Cardiovascular System - Coronary Circulation Development
| Embryology - 2 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The coronary circulation provides the blood supply to the heart required for the normal muscular function. From recent mouse studies, the origin of this specialised vasculature is from the sinus venosus.
Development of the heart and vascular system begins very early in mesoderm both within (embryonic) and outside (extra embryonic) the embryo. Vascular development therefore occurs in many places, the most obvious though is the inflow and outflow in the forming heart, which grows rapidly creating an externally obvious cardiac "bulge" on the early embryo.
The coronary circulation an important medical topic postnatally. Blockage and failure of this system leads initially to angina, continued ischemia leads to hypoxic death of cardiac muscle and myocardial infarction, a heart attack.
For general information about blood vessel development, see Blood Vessel Development and note that blood vessels also occurs outside the embryo in the extra-embryonic mesoderm of the yolk sac (vitelline) and in the villi of the placenta.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Coronary Circulation Embryology <pubmed limit=5>Coronary Circulation Embryology</pubmed> |
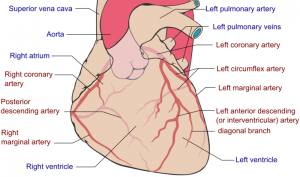
Coronary Vessel Anatomy
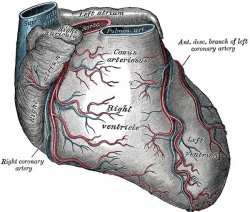
Coronary Arteries
Three aortic sinuses, or sinuses of Valsalva, are the left, right and posterior that give rise to the two coronary arteries, the posterior aortic sinus is the non-coronary sinus.
|
|
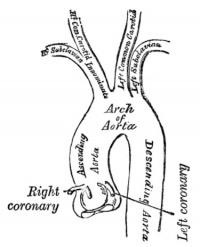
|
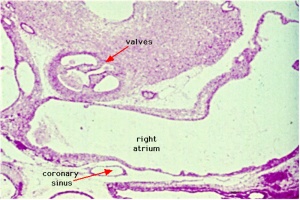
Coronary Veins
|
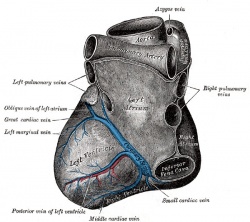
|
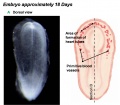
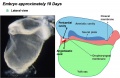
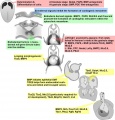
Human Coronary Vessel Development
An early research paper identified development of the coronary arteries in the embryonic human heart using embryos from the Carnegie Collection.[5]
- six sinuses of the embryonic great arteries
- Carnegie Embryological Collection coronary vasculature in 351 staged, serially sectioned human embryos (Carnegie stages 9 to 23).
stage 14 or 15 - A plexus of blind epicardial capillaries appears on the heart in Carnegie
stage 15, 16, or 17 - acquires a coronary sinus connection
stage 18 - connection of the proximal coronary arteries to the aorta.
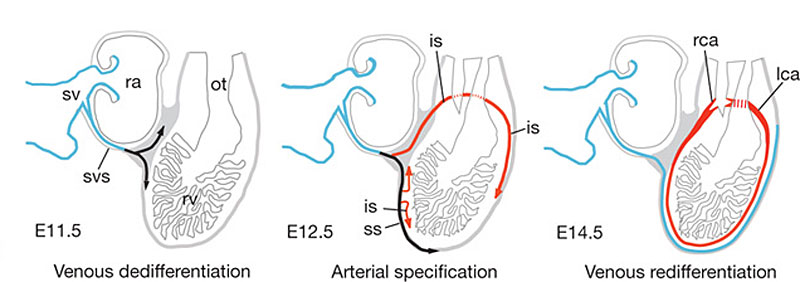
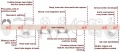
Mouse Coronary Vessel Development
Image showing changes in venous (blue) and arterial (red) marker expression during coronary development; black indicates dedifferentiated venous cells.[3]
Abnormalities
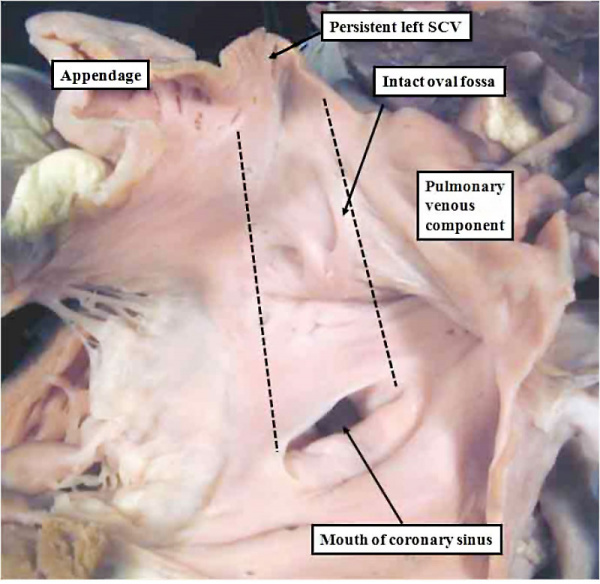
There are several abnormalities associated with the anatomical variation in vascular pattern of vessels including abnormal origins and absence.
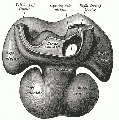
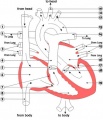
The image shows a human coronary sinus defect as viewed from the left atrial aspect.
There is a persistent left superior caval vein (SCV) opening to the leftward and cranial margin of the left atrium. The caval vein would normally have been incorporated into the left atrioventricular groove, but the walls which would normally extend to its opening to the right atrium (black dotted lines) have disappeared. Note the integrity of the oval fossa.
Anomalous Left Coronary Artery from the Pulmonary Artery
Anomalous origin of left coronary artery from pulmonary artery (ALCAPA) is a rare malformation (incidence of 0.25–0.50% )in children with abnormal cardiac development leading to a mortality rate of 90% in unoperated infants.[6] There are a variety of surgical repair techniques including: direct aortic implantation, a modified tubular extension technique, and an intrapulmonary baffling technique.[7]
Anomalous Right Coronary Artery from the Left Sinus
Anomalous Single Coronary Artery from the left sinus of Valsalva
This abnormality has been reported as occurring with an absence of the right coronary artery. [8]
Ectopic Origin of the Right Coronary Artery
The right coronary artery arises outside the borders of the right coronary sinus, the most common locations are the left sinus (either the anterior, superior portion) or directly from the left main coronary artery. Anterior displacement is the most prevalent anatomical variant.[9] Also very rarely arising from other anatomical locations including; the non-coronary (posterior) sinus, left anterior descending coronary artery, left circumflex coronary artery, pulmonic artery, and the descending aorta. Clinically seen as cardiac ischemia and arrhythmia.
Anomalous Sinus Node Artery
The sinus node artery normally originates from the proximal segment of the right coronary artery, left circumflex artery, or from both. With anomalous sinus node artery this vessel can originate from the left anterior descending artery.
Congenital Sinus of Valsalva Aneurysm
This aneurysm, a blood-filled dilatation, arises due to a localized weakness of the wall of the sinus bulging that may rupture into an adjacent cardiac chamber, forming an aortocardiac fistula.
References
- ↑ <pubmed>27959616</pubmed>
- ↑ <pubmed>26675596</pubmed>
- ↑ 3.0 3.1 <pubmed>20336138</pubmed>
- ↑ <pubmed>18093989</pubmed>
- ↑ <pubmed>3286038</pubmed>
- ↑ <pubmed>7949665</pubmed>
- ↑ <pubmed>19808708</pubmed>| Interact Cardiovasc Thorac Surg.
- ↑ <pubmed>19693319</pubmed>PMC2720291
- ↑ <pubmed>20197575</pubmed>| J Invasive Cardiol.
Reviews
<pubmed>27959616</pubmed> <pubmed>26675596</pubmed>
<pubmed></pubmed> <pubmed></pubmed>
Articles
<pubmed></pubmed> <pubmed></pubmed> <pubmed></pubmed>
Search Pubmed
Search Pubmed: Coronary Circulation Development
Additional Images
See also Category:Heart ILP and Category:Heart
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 2) Embryology Cardiovascular System - Coronary Circulation Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Cardiovascular_System_-_Coronary_Circulation_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G