Paper - The development of the cloaca in human embryos
| Embryology - 18 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Pohlman AG. The development of the cloaca in human embryos. (1911) Amer. J Anat. 12: 1-26.
| Online Editor | ||||
|---|---|---|---|---|
| A number of Carnegie Embryos and models generated from these embryos are described in this historic study of human cloaca development. Note that Polman numbers the embryos examined in sequence 1-30, but also gives Mall's Carnegie embryo numbers in the tabulation.
See also by this author - Pohlman AG. Has a persistence of the Müllerian ducts any relation to the conditions of cryptorchidism? (1904) Amer. Med. 8: 1003-1006. Carnegie Embryo 186 cloaca - (cloacal cavity) The term describing the common cavity into which the intestinal, genital, and urinary tracts open in vertebrates. Located at the caudal end of the embryo it is located on the surface by the cloacal membrane. In many species this common cavity is later divided into a ventral urogenital region (urogenital sinus) and a dorsal gastrointestinal (rectal) region. cloacal membrane - Forms the external lower membrane limit (caudal end) of the early gastrointestinal tract (GIT). This membrane is formed during gastrulation by ectoderm and endoderm without a middle (intervening) layer of mesoderm. The membrane breaks down to form the initial "anal opening" of the gastrointestinal tract. The upper end of the gastrointestinal tract has a similar embryonic membrane, the buccopharyngeal membrane.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Development of the Cloaca in Human Embryos
Indiana University
From the Anatomical Laboratory, Johns Hopkins University
Introduction
The active work in the embryology of the urogenital system began about 1880 and continued for a period of some fifteen years when Keibel's monograph was published. The writer's investigation of the cloacal region was started in 1903, and was undertaken in part as a control of Keibel's work, and in part with a view toward solving some of the points on which differences in opinion existed. The publication of this report has been delayed in the hope that certain facts in comparative embryology might be established and help to clarify some of the obscure relations found in the human embryo. Inasmuch as the extensive investigations of Fleischmann and his students have come to naught in this respect, the major differences in opinion will be considered, and the doubtful points answered in so far as it is possible. The short literature review covers the essential facts and effort has been made to reduce the description of the material to a concise tabulation. The writer expresses his indebtedness to Prof. Keibel at whose suggestion the development of the later stages in the embryology was undertaken, and to Prof. F. P. Mall for the use of his collection of embryos and for the many courtesies shown him in the Anatomical Laboratory of Johns Hopkins University.
Born's article ('93) reviews the earlier development of the cloacal region in a very complete manner, and the substance is as follows: The entoderm of the enteron comes into direct relation with the surface ectoderm in the pharyngeal and cloacal membranes during the formation of the head and tail folds. Both of these membranes lose their primitive position and become folded into the substance of the embryo through increase in the surrounding mesoderm. The allantois, which is developed dorsally in the mammalian embryo (human and guinea pig excepted), shifts to a ventral position on the gut, and is gradually displaced from its intimate relation to the yolk sac through increase in the amount of mesodermal tissue. The primitive streak is carried to the ventral surface of the body during the formation of the tail fold, and forms the whole or part of the cloacal membrane. Kolliker f'83), Strahl ('83, '84), and Bonnet ('88) believe that the caudal end of the primitive streak is made up of applied layers of ectoand entoderm, and that it enters as such into the formation of the cloacal membrane. Keibel ('88) argues that this primitive relation of the ecto- and entoderm is lost through interposition of mesoderm; the latter disappearing later with restoration of the original two layered condition.
The model of the 4.2 mm. human embryo presented by Keibel ('88) shows the hind gut and widely lumened allantois opening cephalward into the caudal entodermal sac or cloaca. Ventrally, this cloaca is limited as far as the dermal navel by the epithelial cloacal membrane. Caudalward, the limit of the cloacal membrane comes about by a mesodermic separation of the epithelial layers. The gut segment distal to the lower limit of the cloacal membrane may be termed the tail gut and terminates in an undifferentiated cell mass formed by itself, the chorda and neural tube. Born emphasizes the length of the cloacal membrane as follows: "I call particular attention to the original extent of the cloacal membrane. It reaches cephalward to the point where the allantois leaves the body at right angles; i.e., as far as the caudal border of the dermal navel."
The cloacal membrane does not extend as far as the allantois in the model of an 8.0 mm. human embryo presented by Keibel ('91) . Mesodermic tissue has apparently wandered in from above and separated the layers of epithelium. The cloacal membrane is therefore shorter than in the 4.2 mm. stage. The tail gut shows degeneration. The precloacal mesodermic tissue has increased in amount but instead of displacing the cloacal membrane caudal-ward, has folded it into the lower surface of the genital tubercle as was first described by Tourneux ('89), and verified by Retterer ('90) and Reichel ('93). No intermediate stage in the development has been described up to this time ('93). With the increase in size of the genital tubercle, the entodermal cloaca becomes more deeply placed as was demonstrated by Reichel ('93). Born states that the epithelial plate contained within the genital tubercle is ectodermic and that it is continuous with the superficial ectoderm. The epithelial plate occupies the caudal surface of the eminence and is bordered laterally by folds of mesoderm (repli ano-genitaux of Retterer) , while the cloacal membrane itself terminates at the postanal fold (replis postanal)., "The depression arises (as in the mouth region) through increase in the height of the limiting borders. The depression is always closed in by epithelium, and the base of the depression is never separated from the entodermal cloaca by mesodermic tissue." (Born.)
The cloaca is gradually divided into a ventral (bladder-urogenital sinus) segment, and a dorsal (gut) segment. As to the manner of this division, Retterer and Born agree with the Tiedemann-Rathke idea of the gradual separation into two segments through approximation of two lateral folds of mesoderm, while Tourneux believes it to be accomplished by a septal (frontal) downgrowth. Lieberkiihn ('82) and Keibel ('89) dispute the theory of Rathkc ('32) championed bj' von Mihlacovics ('85), that the bladder arises from the allantois, and state that it is made up for the most part from the ventral cloacal segment - agreed to by Retterer and Reichel. Born takes a neutral position and believes that at least the trigone of the human bladder may be developed in a manner like that found in the guinea pig (Keibel). Born and Minot do not think that the anlage of the upper part of the bladder is of particular importance. "We are probably not mistaken when we grant that not only the bladder (as far as the apex) but the male urethra as far as the caput gallinaginis, the entire female urethra, and in the male, also the pars prostatica and the entire pars membranacea are developed from the ventral cloacal segment."
The most important of the recent works is that of Ke bel ('96) who sums up the development of the cloacal region in the following words : "The human embryo possesses a large entodermal cloaca in the early stages of its development which however is never opened to the outside through a cloacal anus ('Cloakenafter' of Prenant), but remains closed through the cloacal membrane (the 'anal membrane' of the earlier writers) as long as it exists as such. An ectodermal cloaca is to be found only in traces if at all. The entodermal cloaca is separated into a ventral and a dorsal segment by a frontal septum. A large part or all of the bladder, the urethra and the urogenital sinus as far as the cloacal membrane are derived from the ventral segment; while the dorsal segment becomes continuous with the ectodermal segment of the rectal canal. The primitive perineum is formed when the frontal septum fuses with the cloacal membrane and the rudimentary ectodermal cloaca is then divided by the permanent perineum. The ectodermal anal pit (protodaeum) is situated behind the permanent perineum, while the ectodermal portion of the urogenital sinus is ventral to it."
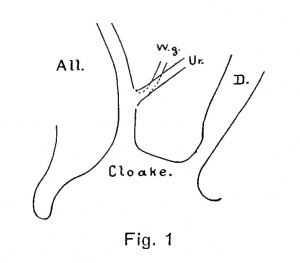
Another investigator, who has been particularly fortunate in the amount of embryological material at his disposal, expresses a somewhat different view. Nagel ('02) practically reiterates his statements of 1894. "The inspection of the tail end of human embryos of 11-13 mm. length reveals an oval pit extending from the coccygeal prominence to the tip of the genital eminence. This pit (cloaca) receives the openings of the gut dorsally, and the Canalis urogenitalis ventrally; the two separated by a partition of some 0.3 mm. thickness. The Wolffian and Mullerian ducts open higher up in the Canalis urogenitalis and will not be considered in the description of this depression. The Canalis urogenitalis and the gut open into this pit (cloaca) which would reach (comparing with adult relations) from the dorsal border of the anus to the ventral border of the urethral opening {i.e. Frenulum clitoridis). Later he states: "In what manner the division of the cloaca is accomplished is not perfectly understood either in man or in mammals. I found the relations in the youngest human embryo that I had opportunity to examine like those pictured in fig, 1, naturally with exception of the form of the bladder. I commit myself therefore, as far as the human embryo is concerned, to the view of Rathke which has recently been substantiated by Retterer and von Mihalcovics in the animals."
The work of von Mihalcovics ('85) referred to, says in part; "This septum between the urogenital canal and the gut arises in part through increase in length of the afore-mentioned perineal Told (septum) to which, distalward, two lateral folds join to the perineal folds. The cloaca takes no part in the formation of the urogenital canal." The opinion of Retterer ('94), mentioned by Nagel, is summarized as follows: "In the guinea pig, as in other mammals studied up to the present time (man, pig, sheep and rabbit), a fold of mesoderm appears at the cephalic extremity of each lateral cloacal wall, and extends little by little toward the caudal end of the cloaca. These lateral folds encroach upon the lumen of the cloaca and divide it into two canals, the one dorsal or rectal, and the other ventral or vesico-urogenital."
That these opinions, while they are in the main contrary to the views of Keibel although they appeared at an earlier date, are accepted at the present time can be illustrated by an extract from Zuckerakndls Handbuch der Urologie ('03): "In the second fetal month, the proximal segment of the allantois widens to form the bladder, while the distal and narrow portion (urachus) obliterates to form the Lig. vesico-umbilicale," "The division of the cloaca is accomplished by three folds, a median and two lateral. The former occupies the angle between the allantois and hind gut, while the latter are developed in the lateral walls of the cloaca."
The investigations of Tourneux ('94) agree with those of Keibel in that he also states the cloaca to be a closed sac. His objections to the method of cloacal division as described by Retterer are as follows: "The form of the inferior border of the recto-urogenital septum is that of a vaulted arch and not that of an elliptical arch with the vertex upward - " a fact easity demonstrated in frontal sections. In addition to this, the transverse sections show that the lateral folds are found only toward the summit of the arch and converge rapidly. Further, the septum shows no signs of an epithelial raphe at the supposed line of fusion to indicate the transition that one encounters, as Keibel states, "at the line of union of the palatine ridges."
Nagel ('96) answers Keibel's article by stating If Keibel did not find the facts as presented by myself in all of his embryos of 11.0 mm. and upwards, then the embryos are at fault." "Furthermore in order that an embryo may be declared of scientific value, I demand that the urogenital canal be open into the cloacal pit in all embryos over 8.0 mm., and that the cloacal membrane have disappeared as far as the tip of the genital eminence. Inasmuch as th& allantois contained within the umbilical cord is practically obliterated at this stage, where could the secretion from the mesonephros be stored up if the cloacal membrane were intact?"
The questions that appeal to the writer as doubtful ones may be expressed as follows:
- Does the cloaca proper ever open to the outside?
- Does the cloacal membrane arise directly by displacement of applied layers of ecto- and entoderm in the primitive streak or is it formed in situ through disappearance of the intervening mesoderm?
- In what manner is the cloacal division effected?
- What is the anlage for the bladder?
- Can the urinary function of the mesonephros assumed by Nagel be demonstrated?
Material
The study of the cloacal region was done in part by working out the relations in serial sections, and in part through reconstruction. Thirty embryos in all were examined; reconstruction employed in thirteen, of which six stages will be presented. The material, with the exception of these six , is given entirely in the tabulation. The serial number, used throughout this paper, refers to the age of the embryo based on the development of the urogenital tract and with no particular reference to its length. It is interesting to note that with the exception of nos. 4, 11, 13, and 21, the development of the tract has proved to be an excellent check on the determination of the age by the greatest length method. The Mall number refers to the catalogue number of the collection of human embryos at Johns Hopkins University and the section thickness (indicated in microns) ; the section direction (+ for transverse, = for sagittal, and || for coronal; and the condition (f for fair and g for good) are recorded in this manner in the Mall catalogue. The modelling of the embryo is indicated by the magnification of the same, and where the model is presented in this paper it is designated by a capital letter. The brief normentafel shows the major points of difference in the development of the cloacal region. The embryo 30 is the Piper II embryo of the collection, and will be presented later in connection with the origin of the bulbovestibular glands. This embryo and also no. 20 have been reported in connection with the condition of complete double ureter (Johns Hopkins Bull., vol. 16, Feb. '04).
Tabulation
- Online editor - Carnegie stages have been added to the original tabulation and the text information is now edited in a sortable table.
| Embryo | Mall | Stage | CRL | Plane | Thickness (microns) | Quality | Notes |
|---|---|---|---|---|---|---|---|
| 1 | 12 | 11 | 2.1 mm | transverse | 10 | good | Modelled by Mall ('97). Allantois of small lumen and in relation with the caudal border of the yolk sac. No evidences of a cloacal membrane. |
| 2 | 391 | 10 | 2.0 mm | transverse | 10 | Good | Appearance of hind gut. An interval of several sections between allantois and yolk sac. No cloacal membrane. |
| 3 | 164 | 11 | 3.5 mm | transverse | 20 | Good | Model A. x 100. Cloaca well developed and limited for caudal half of the ventral border by epithelial cloacal membrane. Allantois comes off at right angles and at lower border of dermal navel. Lateral furrow along later line of division. |
| 4 | 209 | 12 | 3.0 mm | transverse | 50 | Fair | Relations about the same as the fore-going stage. Sections too thick for detailed study. Carnegie stage 12 |
| 5 | 186 | 13 | 3.5 mm | transverse | 20 | Good | Model B. x 100. Allantois wide lumened and no distinct line of demarkation from the ventral cloacal segment. Wolflian ducts have arrived but not as yet opened. Tail gut maximum of development. Probably the first signs of cloacal division. |
| 6 | 98 | 4.0 mm | transverse | 20 | Fair | About the same as the foregoing. 13 | |
| 7 | 148 | 13 | 4.3 mm | transverse | 10 | Good | x 100 Wolffian ducts open into cloaca. Allantois of small lumen. Ventral cloacal segment widening. Evidences of division quite apparent. |
| 8 | 76 | 13 | 4.5 mm | transverse | 20 | Good | x 100. Widened ventral cloacal segment goes over indistinctly into allantois. Tail gut still quite large. Renal buds well developed. With exception of large allantois and wider tail gut, quite similar to Model C. |
| 9 | 80 | 14 | 5.0 mm | transverse | 10 | Good | Model C. x 100. About the same as the foregoing stage. Tail gut undergoing degenerative changes. |
| 10 | 371 | 15 | 6.6 mm | sagittal | 10 | Good | Tail gut rudimentary. Development of cloaca and renal buds about the same as in no. 12. |
| 11 | 116 | 13 | 5.0 mm | sagittal | 20 | Good | x 100. Intracloacal epithelial plug well marked. Stage about the same as that found in no. 12. |
| 12 | 2 | 15 | 7.0 mm | transverse | 15 | Good | Model D x 100. Marked development of renal buds. Kidney and ureter segments evident. Ventral cloacal segment much widened. Allantois of narrow lumen. Intracloacal epithelial plug marked. Tail gut maximum in length and slightly widened at caudal end. |
| 13 | 241 | 15 | 6.0mm | transverse | 10 | Goo(l | Tailgutmore rudimentary than in no. 12. Cloacal membrane wide and shows intracloacal epithelial plug. Otherwise about the same as in no. 12. |
| 14 | 383 | 16 | 7.0 mm | transverse | 10 | Good | State similar to no. 12. Cloacal membrane intact. |
| 15 | 397 | 16 | 8.0 nma. | transverse | 10 | Good | Similar to foregoing. |
| 16 | 113 | 15 | 8.0 mm | sagittal | 10 | Fair | x 100. Stage of cloacal division between than found in no. 12 and that found in no. 19. Cloacal membrane intact. Division of ureteral pelvis. Ureter segment elongated. |
| 17 | 114 | 10.0 mm | sagittal | 10 | Fair | x 100. Stage of cloacal division about the same as the foregoing. Cloacal membrane intact. Kidney a little higher up. Upper and lower pelvis evident. | |
| 18 | 109 | 18 | 11.0 mm | transverse | 20 | Good | Sections directly through lower part of cloacal region somewhat damaged. Otherwise about the same as no. 19. |
| 19 | 221 | 16 | 12.0 mm | sagittal | 20 | Good | Model E x 100. All traces of tail gut lost. Cloacal membrane somewhat depressed from surface. Cloacal segment of Wolffian duct shortened and opening of ureter and duct common into ventral cloacal segment. Beginning formation of genital eminence and lengthening of the ventral cloacal wall. Cloacal membrane intact. |
| 20 | 175 | 13.0 mm | transverse | 20 | Good | x 100. Cloaca about divided. Complete double ureter. Orifices of lateral, and medial ureter and Wolffian duct at same level into cloaca. | |
| 21 | 353 | 17 | 11.0 mm | transverse | 10 | Good | Cloaca completely divided. Anal and urogenital membranes intact. Ureteral orifice independent and on level with Wolffian opening. |
| 22 | 317 | 18 | 15.0 mm | coronal | 20 | Good | About the same stage as in no. 21. |
| 23 | 350 | 19 | 15.0 mm | coronal | 10 | Good | About the same as the foregoing, and no. 24. |
| 24 | 43 | 19 | 16.0mm | sagittal | 50 | Good | Model F . x 50. Anal and urogenital membranes separated. Genital eminence more marked. Ectodermic inclusion in furrow on caudal surface. Ureteral orifice on same level but some distance lateral to Wolffian orifice. |
| 25 | 256 | 20 | 16.0 mm | sagittal | 50 | Good | Urogenital sinus open to outside through rupture of urogenital membrane. Kidney nearly up to normal position and rotated. |
| 26 | 296 | 18 | 7.0mm | coronal | 20 | Good | Slightly older than above. Appearance of secondary genital folds at the sides of the phallus. |
| 27 | 128 | 21 | 20.0 mm | coronal | 50 | Good | About same as above. |
| 28 | 240 | 20 | 20.0 mm | coronal | 20 | Good | |
| 29 | 22 | 21 | 20.0 mm | transverse | 50 | Good | One Mullerian duct has reached the urogenital sinus. |
| 30 | Embryo KP | 22.0 mm | transverse | 15 | Good | X 66. Both Mullerian ducts at sinus. |
The last six embryos of this series have been introduced merely to carry out the idea of a brief arrangement according to the development of the tract in so far as it was possible and to indicate the position of Model of no. 30 referred to in reference to the measurements on the inter-ureteral and inter- Wolffian widths mentioned in the discussion of the anlage for the trigonum. Embryo no. 30 has additional interest in that it like no. 20 has a complete duplication of the ureter on one side.
The description of the models has been made in as brief a manner as feasible, and a discussion of the various points in question will be brought up after they have been described. All of these models represent epithelial casts, and all of them with the exception of Model F. are X 100.
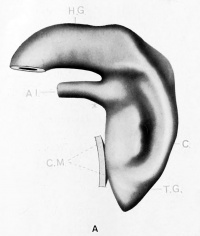
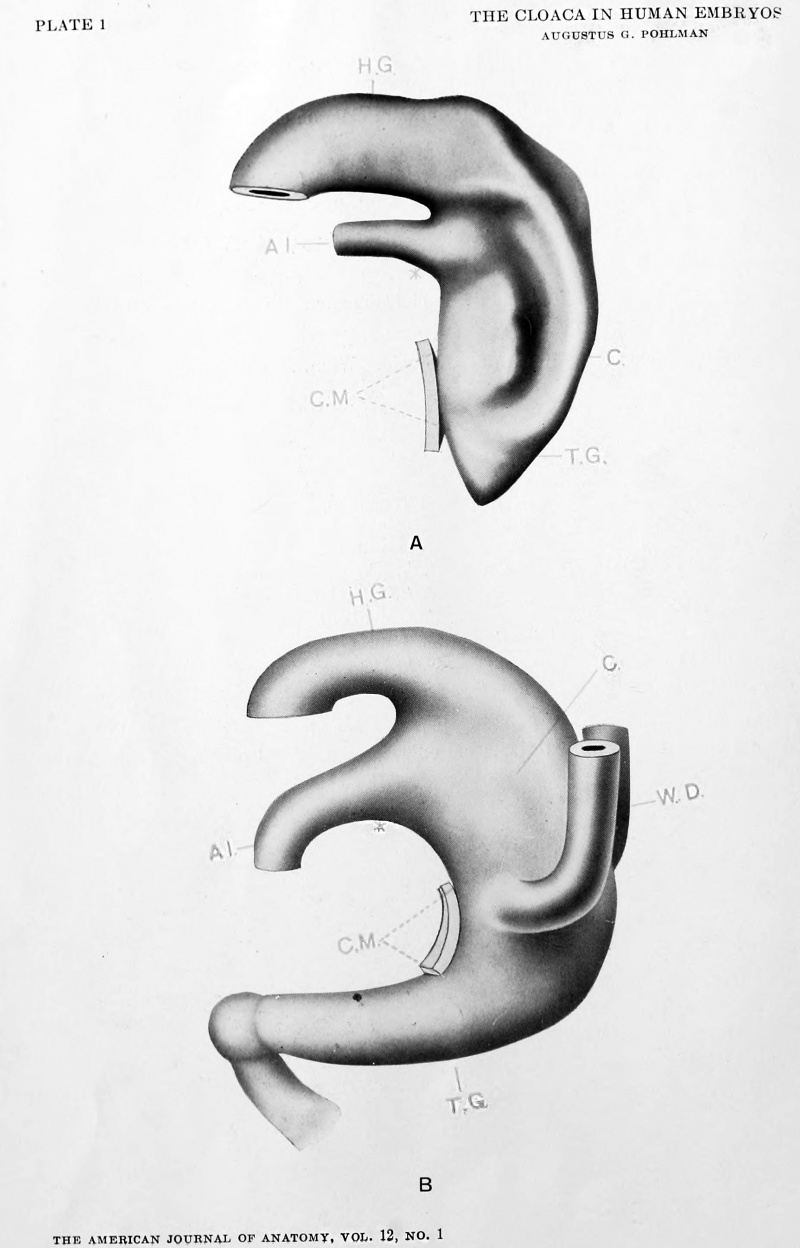
| Model A. {No. 3; Mall no. 164; 3.5 mm.) This model shows the cloaca to be a sort of conical sac receiving the wider lumened hind gut dorsally, and the narrow lumened allantois ventrally. The latter joining the cloaca at about a right angle. The cloaca is much wider dorsally and is somewhat wedged-shaped in section. Ventrally, it is limited for about one-half of its extent by the epithelial cloacal membrane which is indicated by a surface contact. The right wall of the cloaca is concaved from before backwards while the left side (shown) is correspondingly convex, and presents a well marked furrow extending from the saddle between the allantois and hind gut to about the mid area of the cloacal membrane. This furrow is interesting because while it indicates the probable line of the later division, it was found in but two embryos of the series examined. The part of the cloaca caudal to the cloacal membrane - the tail gut - is quite short and cone-shaped. The model with the exception of the furrow conforms to Keibel's model of the His embryo EB (3 mm.) |
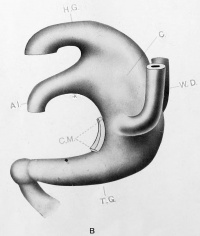
| Model B {No. 5; Mall no. 186; 3.5 mm.) This model, although made from an embryo of the same length as the above, shows a distinctly older stage in the development of the tract. The allantois has a much wider lumen and the angle between it and the hind gut is much more acute. The Wolffian ducts have reached the cloaca, and attach but do not open into it ventrally near the upper limit of the cloacal membrane. The tail gut is much larger both in length and in thickness, and terminates in an undifferentiated cell mass formed by itself, the chorda and the neural tube (shown as a knob-like ending). The cloaca is wider dorsally than ventrally. A stage similar to the one described was found in two other embryos (nos. 7 and 8), in both of which the allantois was wide at its apparent opening into the cloaca. The possibility of some minor degree of development abnormality suggested itself but on careful study, it was decided that the first evidences of cloacal division were at hand.
|
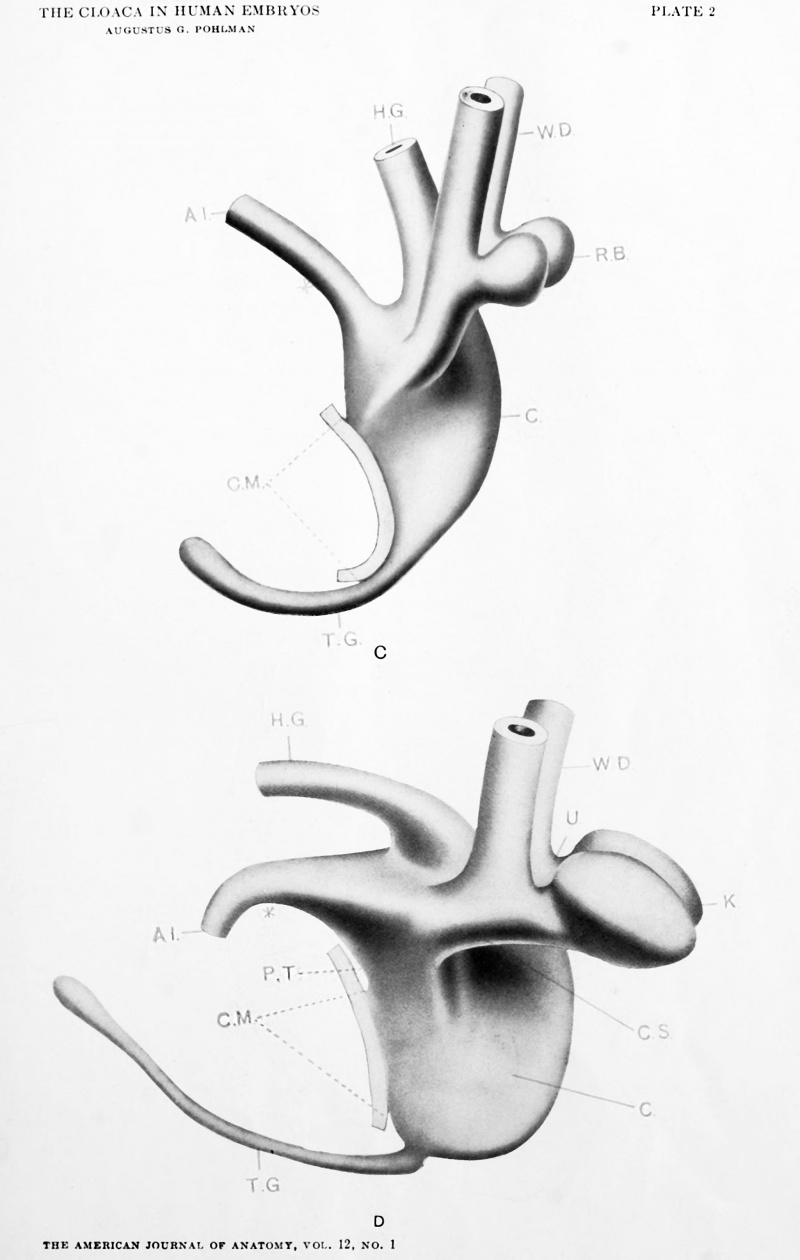
| Model C. {No. 9; Mall no. 80; 5.0 mm..) This model shows marked degeneration of the tail gut. The allantois is narrow lumened, while the widely lumened ventral cloacal segment lies ventral to the hind gut and dorsal segment, and is separated from them by peritoneum. The Wolffian ducts open in the same situation as in the foregoing model, and present dorsal diverticulae, the renal buds. The ventral portion of the cloaca, especially the area above the cloacal membrane, is markedly widened and flattened. The lateral furrow of Model A was not present. This model conforms to the Keibel model of a 6.5 mm. embryo (a considerably older stage if we go by the greatest length rule). The cloaca has lost its more or less even width and attains its greatest lateral diameter at the level of the Wolffian orifices. There is no evidence of displacement of the Wolffian ducts from their primitive position in relation with the upper limit of the cloacal membrane. |
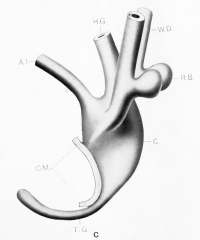
| Model D {No. 12; Mall no. 2; 7.0 mm.) This model fills in a gap in the Keibel series which may be considered quite an important one. The hind gut and all that pertains to the dorsal segment of the cloaca has retained an even caliber, while the ventral segment has widened progressively. The division of the cloaca may be traced to the level of the Wolffian orifices. The Wolffian ducts are much better developed and the renal anlage has resolved itself into distinct ureter and kidney segments. The segment of Wolffian duct between the orifice of the ureter and the cloaca is relatively shorter. The renal anlagen have assumed the position of dorsal convergence (mentioned by Keibel) but this is only a relative matter (see later). The tail gut has undergone further degeneration and has lost its lumen in part. |
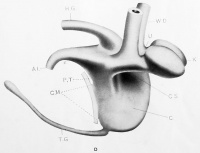
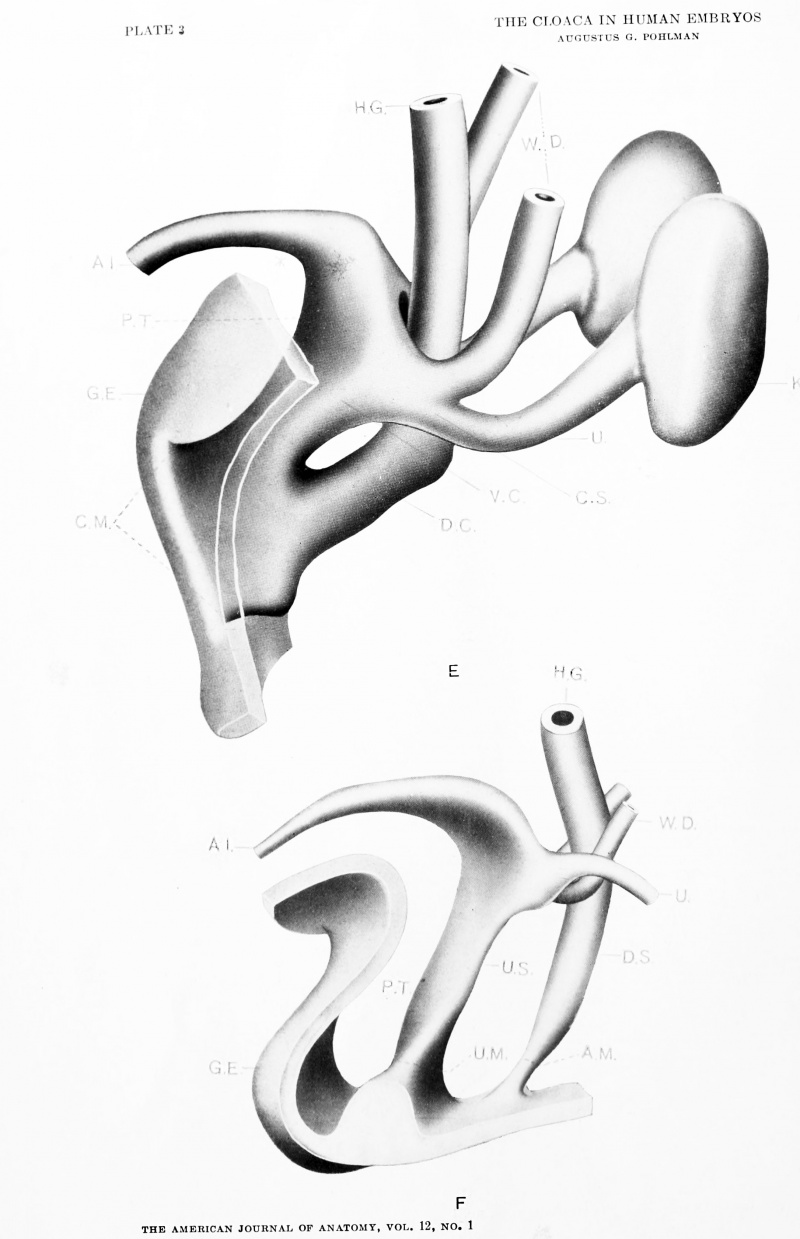
| Model E. {No. 19; Mall no. 221; 12.0 mm.) This stage shows the cloaca about half divided along the furrow line indicated in Model A. The cloacal membrane has thickened but mostly through addition of ectodermal cells, and has been displaced through development of the ' precloacal mesoderm' from its primitive position parallel to the dorsal line of the cloaca to one more nearly at right angles to it. In so far as it was possible to ascertain, this downward displacement of the cloacal membrane in no way affects its caudal limit. The dorsal segment has retained the original proportions while the ventral segment has widened - most marked again at the level of the Wolffian orifices. The further development of the kidney and ureter will be noted and the gradual approach of the ureteral orifice to the cloaca proper is evident. The ureter shows signs of shifting from its primitive dorsal position on the Wolffian duct to a more lateral one. The peritoneum has descended to the level of the ducts while the cloacal division is relatively far advanced. The model agrees with Keibel's model of an 11.5 mm. embryo. |
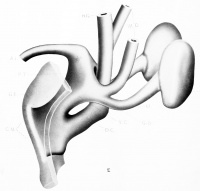
| Model F. {No. 24; Mall no. 43; 16.0 mm. X 50.) Unfortunately this embryo was cut too thick for minute reconstruction. The stage however fills in another gap in the Keibel series. Here the cloaca and the cloacal membrane are completely divided - the ventral segment limited by the urogenital plate and the dorsal segment by the anal plate. The ureter is displaced from its dorso-lateral position on the Wolffian duct to a supero-lateral one and opens distinctly into the ventral cloacal segment. The marked increase in precloacal tissue has resulted not only in the large genital eminence, but the urogenital plate has been dislocated deeper into the base of the phallus and a marked heaping up of ectodermal cells has occurred in the furrow on the caudal surface of the eminence. The two resultant segments of the cloacal membrane, the urogenital and anal plates are apparently no longer than they were in much younger stages; a point that will be brought out in greater detail later. |
The Cloacal Membrane
While authorities seem to agree that the cloacal membrane arises from the primitive streak, or at least that portion which has been displaced ventrally in the formation of the tailfold, they disagree on the point whether this part of the primitive streak consists in applied layers of ecto- and entoderm, or whether it is composed of all three germ layers. Stated in an simple manner, is the cloacal membrane formed dorsally as an epithelial membrane or is it formed ventrally through disappearance of intervening mesodermal tissue? One of the earliest stages in the formation of the tail fold is found in the Spee reconstruction ('96) of embryo Gle (2.0 mm.). A schematic sagittal section is presented in his fig. 1, and emphasis is laid on the point that the primitive streak is composed of all three germ layers. No. 1 of the present tabulation was reconstructed by Mall ('97), and while a slightly older stage in the development, shows the cloacal sac limited ventrally by all three germ layers. No. 2 presents a similar condition. The first embryo of this series to show a cloacal membrane is no. 3 (3.5 mm.) which is quite a little farther advanced in the development than no. 2; unfortunately the transition stages are wanting. It is the writer's opinion therefore that the view held by Keibel is the correct one, and that while the cloacal membrane is derived from the primitive streak area, it is formed in situ as an epithelial membrane on the ventral cloacal surface, naturally after the formation of the tail fold.
The sagittal width of the cloacal membrane varies somewhat with the stage in the development. Born's statement that it extends as far cephalward as the dermal navel is based on Keibel's model of a 4.2 mm. embryo ('88). A somewhat similar condition is noted in two of the embryos of this series. The writer does not believe this to be the normal relation, but rather one which through persistence may have a decided bearing on bladder exstrophy and epispadias. The epithelial plate, if it extends to the dermal navel, and persists in this relation, separates the precloacal tissue into two lateral halves, and accounts quite satisfactorily for the deficience in the abdominal wall and for the gutter on the upper surface of the phallus. In practically all of the embrj'os exam- ined, the cloacal membrane did not extend higher than the level of the Wolffian orifices. The membrane is normally shortest in length at the time of its formation but attains its greatest length in Model C. (0.29 mm.); in Model D (0.27 mm.); in Model E (0.34 mm.). This increase is not marked when we consider that these stages represent 5, 7 and 12 mm. embryos respectively.
The width of the cloacal membrane is a rather indefinite quantity to measure ; it widens progressively however with the widening of the ventral cloacal segment, and in the proportion of 5 in B to 8 in C to 12 in D or in other words in proportion to the greatest length measurement of the embryo.
The position of the cloacal membrane is about parallel to the long axis of the embryo in Models A, B and C. With the development of the precloacal tissue, the cloacal membrane is displaced so that its upper end is pushed caudalward — beginning in D; more marked in E ; and culminating in F where the upper end of the membrane has been displaced through practically 90° with the caudal end as the fixed point of the radius. The displacement is probably due to the active lengthening of the ventral cloacal wall and to the development of the genital eminence. The membrane is divided into two parts in Model F — a ventral or urogenital membrane and a dorsal or anal membrane; both of which have been displaced from the surface in the manner suggested by Born. The anal membrane persists beyond the stages described in this paper, and is dealt with in sufficient detail in Keibels' article. The urogenital membrane ruptures with its formation and is of particular interest in its relation to the position of the bulbovestibular glands ; a point to be brought in a paper which is to appear shortly. It may be noted that in Model D the cloacal membrane is much thicker in its midarea, and not infrequently one finds a heaping up of cells in this region and a corresponding eminence on the inner surface of the cloacal (intracloacal epithelial plug) . The significance of this is not understood but it may be due to the more active proliferation of the ectodermal cells which contribute to the formation of the cloacal membrane.
The Ectodermal Cloaca
An ectodermal cloaca has been stated by Keibel to be present only in traces, if at all. None of the embryos examined showed a surface depression, and we may therefore critically examine Nagel's fig. 1 and his contention concerning the cloacal membrane. It will be seen that in fig. 1, while the embryo shows a cloacal division comparable to our Model E; the phallus is developed to the extent of that found in Model F ; while the cloacal membrane is ruptured. None of the embryos of this stage, or younger than this stage, showed rupture of the cloacal membrane, and we may therefore agree with Keibel that the cloacal membrane remains intact normally as long as it persists as such. Nagel believes that the rupture of the membrane is essential or where would the secretion of the mesonephros be passed? We may answer this question. The ventral cloacal segment and the allantois do not show the degree of widening necessary to accommodate the urinary excretion, and we call to mind Hill's work on the pig embryo ('04) and our own ('04) on the human embrj'o, in both of which it is conclusively shown that the mesonephros is actively degenerate long before the kidney is developmentally fitted to take up a urinary function. Cases of atresia urethrae commonly occur without bladder distension ; monstrosities may go to term without kidneys and with no undue persistence of the mesonephros; and experimentally, it has been shown that there is no evidence of urinary excretion into the amniotic fluid even in term children. We do not believe it necessary to assume that the mesonephros functionates as an organ of excretion in the placental mammals, and do not consider Nagel's contention for a functional rupture of the cloacal membrane as a serious one.
The Tail Gut
The tail gut is that portion of the enteron that lies caudal to the lower limit of the cloacal membrane and before the formation of the cloacal membrane has no definite line of demarkation from the cloaca proper. The tail gut is represented by a conical sac in Model A and attains its largest proportions in Model B or at about the time that the Wolffian ducts reach the cloaca. Its relation to the neural tube and to the chorda is most marked at this stage in the development but the histogenesis of this region (shown in ]\Iodel B as a terminal knob) together with its possible connection with the ano-coccygeal body is not definitely known. The degenerative changes have already set in in Model C and are more apparent in Model D. The shrinkage in size does not affect the tail gut evenly throughout its length, but is less marked at the caudal extremity where a cord of cells may be found even after all traces of the primitive connection with the cloaca have disappeared. In general, the tail gut arises with the tail and accompanies the tail in its development and resorbtion. It appears at a 3.5 mm. stage and has almost completely disappeared in an 8.0 mm. embryo. The comparative embryology of this rudimentary and transient segment of the gut deserves careful study.
The Cloaca
Morphologically, we may hardly consider that portion of the gut caudal to the orifices of the allantois and hind gut as cloaca until the arrival of the Wolffian ducts contribute a connection with. the urogenital system, and practically we may not consider the sac as such until the formation of the cloacal membrane indicates how much is cloaca and how much is tail gut. The condition in the development which antedates these structures might be termed the precloacal stage.
Born states that the allantois arises ventrally in the human embryo and in close relation to the yolk sac, and that the allantois is displaced caudalward through growth in the intervening mesoderm. We may agree with the first part of this statement from the relations demonstrated in the Spee embryo and in Mall's reconstruction of no. 1. The last point, the downward displacement of the allantois, may be seriously questioned. In the formation of the hind gut, either the allantois is displaced caudalward, or the yolk sac is pushed upward on the gut. A comparison of embryos 1 and 3 favors the latter view. The development of the hind gut does not seem to take place at the expense of the cloaca, but the allantois appears to occupy a relatively fixed position at the lower border of the dermal navel. This argument is strengthened by the active increase in length of the hind gut, and by such conditions of strictly pathological nature such as bladder exstrophy. The line of demarkation however between what is allantois and what is ventral cloacal segment is largely a matter of topography, but is after all of some importance in a definite determination of the anlage for the urinary bladder.
The Division Of The Cloaca
The division of the cloaca manifests itself in a separation into ventral and a dorsal segment. According to Tourneux, the division is accomplished through the downgrowth of a single mesodermic fold (the saddle between the allantois and hindgut), while Retterer maintains that two lateral mesodermic folds encroach upon the lumen of the cloaca and pinch it off, as it were, into the two resulting segments. Minot ('97) and Keibel hold the manner of division to be of little moment, and even Fleischmann ('07) appears to have given up hope of solving the question from the standpoint of comparative embryology. Tourneux's idea is, according to the writer, a much better description of the process than that of Retterer.
Taking the Wolffian ducts as a fixed point, it is easily seen that a coronal septum grows down dorsal to the Wolffian orifices until finally the entoderm covering the septum comes into relation with the entoderm of the cloacal membrane. With the disappearance of the epithelium in this fused area, the mesoderm of the setpum touches the ectoderm of the surface and the cloacal membrane is split into its two resulting segments. While this division of the cloaca results in two segments that are about of equal size as far as their antero-posterior measurements are concerned, and inasmuch as the cloacal membrane does not materially increase in length, it may be assumed that the cloacal membrane is split into about equal parts in its transformation into the urogenital and anal membranes. This would mean that the increase in the apparent length of the urogenital membrane is due to proliferation of the ectodermal cells or to inclusion of the surface ectoderm as Born states it. The difference in opinion on this point will be brought out in a later article.
The development of the ventral cloacal segment interests us because it has to do with the formation of the bladder. Detailed measurements of this region have been suppressed because of the variable size of the cloaca proper, but the union of the allantois to the cloaca seems to be fairly definite in Models C, D and E. In all of these embryos the Wolffian ducts open a little above the center of a line drawn from the caudal extremity of the cloacal membrane to the apparent line of union of allantois with cloaca. We have therefore no material reason for not believing, inasmuch as this is the primitive relation of the Wolffian orifices and there is no evidence of downward displacement of the allantois, that the gradually constricting area above the Wolffian orifices is ventral cloacal segment (see Model B).
The active widening of the ventral cloacal segment, particularly at the level of the Wolffian orifices, is evident to every observer of the cross sections in this region, and in the dorsal convergence of the ureters — a fact first noted by Lieberkuhn and Keibel. It is evident also, as suggested by them, that the distal segment of the Wolffian ducts may become incorporated into the anlage for the bladder. This alone does not however account for the lateral rotation of the ureter to gain the position noted in Model F, nor does it explain the later upper displacement of the ureter to its normal position in the bladder.
It is seen in our models that the renal buds appear on the dorsal aspect of the Wolffian ducts at some distance from the cloaca, and the intervening segment of duct may be termed the ' cloacal segment' merely for convenience in the description. It is well known that this gradual shifting of the ureter is not due alone to the resorption of the cloacal segment of the Wolffian duct, for when two ureters arise from the same duct, the one with the orifice nearest the cloaca assumes the more lateral position. In this region we have to deal with the relations as they appear from their topography for it is not possible as yet to differentiate mesodermal epithelium from that of entodermal origin.
The widening of the ventral cloacal segment is made evident from the following measurements. A comparison of the inter-Wolfiian width and the width of the dorsal segment gives a proportion of 20 : 9 in B; 28 : 8 in C ; 36 : 9 in D ; and 46 : 9 in E. This means that the ventral cloacal segment increases markedly in width while the dorsal segment remains about the same. Observed in transverse sections, this active widening gives the impression of two lateral mesodermic folds encroaching upon the cloacal lumen (Retterer's idea.) The widening of the ventral cloacal segment is most marked at the level of the Wolffian orifices and progresses equally both upward and downward giving the idea coronal section of the cloaca in Model E a diamond shape.
In conclusion we may sum up the points that have been investigated in this paper in a review of the development of the cloaca and of the structures in relation with it. The article has been written largely as a check on the earlier stages in the development in order that the writer might have first hand information regarding this important segment of the enteron.
Summary
The cloaca is a closed entodermal sac and is never normally opened to the outside (amniotic cavity) through rupture of the cloacal membrane. It represents that portion of the enteron which lies caudal to the opening of the allantois and may not be separated from the tail gut until the formation of the cloacal membrane, when the inferior border of the membrane forms an arbitrary limit between the cloaca and the tail gut. The cloacal membrane arises from the primitive streak area. The primitive streak at the time of its ventral displacement in the formation of the tail fold, is composed of three germ layers, and the cloacal membrane is formed in situ through disappearance of the intervening mesoderm. Normally, the cloacal membrane occupies the caudal half of the ventral cloacal border, but in some cases, the membrane may extend as far cephalward as the dermal navel.. This latter condition has an important bearing on the embryology of bladder exstrophy and epispadias. The increase in width and thickness of the cloacal membrane is concomitant with the corresponding changes in the ventral segment of the cloaca, and is due to ectodermic proliferation. The membrane is displaced in the process of cloacal division through development of the precloacal mesodermic tissue to form the genital eminence, and at the same time, the original membrane comes to lie deeper in the substance of the embryo. The division of the cloaca begins before the arrival of the Wolffian ducts, and is effected by the downgrowth of a coronal septum. The division, when completed, results in a ventral, (or bladder-urogenital sinus,) segment and a dorsal, (or rectal,) segment which are about of equal size as far as their antero-posterior measurements are concerned. The same holds true of the resultants of the division of the cloacal membrane - the urogenital and anal membranes ; both of which are about of the same length and both of which are displaced from the surface through upgrowth of the surrounding mesoderm. The ventral cloacal segment enlarges progressively in length and in width during the division of the cloaca, and as the widening is most marked at the orifices of the Wolffian ducts, the cloacal segment of the ducts may contribute to this active enlargement, or in other words, some of the epithelium of the bladder may be of mesodermic origin. The displacement of the ureter on the Wolffian duct, and the later upward displacement of its orifice from that of the duct, is due in part to the disappearance of the cloacal segment of the duct. The orifices of both ureter and Wolffian duct are however shifted medianward after the completion of the cloacal division, so that the trigonum may be spoken of as distinctly of entodermic origin. The greater part of the bladder and urachus are developed from the ventral segment including the mesodermic contributions from the Wolffian ducts. The allantois probably contributes no part in the formation of the bladder and retains its original relation to the lower border of the dermal navel. The urogenital membrane ruptures normally when the division of the cloaca is completed, or in other words, with its formation, and before the arrival of the Mullerian ducts at the urogenital sinus. The contribution of the entoderm and ectoderm in the formation of the urethra, together with the developmental relations of the bulbovestibular glands, will be considered in a later paper.
Born has stated that the trigonum may be of mesodermic origin. We have measured Model F and compared it wdth Model 30 and find the following: that the inter-ureteral distance (between the orifices) is greater in F (0.55 mm.) than in no. 30 (0.34) ; while the Wolffian ducts in the former are almost twice as far apart as in the latter. This implies that the ureter is not displaced lateral-ward from the duct but that both are displaced medianward, and that if there is any part of the bladder to which the mesoderm has definitely not contributed it is the trigonum.
We are of the opinion that the ventral segment of the cloaca probably forms the anlage for the bladder including the urachus, as was stated in Born's article, and that the cloacal segment of the Wolffian ducts (mesodermic) contributes some part. How much is impossible to determine. When the cloacal membrane extends as far upward as the dermal navel and persists as an epithelial membrane, we have the origin of the cases of complete bladder exstrophy which take in the whole ventral wall of the bladder and urachus. The present champion of the allantoigen origin of the bladder is not supported by our investigations. Rather we agree with Disse ('02) who states that Nagel misunderstood the relations found in the earlier stages through examination of embryos too far advanced in the development.
Bibliography
Bonnet, K. 1888 Ucber die Entwickelung der Allantois und die Bildung des Afters bei den Wiederkiiuem. Anat. Anz.
Born, G. 1893 Die Entwickelung der Ableitungswege, etc. Ergebnisse der Anat. und Entwickelungsgesch.
DissE, J. 1902 Harnorgane. In von Bardeleben's Handbuch der Anatomie.
Hill, E. C. 1904 On the first appearance of the renal artery, etc. Johns Hopkins Bull., vol 16, February.
Fleischmann, 1907 Die Stilcharactere am Urodaeum und Phallus. Morph-Jahrbuch, vol. 36.
Keibel, F. 1888 Die Entwickelungsvorgange am hinteren Ende des Meersch" weinchenembryo. Arch. f. Anat. und Entw.
1891 Zur Entwickelungsgeschichte der Harnblase. Anat. Anz.
1896 Zur Entwickelungsgeschichte desmensch. Urogenitalapparates. Arch. f. Anat. und Phys. Anat. Abt.
Kolliker, A. Von 1898 Entwickelungsgeschichte des Menschen.
Lieberkuhn, N. 1882 Qucrschnittc von der Anlage der AUantois und der Harnblase, etc. Marburger Sitzungsberichte.
Mall FP. Human embryos. (1893) Ref. Handb. Med. Sci. Supplement, 9: 268-269.
Mall FP. Hosp. Bull., XIV, 1903. Catalogue of the collection of human embryos in the Anatomical Laboratory of the Johns Hopkins University. Johns Hopkins Hopkins University. Baltimore, 1904.
Mall FP. Development of the human coelom. (1897) J Morphol. 12: 395-453.
Mihaicovics, G. von 1885 Untersuchungen uber die Entw. des Harn- und Geschlechtsapparates der Amnioten. Internat. Monatschrift. f. Anat. u. Hist.
Minot C. S. 1894 Human Embryology.
Nagel, W. 1894 Ueber die Entw. der inneren u. ausseren Genitalien beim mensch lichen Weibe. Arch. f. Gynilkologie.
1896 Zur Entwickelungsgeschichte des Urogenitalsystems beim Menschen. Arch. f. Anat. u. Phys. Anat. Abt.
Pohlman AG. Has a persistence of the Müllerian ducts any relation to the conditions of cryptorchidism? (1904) Amer. Med. 8: 1003-1006.
Ratuke, 1832 Abhandlungen zur Bildungs- u. Entwickelungsgeschichte des Menschen u. der Thiere. Leipzig.
Reichel, P 1893 Die Entwickelung der Harnblase und Harnrohre. Verhand. d. mod. -phys. Gcsell. zu Wiirzburg.
Retterer, E. 1890 Sur I'origin et de revolution de la region ano-genitale des mammifreres. Jr. de I'anat. et de la phys.
1893-4 Mode de cloisonnement du cloaque chez le cobaye. Bibliog. Anat.
Spee, Graf 1896 Neue Beobachtungen uber sehr friihe Entwickelungsstufen des menschlichen Eies. Arch. f. Anat. u. Phys. Anat. Abt.
Strahl 1886 Zur Bildung der Cloake des Kaninchens. Arch. f. Anat. u. Phys. Anat. Abt.
Tourneux, F. 1893-4 Sur le mode de cloisonnement du cloaque, etc. Bibliog. Anat.
Zuckenkandl 1903 Handbuch der Urologie. Wien, Austria.
Plates
Key to Lettering
A., Allantois K., Kidney
A.M., Position of anal membrane P.T., Precloacal mesodermic tissue
C, Cloaca R.B., Renal bud
CM., Cloacal membrane T.G., Tail cut
C.S., Cloacal segment of Wolffian duct U., Ureter
G.E., Genital eminence U.G., Urogenital sinus
H.G., Hind gut U.M., Position of urogenital membrane
- Probable position of point where allantois joins cloaca.
All drawings represent 100 diameters enlargement except of Model F which is 50 diameters.
Plate 1
Plate 2
Plate 3
Cite this page: Hill, M.A. (2026, April 18) Embryology Paper - The development of the cloaca in human embryos. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_development_of_the_cloaca_in_human_embryos
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G