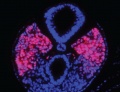
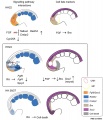
Somitogenesis
| Embryology - 21 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
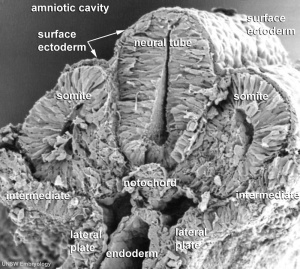
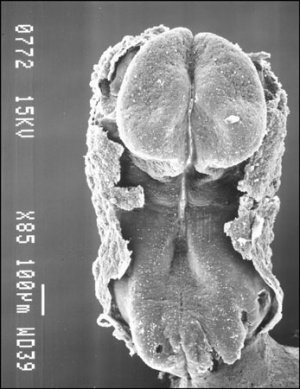
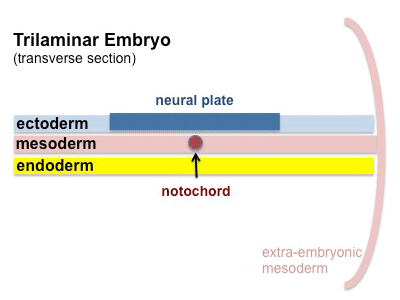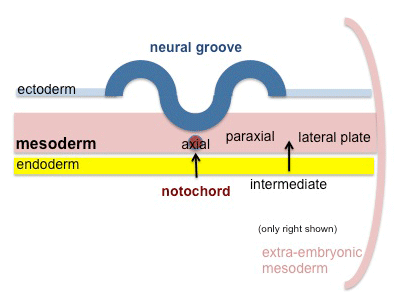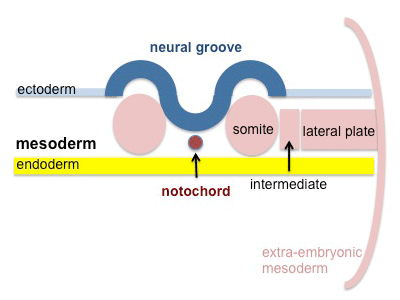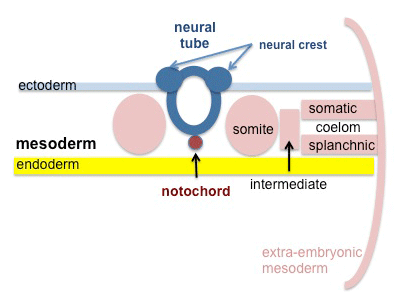
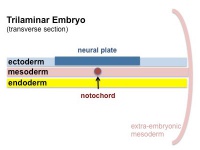
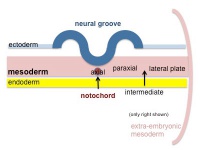
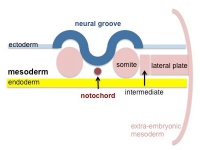
The term somitogenesis is used to describe the process of segmentation of the paraxial mesoderm within the trilaminar embryo body to form pairs of somites, or balls of mesoderm. In humans, the first somite pair appears at day 20 and adds caudally at 1 somite pair/90 minutes until on average 44 pairs eventually form.
A somite is added either side of the notochord (axial mesoderm) to form a somite pair. The segmentation does not occur in the head region, and begins cranially (head end) and extends caudally (tailward) adding a somite pair at regular time intervals. The process is sequential and therefore used to stage the age of many different species embryos based upon the number visible somite pairs.
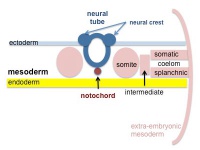
A mesenchymal to epithelial transition defines the outer cellular "shell" of the developing somite, with the core cells remain as a mesenchymal organisation. During early somite development a transient fluid-filled space, the somitocoel, can be identified in each somite and is later lost by cell proliferation. Neural crest cells later also enter and mix with the somatic cells.
Somites give rise to many different connective tissues including: cartilage, bone, muscle and tendon.
Cranial paraxial mesoderm remains unsegmented, and does not form somites, and will form the pharyngeal muscle progenitor field, the embryonic origin of facial and neck muscles.
| Mesoderm Links: endoderm | mesoderm | ectoderm | Lecture - Mesoderm | Lecture - Musculoskeletal | 2016 Lecture | notochord | somitogenesis | somite | splanchnic mesoderm | skeletal muscle | smooth muscle | heart | Notochord Movie | musculoskeletal | cartilage | bone | sonic hedgehog | Category:Mesoderm | ||||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Somitogenesis | Somite | Paraxial mesoderm | Presomitic Mesoderm | Sclerotome | Myotome | Dermatome | |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Presomitic Mesoderm
|
|
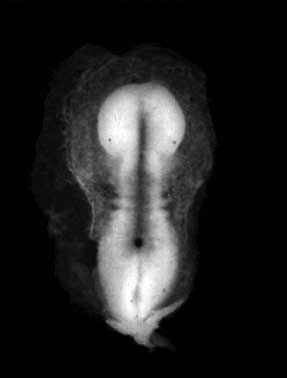
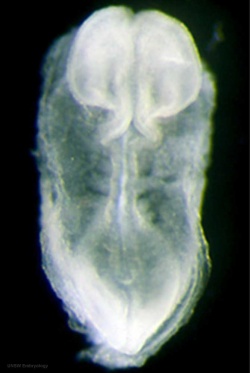
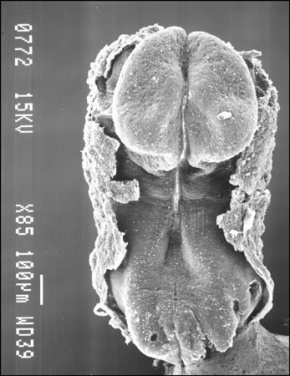
Human First Somites
|
|
|
Human embryo first somite pairs (week 4, Carnegie stage 9)
Somite Number
Week 4 to 5 (GA 6 to 7) covers the main period of human somitogenesis.
| Week | Days | Carnegie Stage | Somite Number (pairs) |
|---|---|---|---|
| Week 3 | 19 - 21 | 9 image | 1 - 3 |
| Week 4 | 22 - 23 | 10 image | 4 - 12 |
| Week 4 | 23 - 26 | 11 image | 13 - 20 |
| Week 4 | 26 - 30 | 12 image | 21 - 29 |
| Week 5 | 28 - 32 | 13 image | 30 |
| Week 5 | 31 - 35 | 14 image | 30+ |
Seel also Other species Somitogenesis
Mesoderm to Somite
Mesoderm means the "middle layer" and it is from this layer that nearly all the bodies connective tissues are derived. In early mesoderm development a number of transient structures will form and then be lost as tissue structure is patterned and organised. Humans are vertebrates, with a "backbone", and the first mesoderm structure we will see form after the notochord will be somites.
- During segmentation the outer cell layer forms an epithelial layer over a still mesenchymal organization of cells at the core.
- The early forming somite has a cavity at its core called a "somitocoel" that later fills with proliferating mesoderm cells.
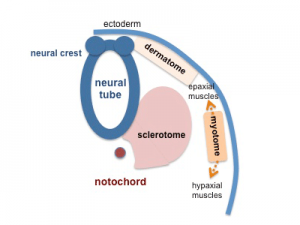
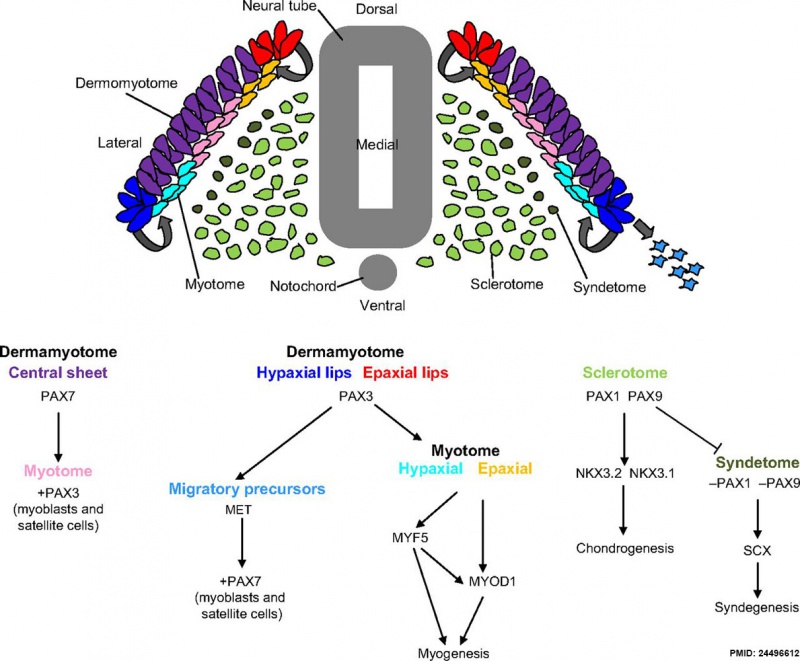
Somite to Sclerotome and Dermomyotome
| Sclerotome | Dermatome |
|---|---|
|
|
| Myotome | |
|
Somite initially forms 2 main regional components
- ventromedial region - sclerotome forms vertebral body and intervertebral disc
- dorsolateral region - dermomyotome forms dermis and skeletal muscle
Sclerotome
- The left and right sclerotomes from the same segmental level engulf the notochord.
- Each segmental level is then resegmented in a rostrocaudal direction.
Dermomyotome
- The dermomyotome is divided into a dorsal and ventral half.
- Dorsal - dermatome.
- Ventral - myotome, this will also divide into a dorsal and ventral half that contribute the epaxial and hypaxial skeletal muscle groups respectively.
- hypaxial - muscles of the ventrolateral body wall, girdle, limb and tongue.[11]
- Muscle cells of the limb, tongue and lateral shoulder girdle muscles - derived from somite migrating myogenic precursor cells.
- Muscle cells of the ventrolateral body wall muscles (intercostal and abdominal muscles) and the medial shoulder girdle muscles - derived from the myotome.
Part of the shoulder girdle muscles (trapezius and sternocleidomastoideus) - derived from the lateral plate mesoderm.
Molecular
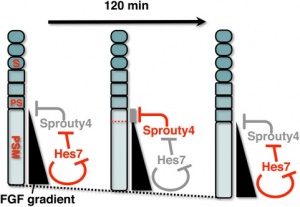
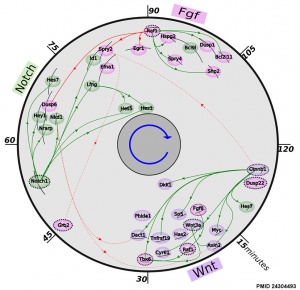
During somitogenesis Dll1 protein appears to regulate clock genes, such as Hes7, expression oscillation in the presomitic mesoderm (PSM).[12]
Hes7
Hairy/Enhancer Of Split, Drosophila, Homolog Of, 7 (HES7) 17p13.1 is a transcriptional repressor protein with teh structure containing a basic helix-loop-helix-Orange domain. It is a direct target of the Notch signaling pathway and also part of a negative feedback required to attenuate Notch signaling.
- Links: OMIM - Hes7
Pax
Mesoderm Development and Pax[13]
- Links: Developmental Signals - Pax
Mesogenin 1
A master regulator of paraxial presomitic mesoderm differentiation.[6]
Other Species
| Animal Species - Average Somite Pair Number | |
|---|---|
| Species | Somites Number |
| Human | 44 |
| Mouse | 65 |
| Chicken | 55 |
| Lizard (anole) | 72-73 |
| Xenopus | 42 |
| Zebrafish | 32 |
Chicken
| Chicken Somitogenesis | ||
|---|---|---|
| 23-26 hr | 1 somite | |
| ca. 23-26 hr | 1-3 | |
| 26-29 hr | 4 | |
| 29-33 hr | 7 | |
| ca. 33 hr | 8-9 | |
| 33-38 hr | 10 | |
| 40-45 hr | 13 | |
| 45-49 hr | 16 | |
| 48-52 hr | 19 | |
| ca. 50-52 hr | 20-21 | |
| 50-53 hr | 22 | |
| ca. 50-54 hr | 23 | |
| 50-55 hr | 24-27 | |
| 51-56 hr | 26-28 | |
| 52-64 hr | 29-32 | |
| 3 da | 30-36 | |
| 3.0-3.5 da | 37- 40 extending into tail | |
| 3.0-3.5 da | 40-43 | |
| 3.5 da | 43-44 | |
| 3.5-4.0 da | Somites extend to tip of tail | |
| Hamburger Hamilton Stages | Chicken Development | ||
| Chicken Somitogenesis | ||
|---|---|---|
| 23-26 hr | 1 somite | |
| ca. 23-26 hr | 1-3 | |
| 26-29 hr | 4 | |
| 29-33 hr | 7 | |
| ca. 33 hr | 8-9 | |
| 33-38 hr | 10 | |
| 40-45 hr | 13 | |
| 45-49 hr | 16 | |
| 48-52 hr | 19 | |
| ca. 50-52 hr | 20-21 | |
| 50-53 hr | 22 | |
| ca. 50-54 hr | 23 | |
| 50-55 hr | 24-27 | |
| 51-56 hr | 26-28 | |
| 52-64 hr | 29-32 | |
| 3 da | 30-36 | |
| 3.0-3.5 da | 37- 40 extending into tail | |
| 3.0-3.5 da | 40-43 | |
| 3.5 da | 43-44 | |
| 3.5-4.0 da | Somites extend to tip of tail | |
| Hamburger Hamilton Stages | Chicken Development | ||
Mouse
| Mouse Somitogenesis | |||||
|---|---|---|---|---|---|
Number |
Stage |
||||
| 12 | 8 | 1 - 7 | 14-15 | 9 | |
| 13 | 8.5 | (8 - 9.25) | 8 - 12 | 15 | 10 |
| 14 | 9 | (8.5 - 9.75) | 13 - 20 | 16 | 11 |
| 15 | 9.5 | (9 - 10.25) | 21 - 29 | 17 - 19 | 12 |
| 16 | 10 | (9.5 - 10.75) | 30 - 34 | 20 - 21 | 13 - 15 |
| 17 | 10.5 | (10 - 11.25) | 35 - 39 | 24 - 25 | 13 - 15 |
| 18 | 11 | (10.5 - 11.25) | 40 - 44 | 25 - 26 | 13 - 15 |
| 19 | 11.5 | (11 - 12.25) | 45 - 47 | 26 - 27 | 16 |
| 20 | 12 | (11.5 - 13) | 48 - 51 | 28 | 17 |
| 21 | 13 | (12.5-14) | 52 - 55 | 29 - 30 | 18 - 19 |
| 22 | 14 | (13.5-15) | 56 - 60 | 31 | 20 - 23 |
| 23 | 15 | 60 + | 32 | Fetal period | |
| Mouse Somitogenesis | |||||
|---|---|---|---|---|---|
Stages |
Number |
Stage |
|||
| 12 | 8 | 1 - 7 | 14-15 | 9 | |
| 13 | 8.5 | (8 - 9.25) | 8 - 12 | 15 | 10 |
| 14 | 9 | (8.5 - 9.75) | 13 - 20 | 16 | 11 |
| 15 | 9.5 | (9 - 10.25) | 21 - 29 | 17 - 19 | 12 |
| 16 | 10 | (9.5 - 10.75) | 30 - 34 | 20 - 21 | 13 - 15 |
| 17 | 10.5 | (10 - 11.25) | 35 - 39 | 24 - 25 | 13 - 15 |
| 18 | 11 | (10.5 - 11.25) | 40 - 44 | 25 - 26 | 13 - 15 |
| 19 | 11.5 | (11 - 12.25) | 45 - 47 | 26 - 27 | 16 |
| 20 | 12 | (11.5 - 13) | 48 - 51 | 28 | 17 |
| 21 | 13 | (12.5-14) | 52 - 55 | 29 - 30 | 18 - 19 |
| 22 | 14 | (13.5-15) | 56 - 60 | 31 | 20 - 23 |
| 23 | 15 | 60 + | 32 | Fetal period | |
Rat
| Maximum Variation | |||||||
|---|---|---|---|---|---|---|---|
| Age Days-Hrs. |
Number of Litters |
Number of Embryos Counted |
Means | All Embryos | One Litter | Standard Deviation |
Coefficient of Variation % |
| 10-12 | 1 | 4 | 8.7 | 6-11 | 6-11 | 2.8 | 32 |
| 10-13 | 1 | 6 1 | 0. 2 | 8-13 | 8-13 | 1.9 | 19 |
| 10-16 | 2 | 12 | 12.4 | 6-26 | 6-26 | 5.8 | 47 |
| 10-17 | 6 | 48 | 12.9 | 4-26 | 7-26 | 3 .3 | 26 |
| 10-18 | 4 | 23 | 14.1 | 6-18 | 9-18 | 3.1 | 22 |
| 10-19 | 1 | 6 | 14 .3 | 12-17 | 12-17 | 1.2 | 8 |
| 10-22 | 3 | 16 | 13.1 | 6-19 | 6-14 | 3.9 | 30 |
| 10-23 | 3 | 17 | 15.2 | 8-18 | 8-18 | 2.4 | 16 |
| 11-00 | 2 | 0 | 14.6 | 10-17 | 10-17 | 1.9 | 13 |
| 11-02 | 2 | 15 | 20 . 9 | 17-22 | 17-22 | 1. 3 | 6 |
| 11-04 | 1 | 10 | 25. 3 | 23-26 | 23-26 | 1.2 | 5 |
| 11-06 | 1 | 2 | 18.0 | 18-18 | 18-18 | .0 | 0 |
| 11-08 | 1 | 8 | 25 0 | 23-28 | 23-28 | 1.9 | 8 |
| 11-09 | 1 | 3 | 22 .0 | 20-25 | 20-25 | 2 . 2 | 10 |
| 11-10 | 2 | 18 | 26.2 | 21-31 | 21-31 | 2.9 | 11 |
| 11-11 | 3 | 26 | 25.6 | 14-29 | 14-27 | 3.2 | 12 |
| 11-12 | 1 | 3 | 26. 7 | 25-28 | 25-28 | 1.2 | 4 |
| 11-14 | 1 | 3 | 21.3 | 21-22 | 21-22 | .6 | 2 |
| 11-15 | 1 | 8 | 23.1 | 17-28 | 17-28 | 4 . 0 | 17 |
| 11-16 | 1 | 7 | 26.7 | 24-28 | 24-28 | 1.3 | 5 |
| 11-17 | 1 | 7 | 26.1 | 25-28 | 25-28 | 1.1 | 4 |
| 11-18 | 3 | 25 | 25 .4 | 19-28 | 19-28 | 1.9 | 7 |
| 11-20 | 2 | 16 | 27.2 | 25-29 | 25-29 | 1.2 | 4 |
| 23 Ages | 44 | 292 | Av 2.2 | Av 13% | |||
| Data Reference[14] Links: rat | somitogenesis | |||||||
| Table III Showing Means And Variation For Each Age | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reference: Landacre FL. and Amstutz MM. Data on the number of somites compared with age in the white rat. (1929) Ohio J. Science. 29(6): 253-259. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Additional Images
References
- ↑ 1.0 1.1 Fongang B & Kudlicki A. (2013). The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network. BMC Dev. Biol. , 13, 42. PMID: 24304493 DOI.
- ↑ Diaz-Cuadros M, Wagner DE, Budjan C, Hubaud A, Tarazona OA, Donelly S, Michaut A, Al Tanoury Z, Yoshioka-Kobayashi K, Niino Y, Kageyama R, Miyawaki A, Touboul J & Pourquié O. (2020). In vitro characterization of the human segmentation clock. Nature , , . PMID: 31915384 DOI.
- ↑ Yoshioka-Kobayashi K, Matsumiya M, Niino Y, Isomura A, Kori H, Miyawaki A & Kageyama R. (2020). Coupling delay controls synchronized oscillation in the segmentation clock. Nature , , . PMID: 31915376 DOI.
- ↑ Juul JS, Jensen MH & Krishna S. (2019). Constraints on somite formation in developing embryos. J R Soc Interface , 16, 20190451. PMID: 31530134 DOI.
- ↑ Maschner A, Krück S, Draga M, Pröls F & Scaal M. (2016). Developmental dynamics of occipital and cervical somites. J. Anat. , 229, 601-609. PMID: 27380812 DOI.
- ↑ 6.0 6.1 Chalamalasetty RB, Garriock RJ, Dunty WC, Kennedy MW, Jailwala P, Si H & Yamaguchi TP. (2014). Mesogenin 1 is a master regulator of paraxial presomitic mesoderm differentiation. Development , 141, 4285-97. PMID: 25371364 DOI.
- ↑ Tiedemann HB, Schneltzer E, Zeiser S, Hoesel B, Beckers J, Przemeck GK & de Angelis MH. (2012). From dynamic expression patterns to boundary formation in the presomitic mesoderm. PLoS Comput. Biol. , 8, e1002586. PMID: 22761566 DOI.
- ↑ Naiche LA, Holder N & Lewandoski M. (2011). FGF4 and FGF8 comprise the wavefront activity that controls somitogenesis. Proc. Natl. Acad. Sci. U.S.A. , 108, 4018-23. PMID: 21368122 DOI.
- ↑ Aulehla A & Pourquié O. (2006). On periodicity and directionality of somitogenesis. Anat. Embryol. , 211 Suppl 1, 3-8. PMID: 17024300 DOI.
- ↑ Resende TP, Ferreira M, Teillet MA, Tavares AT, Andrade RP & Palmeirim I. (2010). Sonic hedgehog in temporal control of somite formation. Proc. Natl. Acad. Sci. U.S.A. , 107, 12907-12. PMID: 20615943 DOI.
- ↑ Pu Q, Abduelmula A, Masyuk M, Theiss C, Schwandulla D, Hans M, Patel K, Brand-Saberi B & Huang R. (2013). The dermomyotome ventrolateral lip is essential for the hypaxial myotome formation. BMC Dev. Biol. , 13, 37. PMID: 24138189 DOI.
- ↑ Takagi A, Isomura A, Yoshioka-Kobayashi K & Kageyama R. (2019). Dynamic Delta-like1 expression in presomitic mesoderm cells during somite segmentation. Gene Expr. Patterns , 35, 119094. PMID: 31899345 DOI.
- ↑ Blake JA & Ziman MR. (2014). Pax genes: regulators of lineage specification and progenitor cell maintenance. Development , 141, 737-51. PMID: 24496612 DOI.
- ↑ 14.0 14.1 Landacre FL. and Amstutz MM. Data on the number of somites compared with age in the white rat. (1929) Ohio J. Science. 29(6): 253-259.
Reviews
Takahashi Y & Sato Y. (2008). Somitogenesis as a model to study the formation of morphological boundaries and cell epithelialization. Dev. Growth Differ. , 50 Suppl 1, S149-55. PMID: 18482400 DOI.
Turnpenny PD. (2008). Defective somitogenesis and abnormal vertebral segmentation in man. Adv. Exp. Med. Biol. , 638, 164-89. PMID: 21038776
Kusumi K, Sewell W & O'Brien ML. (2008). Mouse mutations disrupting somitogenesis and vertebral patterning. Adv. Exp. Med. Biol. , 638, 140-63. PMID: 21038775
Mara A & Holley SA. (2007). Oscillators and the emergence of tissue organization during zebrafish somitogenesis. Trends Cell Biol. , 17, 593-9. PMID: 17988868 DOI.
Cinquin O. (2007). Understanding the somitogenesis clock: what's missing?. Mech. Dev. , 124, 501-17. PMID: 17643270 DOI.
Shifley ET & Cole SE. (2007). The vertebrate segmentation clock and its role in skeletal birth defects. Birth Defects Res. C Embryo Today , 81, 121-33. PMID: 17600784 DOI.
Aulehla A & Pourquié O. (2006). On periodicity and directionality of somitogenesis. Anat. Embryol. , 211 Suppl 1, 3-8. PMID: 17024300 DOI.
Brent AE. (2005). Somite formation: where left meets right. Curr. Biol. , 15, R468-70. PMID: 15964269 DOI.
Christ B, Huang R & Scaal M. (2004). Formation and differentiation of the avian sclerotome. Anat. Embryol. , 208, 333-50. PMID: 15309628 DOI.
Scaal M & Christ B. (2004). Formation and differentiation of the avian dermomyotome. Anat. Embryol. , 208, 411-24. PMID: 15338303 DOI.
Articles
Chu LF, Mamott D, Ni Z, Bacher R, Liu C, Swanson S, Kendziorski C, Stewart R & Thomson JA. (2019). An In Vitro Human Segmentation Clock Model Derived from Embryonic Stem Cells. Cell Rep , 28, 2247-2255.e5. PMID: 31461642 DOI.
O'Rahilly R & Müller F. (2003). Somites, spinal Ganglia, and centra. Enumeration and interrelationships in staged human embryos, and implications for neural tube defects. Cells Tissues Organs (Print) , 173, 75-92. PMID: 12649586 DOI.
Search PubMed
Search NLM Online Textbooks: "Somitogenesis" : Developmental Biology | The Cell- A molecular Approach | Molecular Biology of the Cell | Endocrinology
Search Pubmed: Somitogenesis | Formation | Sclerotome | Hes7
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology Somitogenesis. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Somitogenesis
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G