Abnormal Development - Teratogens: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (2 intermediate revisions by the same user not shown) | |||
| Line 32: | Line 32: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Nanoparticle-induced neuronal toxicity across placental barriers is mediated by autophagy and dependent on astrocytes'''{{#pmid:29610530|PMID29610530}} The potential for maternal nanoparticle (NP) exposures to cause developmental toxicity in the fetus without the direct passage of NPs has previously been shown, but the mechanism remained elusive. We now demonstrate that exposure of cobalt and chromium NPs to BeWo cell barriers, an in vitro model of the human placenta, triggers impairment of the autophagic flux and release of interleukin-6. This contributes to the altered differentiation of human neural progenitor cells and DNA damage in the derived neurons and astrocytes." | |||
* '''Dispensing of potentially teratogenic drugs before conception and during pregnancy: a population-based study'''{{#pmid:25316196|PMID25316196}} "To study the dispensing of potentially teratogenic drugs in the 12-month period before as well as during pregnancy in the Netherlands. Drug-dispensing information was identified from the PHARMO Database Network for the 12-month period before conception and during pregnancy. Drugs with either a Swedish FASS 'D' classification, an Australian ADEC or American FDA 'D' or 'X' classification were considered potentially teratogenic (n = 202). ...Five percent of the pregnancies received a potentially teratogenic drug during pregnancy and 0.66% received a drug from the risk category X. It may be possible to reduce these proportions when reasons for prescription have been explored." (More? [[Abnormal Development - Drugs]] | [[Australian Drug Categories]] | [[USA Drug Categories]]) | * '''Dispensing of potentially teratogenic drugs before conception and during pregnancy: a population-based study'''{{#pmid:25316196|PMID25316196}} "To study the dispensing of potentially teratogenic drugs in the 12-month period before as well as during pregnancy in the Netherlands. Drug-dispensing information was identified from the PHARMO Database Network for the 12-month period before conception and during pregnancy. Drugs with either a Swedish FASS 'D' classification, an Australian ADEC or American FDA 'D' or 'X' classification were considered potentially teratogenic (n = 202). ...Five percent of the pregnancies received a potentially teratogenic drug during pregnancy and 0.66% received a drug from the risk category X. It may be possible to reduce these proportions when reasons for prescription have been explored." (More? [[Abnormal Development - Drugs]] | [[Australian Drug Categories]] | [[USA Drug Categories]]) | ||
| Line 52: | Line 54: | ||
When studying this topic remember the concept of "critical periods of development" that will affect the overall impact of the above listed factors, as outlined in the table below. This can be extended to the potential differences between prenatal and postnatal effects, for example with infections and outcomes. | When studying this topic remember the concept of "critical periods of development" that will affect the overall impact of the above listed factors, as outlined in the table below. This can be extended to the potential differences between prenatal and postnatal effects, for example with infections and outcomes. | ||
{{Critical Periods table}} | |||
==References== | ==References== | ||
Latest revision as of 09:45, 19 April 2018
| Embryology - 3 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Educational Use Only - Embryology is an educational resource for learning concepts in embryological development, no clinical information is provided and content should not be used for any other purpose. |
Introduction
How and why do things go wrong in development? Embryological development is a robust biological system able to cope with many stresses without long-term consequences. When development does go wrong there are generally 3 major types groups: Genetic (inherited), Environmental (maternal) derived and Unknown (not determined or known) abnormalities. Also often not considered, is that pregnancy itself can also expose abnormalities in the mother (congenital heart disease, diabetes, reproductive disorders) that until the pregnancy had gone undetected.
- Infections collectively grouped under the acronym TORCH for Toxoplasmosis, Other organisms (parvovirus, HIV, Epstein-Barr, herpes 6 and 8, varicella, syphilis, enterovirus) , Rubella, Cytomegalovirus and Hepatitis. See also the related topics on maternal hyperthermia and bacterial infections.
- Maternal diet the best characterised is the role of low folic acid and Neural Tube Defects (NTDs) see also abnormal neural development and Neural Tube Defects (NTDs). More recently the focus has been on dietary iodine levels and the role they also play on neural development.
- Maternal drugs effects either prescription drugs (therapeutic chemicals/agents, thalidomide limb development), non-prescription drugs (smoking), and illegal drugs (Cannabis/Marijuana, Methamphetamine/Amphetamine, Cocaine, Heroin, Lysergic Acid Diethylamide). Some therapeutic compounds have teratogenic effects because they are also naturally occurring developmental signals, for example retinoic acid.
- Environment (smoking, chemicals, heavy metals, radiation) and maternal endocrine function (maternal diabetes, thyroid development) and maternal stress.
- Teratogen synergism, different environmental effects can act individually or in combination on the same developing system. For example, neural development can be impacted upon by alcohol (fetal alcohol syndrome), viral infection (rubella) and/or inadequate dietry folate intake (neural tube defects). These effects may also not be seen as a direct effect on a system or systems but result in a reduced birth weight and the potential postnatal developmental effects. Consider also this in relation to the increasing support to the fetal origins hypothesis.
Use the page links below to explore specific teratogens.
| Bacterial Links: bacterial infection | syphilis | gonorrhea | tuberculosis | listeria | salmonella | TORCH | Environmental | Category:Bacteria |
| Abnormality Links: abnormal development | abnormal genetic | abnormal environmental | Unknown | teratogens | ectopic pregnancy | cardiovascular abnormalities | coelom abnormalities | endocrine abnormalities | gastrointestinal abnormalities | genital abnormalities | head abnormalities | integumentary abnormalities | musculoskeletal abnormalities | limb abnormalities | neural abnormalities | neural crest abnormalities | placenta abnormalities | renal abnormalities | respiratory abnormalities | hearing abnormalities | vision abnormalities | twinning | Developmental Origins of Health and Disease | ICD-11 | ||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: teratogen <pubmed limit=5>teratogen</pubmed> |
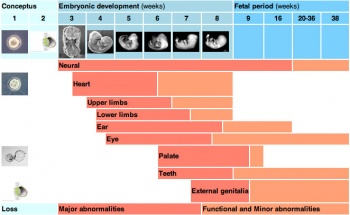
Critical Periods of Development
When studying this topic remember the concept of "critical periods of development" that will affect the overall impact of the above listed factors, as outlined in the table below. This can be extended to the potential differences between prenatal and postnatal effects, for example with infections and outcomes.
| Critical Periods of Human Development | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Conceptus | Embryonic development (weeks) | Fetal period (weeks) | ||||||||||||||||||
|
||||||||||||||||||||
| Neural | ||||||||||||||||||||
| Heart | ||||||||||||||||||||
| Upper limbs | ||||||||||||||||||||
| Lower limbs | ||||||||||||||||||||
| Ear | ||||||||||||||||||||
| Eye | ||||||||||||||||||||
| Palate | ||||||||||||||||||||
| Teeth | ||||||||||||||||||||
| External genitalia | ||||||||||||||||||||
| Loss | Major abnormalities | Functional and Minor abnormalities | ||||||||||||||||||
| ||||||||||||||||||||
References
- ↑ Hawkins SJ, Crompton LA, Sood A, Saunders M, Boyle NT, Buckley A, Minogue AM, McComish SF, Jiménez-Moreno N, Cordero-Llana O, Stathakos P, Gilmore CE, Kelly S, Lane JD, Case CP & Caldwell MA. (2018). Nanoparticle-induced neuronal toxicity across placental barriers is mediated by autophagy and dependent on astrocytes. Nat Nanotechnol , , . PMID: 29610530 DOI.
- ↑ Zomerdijk IM, Ruiter R, Houweling LM, Herings RM, Straus SM & Stricker BH. (2015). Dispensing of potentially teratogenic drugs before conception and during pregnancy: a population-based study. BJOG , 122, 1119-29. PMID: 25316196 DOI.
- ↑ Mayshar Y, Yanuka O & Benvenisty N. (2011). Teratogen screening using transcriptome profiling of differentiating human embryonic stem cells. J. Cell. Mol. Med. , 15, 1393-401. PMID: 20561110 DOI.
- ↑ Lim JH, Kim SH, Shin IS, Park NH, Moon C, Kang SS, Kim SH, Park SC & Kim JC. (2011). Maternal exposure to multi-wall carbon nanotubes does not induce embryo-fetal developmental toxicity in rats. Birth Defects Res. B Dev. Reprod. Toxicol. , 92, 69-76. PMID: 21254368 DOI.
Journals
- Birth Defects Research Part A: Clinical and Molecular Teratology
- Birth Defects Research Part B: Developmental and Reproductive Toxicology
- Part C: Embryo Today: Reviews
Reviews
Articles
Search Pubmed
Search Pubmed: teratogen | teratogenesis
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 3) Embryology Abnormal Development - Teratogens. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Teratogens
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G