Lecture - Early Vascular Development
| Embryology - 9 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This lecture has 2 parts; firstly introducing an overview of early vascular development, secondly the key events in heart development.
- Vasculogenesis - formation of new blood vessels assemble from individual precursor cells.
- Angiogenesis - sprouting of new vessels occurs from pre-existing vessels.
Lecture Objectives
- Understanding of early blood vessel and blood development
- Brief understanding of vascular growth and regression
- Brief understanding of vascular growth factors
- Understanding of heart development
- Understanding of heart timeline
- Understanding of heart septation
Lecture Resources
| Movies | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
Early Development Overview
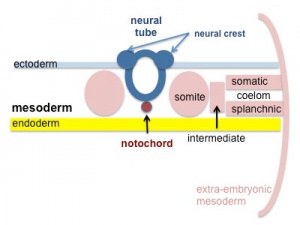
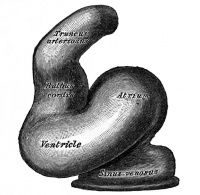
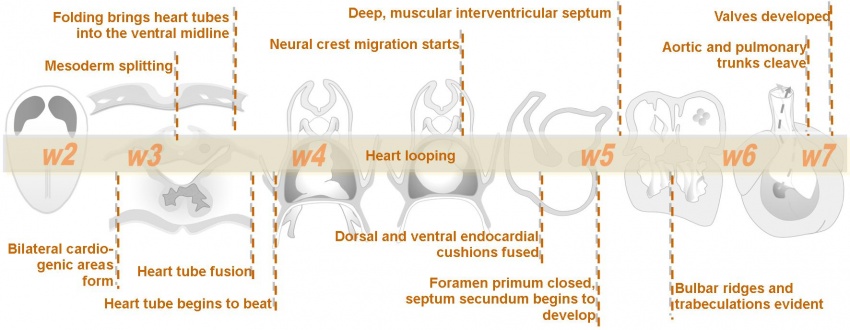
The heart develops from cardiogenic mesoderm that originally lies above the cranial end of the developing neural tube. Enlargement of the cranial neural fold brings this region ventrally to its correct anatomical position. The original paired cardiac tubes fuse, with the "ventricular" primordia initially lying above the "atria". Growth of the cardiac tube flexes it into an "S-shape" tube, rotating the "ventricles" downward and pushing the "atria" upward.
This is then followed by septation, a complex process which converts this simple tube into a four chambered heart (see Heart Development later in this lecture). A key part of this process is the separation of cardiac outflow (truncus arteriosus) into a separate pulmonary and aortic arch outflow. During embryonic development there is extensive remodelling of the initially right and left symmetrical cardiovascular system and a contribution from the neural crest to some vessels.
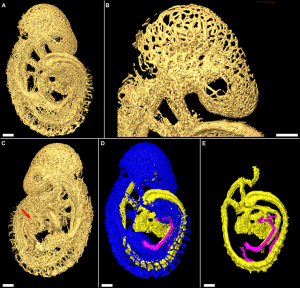
|
| The Human Heart from day 10 to 25 (scanning electron micrograph) |
Timecourse
|
|
Vasculogenesis and Angiogenesis
| Vasculogenesis | Angiogenesis |
|---|---|
| formation of new blood vessels (endothelium from mesoderm) |
formation of blood vessels from pre-existing vessels (occurs in development and adult) |
- Begins week 3 in extraembryonic mesoderm and then embryonic splanchnic mesoderm
- Begins as the formation of blood islands
- Earliest islands - yolk sac, connecting stalk and chorion (Area vasculosa)
- Growth factors stimulate growth and development - Vascular Endothelial Growth Factor (VEGF) and Placental Growth Factor (PlGF, PGF)
- Growing blood vessels follow a gradient generated by target tissues/regions of Vascular Endothelial Growth Factor (VEGF) to establish a vascular bed. Recent findings suggest that Notch signaling acts as an inhibitor for this system, preventing sprouting of blood vessels. Notch is a transmembrane receptor protein involved in regulating cell differentiation in many developing systems.
- PIGF is also a VEGF released from the placental trophoblast cells.
- angioblasts form clusters called "blood islands"
- blood islands extend and fuse together to form a primordial vascular network
Blood islands
- Blood islands contain cells (haemangioblasts) which are capable of differentiating into 2 populations of cells
- Vascular precursors (angioblasts) - form endothelial cells
- Blood cell precursors (haemocytoblasts)
- These angioblasts migrate, coalesce into cords and form a lumen. This process of vessel formation is called vasculogenesis and is dominant in very early embryogenesis e.g. formation of the dorsal aorta
- Sprouting from pre-existing vessels is called angiogenesis e.g. brain is an organ which is vascularized by this process
- Note: the vascular tree undergoes constant remodeling as the embryo grows.
Blood vessel lumen formation
Blood formation
|
Mouse hematopoietic stem cell location |
Red blood cells
| The only cells in the blood are nearly entirely fetal red blood cells.
These cells differ from adult red blood cells in:
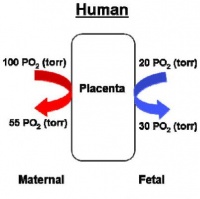
Fetal Oxygen levels |
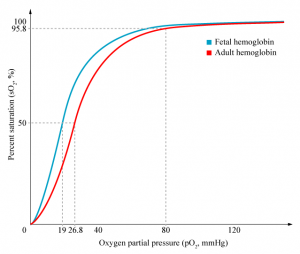
Red Blood Cell haemoglobin oxygen dissociation curves |
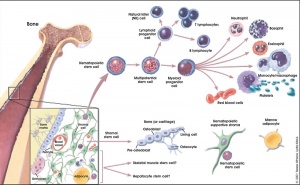
Blood stem cells
Hematopoietic and stromal cell differentiation (adult)
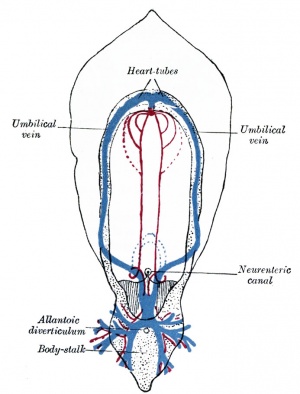
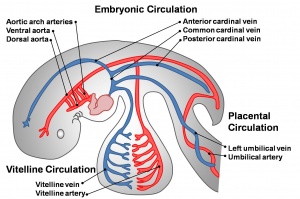
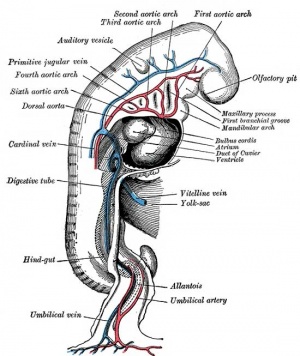
Early vascular systems
- one vascular system with 3 components - vitelline, embryonic (system) and placental
- each component has own system of artery and vein
Vitelline blood vessels
- Angioblasts form a network of vessels over the yolk sac and connecting stalk
- Join into two main vessels, the vitteline veins (omphalomesenteric)
- Pass through vitello-intestinal duct (yolk sac stalk)
- Enter caudal end of cardiac tube
- Vitelline Arteries - arises from dorsal aorta, contribute to adult GIT arteries (fuse to become superior mesenteric artery (midgut)
- Vitelline Veins - empties into sinus venosus, contribute to the adult portal system
Embryo blood vessels
- (systemic) will form the most of the cardiovascular system
- some vessels have neural crest contribution
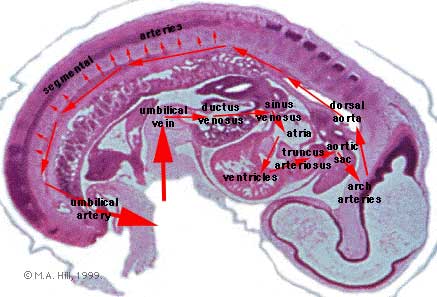
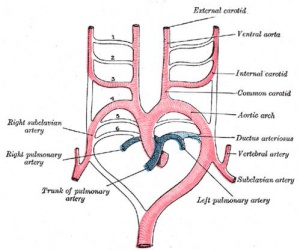
- Arterial blood flow - aortic sac → aortic arches → dorsal aorta →umbilical artery
- dorsal aorta,
- paired initially , later fuses from T4 to L4 (gives off segmental arteries)
- connect to ventral aorta via pharyngeal arches arteries.
- caudally, give rise to umbilical arteries
- laterally, give rise to intersegmental arteries
- dorsal aorta,
- Veins - 3 pairs of veins empty into the sinus venosus of the heart
- vitelline, umbilical (right and left from developing placenta enter caudal cardiac tube; only left persists)
- cardinal veins - anterior, common, posterior
|
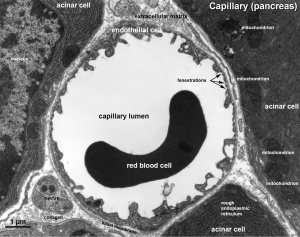
|
| Vein histology | Blood capillary (EM) |
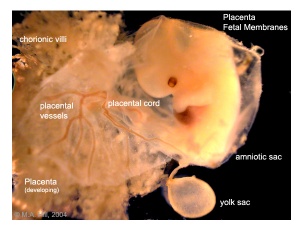
Placental blood vessels
(Placenta development covered in next lecture)
- form initially in the connecting stalk (then umbilical cord) and anastomose in chorion
- extend maternally - toward the chorionic villi
- extend embryonically - toward the sinus venosus and dorsal aorta
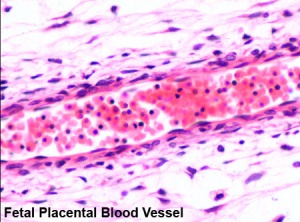
- Arteries - paired and carry deoxygenated blood (from dorsal aorta) and waste products to the placental villi
- Veins - paired initially then only left at end of embryonic period and carry oxygenated blood to the embryo (sinus venosus)
Blood flow through the embryo
High pressure pathway
Maternal Blood | -> umbilical vein -> liver -> anastomosis -> sinus venosus -> atria ventricles-> truncus arteriosus -> aortic sac -> aortic arches-> dorsal aorta-> pair of umbilical arteries | Maternal Blood.
Low pressure pathway
- Head - Large veins lateral to dorsal aortae. These are the superior or anterior cardinal veins. Their function is to drain the head region.
- Body - Large veins lateral to dorsal aortae. These are the inferior or posterior cardinal veins. Their function is to drain the lower part of the embryo.
Blood vessel remodeling
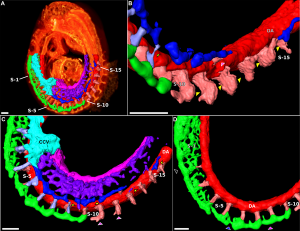
|
|
| Early vascular development is laterally symmetrical (paired left and right). | Embryo development extensively remodelled leading to an asymmetric adult system in the body. Complex balance between Stimulators and Inhibitors of Angiogenisis. |
Vascular Endothelial Growth Factor (VEGF)
- belongs to the platelet derive growth factor (PDGF) family.
- required for early stages of blood vessel patterning.
- required later for endothelial cell maintenance in tissues.
- autocrine VEGF loop from endothelial cell secretion involved in vascular growth.
- 4 protein isoforms generated from a single gene.
VEGF protein family - VEGF (or VEGF-A), VEGF-B, VEGF-C, VEGF-D and placental growth factor (PGF),
VEGF receptors - VEGFR-1, -2 and -3.
- Cells expressing the receptors are directed in their growth.
- Note that there are other growth factor families (FGF, Tie, TGF-β, netrins, semaphorins) that can also influence vessel growth.
- Some angiogenic factors also involved in organ development (liver).
Heart Development
Septation
There are several key developmental events that divide the heart into 4 separate chambers (right atria, right ventricle, left atria, left ventricle) and also divide the outflow tract into 2 (aortic and pulmonary trunks). You should understand the sequence and timing of these septation events. The linked online tutorial is a useful supplement to your understanding of these events.
- atrioventricular separation
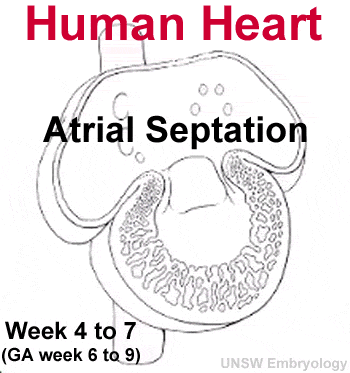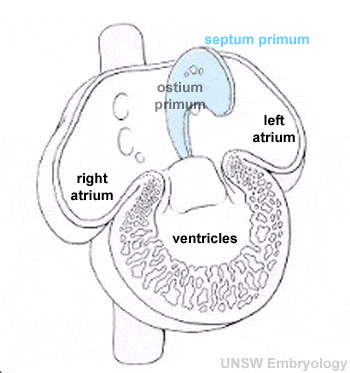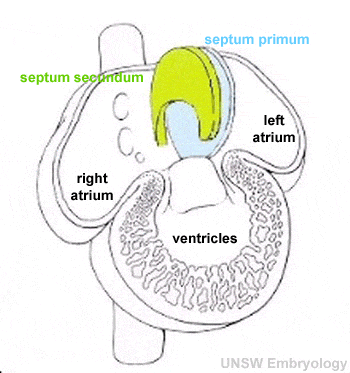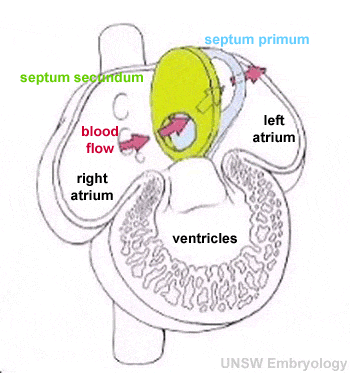
- atrial septation
- ventricular septation
- outflow tract division
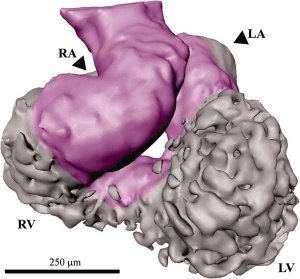
| Week 4 - Stage 10 Human Heart tube | Week 5 - Stage 13 Human Heart (sagittal view) |

|
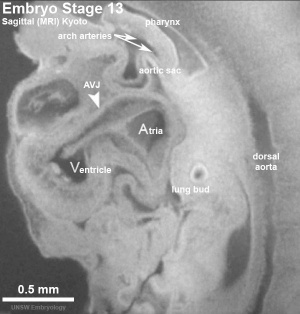
|
| from a pair of heart tubes | to the heart forming chambers |
| Begin Intermediate: | Primordial Heart Tube | Heart Tube Looping | Atrial Ventricular Septation | Outflow Tract | Heart Valves | Cardiac Abnormalities | Vascular Overview |
The cardiovascular system is the first major system to function within the embryo, with the heart beginning to function during the fourth week. Congenital heart defects affect 8-10 of every 1000 births in the United States. Hence in both embryological and clinical contexts it is important to consider heart development.
The intermediate section of this module on cardiac embryology is directed at university level students with some previous study of embryology but minimal previous study of cardiac development specifically. This unit takes approximately 30-40 minutes to complete. The intermediate cardiac module contains 7 pages which should be worked through in order.
- Primordial Heart Tube
- Looping of the Heart Tube
- Atrial and Ventricular Septation
- Development of the Outflow Tract
- Development of Heart Valves
- Abnormalities of Cardiac Development
- Overview of Vascular Development
| Early Heart Description |
|---|
|
Heart layers
- pericardium - covers the heart. Formed by 3 layers consisting of a fibrous pericardium and a double layered serous pericardium (parietal layer and visceral epicardium layer).
- myocardium - muscular wall of the heart. Thickest layer formed by spirally arranged cardiac muscle cells.
- endocardium - lines the heart. Epithelial tissue lining the inner surface of heart chambers and valves.
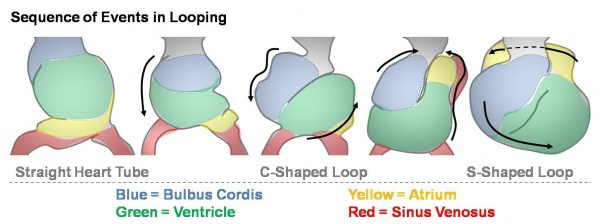
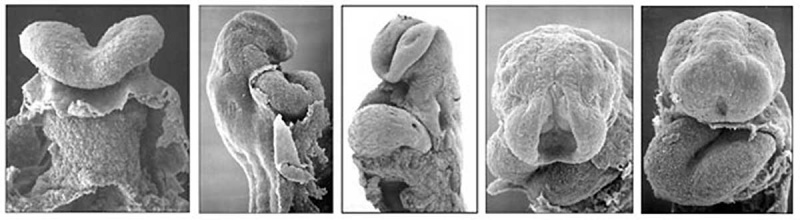
Heart looping
|
|
|
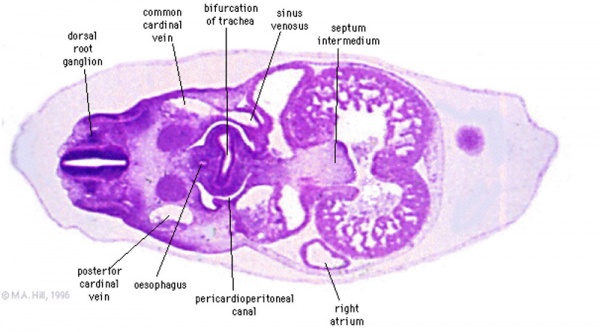
Transverse section- Heart is 2 tubes that fuse in the midline anterior to pharynx.
The pericardial cavity can be imagined as the top of the "horseshoe" of the intraembryonic coelom. (where the arms become the pleural cavity and the ends fuse anteriorly to form a single peritoneal cavity).
This view shows the initial positioning of the ventricles above the atria. The ventricles are rotated into their correct anatomical position by the growth of the heart tube, bending into an "S" shape.
Initially...
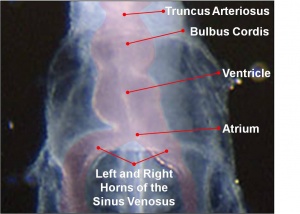
- Heat tube develops a series of constrictions:
- Truncus arteriosus – ventral aortae meet - OUTFLOW
- Bulbus cordis
- Ventricle
- Atrium
- Sinus venosus – caudal end of tube, receiving 4 veins - INFLOW
- Rapid growth – ‘buckling’ and ‘twisting’
- Heart tube bends ventrally into pericardial coelom
- Ventricle enlargens, absorbs lower part of bulbus cordis (bulboventricular loop)
- Ventricle also twists to left- atrium and sinus venosus come to lie dorsal to bulbus cordis and lower part of truncus arteriosus
- Venous inflow comes to lie directly dorsal to the arterial outflow.
- Possible abnormality – dextro-rotation, the heart bends or twists to the right. Maybe associated by other abnormalities.
- Later, sinus venosus becomes absorbed into atrium
Heart neural crest
- The mouse model shows that the heart also has contributions from neural crest E8.5 mouse neural crest
- between the levels of post-otic hindbrain to somite 4, with the most contribution from somite 2 level.
- 7 somite stage - Migration of cardiac neural crest from the neural tube begins. (level post-otic hindbrain and somite 4)
- Pathways dorsolateral, medial, and between somites.
- Then through peri-aortic mesenchyme (lateral to pharynx), through pharyngeal arches (3, 4, 6) into the aortic sac.
- 32 somite stage: Colonisation of the outflow tract mesenchyme.
Data from: Chan WY, Cheung CS, Yung KM, Copp AJ. Chan WY, Cheung CS, Yung KM, Copp AJ. Cardiac neural crest of the mouse embryo: axial level of origin, migratory pathway and cell autonomy of the splotch (Sp2H) mutant effect. Development. 2004 Jul;131(14):3367-79. PMID: 15226254
Embryonic heart rate
- Ultrasonographic measurement of embryonic heart rate (EHR) shows a steady increase from Stage 9-10 (75 beats/minute) to Stage 18 (130 beats/minute) and on to Stage 20, following which a gradual decrease in EHR occurs
- Maximal EHR is reached when morphological development of the embryonic heart is completed.
- Links: Embryonic Heart Rate
Internet Links
Embryo Images Unit: Embryo Images Online Early Cell Populations (cardiogenic section) | Cardiovascular Development | Week 3 Development | Week 4 Development | Heart Chambers and Outflow Tract | Atrioventricular Septation | Outflow Tract Septation | Ventricular Septation | Atrial Septation | Atrial Walls Aortic Arch Vessels | Changes at Birth
References
Online Textbooks
- Developmental Biology by Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000 The Heart | Figure 15.6. Cascade of heart development} | [http://www.ncbi.nlm.nih.gov/books/bv.fcgi?&rid=dbio.figgrp.3698 Figure 15.3. Formation of the chick heart from the splanchnic lateral plate mesoderm | Figure 15.4. Fusion of the right and left heart rudiments to form a single cardiac tube | Figure 15.5. Specification of the atrium and ventricles occurs even before heart looping
- Molecular Biology of the Cell 4th ed. Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York and London: Garland Science; c2002 - Figure 21-35. The vertebrate body plan as a dorsoventral inversion of the insect body plan Figure 22-40. The four classes of muscle cells of a mammal
Terms
| Cardiovascular Terms |
|---|
Cardiovascular System Development See also Heart terms, Immune terms and Blood terms.
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
| 2017 ANAT2341 - Timetable | Course Outline | Group Projects | Moodle | Tutorial 1 | Tutorial 2 | Tutorial 3 |
Labs: 1 Fertility and IVF | 2 ES Cells to Genome Editing | 3 Preimplantation and Early Implantation | 4 Reproductive Technology Revolution | 5 Cardiac and Vascular Development | 6 CRISPR-Cas9 | 7 Somitogenesis and Vertebral Malformation | 8 Organogenesis | 9 Genetic Disorders | 10 Melanocytes | 11 Stem Cells | 12 Group |
|
Lectures: 1 Introduction | 2 Fertilization | 3 Week 1/2 | 4 Week 3 | 5 Ectoderm | 6 Placenta | 7 Mesoderm | 8 Endoderm | 9 Research Technology | 10 Cardiovascular | 11 Respiratory | 12 Neural crest | 13 Head | 14 Musculoskeletal | 15 Limb | 16 Renal | 17 Genital | 18 Endocrine | 19 Sensory | 20 Fetal | 21 Integumentary | 22 Birth | 23 Stem cells | 24 Revision |
| Student Projects: 1 Cortex | 2 Kidney | 3 Heart | 4 Eye | 5 Lung | 6 Cerebellum |