Renal System - Molecular: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (16 intermediate revisions by the same user not shown) | |||
| Line 6: | Line 6: | ||
In the embryo, nephron development, '''nephrogenesis''', occurs through several stages involving classical epithelial/mesenchyme type of interactions. Nephrogenesis continues into the late fetal period ({{GA}} week 34–35) and while the fetal kidney does produce urine, not until after birth does the glomerular filtration rate (GFR) increases rapidly due to a postnatal drop in kidney vascular resistance and an increase in renal blood flow. | In the embryo, nephron development, '''nephrogenesis''', occurs through several stages involving classical epithelial/mesenchyme type of interactions. Nephrogenesis continues into the late fetal period ({{GA}} week 34–35) and while the fetal kidney does produce urine, not until after birth does the glomerular filtration rate (GFR) increases rapidly due to a postnatal drop in kidney vascular resistance and an increase in renal blood flow. | ||
{{Renal Links}} | {{Renal Links}} | ||
{{Molecular System Links}} | |||
{{Factor Links}} | {{Factor Links}} | ||
{{Mechanism Links}} | |||
==Some Recent Findings== | ==Some Recent Findings== | ||
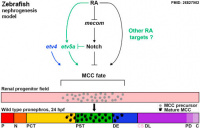
[[File:Zebrafish nephrogenesis signaling01.jpg|thumb|200px|alt=Zebrafish nephrogenesis signaling|Zebrafish nephrogenesis signaling<ref name"PMID26827902"><pubmed>26827902</pubmed></ref>]] | |||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Epithelial cell fate in the nephron tubule is mediated by the ETS transcription factors etv5a and etv4 during zebrafish kidney development'''<ref name"PMID26827902"><pubmed>26827902</pubmed></ref> "Kidney development requires the differentiation and organization of discrete nephron epithelial lineages, yet the genetic and molecular pathways involved in these events remain poorly understood. The embryonic zebrafish kidney, or pronephros, provides a simple and useful model to study nephrogenesis. The pronephros is primarily comprised of two types of epithelial cells: transportive and multiciliated cells (MCCs). Transportive cells occupy distinct tubule segments and are characterized by the expression of various solute transporters, while MCCs function in fluid propulsion and are dispersed in a "salt-and-pepper" fashion within the tubule. Epithelial cell identity is reliant on interplay between the Notch signaling pathway and retinoic acid (RA) signaling, where RA promotes MCC fate by inhibiting Notch activity in renal progenitors, while Notch acts downstream to trigger transportive cell formation and block adoption of an MCC identity. ...abrogation of Notch with the small molecule inhibitor DAPT increased the renal progenitor etv5a expression domain as well as MCC density in etv5a deficient embryos, suggesting Notch acts upstream to inhibit etv5a. In contrast, etv4 levels in renal progenitors were unaffected by changes in RA or Notch signaling levels, suggesting a possible non-cell autonomous role during pronephros formation. Taken together, these findings have revealed new insights about the genetic mechanisms of epithelial cell development during nephrogenesis." [[Developmental_Signals_-_Retinoic_acid|Retinoic acid]] | [[Developmental Signals - Notch|Notch]] | |||
* '''Histone deacetylase 1 and 2 regulate Wnt and p53 pathways in the ureteric bud epithelium'''<ref name'"PMID25758227"><pubmed>25758227</pubmed></ref> "Histone deacetylases (HDACs) regulate a broad range of biological processes through removal of acetyl groups from histones as well as non-histone proteins. Our previous studies showed that Hdac1 and Hdac2 are bound to promoters of key renal developmental regulators and that HDAC activity is required for embryonic kidney gene expression. However, the existence of many HDAC isoforms in embryonic kidneys raises questions concerning the possible specificity or redundancy of their functions. We report here that targeted deletion of both the Hdac1 and Hdac2 genes from the ureteric bud (UB) cell lineage of mice causes bilateral renal hypodysplasia. One copy of either Hdac1 or Hdac2 is sufficient to sustain normal renal development." | * '''Histone deacetylase 1 and 2 regulate Wnt and p53 pathways in the ureteric bud epithelium'''<ref name'"PMID25758227"><pubmed>25758227</pubmed></ref> "Histone deacetylases (HDACs) regulate a broad range of biological processes through removal of acetyl groups from histones as well as non-histone proteins. Our previous studies showed that Hdac1 and Hdac2 are bound to promoters of key renal developmental regulators and that HDAC activity is required for embryonic kidney gene expression. However, the existence of many HDAC isoforms in embryonic kidneys raises questions concerning the possible specificity or redundancy of their functions. We report here that targeted deletion of both the Hdac1 and Hdac2 genes from the ureteric bud (UB) cell lineage of mice causes bilateral renal hypodysplasia. One copy of either Hdac1 or Hdac2 is sufficient to sustain normal renal development." | ||
* '''Bmp7 functions via a polarity mechanism to promote cloacal septation'''<ref name'"PMID22253716"><pubmed>22253716</pubmed></ref> "During normal development in human and other placental mammals, the embryonic cloacal cavity separates along the axial longitudinal plane to give rise to the urethral system, ventrally, and the rectum, dorsally. Defects in cloacal development are very common and present clinically as a rectourethral fistula in about 1 in 5,000 live human births. Yet, the cellular mechanisms of cloacal septation remain poorly understood. ...Our results strongly indicate that Bmp7/JNK signaling regulates remodeling of the cloacal endoderm resulting in a topological separation of the urinary and digestive systems. Our study points to the importance of Bmp and JNK signaling in cloacal development and rectourethral malformations." | * '''Bmp7 functions via a polarity mechanism to promote cloacal septation'''<ref name'"PMID22253716"><pubmed>22253716</pubmed></ref> "During normal development in human and other placental mammals, the embryonic cloacal cavity separates along the axial longitudinal plane to give rise to the urethral system, ventrally, and the rectum, dorsally. Defects in cloacal development are very common and present clinically as a rectourethral fistula in about 1 in 5,000 live human births. Yet, the cellular mechanisms of cloacal septation remain poorly understood. ...Our results strongly indicate that Bmp7/JNK signaling regulates remodeling of the cloacal endoderm resulting in a topological separation of the urinary and digestive systems. Our study points to the importance of Bmp and JNK signaling in cloacal development and rectourethral malformations." [[Developmental_Signals_-_Bone_Morphogenetic_Protein|BMP]] | ||
*''Characterization of Mesonephric Development and Regeneration Using Transgenic Zebrafish.''' <ref><pubmed>20810610</pubmed></ref> "The majority of previous studies have focused on the pronephros of zebrafish, which consists of only two nephrons and is structurally simpler than the mesonephros of adult fish and the metanephros of mammals. To evaluate the zebrafish system for more complex studies of kidney development and regeneration, we investigated the development and post-injury regeneration of the mesonephros in adult zebrafish." (See also [[Zebrafish Development]]) | *''Characterization of Mesonephric Development and Regeneration Using Transgenic Zebrafish.''' <ref><pubmed>20810610</pubmed></ref> "The majority of previous studies have focused on the pronephros of zebrafish, which consists of only two nephrons and is structurally simpler than the mesonephros of adult fish and the metanephros of mammals. To evaluate the zebrafish system for more complex studies of kidney development and regeneration, we investigated the development and post-injury regeneration of the mesonephros in adult zebrafish." (See also [[Zebrafish Development]]) | ||
|} | |} | ||
| Line 32: | Line 32: | ||
<pubmed limit=5>Renal Embryology</pubmed> | <pubmed limit=5>Renal Embryology</pubmed> | ||
|} | |} | ||
== Nephron == | == Nephron == | ||
| Line 81: | Line 48: | ||
{| | {| | ||
|-bgcolor="CEDFF2" | |-bgcolor="CEDFF2" | ||
! Abbreviation | ! Abbreviation<br>([http://omim.org OMIM link]) | ||
! Growth Factor | ! Growth Factor<br>(Factor page link) | ||
! Renal Development | ! Renal Development | ||
! Expression Location | ! Expression Location | ||
|- | |- | ||
| BMP4 | | [http://omim.org/entry/112262 BMP4] | ||
| Bone Morphogenetic Protein 4 | | [[Developmental Signals - Bone Morphogenetic Protein|Bone Morphogenetic Protein 4]] | ||
| prevents ectopic ureteric bud outgrowth and extra ureteric bud divisions | | prevents ectopic ureteric bud outgrowth and extra ureteric bud divisions | ||
| mesenchymal cells surrounding mesonephric duct and stromal mesenchyme surrounding | | mesenchymal cells surrounding mesonephric duct and stromal mesenchyme surrounding uteric bud stalks | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| BMP7 | | [http://omim.org/entry/112267 BMP7] | ||
| Bone Morphogenetic Protein 7 | | [[Developmental Signals - Bone Morphogenetic Protein|Bone Morphogenetic Protein 7]] | ||
| survival of metanephric mesenchyme | | survival of metanephric mesenchyme | ||
| metanephric mesenchyme | | metanephric mesenchyme | ||
|- | |- | ||
| Fgf8 | | [http://omim.org/entry/600483 Fgf8] | ||
| Fibroblast Growth Factor 8 | | [[Developmental Signals - Fibroblast Growth Factor|Fibroblast Growth Factor 8]] | ||
| transition of the induced cap mesenchyme into RVs | | transition of the induced cap mesenchyme into RVs | ||
| cap mesenchyme | | cap mesenchyme | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| GDNF | | [http://omim.org/entry/600837 GDNF] | ||
| Glial-cell derived neurotrophic factor | | Glial-cell derived neurotrophic factor | ||
| induces | | induces uteric bud outgrowth from mesonephric duct, interacts with Ret | ||
| metanephric mesenchyme | | metanephric mesenchyme | ||
|- | |- | ||
| VEGF | | [http://omim.org/entry/192240 VEGF] | ||
| Vascular endothelial growth factor | | [[Developmental Signals - Vascular Endothelial Growth Factor|Vascular endothelial growth factor]] | ||
| promotes endothelial cell proliferation, differentiation | | promotes endothelial cell proliferation, differentiation | ||
| s-shaped body | | s-shaped body | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| Wnt4 | | [http://omim.org/entry/603490 Wnt4] | ||
| Wingless-Type MMTV Integration Site Family, Member 4 | | [[Developmental Signals - Wnt|Wingless-Type MMTV Integration Site Family, Member 4]] | ||
| mesenchymal-to-epithelial transition | | mesenchymal-to-epithelial transition | ||
| cap metanephric mesenchyme, pre-tubular aggregate, nephron progenitors | | cap metanephric mesenchyme, pre-tubular aggregate, nephron progenitors | ||
|- | |- | ||
| Wnt5a | | [http://omim.org/entry/164975 Wnt5a] | ||
| Wingless-Type MMTV Integration Site Family, Member 5a | | [[Developmental Signals - Wnt|Wingless-Type MMTV Integration Site Family, Member 5a]] | ||
| nephrogenesis induction, ectopic bud formation | | nephrogenesis induction, ectopic bud formation | ||
| | | uteric bud, metanephric mesenchyme | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| Wnt9b | | [http://omim.org/entry/602864 Wnt9b] | ||
| Wingless-type MMTV integration site family, Member 9B | | [[Developmental Signals - Wnt|Wingless-type MMTV integration site family, Member 9B]] | ||
| renewal and differentiation of nephron progenitors and normal ureteric bud branching, mesenchymal-to-epithelial transition | | renewal and differentiation of nephron progenitors and normal ureteric bud branching, [[Developmental Mechanism - Mesenchymal Epithelial Transition|mesenchymal-to-epithelial transition]] | ||
| | | uteric bud stalk epithelial cells | ||
|} | |} | ||
| Line 131: | Line 98: | ||
:'''Links:''' [http://omim.org/entry/601091 OMIM Foxd1] | :'''Links:''' [http://omim.org/entry/601091 OMIM Foxd1] | ||
==Intermediate Mesoderm== | |||
==Mesonephric Duct== | |||
==Uteric Bud== | |||
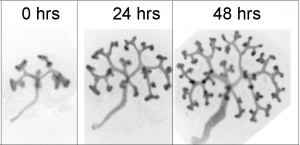
[[File:Mouse-kidney in vitro.jpg|thumb|Mouse E12.5 kidney in vitro]] | |||
* arise near the cloacal connection of the mesonephric duct | |||
* branch from the mesonephric duct laterally into the intermediate mesoderm | |||
* induce the surrounding mesoderm to differentiate - metanephric blastema | |||
** this mesoderm will in turn signal back to differentiate the uteric bud | |||
'''Epithelial - mesenchymal interaction''' | |||
Uteric Bud forms - ureter, pelvis, calyces, collecting ducts | |||
==Nephros Development== | |||
Three pairs appearing in sequence within intermediate mesoderm during development. | |||
# pronephros | |||
# mesonephros | |||
# metanephros | |||
===Pronephros=== | |||
===Mesonephros=== | |||
===Metanephros=== | |||
==Renal Collecting Duct Tree== | |||
A recent in vitro experimental system and a computer model suggests that BMP7 also acts as a repelling signal to establish the branched collecting duct tree.<ref name'"PMID25205115"><pubmed>25205115</pubmed></ref> | |||
:'''Links:''' [[Developmental Signals - Bone Morphogenetic Protein|Bone Morphogenetic Protein]] | |||
== References == | == References == | ||
<references/> | <references/> | ||
| Line 147: | Line 145: | ||
===Reviews=== | ===Reviews=== | ||
<pubmed>25737276</pubmed> | <pubmed>25737276</pubmed> | ||
<pubmed>25608807</pubmed> | |||
<pubmed>20691850</pubmed> | <pubmed>20691850</pubmed> | ||
<pubmed>19906853</pubmed> | <pubmed>19906853</pubmed> | ||
| Line 154: | Line 154: | ||
<pubmed>18184729</pubmed> | <pubmed>18184729</pubmed> | ||
<pubmed>17442697</pubmed> | <pubmed>17442697</pubmed> | ||
<pubmed>9152004</pubmed> | |||
[http://www.nature.com/ng/meetings/nephrogenetics/index.html Forefronts Symposium on Nephrogenetics: from development to physiology March 8-11, 2007 Danvers, MA] A meeting to synthesize an integrated view of the normal development and function of the kidney from the genetic standpoint. | [http://www.nature.com/ng/meetings/nephrogenetics/index.html Forefronts Symposium on Nephrogenetics: from development to physiology March 8-11, 2007 Danvers, MA] A meeting to synthesize an integrated view of the normal development and function of the kidney from the genetic standpoint. | ||
| Line 159: | Line 161: | ||
===Articles=== | ===Articles=== | ||
<pubmed></pubmed> | |||
<pubmed></pubmed> | |||
<pubmed>25205115</pubmed> | |||
<pubmed>18846389</pubmed> | <pubmed>18846389</pubmed> | ||
<pubmed>12783789</pubmed> | |||
<pubmed>9690097</pubmed> | |||
===Search PubMed=== | ===Search PubMed=== | ||
'''Search Pubmed:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=renal+system+development Renal System Development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=renal+development Renal Development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=intermediate%20mesoderm intermediate mesoderm] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=kidney%20development kidney development] | [http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=renal_development renal development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=ureteric%20bud ureteric bud] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=nephron%20development nephron development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=bladder%20development bladder development] | '''Search Pubmed:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=renal+system+development Renal System Development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=renal+development Renal Development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=intermediate%20mesoderm intermediate mesoderm] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=kidney%20development kidney development] | [http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=renal_development renal development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=ureteric%20bud ureteric bud] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=nephron%20development nephron development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=bladder%20development bladder development] | ||
| Line 173: | Line 174: | ||
<gallery> | <gallery> | ||
</gallery> | </gallery> | ||
== Terms == | == Terms == | ||
{{Renal terms}} | {{Renal terms}} | ||
==External Links== | ==External Links== | ||
Latest revision as of 10:51, 31 August 2016
| Embryology - 26 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The paired adult kidneys consist of a functional unit called the "nephron", that filters blood, excretes waste, reabsorbs water (and other compounds) and has endocrine functions. Each adult human kidney typically contains about 750,000 nephrons, though the total number can vary significantly from as few as 250,000 to as many as 2,000,000.[1][2]
In the embryo, nephron development, nephrogenesis, occurs through several stages involving classical epithelial/mesenchyme type of interactions. Nephrogenesis continues into the late fetal period (GA week 34–35) and while the fetal kidney does produce urine, not until after birth does the glomerular filtration rate (GFR) increases rapidly due to a postnatal drop in kidney vascular resistance and an increase in renal blood flow.
- Molecular System Links: Heart | Neural | Renal | Respiratory | Mechanisms | Factors | Molecular
| Factor Links: AMH | hCG | BMP | sonic hedgehog | bHLH | HOX | FGF | FOX | Hippo | LIM | Nanog | NGF | Nodal | Notch | PAX | retinoic acid | SIX | Slit2/Robo1 | SOX | TBX | TGF-beta | VEGF | WNT | Category:Molecular |
| Mechanism Links: mitosis | cell migration | cell junctions |epithelial invagination | epithelial mesenchymal transition | mesenchymal epithelial transition | epithelial mesenchymal interaction | morphodynamics | tube formation | apoptosis | autophagy | axes formation | time | molecular |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Renal Embryology <pubmed limit=5>Renal Embryology</pubmed> |
Nephron
In humans, nephrogenesis only occurs before birth, though nephron maturation continues postnatally. Mean glomerular number shown to level at 36 weeks, increasing from about 15,000 at 15 weeks to 740,000 at 40 weeks.
Nephron development has four identifiable developmental stages:
|
|
| Abbreviation (OMIM link) |
Growth Factor (Factor page link) |
Renal Development | Expression Location |
|---|---|---|---|
| BMP4 | Bone Morphogenetic Protein 4 | prevents ectopic ureteric bud outgrowth and extra ureteric bud divisions | mesenchymal cells surrounding mesonephric duct and stromal mesenchyme surrounding uteric bud stalks |
| BMP7 | Bone Morphogenetic Protein 7 | survival of metanephric mesenchyme | metanephric mesenchyme |
| Fgf8 | Fibroblast Growth Factor 8 | transition of the induced cap mesenchyme into RVs | cap mesenchyme |
| GDNF | Glial-cell derived neurotrophic factor | induces uteric bud outgrowth from mesonephric duct, interacts with Ret | metanephric mesenchyme |
| VEGF | Vascular endothelial growth factor | promotes endothelial cell proliferation, differentiation | s-shaped body |
| Wnt4 | Wingless-Type MMTV Integration Site Family, Member 4 | mesenchymal-to-epithelial transition | cap metanephric mesenchyme, pre-tubular aggregate, nephron progenitors |
| Wnt5a | Wingless-Type MMTV Integration Site Family, Member 5a | nephrogenesis induction, ectopic bud formation | uteric bud, metanephric mesenchyme |
| Wnt9b | Wingless-type MMTV integration site family, Member 9B | renewal and differentiation of nephron progenitors and normal ureteric bud branching, mesenchymal-to-epithelial transition | uteric bud stalk epithelial cells |
- Foxd1 - (Brain Factor-2) transcription factor that is a renal stroma specific gene.
- Links: OMIM Foxd1
Intermediate Mesoderm
Mesonephric Duct
Uteric Bud
- arise near the cloacal connection of the mesonephric duct
- branch from the mesonephric duct laterally into the intermediate mesoderm
- induce the surrounding mesoderm to differentiate - metanephric blastema
- this mesoderm will in turn signal back to differentiate the uteric bud
Epithelial - mesenchymal interaction
Uteric Bud forms - ureter, pelvis, calyces, collecting ducts
Nephros Development
Three pairs appearing in sequence within intermediate mesoderm during development.
- pronephros
- mesonephros
- metanephros
Pronephros
Mesonephros
Metanephros
Renal Collecting Duct Tree
A recent in vitro experimental system and a computer model suggests that BMP7 also acts as a repelling signal to establish the branched collecting duct tree.[8]
- Links: Bone Morphogenetic Protein
References
Textbooks
- The Developing Human: Clinically Oriented Embryology (8th Edition) by Keith L. Moore and T.V.N Persaud - Moore & Persaud Chapter 13 p303-346
- Larsen’s Human Embryology by GC. Schoenwolf, SB. Bleyl, PR. Brauer and PH. Francis-West - Chapter 10 p261-306
- Before We Are Born (5th ed.) Moore and Persaud Chapter14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
Online Textbooks
Search Bookshelf intermediate mesoderm | kidney development | renal development | ureteric bud | nephron development | bladder development
- Developmental Biology by Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000 Chapter 14 Intermediate Mesoderm | Figure 14.18. General scheme of development in the vertebrate kidney | Figure 23-23. Mechanism of mesenchymal inductive effect on the ureteric bud | Figure 14.21. Ureteric bud growth is dependent on GDNF and its receptor
- Molecular Cell Biology by Lodish, Harvey; Berk, Arnold; Zipursky, S. Lawrence; Matsudaira, Paul; Baltimore, David; Darnell, James E. New York: W. H. Freeman & Co.; c1999 Reciprocal Epithelial-Mesenchymal Interactions Regulate Kidney Development | Figure 23-21. Embryonic development of the kidney
Reviews
<pubmed>25737276</pubmed> <pubmed>25608807</pubmed> <pubmed>20691850</pubmed> <pubmed>19906853</pubmed> <pubmed>19828308</pubmed> <pubmed>19615554</pubmed> <pubmed>18184729</pubmed> <pubmed>17442697</pubmed> <pubmed>9152004</pubmed>
Forefronts Symposium on Nephrogenetics: from development to physiology March 8-11, 2007 Danvers, MA A meeting to synthesize an integrated view of the normal development and function of the kidney from the genetic standpoint.
<pubmed>16916378</pubmed>
Articles
<pubmed></pubmed> <pubmed></pubmed> <pubmed>25205115</pubmed> <pubmed>18846389</pubmed> <pubmed>12783789</pubmed> <pubmed>9690097</pubmed>
Search PubMed
Search Pubmed: Renal System Development | Renal Development | intermediate mesoderm | kidney development | renal development | ureteric bud | nephron development | bladder development
Additional Images
Terms
| Renal Terms | ||
|---|---|---|
| ||
|
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Australia - Network for Genes & Environment in Development Professor John F. Bertram
- GenitoUrinary Development Molecular Anatomy Project (GUDMAP) Renal Development Tutorial | Genital Development Tutorial
- Urine Metabolome database is a freely available electronic database containing detailed information about ~3100 small molecule metabolites found in human urine along with ~3900 concentration values.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 26) Embryology Renal System - Molecular. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Renal_System_-_Molecular
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G