Paper - The ductus venosus in the fetus and in the adult (1923)
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Stewart WB. The ductus venosus in the fetus and in the adult. (1923) Anat. Rec. 25(4): 225-235.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Ductus Venosus in the Fetus and in the Adult
Walter Blair Stewart
Department of Pathological Anatomy, The Mayo Clinic, Rochester, Minnesota
Two Figures (1923)
Introduction
Text-books state that the ductus venosus of the fetus, following its postnatal occlusion, persists through adult life as a fine fibrous cord termed the ligamentum venosum. “The umbilical vein and the ductus venosus become completely obliterated between the second and fifth days after birth, and ultimately dwindle to fibrous cords, the former becoming the round ligament of the liver, and the latter the fibrous cord, which in the adult may be traced along the fissure of the ductus venosus” (Gray’s Anatomy).
The ductus venosus is a fetal blood vessel which has been formed secondarily by a longitudinal anastomosis of capillary sinusoids of the liver during the early weeks of intra—uterine life, and which, in the mature fetus, as a direct continuation of the umbilical vein, conductsa large part of the placental blood into the left hepatic vein just before its union with the inferior vena cava, thus short-circuiting the liver. The purpose of this short cut during fetal life is to permit the richly oxygenated, placental blood to pass rapidly to the body of the fetus, through which it may be distributed after only slight previous admixture with the hepatic and somatic circulations. The size of the liver, relative to that of the entire body, is muchgreater during early, than during late fetal life. This is attributable to the fact that all the placental blood must pass through the liver sinusoids up to the time of formation of the ductus, and only a small part after its formation.
The literature on the subject of the ductus venosus is limited to the abbreviated statements found in current anatomical, embryological, and obstetrical text—books and articles scattered over the last seventy years of medical writing. In 1856, Meadows reported an instance of fatal hemorrhage from the ductus venosus occurring in a child twenty hours after birth, although the evidence on which he localized the lesion to the ductus is not clear. He says: “ But on examining the liver an enormous clot was found lying on its upper surface, between it and the peritoneum which had been stripped off by the effused blood; the same thing was also seen on the under surfacepof the right lobe; these two clots were much thicker behind than before, and seemed to have originated from the ductus venosus; but how the blood got into the cavity of the peritoneum could not be positively ascertained.”
No cause for the accident could be allocated. There was no history of intrapartum or postpartum injury to the child. The labor and the first twenty hours of extra-uterine life were normal.
Jackson, as early as 1859, mentions the error of most textbooks of anatomy, including the volume ‘recently produced by Gray,’ in which it is said that the ductus venosus enters into the vena cava. According to Jackson’s own observations on dissections, “it terminates in one of the hepatic veins near and sometimes close tothe opening of this last into the vena cava.” 8 He said that Mr. John Bell, in his anatomy (1802) was one of the few previous writers who had formulated this fact correctly.
Also in a small volume by Mr. John Struthers of Edinburgh (’54) it was noted that the ductus venosus “enters not the vena cava but the left hepatic vein, about a quarter of an inch before the latter ends in the vena cava.” Jackson says that the present instance is one of too many in which errors are perpetuated in the text-books of medical science.
The interesting anomaly of absence of the ductus venosus with a description of the consequences and a summary of four other reported cases is recorded by Paltauf in 1888. The condition is very rare, Kiister reporting one case and Otto three cases. Kiister’s case was complicated by the presence of abdominal fissure, and Otto’s showed unusual points of entry of the umbilical vein. Paltauf’s was a normal, full-term child, except that such a huge ascites was present that it formed an obstruction to the natural course of labor, andparacentesis had to be resorted to. Moreover, hydramnios had been present. The placenta was very large, the umbilical cord long, its veins tortuous and showing varicose dilatations; the subperitoneal veins were very large and somewhat tortuous. The umbilical vein led obliquely into the portal vein. No sign, even such as a fibrous strand, remained of the duetus. Thus a condition of portal obstruction in the fetus arose, analogous to the portal cirrhosis of the adult, with a resulting hydramnios, ascites, and attempts at collateral circulation. In this instance prominent anastomoses had been produced on either side between the spermatic vein, emptying into the vena cava, and the colic veins, emptying into the portal vein, and further anastomoses with veins of the abdominal wall. This is a type of collateral circulation unknown in acquired portal obstruction, since in the adult the portal and caval circulations are too widely separated, and the venous channels are no longer patulous, as during the process of development. In explaining this absence of the duetus Paltauf invokes the statement of His, that the duetus venosus is a secondary formation, a short cut of the capillary sinusoids of the liver among the islands of liver cells. In this case such a channel has not been opened up, with the result that all the placental blood must pass through the capillaries of the liver and hence must be under increased pressure.
As to the chief function of the duetus venosus, Paltauf believes that it serves to maintain a free passage for the blood in the umbilical and portal veins, equalizing rapidly the differences in pressure which probably prevail in the two channels, and avoiding any detrimental influence of the umbilical venous flow, which is under greater pressure, on the portal flow. The liver sinusoids of the late fetus, even as the embryonic liver sinusoids, should suffice for the passage of the umbilical blood, provided no secondary disturbances arise. Instances of hydrops and ascites with hydramnios in cases of fetal hepatitis, in spite of the presence of the duetus venosus, serve only to emphasize the prominent part of the sinusoidal circulation even in fetal life.
Barge asks why the duetus venosus closes after birth. It is natural enough that the umbilical vein should become obliterated after ligation of the cord, for blood vessels usually close distal to a ligature. The ductus has been regarded as a continuation of the umbilical vein—~——a point which would afford a simple explanation were it not for the fact that the portion of the umbilical vein, known in the adult as the recessus umbilicalis of the portal vein, persists and functions throughout life. In this case the circulation disobeys the law of hydrodynamics which would have it flow in the line of least resistance, namely, through the ductus; but after ligation of the umbilical vein it chooses to pass through the sinusoids of the liver where it meets much more resistance.
This raises two questions: Is it not possible that the ductus closes during fetal life? There is no evidence to support this view, apart from Gegenbauer’s observation that the lumen of the duct gets narrower during fetal life. Is the closing of the duct a useful occurrence? Surely it is, for it does not seem practical that a large volume of blood should dodge the liver, one of the main organs of general metabolism. But neither this practical side, nor the hereditary tendency defended by Roux, explains the mechanism of closure of the duct. Barge emphasizes the initiation of respiration after birth and the cessation of the fetal circulation, especially the former. During inspiration the sinusoids of the liver are compressed by the diaphragm, while during expiration the pressure lets up and a kind of suction action then draws the main part of the blood into the liver, resulting in a reduction of the volume of blood in the ductus which furthers the closing of the ductus. DeVries objects to this hypothesis on the ground that respiration is too rapid a process to permit the endothelial lining of the duct to stick together long enough to result in a ‘growing together’ of the duct walls. Bolk attributes the collapse of the ductus to the negative venous pressure arising after birth. Boeke has found the ductus collapsed without obliteration in several instances among older children.
Richter says that the ductus venosus passes through the fissure between the Spigelian lobe and the left lobe of the liver, from the juncture of the umbilical vein with the left branch of the portal vein, to the left branch of the hepatic vein immediately before it empties into the inferior vena cava. During the first week of life the ductus can be injected, and shows a lumen of 1 to 2 mm. in diameter. Closure begins at the portal end of the ductus at the end of the first, or during the second week. By the fourth to the sixth week it is completely obliterated except for a small pit at the hepatic end. Although the lumen is obliterated, one often finds endothelial-lined spaces filled with blood in the connective tissue filling the original lumen, and shoots of vessels grown through the original vessel wall into the connective tissue of the lumen and supplying it. In the process of closure the incompletely obliterated lumen is in part distended with thrombi which are in process of being replaced by a proliferating network of fibers. In some instances openings of the lumen of the ductus may occur, and even communications with portal branches, which may form a basis for later communication between the portal vein and the vena cava in cases of cirrhosis of the liver.
Embryology
The first paired vessels of human embryos (according to Prentiss) are formed as longitudinal anastomoses of capillary networks which originate first in the angioblast of the yolk-sac and chorion. In embryos of 1.3 mm. umbilical veins from the chorion, fused in the body-stalk and separating to pass forward in the somatopleure on either side to the paired tubular heart, are already formed. At 2.5 mm. a longitudinal fusion of the paired heart has occurred, and numerous tributary veins from the yolk sac, united into a single vessel on either side, join the umbilical Veins to empty into the sinus Venosus, one of the three primitive dilatations of the heart tube, at its base. At 4.2 mm. the umbilical vein on either side has been joined by the common cardinal vein, the resultant trunk entering the sinus laterally. At this stage the liver diverticulum has grown out ventrally from the foregut cranial to the wall of the yolk-sac, and is beginning to grow into the walls of the vitelline veins, producing minute isolated islands of liver cells surrounded by the vitelline sinusoids. Between the liver diverticulum and the yolk sac three transverse anastomoses between the vitelline veins, one dorsal and two ventral to the primitive gut, arise. Shortly after this an oblique venous passage is opened up among the islands of liver cells, passing from the juncture of the left vitelline and umbilical vein (for the latter has now approached and tapped the former on each side) forward to the point of entry of the right vitelline vein into the sinus venosus. This secondary formation and short cut for the main bulk of the placental blood, which now enters largely through the left umbilical vein (the proximal end of this vessel and its union with the sinus venosus disappearing), becomes the ductus venosus. The vitelline veins distal ‘to the liver, through the atrophy of the parts of the two venous rings encircling the gut and the growth of others, come to form the portal vein of the adult.
The Postnatal Ductus Venosus
The present study is based on a survey of material removed at fifty consecutive postmortem examinations at the Mayo Clinic. In one case-, that of a still-born child at full term (case A400979) the ductus venosus was completely patent, although the first signs of closure were evident. There was a sharp line of demarcation between the intima of the umbilical vein and that of the ductus, the two being of notably different shades of color. The circumference of the ductus at the end adjoining the portal vein was much less than that at the end adjoining the left hepatic vein, '7 mm. as compared with 12 mm. The circumference of the left branch of the portal vein at its point of union with the urnbilical vein was only 3.5 mm.-~a fact which would lead one to suspect that twice as much of the umbilical blood flowed through the ductus as did through the circuitous route of the liver capillaries. The umbilical vein, too, was completely patent and showed no narrowing, although the intima distal to the point of entry of the veins from the quadrate lobe and the left lobe of the liver (the future recessus umbilicalis of the portal vein) showed distinct roughening and absence of the normal intimal sheen.
In one child of eleven hours (case A398634) there were no signs of closure, or of narrowing of the ductus, except that the sa.me sharp line of demarcation existed between the intima of the umbilical vein and the ductus as in the still—born child.
In another child, who had lived two days (case A401005), there was no opening or even a trace of a scar indicating an opening at the umbilical end (now the portal end) of the ductus, although the hepatic end was widely open, as in the fetus, and the duct itself was freely patent almost to its point of union with the portal vein. Several small veins which drained the surrounding liver tissue emptied into the hepatic half of the ductus, the embryologic channel thus serving also as a hepatic vein. The umbilical vein was completely closed except for a minute pinhole opening into the recessus umbilicalis and a channel of similar size present in the round ligament.
In a case of congenital heart disease with pulmonary atresia in a child of four and one-half months (case A395079), the ductus was completely obliterated with neither opening nor scar at either end.
In a girl of thirteen years (case A400834) a ductus patent in its hepatic half, with an opening 2 mm. in diameter into the hepatic vein, was found. There was no sign of the portal end of the ductus. I injected India ink into the patent end of the ductus and the ink passed out into and distended certain small hepatic veins draining the left lobe of the liver, small vessels running in the hepatogastric ligament, and the vasa vasorum of the ductus itself. In the open part of the ductus five small veins were seen to enter its lumen, through which the injectionmass had passed. The portal half of the ductus did not take the India ink.
In an analysis of the cases seen in adult life, the most striking fact is that there persistsa definite, although often semiobliteratecl duct, lying close to the liver in the fossa for the ductus venosus.
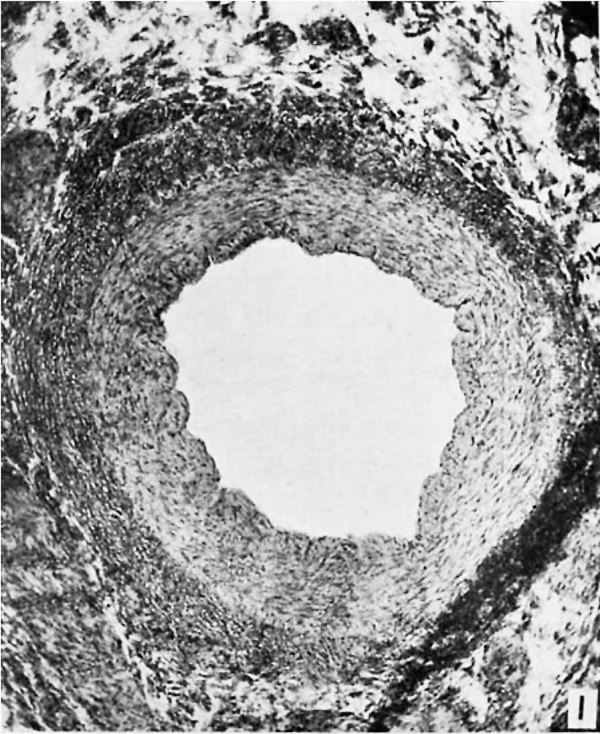
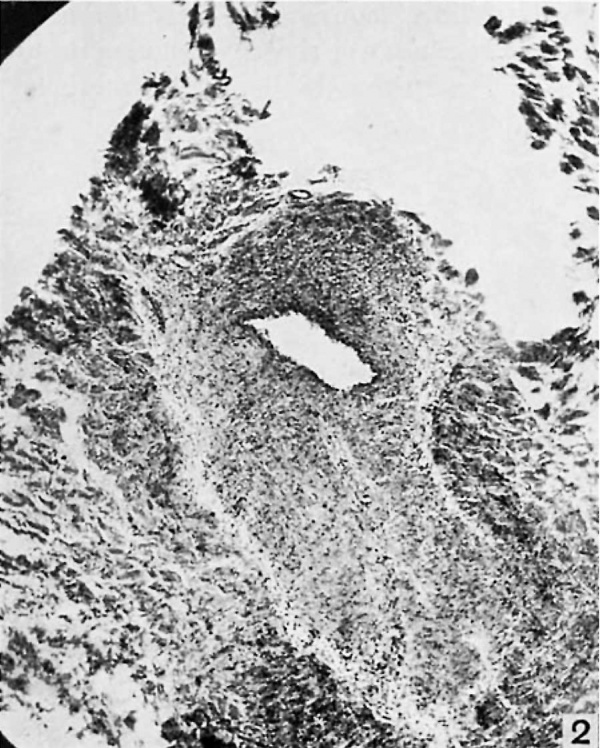
It appears as a scant bundle of fibrous strands, semi-translucent, yet denser than the thin fibrous sheet, the hepatogastric ligament, near the bottom of whose meshes it extends from portal to hepatic vein. It is extremely difiicult to locate. But in the center of this apparently insignificant, flat, fibrous band there may be traced in each case a definite duct, often intima—lined and glistening, especially in its hepatic half, even though there is no connection with the veins at either end. In a few instances such a complete patency is shown through the whole length, but more often it is only near the hepatic end. The remainder of the ductus may present a lumen after a passage has been gently forced by a probe. But in every case a pathway which once represented the lumen of the ductus venosus may be dissected out grossly. Microscopic examination confirms this, for the entirely patent sections present a vein of normal appearance, a thick wall with all its coats, and a large circular lumen (fig. 1). The sections from the semiobliterated zones show a minute, circular, or a flat, elongated lumen, endotheliuin-lined in some places, and with rough, fibrous elevations or irregular projections from the in.tima at others (fig. 2). In no case was an open passage persistent from the portal vein to the hepatic vein. It is interesting to speculate as to what clinical syndrome, if any, a ductus venosus, completely patent throughout adult life, would give. Certainly it would lead to some metabolic disorder, possibly to a condition similar to that resulting from an Eck’s fistula. It is worthy of note that a patent ductus is not found as one of the channels of collateral circulation in cases of secondary portal obstruction, such as cirrhosis of the liver.
The duct ranged between 35 and 65 mm. in length in the adult cases, the average being 45 mm. The livers from these weighed between 825 and 2600 grams, with an average weight of 1680 grams. There seemed to be no correlation between the length of the ductus and the weight of the liver. In the portal vein at the end of the ductus there was almost invariably a smooth intimal lining showing no opening into the ductus and no pit or scar to mark the site of the former opening, except that by traction on the ductus the intima could be depressed, revealing its former location. One case presented a minute depression. Another showed a minute, pinhole opening leading into the ductus, which was widely patent down to within 1 cm. of its hepatic end, where it had closed without leaving a scar.
Fig. 1 (A401361) Patient aged thirty-three- Section through hepatic end of ductus venosus. Both openings closed without scars. X 50.
Fig. 2. (A397718.) Patient aged seventy-one. Section through middle of ductus venosus. "Both. openings closed without scars. X 50.
In fifteen cases the hepatic vein presented an opening into the ductus, varying from the size of a pin-point to 2 mm. in diameter. The lumen of the ductus was either definitelypatent, or could be forced open by the probe in most of these cases. In a few instances an oval thinning of the intima of the hepatic vein was evident; in a few there were fatty changes in this intima ; in one, a narrow, fibrous, intima-covered bridge, 5 mm. long, across the closed oval, and in one, two circular depressions in the intima, side by side, partly covered over by a thin sheet of intima. The location of this hepatic end of the ductus is quite constant, usually just above, occasionally beside the juncture of the upper and lower branches of the left hepatic vein, this common left hepatic vein being a very short vessel which empties almost at once into the inferior vena cava. The portal end of the ductus lies in the upper part of the left main branch of the portal vein, directly opposite to, or a little to the left of the point of juncture of the round ligament with the processus globularis of the portal vein.
In eight instances a definite opening, in one 7 mm. in circumference, led from the portal vein into a patent channel in the round ligament, usually soon ending in a fibrous obliteration, but in one case of Laennec’s cirrhosis, extending as a wide, somewhat tortuous channel through the whole length of the round ligament.
Summary
- The ductus venosus extends from the left branch of the portal vein, opposite the point of entry of the umbilical vein, in the left posterior f ossa of the liver in the folds of the hepatogastric ligament, to the point of union of the upper and lower branches of the left hepatic vein, just before the common trunk empties into the inferior vena cava. T
- The portal end of the ductus venosus closes within the first two days after birth, the hepatic end may present a functioning stoma throughout life. The hepatic half of the ductus venosus may remain open, receive tributaries from the parenchyma of the liver and the hepatogastric ligament, and thus function as a hepatic vein in the adult.
- In every adult the remains of the fetal ductus venosus persist as a fine, flat, fibrous cord with a lumen only semiobliterated even though both ends are completely closed.
- No cases of complete patency of the entire ductus in adult life were observed personally or seen recorded in the literature.
- The ductus venosus is a short cut around the liver for the oxygenated fetal blood in its passage from the placenta to the heart.
Bibliography
BARGE, J. A. J. 1919 Waarom sluit zich de ductus venosus Arantii. Nederl. Tijdschr. v. Geneesk., vol. 1, pp. 718-722.
Jackson JBS. The ductus venosus. (1859) Extra. Rec. Bost. Soc. Med. Impr. 3: 296.
MEADOWS, A.. 1856-1857 Case of fatal hemorrhage from the ductus venosus occurring in a child twenty hours after birth. Tr. Med. Soc. King’s Coll., London, vol. 1, pp. 224-226.
PALTAUF, R. 1888 Ein Fall von Mangel des Duct-us venos. Arantii. Wien. klin. W'chnschr., Bd. 1, S. 165-167.
RICHTER, E. 1911 Uber den Verschluss des Ductus venosus Arantii nebst Bemerkungen fiber die Anatomic der Pfortader. Virchows Arch. f. path. Anat._, Bd. 205, s. 257-263.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - The ductus venosus in the fetus and in the adult (1923). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_ductus_venosus_in_the_fetus_and_in_the_adult_(1923)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G