Hearing - Inner Ear Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (25 intermediate revisions by the same user not shown) | |||
| Line 4: | Line 4: | ||
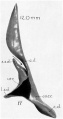
[[File:Stage11_sem20a.jpg|thumb|alt=Otic placodes electron micrograph|Otic placodes ([[Carnegie_stage_11|Stage 11]] dorsal view)]] | [[File:Stage11_sem20a.jpg|thumb|alt=Otic placodes electron micrograph|Otic placodes ([[Carnegie_stage_11|Stage 11]] dorsal view)]] | ||
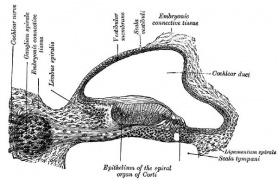
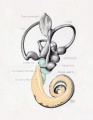
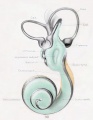
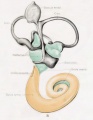
[[File:Streeter028-30.jpg|thumb|alt=Historic images - The membranous labyrinth|Historic images - The membranous labyrinth]] | [[File:Streeter028-30.jpg|thumb|alt=Historic images - The membranous labyrinth|Historic images - The membranous labyrinth]] | ||
The inner ear is derived from a pair of surface sensory [[Placodes|placodes]] ('''otic placodes''') that appear in human development during week 4 ({{GA}} week 6) in the head region lying behind the second pharyngeal arch. | The {{inner ear}} is derived from a pair of surface sensory [[Placodes|placodes]] ('''otic placodes''') that appear in human development during week 4 ({{GA}} week 6) in the head region lying behind the second pharyngeal arch. | ||
These otic placodes fold inwards forming initially a depression, then pinch off entirely from the surface forming an epithelium surrounding a fluid-filled sac or vesicle ('''otic vesicle''', '''otocyst''', auditory vesicle). The vesicle sinks into the head mesenchyme some of which closely surrounds the otocyst forming the otic capsule. | These otic placodes fold inwards forming initially a depression, then pinch off entirely from the surface forming an epithelium surrounding a fluid-filled sac or vesicle ('''otic vesicle''', '''otocyst''', auditory vesicle). The vesicle sinks into the head mesenchyme some of which closely surrounds the otocyst forming the otic capsule. | ||
The otocyst finally lies close to the early developing hindbrain ( | The otocyst finally lies close to the early developing hindbrain ({{rhombencephalon}}) and the developing vestibulo-cochlear-facial ganglion complex. | ||
The otocyst epithelium then undergoes a series of morphological changes, forming the primitive membranous labyrinth. During the human fetal period this will differentiate into the inner ear components for hearing (cochlea) and balance (semi-circular canals). Cochlear development involving a transcription factor spatiotemporal gradient continues through to {{GA}} week 16.{{#pmid:31788757|PMID31788757}} | |||
The adult cochlear has a "snail-shell" appearance, with the total number of turns differing between species. The adult human cochlear is typically described as having 2.5 turns, but this can vary up to 2.75 or even 3 turns.{{#pmid:19225438|PMID19225438}} | The adult cochlear has a "snail-shell" appearance, with the total number of turns differing between species. The adult human cochlear is typically described as having 2.5 turns, but this can vary up to 2.75 or even 3 turns.{{#pmid:19225438|PMID19225438}} | ||
The "organ of corti" that develops within the cochlea was first identified by Alfonso Giacomo Gaspare Corti (1822–1876), an Italian anatomist, in 1851. | The "organ of corti" that develops within the cochlea was first identified by Alfonso Giacomo Gaspare Corti (1822–1876), an Italian anatomist, in 1851. | ||
{{Hearing Links}} | {{Hearing Links}} | ||
== Some Recent Findings == | == Some Recent Findings == | ||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | * '''Characterization of the development of the mouse cochlear epithelium at the single cell level'''{{#pmid:32404924|PMID32404924}} "Mammalian hearing requires the development of the organ of Corti, a sensory epithelium comprising unique cell types. The limited number of each of these cell types, combined with their close proximity, has prevented characterization of individual cell types and/or their developmental progression. To examine cochlear development more closely, we transcriptionally profile approximately 30,000 isolated mouse cochlear cells collected at four developmental time points. Here we report on the analysis of those cells including the identification of both known and unknown cell types. Trajectory analysis for OHCs indicates four phases of gene expression while fate mapping of progenitor cells suggests that OHCs and their surrounding supporting cells arise from a distinct (lateral) progenitor pool. Tgfβr1 is identified as being expressed in lateral progenitor cells and a Tgfβr1 antagonist inhibits OHC development." | ||
* ''' | * '''β-Catenin is required for radial cell patterning and identity in the developing mouse cochlea'''{{#pmid:31570588|PMID31570588}} "Development of multicellular organs requires the coordination of cell differentiation and patterning. Critical for sound detection, the mammalian organ of Corti contains functional units arranged tonotopically along the cochlear turns. Each unit consists of sensory hair cells intercalated by nonsensory supporting cells, both specified and radially patterned with exquisite precision during embryonic development. However, how cell identity and radial patterning are jointly controlled is poorly understood. Here we show that β-catenin is required for specification of hair cell and supporting cell subtypes and radial patterning of the cochlea in vivo. In 2 mouse models of conditional β-catenin deletion, early specification of Myosin7-expressing hair cells and Prox1-positive supporting cells was preserved. While β-catenin-deficient cochleae expressed {{FGF}}8 and {{FGF}}R3, both of which are essential for pillar cell specification, the radial patterning of organ of Corti was disrupted, revealed by aberrant expression of cadherins and the pillar cell markers P75 and Lgr6. Moreover, β-catenin ablation caused duplication of FGF8-positive inner hair cells and reduction of outer hair cells without affecting the overall hair cell density. In contrast, in another transgenic model with suppressed transcriptional activity of β-catenin but preserved cell adhesion function, both specification and radial patterning of the organ of Corti were intact. Our study reveals specific functions of β-catenin in governing cell identity and patterning mediated through cell adhesion in the developing cochlea. | ||
* ''' | * '''Formation of the Periotic Space During the Early Fetal Period in Humans'''{{#pmid:29293291|PMID29293291}} "Digital data sets from magnetic resonance images and phase-contrast X-ray tomography images of 24 inner ear organs from 24 human fetuses from the [[Kyoto Collection]] (fetuses in trimesters 1 and 2; crown-rump length: 14.4-197 mm) were analyzed. The membranous labyrinth was morphologically differentiated in samples at the end of the embryonic period (Carnegie stage {{CS23}}), and had grown linearly to more than eight times in size during the observation period. The periotic space was first detected at the 35-mm samples, around the vestibule and basal turn of the cochlea, which elongated rapidly to the tip of the cochlea and semicircular ducts, successively, and almost covered the membranous labyrinth at the 115-mm CRL stage or later. In those samples, several ossification centers were detected around the space." | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 37: | Line 38: | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Inner+Ear+Development ''Inner Ear Development'] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Inner+Ear+Development ''Inner Ear Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Inner+Ear+Embryology ''Inner Ear Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Organ+of+Corti+Development ''Organ of Corti Development''] | | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! Older papers | ! Older papers | ||
|- | |- | ||
| | | {{Older papers}} | ||
* '''Developmental profiling of microRNAs in the human embryonic inner ear'''{{#pmid:29373586|PMID29373586}} "Due to the extreme inaccessibility of fetal human inner ear tissue, defining of the microRNAs (miRNAs) that regulate development of the inner ear has relied on animal tissue. In the present study, we performed the first miRNA sequencing of otic precursors in human specimens. Using HTG miRNA Whole Transcriptome assays, we examined miRNA expression in the cochleovestibular ganglion (CVG), neural crest (NC), and otic vesicle (OV) from paraffin embedded (FFPE) human specimens in the Carnegie developmental stages {{CS13}}, {{CS14}} and {{CS15}}. We found that in human embryonic tissues, there are different patterns of miRNA expression in the CVG, NC and OV. In particular, members of the miR-183 family (miR-96, miR-182, and miR-183) are differentially expressed in the CVG compared to NC and OV at Carnegie developmental stage {{CS13}}. We further identified transcription factors that are differentially targeted in the CVG compared to the other tissues from stages 13-15, and we performed gene set enrichment analyses to determine differentially regulated pathways that are relevant to CVG development in humans. These findings not only provide insight into the mechanisms governing the development of the human inner ear, but also identify potential signaling pathways for promoting regeneration of the spiral ganglion and other components of the inner ear." {{MicroRNA}} | |||
* '''Role of BDNF and neurotrophic receptors in human inner ear development'''{{#pmid:28924861|PMID28924861}} "The expression patterns of the neurotrophin, brain-derived neurotrophic factor, BDNF, and the neurotrophic receptors-p75NTR and Trk receptors-in the developing human fetal inner ear between the gestational weeks ({{GA}}) 9 to 12 are examined via in situ hybridization and immunohistochemistry. BDNF mRNA expression was highest in the cochlea at {{GA}} 9 but declined in the course of development. In contrast to embryonic murine specimens, a decline in BDNF expression from the apical to the basal turn of the cochlea could not be observed. ...Our findings suggest that BDNF and neurotrophin receptors are important players during early human inner ear development. In particular, they seem to be important for the survival of the afferent sensory neurons." | |||
* '''Lineage tracing of Sox2-expressing progenitor cells in the mouse inner ear reveals a broad contribution to non-sensory tissues and insights into the origin of the organ of Corti'''{{#pmid:27090805|PMID27090805}} "The transcription factor [[Developmental Signals - Sox|Sox2]] is both necessary and sufficient for the generation of sensory regions of the inner ear. ...We find that Sox2-expressing cells in the early otocyst give rise to large numbers of non-sensory structures throughout the inner ear, and that Sox2 only becomes a truly prosensory marker at embryonic day (E)11.5. Our fate map reveals the organ of Corti derives from a central domain on the medial side of the otocyst and shows that a significant amount of the organ of Corti derives from a Sox2-negative population in this region." {{Sox}} | {{Mouse}} | |||
* '''Development of the stria vascularis and potassium regulation in the human fetal cochlea'''{{#pmid:25663387|PMID25663387}} "We present an investigation on the development of the stria vascularis in the human fetal cochlea between 9 and 18 weeks of gestation (W9-W18) and show the cochlear expression dynamics of key potassium-regulating proteins. At W12, MITF+/SOX10+/KIT+ neural-crest-derived melanocytes migrated into the cochlea and penetrated the basement membrane of the lateral wall epithelium, developing into the intermediate cells of the stria vascularis. These melanocytes tightly integrated with Na+ /K+ -ATPase-positive marginal cells, which started to express KCNQ1 in their apical membrane at W16. At W18, KCNJ10 and gap junction proteins GJB2/CX26 and GJB6/CX30 were expressed in the cells in the outer sulcus, but not in the spiral ligament." | * '''Development of the stria vascularis and potassium regulation in the human fetal cochlea'''{{#pmid:25663387|PMID25663387}} "We present an investigation on the development of the stria vascularis in the human fetal cochlea between 9 and 18 weeks of gestation (W9-W18) and show the cochlear expression dynamics of key potassium-regulating proteins. At W12, MITF+/SOX10+/KIT+ neural-crest-derived melanocytes migrated into the cochlea and penetrated the basement membrane of the lateral wall epithelium, developing into the intermediate cells of the stria vascularis. These melanocytes tightly integrated with Na+ /K+ -ATPase-positive marginal cells, which started to express KCNQ1 in their apical membrane at W16. At W18, KCNJ10 and gap junction proteins GJB2/CX26 and GJB6/CX30 were expressed in the cells in the outer sulcus, but not in the spiral ligament." | ||
| Line 50: | Line 55: | ||
* '''Auditory ganglion source of Sonic hedgehog regulates timing of cell cycle exit and differentiation of mammalian cochlear hair cells'''{{#pmid:23918393|PMID23918393}} "Neural precursor cells of the central nervous system undergo successive temporal waves of terminal division, each of which is soon followed by the onset of cell differentiation. The organ of Corti in the mammalian cochlea develops differently, such that precursors at the apex are the first to exit from the cell cycle but the last to begin differentiating as mechanosensory hair cells. ...The dynamic relationship between the restriction of Shh expression in the developing spiral ganglion and its proximity to regions of the growing cochlear duct dictates the timing of terminal mitosis of hair cell precursors and their subsequent differentiation." [[Developmental_Signals_-_Sonic_hedgehog|Sonic hedgehog]] | * '''Auditory ganglion source of Sonic hedgehog regulates timing of cell cycle exit and differentiation of mammalian cochlear hair cells'''{{#pmid:23918393|PMID23918393}} "Neural precursor cells of the central nervous system undergo successive temporal waves of terminal division, each of which is soon followed by the onset of cell differentiation. The organ of Corti in the mammalian cochlea develops differently, such that precursors at the apex are the first to exit from the cell cycle but the last to begin differentiating as mechanosensory hair cells. ...The dynamic relationship between the restriction of Shh expression in the developing spiral ganglion and its proximity to regions of the growing cochlear duct dictates the timing of terminal mitosis of hair cell precursors and their subsequent differentiation." [[Developmental_Signals_-_Sonic_hedgehog|Sonic hedgehog]] | ||
* '''Tbx1 and Brn4 regulate retinoic acid metabolic genes during cochlear morphogenesis.''' | |||
* '''Tbx1 and Brn4 regulate retinoic acid metabolic genes during cochlear morphogenesis.'''{{#pmid:19476657|PMID19476657}} "These results indicate that Tbx1 expression in the POM regulates cochlear outgrowth potentially via control of local retinoic acid activity." | |||
|} | |} | ||
==Otic Placode== | ==Otic Placode== | ||
| Line 78: | Line 84: | ||
:'''Links:''' [[Placodes]] | [[Week 4]] | :'''Links:''' [[Placodes]] | [[Week 4]] | ||
== | ==Cochlea Timeline== | ||
The otocyst will differentiate to form all components of the membranous labyrinth. The organ of Corti forms initially from the central domain on the medial side of the otocyst.{{#pmid:27090805|PMID27090805}} Sox2 regulates organ of Corti prosensory progenitor development, through Jag1 ([[Developmental_Signals_-_Notch|Notch ligand]]) signaling. | The otocyst will differentiate to form all components of the membranous labyrinth. The organ of Corti forms initially from the central domain on the medial side of the otocyst.{{#pmid:27090805|PMID27090805}} Sox2 regulates organ of Corti prosensory progenitor development, through Jag1 ([[Developmental_Signals_-_Notch|Notch ligand]]) signaling. | ||
| Line 98: | Line 104: | ||
File:Gray0903.jpg|fetal cat cochlear duct | File:Gray0903.jpg|fetal cat cochlear duct | ||
</gallery> | </gallery> | ||
==Week 5== | ===Week 5=== | ||
Stage 13 embryo (week 5) showing otocyst that will form the inner ear. | Stage 13 embryo (week 5) showing otocyst that will form the inner ear. | ||
| Line 109: | Line 115: | ||
|} | |} | ||
==Week 7== | ===Week 7=== | ||
Based upon Streeter.<ref name=Streeter1957>{{Ref-Streeter1957}}</ref> | |||
* Carnegie stage {{CS18}} - L-shaped cochlear duct. | |||
* Carnegie stage {{CS19}} - Tip of cochlea turns up; short tip. | |||
[[File:Stage_19_ear.jpg|800px]] | [[File:Stage_19_ear.jpg|800px]] | ||
==Week 8== | ===Week 8=== | ||
Based upon Streeter.<ref name=Streeter1957>{{Ref-Streeter1957}}</ref> | |||
* Carnegie stage {{CS20}} - Long tip; transitional stage. | |||
* Carnegie stage {{CS21}} - Return down curve (tip turns down). | |||
* Carnegie stage {{CS22}} - Transitional stage. | |||
* Carnegie stage {{CS23}} - Tip turns up second time. Tip turns down second time (second return down curve). | |||
Stage 22 embryo (week 8) showing the embryo near the end of the embryonic period. | Stage 22 embryo (week 8) showing the embryo near the end of the embryonic period. | ||
| Line 129: | Line 149: | ||
[[File:Gray0924.jpg|600px]] | [[File:Gray0924.jpg|600px]] | ||
Periotic space first appears in [[week 8]] embryos 35 mm {{CRL}} around the vestibule and basal turn of the cochlea.{{#pmid:29293291|PMID29293291}} | |||
==Fetal== | ==Fetal== | ||
| Line 143: | Line 165: | ||
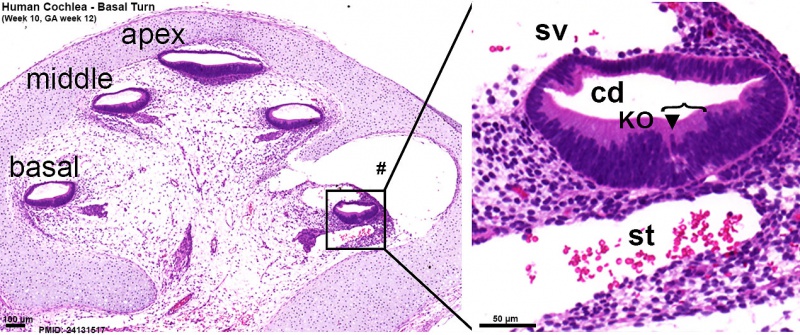
Human fetal cochlea basal turn week 10 Gestational Week {{GA}} 12 {{HE}}{{#pmid:24131517|PMID24131517}} | Human fetal cochlea basal turn week 10 Gestational Week {{GA}} 12 {{HE}}{{#pmid:24131517|PMID24131517}} | ||
===Week 14=== | |||
Periotic space almost covers the membranous labyrinth in the fetus 115 mm {{CRL}} week 14 ({{GA}} week 16).{{#pmid:29293291|PMID29293291}} | |||
===Week 18 - 22=== | ===Week 18 - 22=== | ||
| Line 218: | Line 244: | ||
===Fetal Cochlea Molecular=== | ===Fetal Cochlea Molecular=== | ||
Human fetal cochlea basal turn by Gestational Week {{GA}} | Human fetal cochlea basal turn by Gestational Week {{GA}}{{#pmid:24131517|PMID24131517}} | ||
{| | {| | ||
| [[File:Human cochlea fetal development cartoon.jpg|alt=Human cochlea fetal development cartoon|400px]] | | [[File:Human cochlea fetal development cartoon.jpg|alt=Human cochlea fetal development cartoon|400px]] | ||
| Line 282: | Line 308: | ||
==Organ of Corti== | ==Organ of Corti== | ||
Within the cochlea, the specialised structure required for converting mechanical vibration into an electrical signal occurs at the organ of | Within the cochlea, the specialised structure required for converting mechanical vibration into an electrical signal occurs at the {{organ of Corti}} (spiral organ of Corti). | ||
The images (mouse) below show the detail of the specialised structure, the organ of Corti, that develops through the fetal period. | The images (mouse) below show the detail of the specialised structure, the organ of Corti, that develops through the fetal period. | ||
| Line 311: | Line 337: | ||
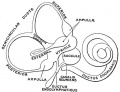
==Cochlear Sac== | ==Cochlear Sac== | ||
[[File:Cochlea stria vascularis cartoon 02.jpg|thumb|Cochlear Sac | [[File:Cochlea stria vascularis cartoon 02.jpg|thumb|Cochlear Sac{{#pmid:25663387|PMID25663387}}]] | ||
* generates coiled cochlear duct (humans 2 1/2 turns) | * generates coiled cochlear duct (humans 2 1/2 turns) | ||
* remainder forms saccule | * remainder forms saccule | ||
| Line 348: | Line 374: | ||
|- | |- | ||
| In the adult the stria vascularis functions to synthesise and secretes endolymph. The three main cell types within the stria that have different embryological origins and are connected by different forms of cell junctions. | | In the adult the stria vascularis functions to synthesise and secretes endolymph. The three main cell types within the stria that have different embryological origins and are connected by different forms of cell junctions. | ||
| Anatomical (upper half) and compartmental (lower half) model of the adult stria vascularis showing the three cellular layers and depicting the location of potassium regulating channels. The stria vascularis is electrochemically isolated from neighboring structures by tight junctions (black bars). | | Anatomical (upper half) and compartmental (lower half) model of the adult stria vascularis showing the three cellular layers and depicting the location of potassium regulating channels. The stria vascularis is electrochemically isolated from neighboring structures by tight junctions (black bars).{{#pmid:25663387|PMID25663387}} | ||
|} | |} | ||
<gallery> | <gallery> | ||
| Line 370: | Line 396: | ||
| derived from epithelia. | | derived from epithelia. | ||
| derived from the neural crest. | | derived from the neural crest. | ||
| derived from otic mesenchyme. | | derived from otic mesenchyme.{{#pmid:21925491|PMID21925491}} | ||
|} | |} | ||
| Line 376: | Line 402: | ||
:''Links:'' [[Neural_Crest_-_Melanocyte_Development#Inner_Ear_Melanocytes|Neural Crest - Inner Ear]] | [[Neural_Crest_-_Melanocyte_Development|Neural Crest - Melanocytes]] | :''Links:'' [[Neural_Crest_-_Melanocyte_Development#Inner_Ear_Melanocytes|Neural Crest - Inner Ear]] | [[Neural_Crest_-_Melanocyte_Development|Neural Crest - Melanocytes]] | ||
===Mouse SEM=== | ===Mouse SEM=== | ||
The gallery below shows scanning electron micrographs of the developing mouse (E18.5) cochlea. | The gallery below shows scanning electron micrographs of the developing mouse (E18.5) cochlea.{{#pmid:16410827|PMID16410827}} | ||
<gallery> | <gallery> | ||
File:Mouse_E18.5_cochlea_sem01.jpg|Cochlea overview E18.5 | File:Mouse_E18.5_cochlea_sem01.jpg|Cochlea overview E18.5 | ||
| Line 462: | Line 489: | ||
* nerogenin 1 affects both ganglia | * nerogenin 1 affects both ganglia | ||
* LIN28B - RNA-binding protein times auditory prosensory cell cycle withdrawal and differentiation through both let-7–dependent and independent mechanisms.{{#pmid:26139524|PMID26139524}} | * LIN28B - RNA-binding protein times auditory prosensory cell cycle withdrawal and differentiation through both let-7–dependent and independent mechanisms.{{#pmid:26139524|PMID26139524}} | ||
* {{FGF}} - {{FGF}}8 and {{FGFR}}3 are essential for pillar cell specification.{{#pmid:31570588|PMID31570588}} | |||
From a recent study of human fetal tissue.{{#pmid:31788757|PMID31788757}} | |||
* Leucine-rich repeat-containing G protein-coupled receptor 5 (LGR5) - apical poles of the sensory epithelium of the cochlear duct and the vestibular end organs {{GA}} week 11. Limited to hair cells of the organ of Corti by {{GA}} week 12. | |||
* transforming growth factor-β-activated kinase-1 (TAK1) - inner hair cells {{GA}} week 12 and colocalized with p75 neurotrophic receptor expression. | |||
* SRY (sex-determining region Y)-box 2 (SOX2) - supporting cells of utricle at the earliest stage examined at {{GA}} week 9. | |||
* GATA binding protein 3 (GATA3) - cochlear sensory epithelium and spiral ganglia at {{GA}} week 9. Midline of both the utricle and saccule in the zone corresponding to the striolar reversal zone where the hair cell phenotype switches from type I to type II. | |||
Tsukushi (TSK) - {{Mouse}} study{{#pmid:32127020|PMID32127020}} | |||
* a small, secreted, leucine-rich-repeat proteoglycan-interacts with and regulates cellular signaling cascades. | |||
* interacts with {{Sox}}2 and {{BMP}}4 to control stereocilia formation in the inner hair cells. | |||
* early embryonic stages - accumulates in nonprosensory regions | |||
* late embryonic stages - and in both nonprosensory and prosensory. | |||
* adult mice - localized in the organ of Corti, spiral ganglion cells, and the stria vascularis. | |||
==Semicircular canal == | ==Semicircular canal == | ||
| Line 515: | Line 560: | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:28603711}} | |||
{{#pmid:25040109}} | {{#pmid:25040109}} | ||
{{#pmid:26052920}} | |||
{{#pmid:20637105}} | {{#pmid:20637105}} | ||
| Line 523: | Line 572: | ||
* '''Bookshelf - Neuroscience''' [http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=neurosci.section.894 Neuroscience - The Inner Ear] | * '''Bookshelf - Neuroscience''' [http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=neurosci.section.894 Neuroscience - The Inner Ear] | ||
===Articles=== | ===Articles=== | ||
{{#pmid:32120242}} | |||
{{#pmid:32127020}} | |||
{{#pmid:31788757}} | |||
{{#pmid:26504244}} | {{#pmid:26504244}} | ||
{{#pmid:18603386}} | {{#pmid:18603386}} | ||
===Search PubMed=== | ===Search PubMed=== | ||
| Line 533: | Line 589: | ||
===Historic=== | ===Historic=== | ||
{{Ref-Keith1902}} | |||
{{Ref-Stricht1919b}} | |||
==Additional Images== | ==Additional Images== | ||
| Line 546: | Line 604: | ||
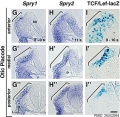
File:Mouse otic placode gene expression 02.jpg|Mouse otic placode Spry1, Spry2, and Wnt | File:Mouse otic placode gene expression 02.jpg|Mouse otic placode Spry1, Spry2, and Wnt | ||
File:Mouse - inner ear cartoon.jpg|Mouse - inner ear cartoon | File:Mouse - inner ear cartoon.jpg|Mouse - inner ear cartoon | ||
File:Human inner ear MicroCT.jpg|Human inner ear MicroCT PMID 30108474 | |||
File:Cat inner ear MicroCT.jpg|Cat inner ear MicroCT PMID 30108474 | |||
</gallery> | </gallery> | ||
===Historic Images=== | ===Historic Images=== | ||
{{Historic Disclaimer}} | {{Historic Disclaimer}} | ||
<gallery> | <gallery> | ||
File:Kollmann731.jpg|Kollmann Fig. 731 | File:Kollmann731.jpg|Kollmann Fig. 731 | ||
| Line 568: | Line 629: | ||
File:Kollmann747.jpg|Kollmann Fig. 747 | File:Kollmann747.jpg|Kollmann Fig. 747 | ||
File:Kollmann748.jpg|Kollmann Fig. 748 | File:Kollmann748.jpg|Kollmann Fig. 748 | ||
</gallery> | |||
{{Ref-Stricht1919b}} | |||
<gallery> | |||
File:Stricht1919b plate1.jpg|Plate 1 - {{cat}}, {{dog}} | |||
File:Stricht1919b plate2.jpg|Plate 2 - {{dog}}, {{cat}} | |||
File:Stricht1919b plate3.jpg|Plate 3 - {{bat}}, {{rat}} | |||
</gallery> | </gallery> | ||
| Line 577: | Line 645: | ||
</gallery> | </gallery> | ||
{{Ref-Anson1934}} | |||
<gallery> | <gallery> | ||
File:Anson1934 fig01-8.jpg|Fig. 1-8. Transverse sections of embryos in the Harvard Embryological Collection | File:Anson1934 fig01-8.jpg|Fig. 1-8. Transverse sections of embryos in the Harvard Embryological Collection | ||
| Line 590: | Line 659: | ||
File:Anson1934 fig18.jpg|Anson Fig. 18 | File:Anson1934 fig18.jpg|Anson Fig. 18 | ||
File:Anson1934 fig19.jpg|Anson Fig. 19 | File:Anson1934 fig19.jpg|Anson Fig. 19 | ||
</gallery> | |||
{{Ref-Streeter1906}} | |||
<gallery> | |||
File:Streeter1906 fig01.jpg|Streeter Fig. 1 | File:Streeter1906 fig01.jpg|Streeter Fig. 1 | ||
File:Streeter1906 fig02.jpg|Streeter Fig. 2 | File:Streeter1906 fig02.jpg|Streeter Fig. 2 | ||
| Line 603: | Line 675: | ||
File:Streeter030.jpg|Fetus 130 mm CRL (lateral) | File:Streeter030.jpg|Fetus 130 mm CRL (lateral) | ||
File:Streeter031.jpg|Fetus 130 mm CRL (median) | File:Streeter031.jpg|Fetus 130 mm CRL (median) | ||
{{Ref-Gray1918}} | |||
<gallery> | |||
File:Gray0924.jpg|Gray Fig 924 | File:Gray0924.jpg|Gray Fig 924 | ||
File:Gray0928.jpg|Gray Fig 928 | File:Gray0928.jpg|Gray Fig 928 | ||
| Line 611: | Line 686: | ||
<gallery> | <gallery> | ||
File:Streeter1957 fig07.jpg|Stage 19-23 | File:Streeter1957 fig07.jpg|Stage 19-23 | ||
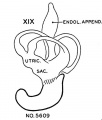
File:Streeter1957 fig7-19.jpg|[[Carnegie stage 19]] ( | File:Streeter1957 fig7-19.jpg|[[Carnegie stage 19]] (Embryo {{CE5609}}) | ||
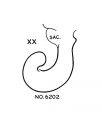
File:Streeter1957 fig7-20.jpg|[[Carnegie stage 20]] ( | File:Streeter1957 fig7-20.jpg|[[Carnegie stage 20]] (Embryo {{CE6202}}) | ||
File:Streeter1957 fig7-21.jpg|[[Carnegie stage 21]] ( | File:Streeter1957 fig7-21.jpg|[[Carnegie stage 21]] (Embryo {{CE22}}) | ||
File:Streeter1957 fig7-22.jpg|[[Carnegie stage 22]] ( | File:Streeter1957 fig7-22.jpg|[[Carnegie stage 22]] (Embryo {{CE840}}) | ||
File:Streeter1957 fig7-23.jpg|[[Carnegie stage 23]] ( | File:Streeter1957 fig7-23.jpg|[[Carnegie stage 23]] (Embryo {{CE86}}) | ||
</gallery> | </gallery> | ||
Latest revision as of 09:46, 28 May 2020
| Embryology - 20 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
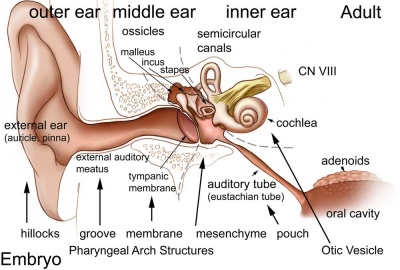
Introduction
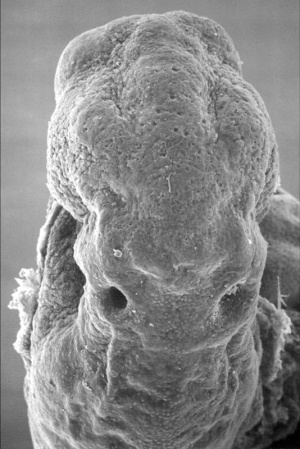
The inner ear is derived from a pair of surface sensory placodes (otic placodes) that appear in human development during week 4 (GA week 6) in the head region lying behind the second pharyngeal arch.
These otic placodes fold inwards forming initially a depression, then pinch off entirely from the surface forming an epithelium surrounding a fluid-filled sac or vesicle (otic vesicle, otocyst, auditory vesicle). The vesicle sinks into the head mesenchyme some of which closely surrounds the otocyst forming the otic capsule.
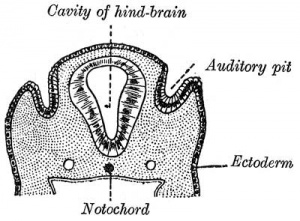
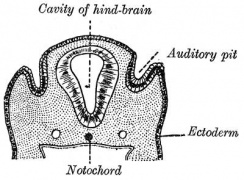
The otocyst finally lies close to the early developing hindbrain (rhombencephalon) and the developing vestibulo-cochlear-facial ganglion complex.
The otocyst epithelium then undergoes a series of morphological changes, forming the primitive membranous labyrinth. During the human fetal period this will differentiate into the inner ear components for hearing (cochlea) and balance (semi-circular canals). Cochlear development involving a transcription factor spatiotemporal gradient continues through to GA week 16.[1]
The adult cochlear has a "snail-shell" appearance, with the total number of turns differing between species. The adult human cochlear is typically described as having 2.5 turns, but this can vary up to 2.75 or even 3 turns.[2]
The "organ of corti" that develops within the cochlea was first identified by Alfonso Giacomo Gaspare Corti (1822–1876), an Italian anatomist, in 1851.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Inner Ear Development | Inner Ear Embryology | Organ of Corti Development | |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Otic Placode
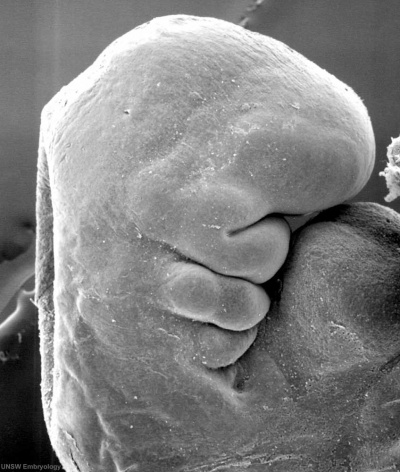
The embryonic surface sensory placode associated with hearing and balance. This will be lost from the embryo surface to form the otocyst or otic vesicle.
- Stage 11 - single layer of ectodermal cells organized in a columnar epithelium, which differs in cell shape from the surrounding cuboidal epithelia that will contribute the epithelia of the skin.
- zebrafish model - a number of specific genes are involved in initial induction of the otic placode including both growth factors (fgf3 and fgf8) and transcription factors (dlx3b, dlx4b, and foxi1).[13]
- Proliferation of the otic placode cells leads to an inward folding, or invagination, giving the external appearance of a depression on the lateral sides of the early developing neck region.
- The epithelium is still a single layer of cells, which continues to invaginate until the edges of the disc of cells come into apposition on the embryo surface.
- mouse model - placodal invagination but not specification requires placodal expression of the transcription factor Sox9.[14]
Sensory Placodes
- Week 4 a series of thickened surface ectodermal patches form in pairs rostro-caudally in the head region.
- Recent research suggests that all sensory placodes may arise from common panplacodal primordium origin around the neural plate, and then differentiate to eventually have different developmental fates.
- Each pair of sensory placodes will later contribute key components of each of our special senses (hearing, vision, smell and taste).
- Otic Placode - one of the first to form and contributes inner ear structures.
- Optic (Lens) Placode - lies on the surface, adjacent to the outpocketing of the nervous system (which will for the retina) and will form the lens.
- Nasal Placode - 2 components (medial and lateral) and will form the nose olefactory epithelium.
- Other species have a number of additional placodes which form other sensory structures (fish, lateral line receptor).
- Note that their initial postion on the developing head is significantly different to their final position in the future sensory system.
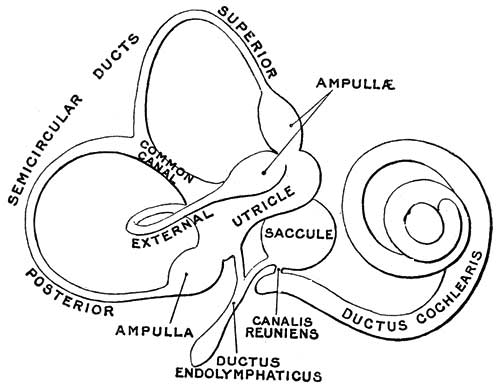
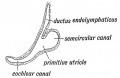
Cochlea Timeline
The otocyst will differentiate to form all components of the membranous labyrinth. The organ of Corti forms initially from the central domain on the medial side of the otocyst.[8] Sox2 regulates organ of Corti prosensory progenitor development, through Jag1 (Notch ligand) signaling.
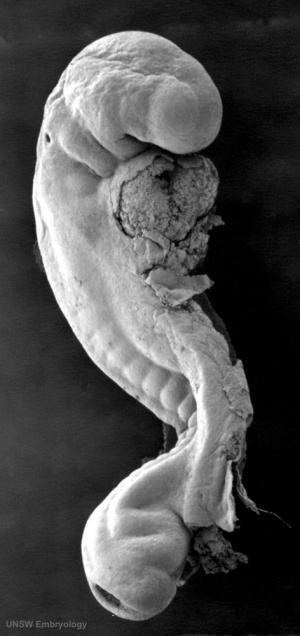
|
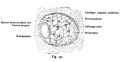
Stage 13 otic vesicle now lies beneath the embryo surface.
|
- Otocyst historic drawings
Week 5
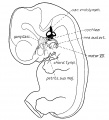
Stage 13 embryo (week 5) showing otocyst that will form the inner ear.
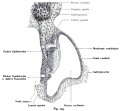
| A. Ventrolateral view of the whole embryo with 5-mm scale bar. At this stage of development no middle or external ear structures are apparent and will be derived later from pharyngeal arches one and two (labeled). | B. The gray bar through the head indicates the plane of cross-section, which is a cross-section of the head showing the size and position of the otic vesicles. At this stage of development they lie within the head mesenchyme behind pharyngeal arch one and two and in close apposition to the developing hindbrain. Note the close position of the otic vesicle to the rhombomeres, hindbrain folds that represent the initial segmentation of the hindbrain. Also shown are developing cranial ganglia and blood vessel lying adjacent to the otic vesicles. The wall of the otic vesicle at this stage is a simple epithelium. |
Week 7
Based upon Streeter.[15]
Week 8
Based upon Streeter.[15]
- Carnegie stage 20 - Long tip; transitional stage.
- Carnegie stage 21 - Return down curve (tip turns down).
- Carnegie stage 22 - Transitional stage.
- Carnegie stage 23 - Tip turns up second time. Tip turns down second time (second return down curve).
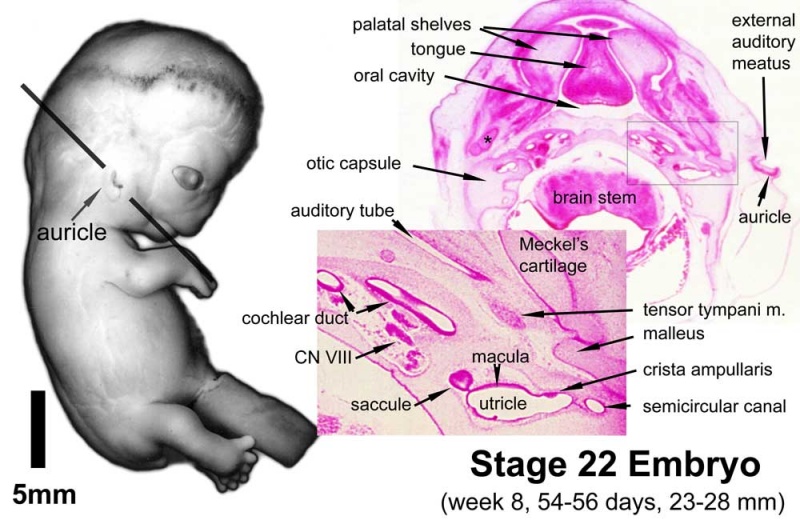
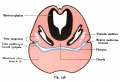
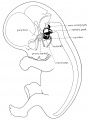
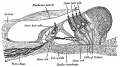
Stage 22 embryo (week 8) showing the embryo near the end of the embryonic period.
| A. Lateral view of the whole embryo with 5 mm scale bar. Note the well developed external ear with simplified adult structure and narrower meatal opening. The grey bar through the head indicates the plane of cross-section for (B) and (C). | B. Cross-section of the head at the plane of the skull base and oral cavity to the top. The otic capsule is well formed by this stage containing all the membranous labyrinth structures. It is still a cartilaginous structure ventral to the brainstem and lying behind the oral cavity. The tongue occupies the floor of the oral cavity with the unfused palatal shelves lying lateral and the auditory tubes clearly shown on the posterior wall. The external ear is visible on the right hand side of the head with a band of cartilage (dark stain) within the auricle.
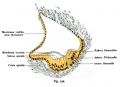
C. The gray box indicates this region: detail of inner and middle ear development. The middle ear cavity has not yet formed and the ossicles (malleus shown) are embedded in mesenchyme that is being lost. The tensor tympani muscle is differentiating in the adjacent mesenchyme. The inner ear membranous labyrinth has formed its adult external structure. The section through the turns of the cochlear duct shows the internal cochlea structure is still underdeveloped; in contrast, the balance region is more developed. |
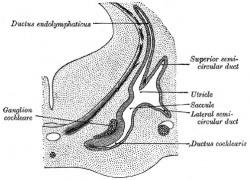
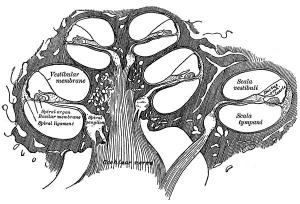
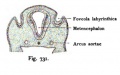
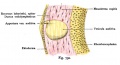
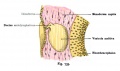
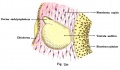
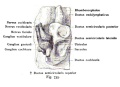
The Membranous Labyrinth
Periotic space first appears in week 8 embryos 35 mm CRL around the vestibule and basal turn of the cochlea.[5]
Fetal
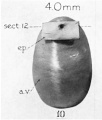
Week 8.4
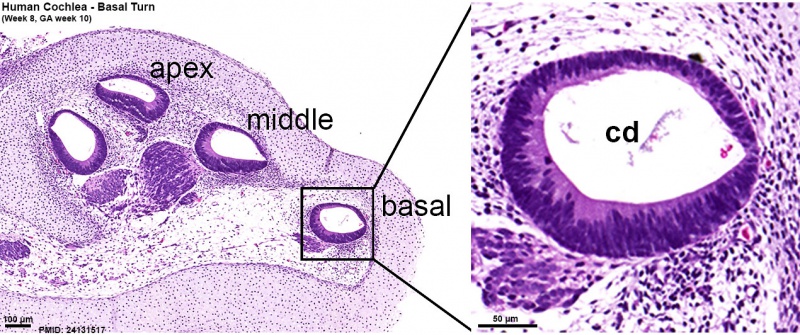
Human fetal cochlea basal turn week 8.4 Gestational Week GA 10.4 (Stain - Haematoxylin Eosin)[16]
Week 10
Human fetal cochlea basal turn week 10 Gestational Week GA 12 (Stain - Haematoxylin Eosin)[16]
Week 14
Periotic space almost covers the membranous labyrinth in the fetus 115 mm CRL week 14 (GA week 16).[5]
Week 18 - 22
Developmental data from an EM study of the human organ of corti.[17] Original GA data has been recalculated as fertilisation ages.
| Week | Event |
|---|---|
| 18 | stria vascularis, tectorial membrane, afferent nerve ending |
| 20 | Nuel space, stereocilia maturation, efferent nerve ending |
| 22 | tunnel of corti, kinocilium, space between hair and supporting cell |
| Week 18 | Week 20 | Week 22 |
|---|---|---|
Historic
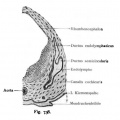
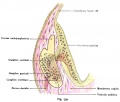
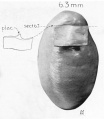
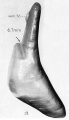
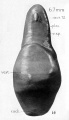
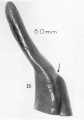
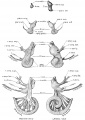
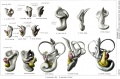
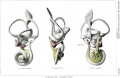
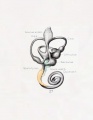
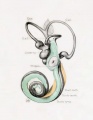
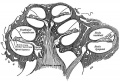
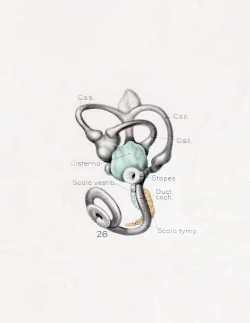
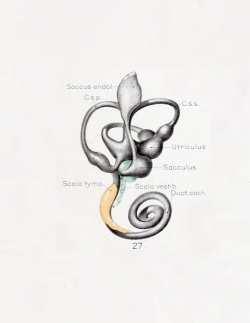
A series of historic wax-plate reconstructions of the membranous labyrinth and the surrounding periotic tissue-spaces showing development stages (median and lateral views) of these spaces at the same scale (ages are only approximations calculated on CRL).
| Human Fetal Membranous Labyrinth Development | |
|---|---|
| Fetus 50 mm CRL 10 weeks (GA 12) | |
|
|
| lateral view | median view |
| Fetus 85 mm CRL 14 weeks (GA 16) | |
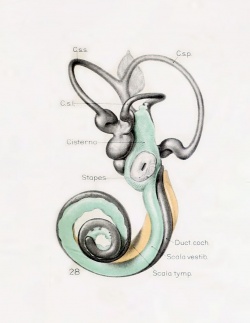
|
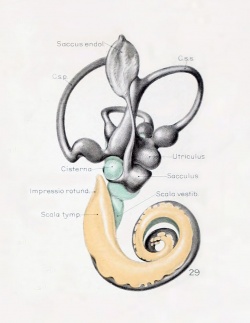
|
| lateral view | median view |
| Fetus 130 mm CRL 15 weeks (GA 17) | |
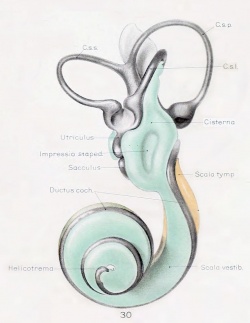
|
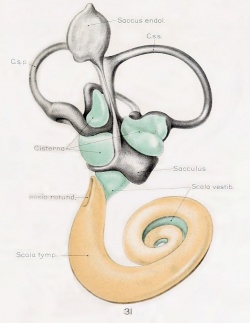
|
| lateral view | median view |
Fetal Cochlea Molecular
Human fetal cochlea basal turn by Gestational Week GA[16]
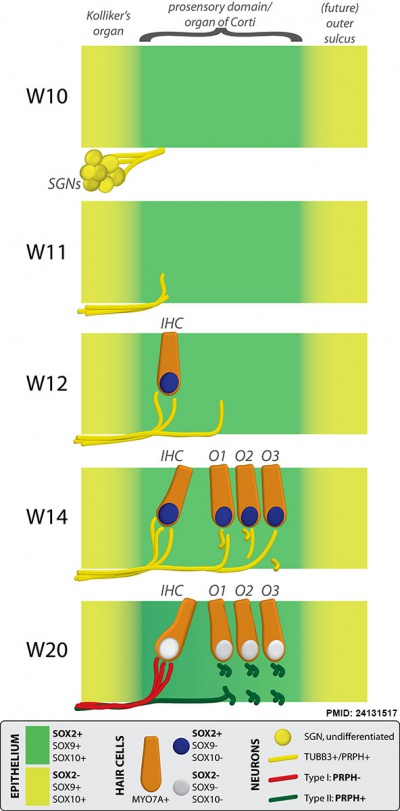
Abbreviations
|
Gestational Week GA
Week 10 (W10) - SOX2 identifies the prosensory domain within the SOX9/SOX10+ cochlear duct epithelium. Neurites from the adjoining TUBB3+/PRPH + SGNs do not yet penetrate into the epithelium.
|
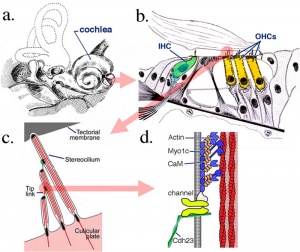
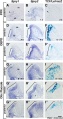
Organ of Corti
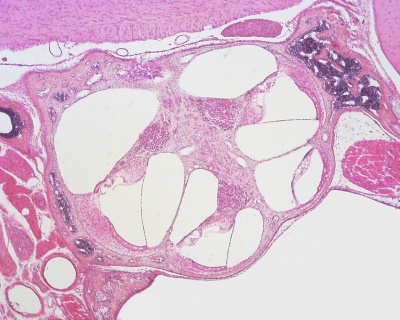
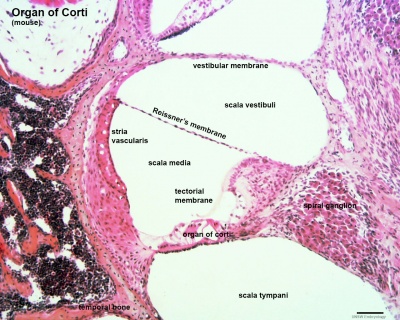
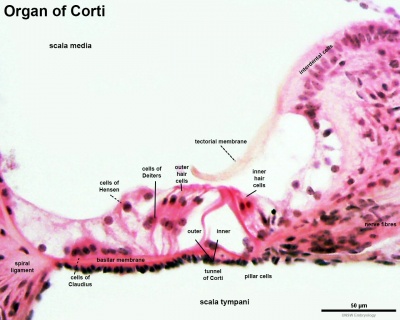
Within the cochlea, the specialised structure required for converting mechanical vibration into an electrical signal occurs at the organ of Corti (spiral organ of Corti).
The images (mouse) below show the detail of the specialised structure, the organ of Corti, that develops through the fetal period.
|
|

|
|
Endolymphatic Sac
|
|
Vestibular Sac
- generates 3 expansions - form semicircular ducts
- remainder forms utricle
- epithelia lining generates - hair cells, ampullary cristae, utricular macula
- Vestibular - Otoconia, otoconin- inner ear biominerals
Cochlear Sac
- generates coiled cochlear duct (humans 2 1/2 turns)
- remainder forms saccule
- epithelia lining generates
- hair cells
- structures of organ of corti
- saccular macula
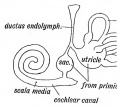
Scala Media
(Latin, medius = middle) spiral of middle cochlear duct lying between scala vestibuli and scala tympani, containing endolymph.
Scala Tympani
(Latin, tympanon = drum) the spiralling cochlear duct below spiral lamina, containing perilymph and ending at round window near tympanic membrane.
Scala Vestibuli
(Latin, vestibulum = cavity at beginning of canal) the spiralling cochlear duct above spiral lamina, containing perilymph, beginning near the vestbule and ending where it communicates with the scala tympani at the helicotrema.
Endolymph
- extracellular fluid secreted by the stria vascularis.
- potassium is the main cation required for depolarizing electrical current in the hair cells.
Perilymph
- extracellular fluid similar in composition to either plasma or cerebrospinal fluid.
- sodium is the main cation.
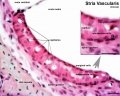
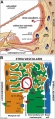
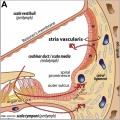
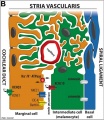
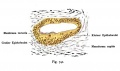
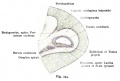
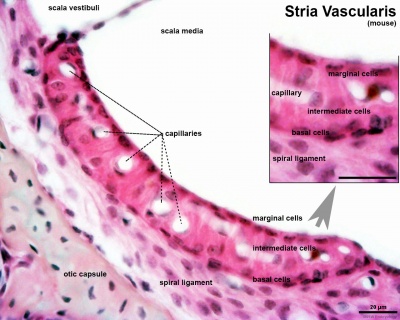
Stria Vascularis
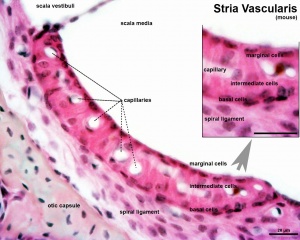
|
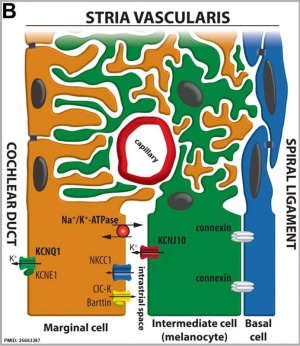
|
| In the adult the stria vascularis functions to synthesise and secretes endolymph. The three main cell types within the stria that have different embryological origins and are connected by different forms of cell junctions. | Anatomical (upper half) and compartmental (lower half) model of the adult stria vascularis showing the three cellular layers and depicting the location of potassium regulating channels. The stria vascularis is electrochemically isolated from neighboring structures by tight junctions (black bars).[9] |
| Marginal cells | Intermediate cells | Basal cells |
|---|---|---|
| line the lumen of the cochlear duct | melanocyte-like cells lie between the marginal and basal cell layers | mesenchymal spiral ligament fibrocytes |
| derived from epithelia. | derived from the neural crest. | derived from otic mesenchyme.[18] |
Mouse SEM
The gallery below shows scanning electron micrographs of the developing mouse (E18.5) cochlea.[19]
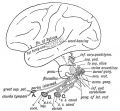
Adult Cochlea
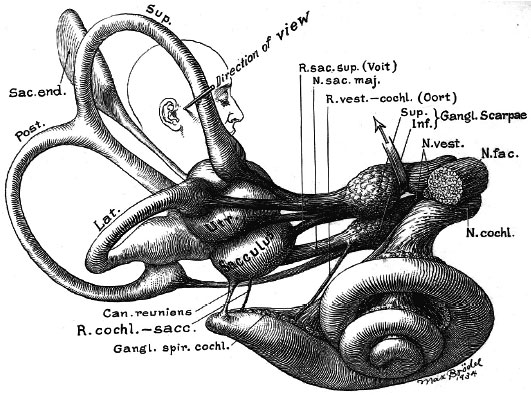
Adult inner ear (Max Brödel 1934)
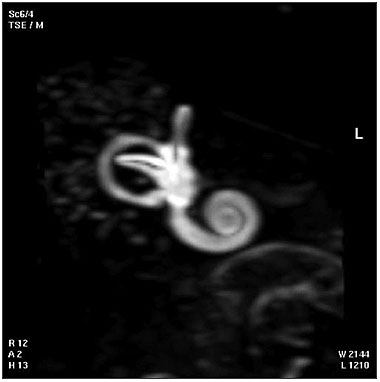
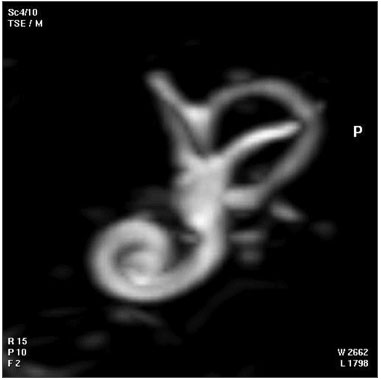
| Magnetic Resonance Images of the Adult Cochlea[20] | |
|---|---|
|
|
The investigators used a method to obtain magnetic resonance imaging (MRI) to measure cochlear length from the temporal bones of 6 cadavers. By overlapping digitalized rulers on these images it was possible to measure cochlear length. Adult cochlear length varied between 17 and 26.5 millimeters.[20]
Mouse cochlea (SEM) showing organization of the hair cells.[19]
- Mouse Cochlea Links: Cochlea overview SEM | Base region SEM | Mid-base and Apex region SEM | Mid-base region SEM | Mid-base hair cells SEM | Mouse Development
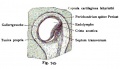
Bony Labyrinth
- formed from chrondified mesoderm
- Periotic Capsule
- mesenchyme within capsule degenerates to form space filled with perilymph
Auditory Neural Pathway
Vestibulocochlear Nerve
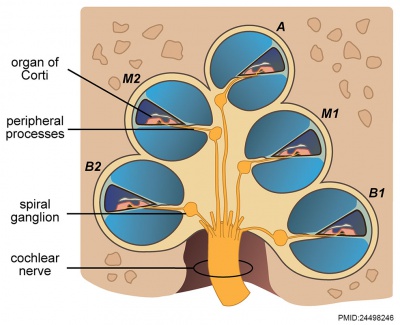
|
| Adult cochlea nerve glia cartoon[10] |
- forms beside otocyst
- from wall of otocyst and neural crest cells
- bipolar neurons
Vestibular Neurons
- outer end of internal acoustic meatus
- innervate hair cells in membranous labyrinth
- axons project to brain stem and synapse in vestibular nucleus
Cochlear Neurons
- cell bodies lie in modiolus
- central pillar of cochlear
- innervate hair cells of spiral organ
- axons project to cochlear nucleus
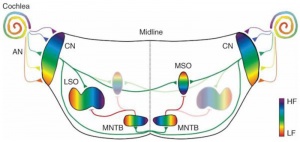
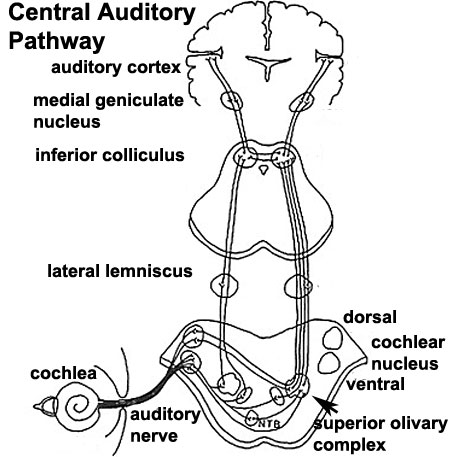
- Links: Hearing - Neural Pathway
Cochlea Glial
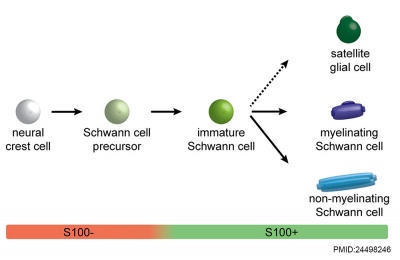
|
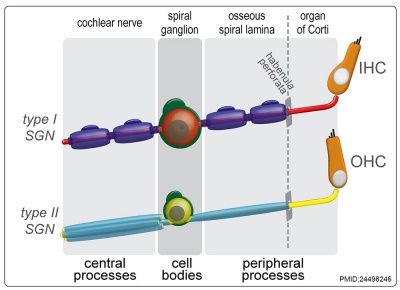
|
| Cochlea glial lineage [10] | Adult cochlea nerve glia cartoon[10] |
- Links: Hearing - Neural Pathway
Inner Ear Genes
- hindbrain segmentation occurs at same time placode arises
- otocyst adjacent to rhombomere 5
- may influence development
- Hoxa1, kreisler, Fgf3
- genes regulating neural crest cells (neural genes)
- Pax2 Ko affects cochlear and spiral ganglion, but not vestibular apparatus
- nerogenin 1 affects both ganglia
- LIN28B - RNA-binding protein times auditory prosensory cell cycle withdrawal and differentiation through both let-7–dependent and independent mechanisms.[21]
- FGF - FGF8 and Template:FGFR3 are essential for pillar cell specification.[4]
From a recent study of human fetal tissue.[1]
- Leucine-rich repeat-containing G protein-coupled receptor 5 (LGR5) - apical poles of the sensory epithelium of the cochlear duct and the vestibular end organs GA week 11. Limited to hair cells of the organ of Corti by GA week 12.
- transforming growth factor-β-activated kinase-1 (TAK1) - inner hair cells GA week 12 and colocalized with p75 neurotrophic receptor expression.
- SRY (sex-determining region Y)-box 2 (SOX2) - supporting cells of utricle at the earliest stage examined at GA week 9.
- GATA binding protein 3 (GATA3) - cochlear sensory epithelium and spiral ganglia at GA week 9. Midline of both the utricle and saccule in the zone corresponding to the striolar reversal zone where the hair cell phenotype switches from type I to type II.
Tsukushi (TSK) - mouse study[22]
- a small, secreted, leucine-rich-repeat proteoglycan-interacts with and regulates cellular signaling cascades.
- interacts with Sox2 and BMP4 to control stereocilia formation in the inner hair cells.
- early embryonic stages - accumulates in nonprosensory regions
- late embryonic stages - and in both nonprosensory and prosensory.
- adult mice - localized in the organ of Corti, spiral ganglion cells, and the stria vascularis.
Semicircular canal
- Otx1- cochlear and vestibular normal
- Hmx3, Prx1, Prx2
Sensory Organs
- thyroid hormone receptor beta
- Zebrafish-mindbomb mutant has excess hair cells but not supporting cells, Notch-Delta signaling
- Gene Expression-inner ear
- Brn-3c and Hair cell development
- Supporting Cells- p27kip
- Thyroid Hormone
- Ganglion neurons require growth factors
- vestibular neurons- BDNF, NT3
- survival not development
Sox9 20346939 Sox2 20071536
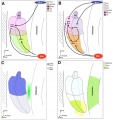
Other Species Overview
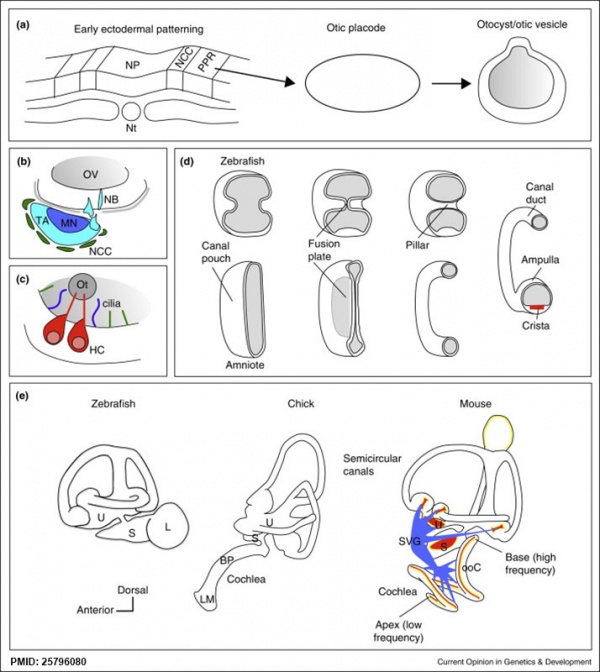
Comparison of zebrafish and late stage chick and mouse embryos[23] Zebrafish Development Chicken Development | Mouse Development |
(a) Formation of the pre-placodal region (PPR), otic placode and otocyst (otic vesicle) from cranial ectoderm. The otocyst is the source of nearly all cell types of the mature ear.
(b) Otic neurogenesis: neuroblasts are specified from otic vesicle epithelium, but delaminate from it and accumulate beneath the ear in a transit amplifying population (light blue). Neurons (dark blue) differentiate from this population, and innervate sensory hair cells in the overlying otic epithelium. The ganglion develops in close association with neural crest cells (green), which give rise to glia. (c) Early otolith formation in the zebrafish otic vesicle. At least three distinct populations of cilia can be distinguished: immotile hair cell kinocilia (red), which tether the otolith at early stages; motile cilia (blue) in the vicinity of the sensory hair cells, which do not bind otolithic material, and shorter immotile cilia (green). (d) Schematic comparison of semicircular canal formation in the zebrafish ear (top row) and a generalised amniote ear (bottom row). A single canal is illustrated for clarity. Epithelia adhere at a fusion plate, from which cells are cleared to make the duct. The end result of both events is the same (right hand image), but the fusion plate is much smaller in the zebrafish. (e) Comparative sketches of inner ears from adult zebrafish and late stage chick and mouse embryos. Sensory (red), neuronal (blue) and endolymph-regulating (yellow) cells are shown for the mouse ear.
(text from original figure legend) |
Mouse
- Dual embryonic origin of the mammalian otic vesicle forming the inner ear[24] "The inner ear and cochleovestibular ganglion (CVG) derive from a specialized region of head ectoderm termed the otic placode. During embryogenesis, the otic placode invaginates into the head to form the otic vesicle (OV), the primordium of the inner ear and CVG. Non-autonomous cell signaling from the hindbrain to the OV is required for inner ear morphogenesis and neurogenesis. In this study, we show that neuroepithelial cells (NECs), including neural crest cells (NCCs), can contribute directly to the OV from the neural tube. ...This study defines a dual cellular origin of the inner ear from sensory placode ectoderm and NECs, and changes the current paradigm of inner ear neurosensory development."
References
- ↑ 1.0 1.1 Johnson Chacko L, Sergi C, Eberharter T, Dudas J, Rask-Andersen H, Hoermann R, Fritsch H, Fischer N, Glueckert R & Schrott-Fischer A. (2020). Early appearance of key transcription factors influence the spatiotemporal development of the human inner ear. Cell Tissue Res. , 379, 459-471. PMID: 31788757 DOI.
- ↑ Biedron S, Westhofen M & Ilgner J. (2009). On the number of turns in human cochleae. Otol. Neurotol. , 30, 414-7. PMID: 19225438 DOI.
- ↑ Kolla L, Kelly MC, Mann ZF, Anaya-Rocha A, Ellis K, Lemons A, Palermo AT, So KS, Mays JC, Orvis J, Burns JC, Hertzano R, Driver EC & Kelley MW. (2020). Characterization of the development of the mouse cochlear epithelium at the single cell level. Nat Commun , 11, 2389. PMID: 32404924 DOI.
- ↑ 4.0 4.1 Jansson L, Ebeid M, Shen JW, Mokhtari TE, Quiruz LA, Ornitz DM, Huh SH & Cheng AG. (2019). β-Catenin is required for radial cell patterning and identity in the developing mouse cochlea. Proc. Natl. Acad. Sci. U.S.A. , 116, 21054-21060. PMID: 31570588 DOI.
- ↑ 5.0 5.1 5.2 Ishikawa A, Ohtsuki S, Yamada S, Uwabe C, Imai H, Matsuda T & Takakuwa T. (2018). Formation of the Periotic Space During the Early Fetal Period in Humans. Anat Rec (Hoboken) , 301, 563-570. PMID: 29293291 DOI.
- ↑ Chadly DM, Best J, Ran C, Bruska M, Woźniak W, Kempisty B, Schwartz M, LaFleur B, Kerns BJ, Kessler JA & Matsuoka AJ. (2018). Developmental profiling of microRNAs in the human embryonic inner ear. PLoS ONE , 13, e0191452. PMID: 29373586 DOI.
- ↑ Johnson Chacko L, Blumer MJF, Pechriggl E, Rask-Andersen H, Dietl W, Haim A, Fritsch H, Glueckert R, Dudas J & Schrott-Fischer A. (2017). Role of BDNF and neurotrophic receptors in human inner ear development. Cell Tissue Res. , 370, 347-363. PMID: 28924861 DOI.
- ↑ 8.0 8.1 Gu R, Brown RM, Hsu CW, Cai T, Crowder AL, Piazza VG, Vadakkan TJ, Dickinson ME & Groves AK. (2016). Lineage tracing of Sox2-expressing progenitor cells in the mouse inner ear reveals a broad contribution to non-sensory tissues and insights into the origin of the organ of Corti. Dev. Biol. , 414, 72-84. PMID: 27090805 DOI.
- ↑ 9.0 9.1 9.2 Locher H, de Groot JC, van Iperen L, Huisman MA, Frijns JH & Chuva de Sousa Lopes SM. (2015). Development of the stria vascularis and potassium regulation in the human fetal cochlea: Insights into hereditary sensorineural hearing loss. Dev Neurobiol , 75, 1219-40. PMID: 25663387 DOI.
- ↑ 10.0 10.1 10.2 10.3 Locher H, de Groot JC, van Iperen L, Huisman MA, Frijns JH & Chuva de Sousa Lopes SM. (2014). Distribution and development of peripheral glial cells in the human fetal cochlea. PLoS ONE , 9, e88066. PMID: 24498246 DOI.
- ↑ Bok J, Zenczak C, Hwang CH & Wu DK. (2013). Auditory ganglion source of Sonic hedgehog regulates timing of cell cycle exit and differentiation of mammalian cochlear hair cells. Proc. Natl. Acad. Sci. U.S.A. , 110, 13869-74. PMID: 23918393 DOI.
- ↑ Braunstein EM, Monks DC, Aggarwal VS, Arnold JS & Morrow BE. (2009). Tbx1 and Brn4 regulate retinoic acid metabolic genes during cochlear morphogenesis. BMC Dev. Biol. , 9, 31. PMID: 19476657 DOI.
- ↑ Solomon KS, Kwak SJ & Fritz A. (2004). Genetic interactions underlying otic placode induction and formation. Dev. Dyn. , 230, 419-33. PMID: 15188428 DOI.
- ↑ Barrionuevo F, Naumann A, Bagheri-Fam S, Speth V, Taketo MM, Scherer G & Neubüser A. (2008). Sox9 is required for invagination of the otic placode in mice. Dev. Biol. , 317, 213-24. PMID: 18377888 DOI.
- ↑ 15.0 15.1 Streeter GL. Developmental Horizons In Human Embryos Description Or Age Groups XIX, XX, XXI, XXII, And XXIII, Being The Fifth Issue Of A Survey Of The Carnegie Collection. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: 167-196.
- ↑ 16.0 16.1 16.2 Locher H, Frijns JH, van Iperen L, de Groot JC, Huisman MA & Chuva de Sousa Lopes SM. (2013). Neurosensory development and cell fate determination in the human cochlea. Neural Dev , 8, 20. PMID: 24131517 DOI.
- ↑ Igarashi Y & Ishii T. (1980). Embryonic development of the human organ of Corti: electron microscopic study. Int. J. Pediatr. Otorhinolaryngol. , 2, 51-62. PMID: 7188054
- ↑ Trowe MO, Maier H, Petry M, Schweizer M, Schuster-Gossler K & Kispert A. (2011). Impaired stria vascularis integrity upon loss of E-cadherin in basal cells. Dev. Biol. , 359, 95-107. PMID: 21925491 DOI.
- ↑ 19.0 19.1 Kiernan AE, Xu J & Gridley T. (2006). The Notch ligand JAG1 is required for sensory progenitor development in the mammalian inner ear. PLoS Genet. , 2, e4. PMID: 16410827 DOI.
- ↑ 20.0 20.1 Pochini Sobrinho F, Lazarini PR, Yoo HJ, Abreu Júnior Ld & Meira Ade S. (2009). A method for measuring the length of the cochlea through magnetic resonance imaging. Braz J Otorhinolaryngol , 75, 261-7. PMID: 19575114
- ↑ Golden EJ, Benito-Gonzalez A & Doetzlhofer A. (2015). The RNA-binding protein LIN28B regulates developmental timing in the mammalian cochlea. Proc. Natl. Acad. Sci. U.S.A. , 112, E3864-73. PMID: 26139524 DOI.
- ↑ Miwa T, Ohta K, Ito N, Hattori S, Miyakawa T, Takeo T, Nakagata N, Song WJ & Minoda R. (2020). Tsukushi is essential for the development of the inner ear. Mol Brain , 13, 29. PMID: 32127020 DOI.
- ↑ Whitfield TT. (2015). Development of the inner ear. Curr. Opin. Genet. Dev. , 32, 112-8. PMID: 25796080 DOI.
- ↑ Freyer L, Aggarwal V & Morrow BE. (2011). Dual embryonic origin of the mammalian otic vesicle forming the inner ear. Development , 138, 5403-14. PMID: 22110056 DOI.
Reviews
Magariños M, Pulido S, Aburto MR, de Iriarte Rodríguez R & Varela-Nieto I. (2017). Autophagy in the Vertebrate Inner Ear. Front Cell Dev Biol , 5, 56. PMID: 28603711 DOI.
Nakajima Y. (2015). Signaling regulating inner ear development: cell fate determination, patterning, morphogenesis, and defects. Congenit Anom (Kyoto) , 55, 17-25. PMID: 25040109 DOI.
Basch ML, Brown RM, Jen HI & Groves AK. (2016). Where hearing starts: the development of the mammalian cochlea. J. Anat. , 228, 233-54. PMID: 26052920 DOI.
Chatterjee S, Kraus P & Lufkin T. (2010). A symphony of inner ear developmental control genes. BMC Genet. , 11, 68. PMID: 20637105 DOI.
Driver EC & Kelley MW. (2009). Specification of cell fate in the mammalian cochlea. Birth Defects Res. C Embryo Today , 87, 212-21. PMID: 19750520 DOI.
- Bookshelf - Neuroscience Neuroscience - The Inner Ear
Articles
Schade-Mann T, Münkner S, Eckrich T & Engel J. (2020). Calcium signaling in interdental cells during the critical developmental period of the mouse cochlea. Hear. Res. , 389, 107913. PMID: 32120242 DOI.
Miwa T, Ohta K, Ito N, Hattori S, Miyakawa T, Takeo T, Nakagata N, Song WJ & Minoda R. (2020). Tsukushi is essential for the development of the inner ear. Mol Brain , 13, 29. PMID: 32127020 DOI.
Johnson Chacko L, Sergi C, Eberharter T, Dudas J, Rask-Andersen H, Hoermann R, Fritsch H, Fischer N, Glueckert R & Schrott-Fischer A. (2020). Early appearance of key transcription factors influence the spatiotemporal development of the human inner ear. Cell Tissue Res. , 379, 459-471. PMID: 31788757 DOI.
Gnedeva K & Hudspeth AJ. (2015). SoxC transcription factors are essential for the development of the inner ear. Proc. Natl. Acad. Sci. U.S.A. , 112, 14066-71. PMID: 26504244 DOI.
Santi PA, Rapson I & Voie A. (2008). Development of the mouse cochlea database (MCD). Hear. Res. , 243, 11-7. PMID: 18603386 DOI.
Search PubMed
May 2010 "Inner Ear Development" All (4027) Review (452) Free Full Text (750)
Search Pubmed: Inner Ear Development Cochlea Development
Historic
Keith A. Human Embryology and Morphology. (1902) London: Edward Arnold.
Van der Stricht O. The development of the pillar cells, tunnel space, and Nuel's spaces in the organ of Corti. (1919) J Comp. Neurol. 30: 283-.
Additional Images
Historic Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Van der Stricht O. The development of the pillar cells, tunnel space, and Nuel's spaces in the organ of Corti. (1919) J Comp. Neurol. 30: 283-.
Anson BJ. The early development of the membranous labyrinth in mammalian embryos, with special reference to the endolymphatic duct and the utriculo—endolymphatic duct. (1934) Anat. Rec. 59: 15-25.
Streeter GL. Developmental Horizons In Human Embryos Description Or Age Groups XIX, XX, XXI, XXII, And XXIII, Being The Fifth Issue Of A Survey Of The Carnegie Collection. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36: 167-196.
Carnegie stage 19 (Embryo 5609)
Carnegie stage 20 (Embryo 6202)
Carnegie stage 21 (Embryo 22)
Carnegie stage 22 (Embryo 840)
Carnegie stage 23 (Embryo 86)
Terms
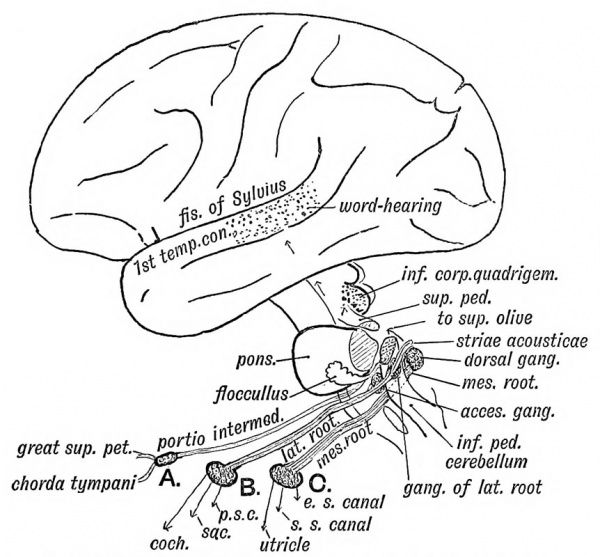
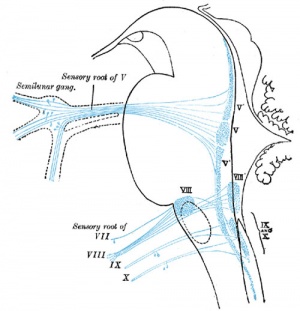
| Hearing Terms | ||
|---|---|---|
Hearing and Balance Development
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Mouse Genomic Database cochlea morphogenesis genes
- Acoustical Society of America ICA/ASA '98 Lay Language Papers
- Promenade around the Cochlea Organ of Corti
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 20) Embryology Hearing - Inner Ear Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Hearing_-_Inner_Ear_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G