Week 2
| Embryology - 3 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
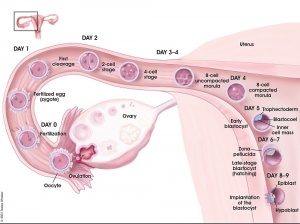
Key events of human development during the second week (week 2) following fertilization or clinical gestational age GA week 4, base on last menstrual period.
Week 2 is about the implantation process and blastocyst differentiation. Note that all cells produced from the initial fertilization event are defined as the "conceptus" and will include cells with both embryonic and extraembryonic futures. In the conceptus, this is a period of blastocyst "hatching" rapid blastocyst differentiation into extraembryonic and embryonic tissues and proliferation. In placental animals, this is the first physical interaction between the conceptus and the maternal uterine wall with adplantation and the commencement of implantaion.
The implanting conceptus releases a hormone (human Chorionic Gonadotropin or hCG) that initiates maternal hormonal changes, stopping the menstrual cycle. Detection of hCG in maternal urine or blood is also the basis of many modern pregnancy tests.
| Week 2 Links: stage 4 | stage 5 | stage 6 | Lecture - Week 1 and 2 | implantation | trophoblast | human chorionic gonadotropin | pregnancy test | twinning | Category:Week 2 | GA week 4 |
Carnegie Stages
| Week: | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Carnegie stage: | 1 2 3 4 | 5 6 | 7 8 9 | 10 11 12 13 | 14 15 | 16 17 | 18 19 | 20 21 22 23 |
Historic Carnegie Stages 5 and 6.
| Stage 5 Links: Week 2 | Implantation | Lecture | Practical | Carnegie Embryos | Category:Carnegie Stage 5 | Next Stage 6 | ||
| Historic Papers: 1941 | 1944 day 9-10 | 1945 day 7.5 | 1945 day 9-10 | ||
|
| Stage 6 Links: Week 2 | Implantation | Lecture | Practical | Carnegie Embryos | Category:Carnegie Stage 6 | Next Stage 7 |
| Historic Papers: 1909 | 1925 | 1937 |
Timeline
| Week 2 - Human Embryo Stages and Events (GA week 4) | ||
|---|---|---|
| Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9 | ||
| Event | ||
| Stage 5 | 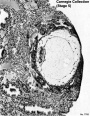 | |
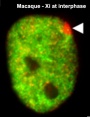 X inactivation X inactivation
| ||
| Stage 6 | ||
| Note - the day timing of stages is only approximate, system names link to first page of that specific system, and events are based upon the literature cited below. | ||
| References
| ||
| Event | ||
| Stage 5 |  | |
 X inactivation X inactivation
| ||
| Stage 6 | ||
Week 2 Movies
|
|
|
- Links: Week 2 Movies | Movies
Implantation
The second week of human development is concerned with the process of implantation and the differentiation of the blastocyst into early embryonic and placental forming structures.
- implantation commences about day 6 to 7
- Adplantation - begins with initial adhesion to the uterine epithelium
- blastocyst then slows in motility, "rolls" on surface, aligns with the inner cell mass closest to the epithelium and stops
- Implantation - migration of the blastocyst into the uterine epithelium, process complete by about day 9
- coagulation plug - left where the blastocyst has entered the uterine wall day 12
Normal Implantation Sites - in uterine wall superior, posterior, lateral
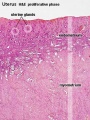
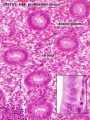
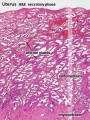
Uterus
- Endometrium - 3 layers in secretory phase of menstrual cycle: compact, spongy, basal
- Myometrium - muscular layer outside endometrium, contracts in parturition
- Perimetrium - tunica serosa of the uterus continuous with the peritoneal wall
Endometrial Layers
- Compact - implantation occurs in this layer, dense stromal cells, uterine gland necks, capillaries of spiral arteries
- Spongy - swollen stromal cells, uterine gland bodies, spiral arteries
- Basal - not lost during menstruation or childbirth, own blood supply
Decidual Reaction
- occurs initially at site of implantation and includes both cellular and matrix changes
- reaction spreads throughout entire uterus, not at cervix
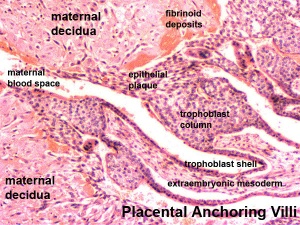
- deposition of fibrinoid and glycogen and epithelial plaque formation (at anchoring villi)
- presence of decidual cells are indicative of pregnancy
Cervix - at mouth of uterus, secretes mucus (CMP), forms a plug/barrier, mechanical and antibacterial Vascular - increased number of blood vessels
Decidua
The endometrium becomes the decidua and forms 3 distinct anatomical regions (at approx 3 weeks)
- Decidua Basalis at implantation site
- Decidua Capsularis enclosing the conceptus
- Decidua Parietalis the remainder of uterus
- Decidua Capsularis and Parietalis fuse eventually fuse and uterine cavity is lost by 12 weeks
Bilaminar Embryo
- Human development about day 8 to 9
The embryoblast (inner cell mass) forms the epiblast and hypoblast layers. This early stage of embryo development is referred to as the bilaminar embryo.
The inner cell mass forms an inner layer of larger cells is also called the "embryoblast" is a cluster of cells located and attached on one wall of the outer trophoblast layer. In week 2 this mass will differentiate into two distinct layers the epiblast and hypoblast, also called the bilaminar embryo.
Epiblast
The epiblast layer will form the entire embryo and undergoes gastrulation in week three to form the 3 germ layers. It also forms an epithelial layer lining the amniotic cavity.
Hypoblast
The hypoblast (or primitive endoderm) is a transient epithelial layer facing towards the blastoceol, it is replaced in week three by the gastrulation migrating endoderm cells.
Syncitiotrophoblasts
- secrete proteolytic enzymes, enzymes break down extracellular matrix around cells
- Allow passage of blastocyst into endometrial wall, totally surround the blastocyst
- generate spaces that fill with maternal blood- lacunae
- secrete Human Chorionic Gonadotropin (hCG), hormone, maintains decidua and corpus luteum. This hormone is diagnostically the basis of pregnancy tests, and is present in maternal.
- Later in development placenta will secrete hCG
Human Chorionic Gonadotropin
(hCG) Placental hormone initially secreted by cells (syncitiotrophoblasts) from the implanting conceptus during week two, supporting the ovarian corpus luteum, which in turn supports the endometrial lining and therefore maintains pregnancy.[1] Hormone can be detected in maternal blood and urine and is the basis of many pregnancy tests. Hormone also stimulates the onset of fetal gonadal steroidogenesis, high levels are teratogenic to fetal gonadal tissues. Other potential cellular sources can include: hyperglycosylated hCG produced by cytotrophoblast cells, free beta-subunit made by multiple primary non-trophoblastic malignancies, and pituitary hCG made by the gonadotrope cells of the anterior pituitary.
- The molecular weight of hCG is approximately 36,000 (36 KDa) for the α-subunit and a β-subunit that are held together by both non-covalent hydrophobic and ionic interactions.
Human Levels
- levels peak at 8 to 10 weeks of pregnancy, then decline and are lower for rest of pregnancy
- 0-1 week: 0-50 mIU/ml
- 1-2 weeks: 40-300 mIU/ml
- 3-4 weeks: 500-6,000 mIU/ml
- 1-2 months: 5,000-200,000 mIU/ml
- 2-3 months: 10,000-100,000 mIU/ml
- 2nd trimester: 3,000-50,000 mIU/ml
- 3rd trimester: 1,000-50,000 mIU/ml
Non-pregnant females: <5.0 mIU/ml Postmenopausal females: <9.5 mIU/ml
Functions
- Promotion of corpus luteal progesterone production
- Angiogenesis of uterine vasculature
- Cytotrophoblast differentiation
- Immuno-suppression and blockage of phagocytosis of invading trophoblast cells
- Growth of uterus in line with fetal growth
- Quiescence of uterine muscle contraction
- Promotion of growth and differentiation of fetal organs
- Umbilical cord growth and development
- Blastocysts signals endometrium prior to implantation
- hCG in sperm and receptors found in fallopian tubes suggesting pre-pregnancy communication
- hCG receptors in adult brain hippocampus, hypothalamus and brain stem, may cause pregnancy nausea and vomiting
- hCG and implantation of pregnancy, hCG stimulates metalloproteinases of cytotrophoblast cell
(Function Data from Table 1[1])
Week 2 Abnormalities
Abnormal Implantation
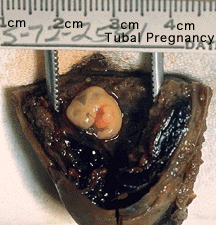
Ectopic Implantation (Pregnancy) | Ectopic pregnancy ultrasound movie
Abnormal implantation sites or Ectopic Pregnancy occurs if implantation is in uterine tube or outside the uterus.
- sites - external surface of uterus, ovary, bowel, gastrointestinal tract, mesentry, peritoneal wall
- If not spontaneous then, embryo has to be removed surgically
Tubal pregnancy - 94% of ectopic pregnancies
- if uterine epithelium is damaged (scarring, pelvic inflammatory disease)
- if zona pellucida is lost too early, allows premature tubal implantation
- embryo may develop through early stages, can erode through the uterine horn and reattach within the peritoneal cavity
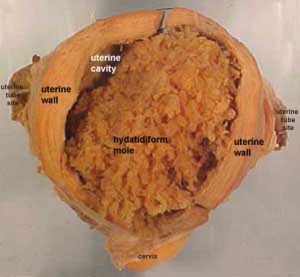
Hydatidiform Mole
Another type of abnormality is when only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole", which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus. The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo.
There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
This topic is also covered in Hydatidiform Mole and Placenta - Abnormalities.
Mole Types
Complete mole - chromosomal genetic material from the ovum (egg) is lost, by an unknown process. Fertilization then occurs with one or two sperm and an androgenic (from the male only) conceptus (fertilized egg) is formed. With this conceptus the embryo (fetus, baby) does not develop at all but the placenta does grow but it is abnormal and forms lots of cysts and has no blood vessels. These cysts look like a cluster of grapes and that is why it is called a hydatidiform mole (grape like). A hydatidiform mole miscarries by about 16 to 18 weeks gestational age. Since the diagnosis can be made by ultrasound before that time, it is better for you to have an evacuation of the uterus (D & C) so that there is no undue bleeding and no infection. Human chorionic gonadotropin (hCG) will assist in making the diagnosis.
Partial mole - three sets of chromosomes instead of the usual two and this is called triploidy. With such a pregnancy the chromosomal (genetic) material from the ovum (egg) is retained and the egg is fertilized by one or two sperm. Since with partial mole there are maternal chromosomes there is a fetus but because of the three sets of chromosomes this fetus is always grossly abnormal and will not survive. (Text modified from: International Society for the Study of Trophoblastic Diseases,see also JRM Gestational Trophoblastic Disease)
Tumour Growth
Like any tumour, unless removed there is a risk of progression:
- Stage I: Tumor confined to uterus (non-metastatic)
- Stage II: Tumor involving pelvic organs and/or vagina
- Stage III: Tumor involving lungs, with or without involving pelvic structures and/or vagina
- Stage IV: Tumor involving distant organs
Placental Mesenchymal Dysplasia
Due to a similar "grape-like" placental appearance, this rare disorder has been mistaken both clinically and macroscopically for a partial hydatidiform molar pregnancy. This disorder also has a high incidence of intrauterine growth restriction (IUGR) and fetal death.
Twin Pregnancy Mole
Hydatidiform mole and co-existent healthy fetus is a very rare condition with only 30 cases documented in detail in the literature.[2]
- Links: International Society for the Study of Trophoblastic Diseases | Sydney Gynaecological Oncology Group Gestational Trophoblastic Disease | The Journal of Reproductive Medicine Gestational Trophoblastic Disease (1998) | Dana-Farber Cancer Institute Gynecologic Oncology Program
Uterus Abnormalities
Endometriosis endometrial tissue located in other regions of the uterus or other tissues. This misplaced tissue develops into growths or lesions which respond to the menstrual cycle hormonal changes in the same way that the tissue of the uterine lining does; each month the tissue builds up, breaks down, and sheds.
- Links: Endometriosis
Monoygotic Twinning
Monozygotic twins (identical) produced from a single fertilization event (one fertilised egg and a single spermatazoa, form a single zygote), these twins therefore share the same genetic makeup. Occurs in approximately 3-5 per 1000 pregnancies, more commonly with aged mothers. The later the twinning event, the less common are initially separate placental membranes and finally resulting in conjoined twins.
| Week | Week 1 (GA week 3) | Week 2 (GA week 4) | |||||||||||||
| Day | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
| Cell Number | 1 | 1 | 2 | 16 | 32 | 128 | bilaminar | ||||||||
| Event | Ovulation | Fertilization | First cell division | Morula | Early blastocyst | Late blastocyst
Hatching |
Implantation starts | X inactivation | |||||||
|
|
|
|
||||||||||||
| Monoygotic
Twin Type |
Diamniotic
Dichorionic |
Diamniotic
Monochorionic |
Monoamniotic
Monochorionic |
Conjoined | |||||||||||
Table based upon recent a recent twinning review.[3] Links: twinning
- Links: Twinning
References
- ↑ 1.0 1.1 Cole LA. (2010). Biological functions of hCG and hCG-related molecules. Reprod. Biol. Endocrinol. , 8, 102. PMID: 20735820 DOI.
- ↑ Piura B, Rabinovich A, Hershkovitz R, Maor E & Mazor M. (2008). Twin pregnancy with a complete hydatidiform mole and surviving co-existent fetus. Arch. Gynecol. Obstet. , 278, 377-82. PMID: 18273627 DOI.
- ↑ Hall JG. (2003). Twinning. Lancet , 362, 735-43. PMID: 12957099 DOI.
Wamaitha SE & Niakan KK. (2018). Human Pre-gastrulation Development. Curr. Top. Dev. Biol. , 128, 295-338. PMID: 29477167 DOI.
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 3) Embryology Week 2. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Week_2
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G