Genetic risk maternal age: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 31: | Line 31: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
{{#pmid:31811307}} "Can preimplantation genetic testing for aneuploidy (PGT-A) improve the live birth rate and reduce the miscarriage rate in patients with recurrent pregnancy loss (RPL) caused by an abnormal embryonic karyotype and recurrent implantation failure (RIF)? ...A large portion of pregnancy losses in the RPL group might be due to aneuploidy, since PGT-A reduced the overall incidence of pregnancy loss in these patients. Although PGT-A did not improve the live birth rate per patient, it did have the advantage of reducing the number of embryo transfers required to achieve a similar number live births compared with those not undergoing PGT-A." | |||
* '''Advanced maternal age and risk of non-chromosomal anomalies: data from a tertiary referral hospital in Turkey'''{{#pmid:28992718|PMID28992718}} "The purpose of this study is to determine if there is a relationship between non-chromosomal fetal anomalies of various organ systems and advanced maternal age. MATERIALS AND METHOD: This study was conducted in 387 women aged 20-53 years who underwent fetal karyotype testing due to positive prenatal test results or advanced maternal age at the Kanuni Sultan Süleyman Training and Research Hospital between September 2011 and March 2015. Fetuses with chromosomal anomalies were excluded from the study. The relationship between non-chromosomal anomalies and maternal age of women aged <35 or ≥35 years was studied. RESULTS: More than 80% (81.7%) of non-chromosomal anomalies were detected in patients aged <35 years, and 18.3% were found in those ≥35 years. There were no statistically significant differences found between the incidence of non-chromosomal anomalies in women aged over 35 years and those under 35 years. When congenital major anomalies were evaluated with respect to various organ systems, the risk of musculo-skeletal system anomalies decreased with advancing maternal age. However, there was no statistically significant difference between the <35 and ≥35-year age groups in the incidence of central nervous system, craniofacial, cardiac, gastrointestinal system, urogenital, respiratory, and limb anomalies. CONCLUSION: The incidence of non-chromosomal anomalies does not increase in fetuses of pregnant women aged over 35 years, in contrast to chromosomal anomalies." | * '''Advanced maternal age and risk of non-chromosomal anomalies: data from a tertiary referral hospital in Turkey'''{{#pmid:28992718|PMID28992718}} "The purpose of this study is to determine if there is a relationship between non-chromosomal fetal anomalies of various organ systems and advanced maternal age. MATERIALS AND METHOD: This study was conducted in 387 women aged 20-53 years who underwent fetal karyotype testing due to positive prenatal test results or advanced maternal age at the Kanuni Sultan Süleyman Training and Research Hospital between September 2011 and March 2015. Fetuses with chromosomal anomalies were excluded from the study. The relationship between non-chromosomal anomalies and maternal age of women aged <35 or ≥35 years was studied. RESULTS: More than 80% (81.7%) of non-chromosomal anomalies were detected in patients aged <35 years, and 18.3% were found in those ≥35 years. There were no statistically significant differences found between the incidence of non-chromosomal anomalies in women aged over 35 years and those under 35 years. When congenital major anomalies were evaluated with respect to various organ systems, the risk of musculo-skeletal system anomalies decreased with advancing maternal age. However, there was no statistically significant difference between the <35 and ≥35-year age groups in the incidence of central nervous system, craniofacial, cardiac, gastrointestinal system, urogenital, respiratory, and limb anomalies. CONCLUSION: The incidence of non-chromosomal anomalies does not increase in fetuses of pregnant women aged over 35 years, in contrast to chromosomal anomalies." | ||
| Line 50: | Line 52: | ||
|- | |- | ||
| {{Older papers}} | | {{Older papers}} | ||
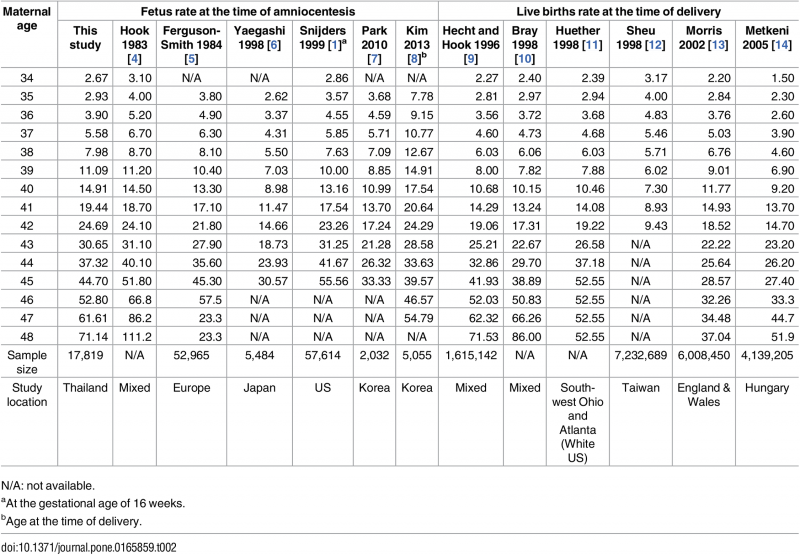
* '''Maternal Age-Specific Rates for Trisomy 21 and Common Autosomal Trisomies in Fetuses from a Single Diagnostic Center in Thailand'''{{#pmid:27812158|PMID27812158}} "To provide maternal age-specific rates for trisomy 21 (T21) and common autosomal trisomies (including trisomies 21, 18 and 13) in fetuses. We retrospectively reviewed prenatal cytogenetic results obtained between 1990 and 2009 in Songklanagarind Hospital, a university teaching hospital, in southern Thailand. Maternal age-specific rates of T21 and common autosomal trisomies were established using different regression models, from which only the fittest models were used for the study. A total of 17,819 records were included in the statistical analysis. The fittest models for predicting rates of T21 and common autosomal trisomies were regression models with 2 parameters (Age and Age2). The rate of T21 ranged between 2.67 per 1,000 fetuses at the age of 34 and 71.06 per 1,000 at the age of 48. The rate of common autosomal trisomies ranged between 4.54 per 1,000 and 99.65 per 1,000 at the same ages. This report provides the first maternal age-specific rates for T21 and common autosomal trisomies fetuses in a Southeast Asian population and the largest case number of fetuses have ever been reported in Asians." | * '''Maternal Age-Specific Rates for Trisomy 21 and Common Autosomal Trisomies in Fetuses from a Single Diagnostic Center in Thailand'''{{#pmid:27812158|PMID27812158}} "To provide maternal age-specific rates for trisomy 21 (T21) and common autosomal trisomies (including trisomies 21, 18 and 13) in fetuses. We retrospectively reviewed prenatal cytogenetic results obtained between 1990 and 2009 in Songklanagarind Hospital, a university teaching hospital, in southern Thailand. Maternal age-specific rates of T21 and common autosomal trisomies were established using different regression models, from which only the fittest models were used for the study. A total of 17,819 records were included in the statistical analysis. The fittest models for predicting rates of T21 and common autosomal trisomies were regression models with 2 parameters (Age and Age2). The rate of T21 ranged between 2.67 per 1,000 fetuses at the age of 34 and 71.06 per 1,000 at the age of 48. The rate of common autosomal trisomies ranged between 4.54 per 1,000 and 99.65 per 1,000 at the same ages. This report provides the first maternal age-specific rates for T21 and common autosomal trisomies fetuses in a Southeast Asian population and the largest case number of fetuses have ever been reported in Asians." | ||
Revision as of 21:29, 9 December 2019
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Educational Use Only - Embryology is an educational resource for learning concepts in embryological development, no clinical information is provided and content should not be used for any other purpose. |
Introduction
The table below shows the correlation of maternal age (mother's age) and the potential risk of human genetic abnormalities in children.
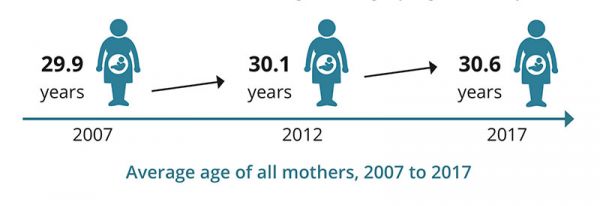
Australian Average Maternal Age Change
The first column shows maternal age, the second column shows the most common human chromosomal abnormality, trisomy 21 (Down syndrome), the third column shows all chromosomal abnormalities. The data below are from papers published in the 1980's.[1][2][3]
Interestingly, recent studies suggest that increasing paternal age (father's age) can also have affects on childhood mortality[4] and neurodevelopmental outcomes.[5]
Age Table
Table Data[1][2][3]
| |||
| Genetic risk maternal age | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Some Recent Findings
|
Sato T, Sugiura-Ogasawara M, Ozawa F, Yamamoto T, Kato T, Kurahashi H, Kuroda T, Aoyama N, Kato K, Kobayashi R, Fukuda A, Utsunomiya T, Kuwahara A, Saito H, Takeshita T & Irahara M. (2019). Preimplantation genetic testing for aneuploidy: a comparison of live birth rates in patients with recurrent pregnancy loss due to embryonic aneuploidy or recurrent implantation failure. Hum. Reprod. , , . PMID: 31811307 DOI. "Can preimplantation genetic testing for aneuploidy (PGT-A) improve the live birth rate and reduce the miscarriage rate in patients with recurrent pregnancy loss (RPL) caused by an abnormal embryonic karyotype and recurrent implantation failure (RIF)? ...A large portion of pregnancy losses in the RPL group might be due to aneuploidy, since PGT-A reduced the overall incidence of pregnancy loss in these patients. Although PGT-A did not improve the live birth rate per patient, it did have the advantage of reducing the number of embryo transfers required to achieve a similar number live births compared with those not undergoing PGT-A."
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Maternal Age Abnormal Development | Maternal Age Genetic Abnormalities
|
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Maternal Age Trisomy 21 Studies
Comparative data compiled by this study.[9]
Ireland
Data from a clinical data study of chromosomal trisomies in the East of Ireland (2011-2013).[8]
Total births 80,894 - 394 trisomy cases (prevalence rate 48.9/10,000)- Trisomy 21 - 289 (prevalence rate 35.7/10,000)
- Trisomy 18 - 75 (prevalence rate 9.3/10,000)
- Trisomy 13 - 30 (prevalence rate 3.7/10,000)
- 90+% trisomies 18/13
- 47% of Trisomy 21
Thailand
The following data is from a recent Thai study of maternal age and trisomies.[9]
References
- ↑ 1.0 1.1 1.2 Hook EB. (1981). Rates of chromosome abnormalities at different maternal ages. Obstet Gynecol , 58, 282-5. PMID: 6455611
- ↑ 2.0 2.1 2.2 Hook EB, Cross PK & Schreinemachers DM. (1983). Chromosomal abnormality rates at amniocentesis and in live-born infants. JAMA , 249, 2034-8. PMID: 6220164
- ↑ 3.0 3.1 3.2 Schreinemachers DM, Cross PK & Hook EB. (1982). Rates of trisomies 21, 18, 13 and other chromosome abnormalities in about 20 000 prenatal studies compared with estimated rates in live births. Hum. Genet. , 61, 318-24. PMID: 6891368
- ↑ Zhu JL, Vestergaard M, Madsen KM & Olsen J. (2008). Paternal age and mortality in children. Eur. J. Epidemiol. , 23, 443-7. PMID: 18437509 DOI.
- ↑ Saha S, Barnett AG, Foldi C, Burne TH, Eyles DW, Buka SL & McGrath JJ. (2009). Advanced paternal age is associated with impaired neurocognitive outcomes during infancy and childhood. PLoS Med. , 6, e40. PMID: 19278291 DOI.
- ↑ Okmen Ozkan B, Köroğlu N, Turkgeldi LS, Cetin BA & Aslan H. (2019). Advanced maternal age and risk of non-chromosomal anomalies: data from a tertiary referral hospital in Turkey. J. Matern. Fetal. Neonatal. Med. , 32, 749-752. PMID: 28992718 DOI.
- ↑ Arya S, Mulla ZD & Plavsic SK. (2018). Outcomes of Women Delivering at Very Advanced Maternal Age. J Womens Health (Larchmt) , , . PMID: 30016194 DOI.
- ↑ 8.0 8.1 McDonnell R, Monteith C, Kennelly M, Martin A, Betts D, Delany V, Lynch SA, Coulter-Smith S, Sheehan S & Mahony R. (2017). Epidemiology of chromosomal trisomies in the East of Ireland. J Public Health (Oxf) , 39, e145-e151. PMID: 27591300 DOI.
- ↑ 9.0 9.1 9.2 Jaruthamsophon K, Sriplung H, Charalsawadi C & Limprasert P. (2016). Maternal Age-Specific Rates for Trisomy 21 and Common Autosomal Trisomies in Fetuses from a Single Diagnostic Center in Thailand. PLoS ONE , 11, e0165859. PMID: 27812158 DOI.
- ↑ Michalski AM, Richardson SD, Browne ML, Carmichael SL, Canfield MA, VanZutphen AR, Anderka MT, Marshall EG & Druschel CM. (2015). Sex ratios among infants with birth defects, National Birth Defects Prevention Study, 1997-2009. Am. J. Med. Genet. A , 167A, 1071-81. PMID: 25711982 DOI.
Articles
Goetzinger KR, Shanks AL, Odibo AO, Macones GA & Cahill AG. (2017). Advanced Maternal Age and the Risk of Major Congenital Anomalies. Am J Perinatol , 34, 217-222. PMID: 27398707 DOI. This study aims to determine if advanced maternal age (AMA) is a risk factor for major congenital anomalies, in the absence of aneuploidy. ...Of 76,156 euploid fetuses, 2.4% (n = 1,804) were diagnosed with a major anomaly. There was a significant decrease in the incidence of major fetal anomalies with increasing maternal age until the threshold of age 35 (p < 0.001). Being AMA was significantly associated with an overall decreased risk for major fetal anomalies (adjusted odds ratio: 0.59, 95% confidence interval: 0.52-0.66). The subgroup analysis demonstrated similar results for women ≥ 40 years of age. Conclusion AMA is associated with an overall decreased risk for major anomalies. These findings may suggest that the "all or nothing" phenomenon plays a more robust role in embryonic development with advancing oocyte age, with anatomically normal fetuses being more likely to survive."
Csermely G, Czeizel AE & Veszprémi B. (2015). Distribution of maternal age and birth order groups in cases with unclassified multiple congenital abnormalities according to the number of component abnormalities: a national population-based case-control study. Birth Defects Res. Part A Clin. Mol. Teratol. , 103, 67-75. PMID: 25224265 DOI.
- "The Hungarian Case-Control Surveillance of Congenital Abnormalities, 1980 to 1996, yielded a large population-based national data set with 22,843 malformed newborns or fetuses ("informative cases") included 1349 UMCA cases with their 2407 matched controls. Case-control comparison of maternal age and birth order was made for cases with UMCA, stratified by component numbers and their controls. In addition, 834 cases with Down syndrome were compared to 1432 matched controls. ...The well-known advanced maternal age with the higher risk for Down syndrome was confirmed. The findings of the study suggest that the young age of mothers associates with the higher risk of UMCA, in addition birth order 4 or more associates with the higher risk for UMCA with 2 and 3 component CAs."
Allen EG, Freeman SB, Druschel C, Hobbs CA, O'Leary LA, Romitti PA, Royle MH, Torfs CP & Sherman SL. (2009). Maternal age and risk for trisomy 21 assessed by the origin of chromosome nondisjunction: a report from the Atlanta and National Down Syndrome Projects. Hum. Genet. , 125, 41-52. PMID: 19050929 DOI.
- "We examined the association between maternal age and chromosome 21 nondisjunction by origin of the meiotic error. We analyzed data from two population-based, case-control studies: Atlanta Down Syndrome Project (1989-1999) and National Down Syndrome Project (2001-2004). Cases were live born infants with trisomy 21 and controls were infants without trisomy 21 delivered in the same geographical regions. We enrolled 1,215 of 1,881 eligible case families and 1,375 of 2,293 controls. We report four primary findings. First, the significant association between advanced maternal age and chromosome 21 nondisjunction was restricted to meiotic errors in the egg; the association was not observed in sperm or in post-zygotic mitotic errors. Second, advanced maternal age was significantly associated with both meiosis I (MI) and meiosis II (MII). For example, compared to mothers of controls, mothers of infants with trisomy 21 due to MI nondisjunction were 8.5 times more likely to be >or=40 years old than 20-24 years old at the birth of the index case (95% CI=5.6-12.9). Where nondisjunction occurred in MII, mothers were 15.1 times more likely to be >or=40 years (95% CI = 8.4-27.3). Third, the ratio of MI to MII errors differed by maternal age. The ratio was lower among women <19 years of age and those >or=40 years (2.1, 2.3, respectively) and higher in the middle age group (3.6). Lastly, we found no effect of grand-maternal age on the risk for maternal nondisjunction. This study emphasizes the complex association between advanced maternal age and nondisjunction of chromosome 21 during oogenesis."
Munné S, Chen S, Colls P, Garrisi J, Zheng X, Cekleniak N, Lenzi M, Hughes P, Fischer J, Garrisi M, Tomkin G & Cohen J. (2007). Maternal age, morphology, development and chromosome abnormalities in over 6000 cleavage-stage embryos. Reprod. Biomed. Online , 14, 628-34. PMID: 17509208 DOI.
- "Previous studies assessing the relationship between embryo development, maternal age and chromosome abnormalities were either small or analysed mostly embryos not suitable for replacement. The present study includes >6000 embryos, including many suitable for replacement. Embryos with the best morphology and development were 44% euploid in patients younger than 35, decreasing to 21% in patients 41 and older. The worst morphology group had only 30% normal embryos from patients younger than 35, and 12% in embryos from patients 41 and older. Thus morphological analysis was able to improve the population of normal embryos only from 30 to 44% in the best of cases. Regarding specific abnormalities, 20% of embryos were aneuploid, 32% aneuploid plus other abnormalities, and the rest had post-meiotic abnormalities. Of those, only aneuploidy increased with maternal age. There were no big differences in the frequency of chromosome abnormalities depending on patient indication, within a similar age group. In summary, previous trends detected in suboptimal embryos were also confirmed in the best embryos for replacement. Although dysmorphism and advanced maternal age are both related to chromosome abnormalities, these parameters can yield at most <50% euploid embryos, and other techniques such as preimplantation diagnosis are required to ensure that only euploid embryos are replaced."
Kuliev A, Cieslak J & Verlinsky Y. (2005). Frequency and distribution of chromosome abnormalities in human oocytes. Cytogenet. Genome Res. , 111, 193-8. PMID: 16192694 DOI.
- "It was previously shown that more than half of the human oocytes obtained from IVF patients of advanced reproductive age are aneuploid, due to meiosis I and meiosis II errors. The present paper further confirms that 61.8% of the oocytes tested by fluorescent probes specific for chromosomes 13, 16, 18, 21 and 22 are abnormal, representing predominantly chromatid errors, which are the major source of aneuploidy in the resulting embryos. Almost half of the oocytes with meiosis I errors (49.3%) are prone to sequential meiosis II errors, which may lead to aneuploidy rescue in 30.8% of the cases. Half of the detected aneuploidies (49.8%) are of complex nature with involvement of two or more chromosomes, or the same chromosome in both meiotic divisions. The aneuploidy rates for individual chromosomes are different, with a higher prevalence of chromosome 21 and 22 errors. The origin of aneuploidy for the individual chromosomes is also not random, with chromosome 16 and 22 errors originating more frequently in meiosis II, and chromosome 18, 13 and 21 errors in meiosis I. There is an age dependence not only for the overall frequency of aneuploidies, but also for each chromosome error, aneuploidies originating from meiosis I, meiosis II, and both meiosis I and meiosis II errors, as well as for different types of aneuploidies. The data further suggest the practical relevance of oocyte aneuploidy testing for detection and avoidance from transfer of the embryos deriving from aneuploid oocytes, which should contribute significantly to the pregnancy outcomes of IVF patients of advanced reproduction age."
Search PubMed
Search PubMed Now: Genetic risk maternal age
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Genetic risk maternal age. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Genetic_risk_maternal_age
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G