Somitogenesis: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 20: | Line 20: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Developmental dynamics of occipital and cervical somites'''<ref name=PMID27380812><pubmed>27380812</pubmed></ref> "Development of somites leading to somite compartments, sclerotome, dermomyotome and myotome, has been intensely investigated. Most knowledge on somite development, including the commonly used somite maturation stages, is based on data from somites at thoracic and lumbar levels. Potential regional differences in somite maturation dynamics have been indicated by a number of studies, but have not yet been comprehensively examined. Here, we present an overview on the developmental dynamics of somites at occipital and cervical levels in the chicken embryo. We show that in these regions, the onset of sclerotomal and myotomal compartment formation is later than at thoracolumbar levels, and is initiated simultaneously in multiple somites, which is in contrast to the serial cranial- to- caudal progression of somite maturation in the trunk." | |||
* '''Mesogenin 1 is a master regulator of paraxial presomitic mesoderm differentiation'''<ref name=PMID25371364><pubmed>25371364</pubmed></ref> "Neuromesodermal (NM) stem cells generate neural and paraxial presomitic mesoderm (PSM) cells, which are the respective progenitors of the spinal cord and musculoskeleton of the trunk and tail. The Wnt-regulated basic helix-loop-helix (bHLH) transcription factor mesogenin 1 (Msgn1) has been implicated as a cooperative regulator working in concert with T-box genes to control PSM formation in zebrafish, although the mechanism is unknown. We show here that, in mice, Msgn1 alone controls PSM differentiation by directly activating the transcriptional programs that define PSM identity, epithelial-mesenchymal transition, motility and segmentation. Forced expression of Msgn1 in NM stem cells in vivo reduced the contribution of their progeny to the neural tube, and dramatically expanded the unsegmented mesenchymal PSM while blocking somitogenesis and notochord differentiation. Expression of Msgn1 was sufficient to partially rescue PSM differentiation in Wnt3a(-/-) embryos, demonstrating that Msgn1 functions downstream of Wnt3a as the master regulator of PSM differentiation." | * '''Mesogenin 1 is a master regulator of paraxial presomitic mesoderm differentiation'''<ref name=PMID25371364><pubmed>25371364</pubmed></ref> "Neuromesodermal (NM) stem cells generate neural and paraxial presomitic mesoderm (PSM) cells, which are the respective progenitors of the spinal cord and musculoskeleton of the trunk and tail. The Wnt-regulated basic helix-loop-helix (bHLH) transcription factor mesogenin 1 (Msgn1) has been implicated as a cooperative regulator working in concert with T-box genes to control PSM formation in zebrafish, although the mechanism is unknown. We show here that, in mice, Msgn1 alone controls PSM differentiation by directly activating the transcriptional programs that define PSM identity, epithelial-mesenchymal transition, motility and segmentation. Forced expression of Msgn1 in NM stem cells in vivo reduced the contribution of their progeny to the neural tube, and dramatically expanded the unsegmented mesenchymal PSM while blocking somitogenesis and notochord differentiation. Expression of Msgn1 was sufficient to partially rescue PSM differentiation in Wnt3a(-/-) embryos, demonstrating that Msgn1 functions downstream of Wnt3a as the master regulator of PSM differentiation." | ||
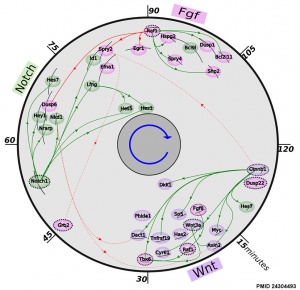
* '''The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network'''<ref name=PMID24304493><pubmed>24304493</pubmed>| [http://www.biomedcentral.com/1471-213X/13/42 BMC Dev Biol.]</ref> "In vertebrate development, the segmental pattern of the body axis is established as somites, masses of mesoderm distributed along the two sides of the neural tube, are formed sequentially in the anterior-posterior axis. This mechanism depends on waves of gene expression associated with the Notch, Fgf and Wnt pathways." | * '''The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network'''<ref name=PMID24304493><pubmed>24304493</pubmed>| [http://www.biomedcentral.com/1471-213X/13/42 BMC Dev Biol.]</ref> "In vertebrate development, the segmental pattern of the body axis is established as somites, masses of mesoderm distributed along the two sides of the neural tube, are formed sequentially in the anterior-posterior axis. This mechanism depends on waves of gene expression associated with the Notch, Fgf and Wnt pathways." | ||
Revision as of 12:10, 15 August 2016
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
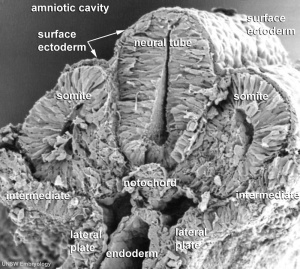
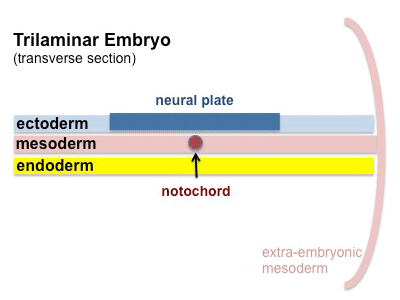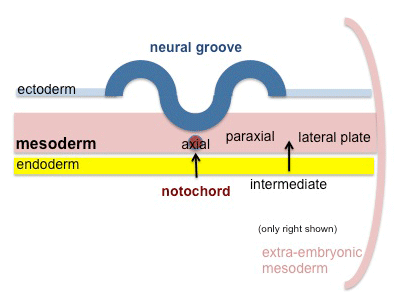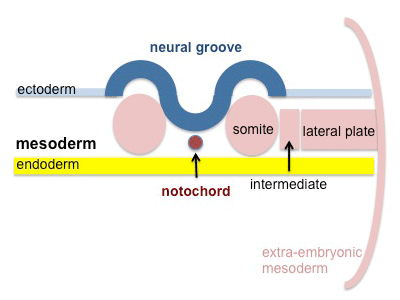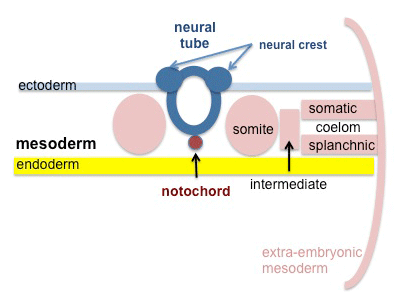
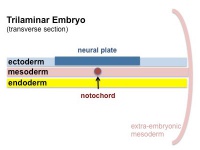
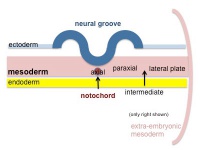
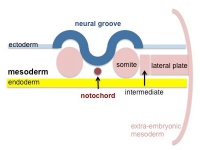
The term used to describe the process of segmentation of the paraxial mesoderm within the trilaminar embryo body to form pairs of somites, or balls of mesoderm. In humans, the first somite pair appears at day 20 and adds caudally at 1 somite pair/90 minutes until on average 44 pairs eventually form.
A somite is added either side of the notochord (axial mesoderm) to form a somite pair. The segmentation does not occur in the head region, and begins cranially (head end) and extends caudally (tailward) adding a somite pair at regular time intervals. The process is sequential and therefore used to stage the age of many different species embryos based upon the number visible somite pairs.
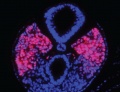
A mesenchymal to epithelial transition defines the outer cellular "shell" of the developing somite, with the core cells remain as a mesenchymal organisation. During early somite development a transient fluid-filled space, the somitocoel, can be identified in each somite and is later lost by cell proliferation. Neural crest cells also enter and mix with the somatic cells.
Somites give rise to many different connective tissues including: cartilage, bone, muscle and tendon.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Somitogenesis <pubmed limit=5>Somitogenesis</pubmed> |
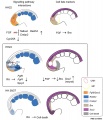
Presomitic Mesoderm
|
|
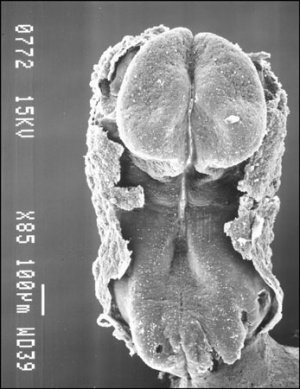
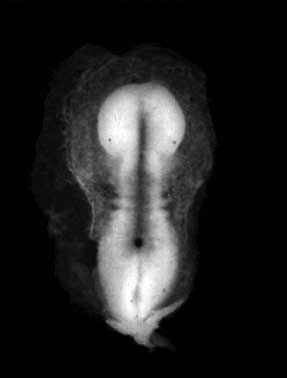
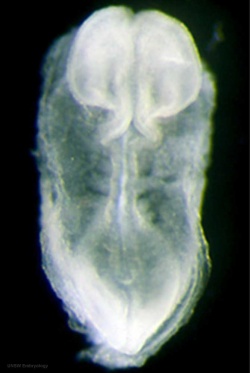
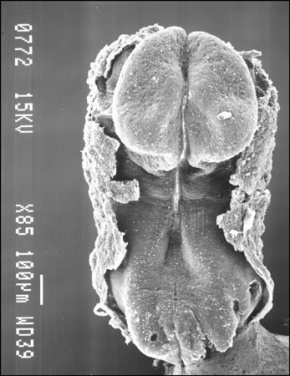
Human First Somites
|
|
|
Human embryo first somite pairs (week 4, Carnegie stage 9)
Somite Number
Week 4 to 5 (GA 6 to 7) covers the main period of human somitogenesis.
| Week | Days | Carnegie Stage | Somite Number (pairs) |
|---|---|---|---|
| Week 3 | 19 - 21 | 9 image | 1 - 3 |
| Week 4 | 22 - 23 | 10 image | 4 - 12 |
| Week 4 | 23 - 26 | 11 image | 13 - 20 |
| Week 4 | 26 - 30 | 12 image | 21 - 29 |
| Week 5 | 28 - 32 | 13 image | 30 |
| Week 5 | 31 - 35 | 14 image | 30+ |
Species Somite Number
| Species | Somites Number |
|---|---|
| Mouse | 65 |
| Chicken | 55 |
| Lizard (anole) | 72-73 |
| Xenopus | 42 |
| Zebrafish | 32 |
Mesoderm to Somite
Mesoderm means the "middle layer" and it is from this layer that nearly all the bodies connective tissues are derived. In early mesoderm development a number of transient structures will form and then be lost as tissue structure is patterned and organised. Humans are vertebrates, with a "backbone", and the first mesoderm structure we will see form after the notochord will be somites.
- During segmentation the outer cell layer forms an epithelial layer over a still mesenchymal organization of cells at the core.
- The early forming somite has a cavity at its core called a "somitocoel" that later fills with proliferating mesoderm cells.
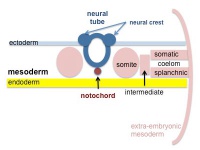
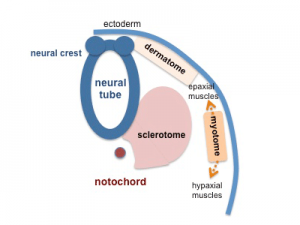
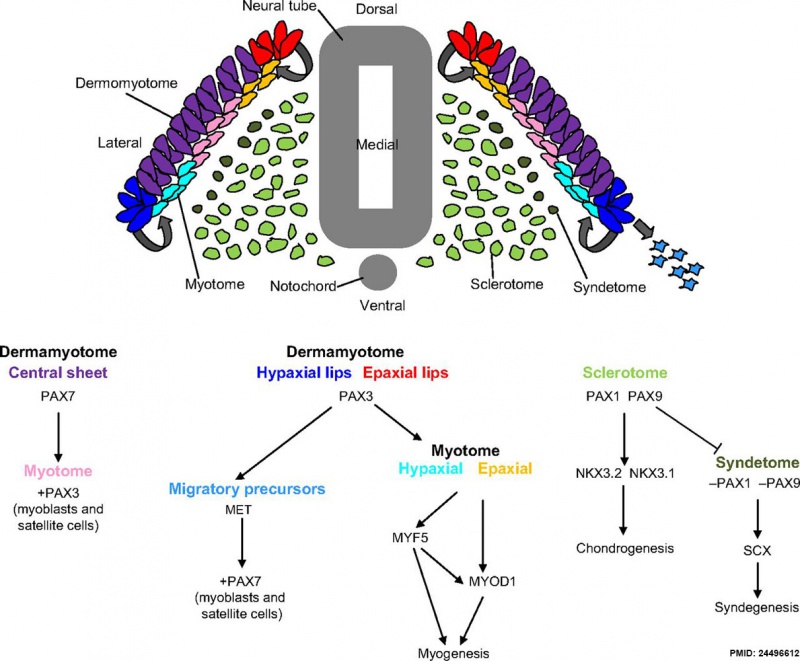
Somite to Sclerotome and Dermomyotome
Somite initially forms 2 main regional components
- ventromedial region - sclerotome forms vertebral body and intervertebral disc
- dorsolateral region - dermomyotome forms dermis and skeletal muscle
Sclerotome
- The left and right sclerotomes from the same segmental level engulf the notochord.
- Each segmental level is then resegmented in a rostrocaudal direction.
Dermomyotome
- The dermomyotome is divided into a dorsal and ventral half.
- Dorsal - dermatome.
- Ventral - myotome, this will also divide into a dorsal and ventral half that contribute the epaxial and hypaxial skeletal muscle groups respectively.
- hypaxial - muscles of the ventrolateral body wall, girdle, limb and tongue.[8]
- Muscle cells of the limb, tongue and lateral shoulder girdle muscles - derived from somite migrating myogenic precursor cells.
- Muscle cells of the ventrolateral body wall muscles (intercostal and abdominal muscles) and the medial shoulder girdle muscles - derived from the myotome.
Part of the shoulder girdle muscles (trapezius and sternocleidomastoideus) - derived from the lateral plate mesoderm.
Molecular
Pax
Mesoderm Development and Pax[9]
- Links: Developmental Signals - Pax
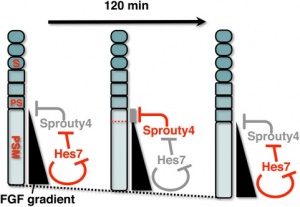
Mesogenin 1
A master regulator of paraxial presomitic mesoderm differentiation.'[3]
Additional Images
References
- ↑ 1.0 1.1 <pubmed>24304493</pubmed>| BMC Dev Biol.
- ↑ <pubmed>27380812</pubmed>
- ↑ 3.0 3.1 <pubmed>25371364</pubmed>
- ↑ <pubmed>22761566</pubmed>| PLoS Comput Biol.
- ↑ <pubmed>21368122</pubmed>
- ↑ <pubmed>17024300</pubmed>
- ↑ <pubmed>20615943</pubmed>
- ↑ <pubmed>24138189</pubmed>
- ↑ <pubmed>24496612</pubmed>| Development
Reviews
<pubmed>18482400</pubmed> <pubmed>21038776</pubmed> <pubmed>21038775</pubmed> <pubmed>17988868</pubmed> <pubmed>17643270</pubmed> <pubmed>17600784</pubmed> <pubmed>17024300</pubmed> <pubmed>15964269</pubmed> <pubmed>15309628</pubmed> <pubmed>15338303</pubmed>
Articles
<pubmed>12649586</pubmed>| Cells Tissues Organs
Search PubMed
Search NLM Online Textbooks: "Somitogenesis" : Developmental Biology | The Cell- A molecular Approach | Molecular Biology of the Cell | Endocrinology
Search Pubmed: Somitogenesis | Formation | Sclerotome | Hes7
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Somitogenesis. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Somitogenesis
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G