicour S, Tatem AJ, Guerra CA, Snow RW & ter Kuile FO. (2010). Quantifying the number of pregnancies at risk of malaria in 2007: a demographic study. PLoS Med. , 7, e1000221. PMID: 20126256 DOI.
- ↑ 2.0 2.1 2.2 McDonald CR, Cahill LS, Ho KT, Yang J, Kim H, Silver KL, Ward PA, Mount HT, Liles WC, Sled JG & Kain KC. (2015). Experimental Malaria in Pregnancy Induces Neurocognitive Injury in Uninfected Offspring via a C5a-C5a Receptor Dependent Pathway. PLoS Pathog. , 11, e1005140. PMID: 26402732 DOI.
- ↑ Ngai M, Weckman AM, Erice C, McDonald CR, Cahill LS, Sled JG & Kain KC. (2020). Malaria in Pregnancy and Adverse Birth Outcomes: New Mechanisms and Therapeutic Opportunities. Trends Parasitol. , 36, 127-137. PMID: 31864896 DOI.
- ↑ Dombrowski JG, Souza RM, Lima FA, Bandeira CL, Murillo O, Costa DS, Peixoto EPM, Cunha MDP, Zanotto PMA, Bevilacqua E, Grisotto MAG, Pedroso de Lima AC, Singer JDM, Campino S, Clark TG, Epiphanio S, Gonçalves LA & Marinho CRF. (2019). Association of Malaria Infection During Pregnancy With Head Circumference of Newborns in the Brazilian Amazon. JAMA Netw Open , 2, e193300. PMID: 31050780 DOI.
- ↑ Umbers AJ, Stanisic DI, Ome M, Wangnapi R, Hanieh S, Unger HW, Robinson LJ, Lufele E, Baiwog F, Siba PM, King CL, Beeson JG, Mueller I, Aplin JD, Glazier JD & Rogerson SJ. (2013). Does malaria affect placental development? Evidence from in vitro models. PLoS ONE , 8, e55269. PMID: 23383132 DOI.
- ↑ Rijken MJ, Papageorghiou AT, Thiptharakun S, Kiricharoen S, Dwell SL, Wiladphaingern J, Pimanpanarak M, Kennedy SH, Nosten F & McGready R. (2012). Ultrasound evidence of early fetal growth restriction after maternal malaria infection. PLoS ONE , 7, e31411. PMID: 22347473 DOI.
- ↑ Westenberger SJ, McClean CM, Chattopadhyay R, Dharia NV, Carlton JM, Barnwell JW, Collins WE, Hoffman SL, Zhou Y, Vinetz JM & Winzeler EA. (2010). A systems-based analysis of Plasmodium vivax lifecycle transcription from human to mosquito. PLoS Negl Trop Dis , 4, e653. PMID: 20386602 DOI.
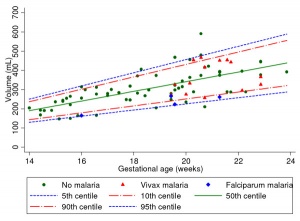
- ↑ Rijken MJ, Moroski WE, Kiricharoen S, Karunkonkowit N, Stevenson G, Ohuma EO, Noble JA, Kennedy SH, McGready R, Papageorghiou AT & Nosten FH. (2012). Effect of malaria on placental volume measured using three-dimensional ultrasound: a pilot study. Malar. J. , 11, 5. PMID: 22222152 DOI.
Books
- Guidelines for the Treatment of Malaria. 3rd edition. Geneva: World Health Organization; 2015. Available from: http://www.ncbi.nlm.nih.gov/books/NBK294440/
- Bioinformatics in Tropical Disease Research: A Practical and Case-Study Approach Gruber, Arthur; Durham, Alan M.; Huynh, Chuong; del Portillo, Hernando A., editors Bethesda (MD): National Library of Medicine (US), NCBI; 2008 Control of Gene Expression in Plasmodium
Reviews
Cutts JC, Agius PA, Zaw Lin R, Powell K, Moore B, Draper JA, Simpson FJI & Fowkes. (2020). Pregnancy-specific malarial immunity and risk of malaria in pregnancy and adverse birth outcomes: a systematic review. BMC Med , 18, 14. PMID: 31941488 DOI.
Bauserman M, Conroy AL, North K, Patterson J, Bose C & Meshnick S. (2019). An overview of malaria in pregnancy. Semin. Perinatol. , , . PMID: 30979598 DOI.
Clark RL. (2019). Genesis of placental sequestration in malaria and possible targets for drugs for placental malaria. Birth Defects Res , , . PMID: 30919596 DOI.
Milner DA. (2018). Malaria Pathogenesis. Cold Spring Harb Perspect Med , 8, . PMID: 28533315 DOI.
Ataíde R, Mayor A & Rogerson SJ. (2014). Malaria, primigravidae, and antibodies: knowledge gained and future perspectives. Trends Parasitol. , 30, 85-94. PMID: 24388420 DOI.
Rogerson SJ, Mwapasa V & Meshnick SR. (2007). Malaria in pregnancy: linking immunity and pathogenesis to prevention. Am. J. Trop. Med. Hyg. , 77, 14-22. PMID: 18165470
Rogerson SJ, Hviid L, Duffy PE, Leke RF & Taylor DW. (2007). Malaria in pregnancy: pathogenesis and immunity. Lancet Infect Dis , 7, 105-17. PMID: 17251081 DOI.
Beeson JG & Duffy PE. (2005). The immunology and pathogenesis of malaria during pregnancy. Curr. Top. Microbiol. Immunol. , 297, 187-227. PMID: 16265906
Articles
Oktavianthi S, Fauzi M, Trianty L, Trimarsanto H, Bowolaksono A, Noviyanti R & Malik SG. (2019). Placental mitochondrial DNA copy number is associated with reduced birth weight in women with placental malaria. Placenta , 80, 1-3. PMID: 31103060 DOI.
Olaleye A, Okusanya BO, Oduwole O, Esu E & Meremikwu M. (2019). A systematic review and meta-analysis of dihydroartemisinin-piperaquine versus sulphadoxine-pyrimethamine for malaria prevention in pregnancy. Int J Gynaecol Obstet , , . PMID: 31050803 DOI.
Lawford HLS, Lee AC, Kumar S, Liley HG & Bora S. (2019). Establishing a conceptual framework of the impact of placental malaria on infant neurodevelopment. Int. J. Infect. Dis. , 84, 54-65. PMID: 31028878 DOI.
Hangi M, Achan J, Saruti A, Quinlan J & Idro R. (2019). Congenital Malaria in Newborns Presented at Tororo General Hospital in Uganda: A Cross-Sectional Study. Am. J. Trop. Med. Hyg. , 100, 1158-1163. PMID: 30860019 DOI.
Dimasuay KG, Gong L, Rosario F, McBryde E, Spelman T, Glazier J, Rogerson SJ, Beeson JG, Jansson T, Devenish RJ & Boeuf P. (2017). Impaired placental autophagy in placental malaria. PLoS ONE , 12, e0187291. PMID: 29125872 DOI.
Search PubMed
Search Pubmed: Placental Malaria
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- CDC Division of Parasitic Diseases and Malaria Malaria
- Toronto General Hospital/Research Institute Kevin Kain
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 25) Embryology Abnormal Development - Malaria. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Malaria
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G