Developmental Signals - Sonic hedgehog: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (33 intermediate revisions by the same user not shown) | |||
| Line 2: | Line 2: | ||
==Introduction== | ==Introduction== | ||
[[File:Sonic.jpg|right]] | [[File:Sonic.jpg|right]] | ||
(SHH, Shh) | The secreted growth factor {{sonic hedgehog}} ({{SHH}}, {{Shh}}) binds the patched (ptc) receptor on cell membrane. This protein has many different functions in different tissues of the developing embryo, the main role would be of establishing "pattern". SHH name derives from homology to the original fruitfly (drosophila) "hedgehog" mutation, where these flies have hairs located in regions (ventral) which are normally hair-free, and therefore have a disrupted body pattern. | ||
| Line 19: | Line 19: | ||
==Some Recent Findings== | ==Some Recent Findings== | ||
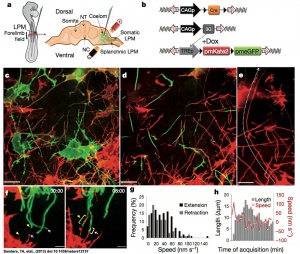
[[File:Mesenchymal_cell_cytoplasmic_extensions_01.jpg|thumb|300px|Mesenchymal cells of the developing limb bud possess long and highly dynamic cytoplasmic extensions. | [[File:Mesenchymal_cell_cytoplasmic_extensions_01.jpg|thumb|300px|Mesenchymal cells of the developing limb bud possess long and highly dynamic cytoplasmic extensions.{{#pmid:23624372|PMID23624372}}]] | ||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | * '''Controlling tissue patterning by translational regulation of signaling transcripts through the core translation factor eIF3c'''{{#pmid:34752747|PMID34752747}} "Although gene expression is tightly regulated during embryonic development, the impact of translational control has received less experimental attention. Here, we find that eukaryotic translation initiation factor-3 (eIF3) is required for Shh-mediated tissue patterning. Analysis of loss-of-function eIF3 subunit c (Eif3c) mice reveal a unique sensitivity to the Shh receptor patched 1 (Ptch1) dosage. Genome-wide in vivo enhanced cross-linking immunoprecipitation sequence (eCLIP-seq) shows unexpected specificity for eIF3 binding to a pyrimidine-rich motif present in subsets of 5'-UTRs and a corresponding change in the translation of these transcripts by ribosome profiling in Eif3c loss-of-function embryos. We further find a transcript specific effect in Eif3c loss-of-function embryos whereby translation of Ptch1 through this pyrimidine-rich motif is specifically sensitive to eIF3 amount. Altogether, this work uncovers hidden specificity of housekeeping translation initiation machinery for the translation of key developmental signaling transcripts." | ||
* | * '''The neocortical progenitor specification program is established through combined modulation of SHH and FGF signaling'''{{#pmid:32737167|PMID32737167}} "Low levels of {{FGF}}15 control progenitor proliferation and differentiation during neocortical development but little is known on how {{FGF}}15 expression is maintained. Our studies identified {{SHH}} signaling as a critical activator of FGF15 expression during corticogenesis. We found that Sufu, via Gli3R, ensured low levels of FGF15 was expressed to prevent abnormal specification of neocortical progenitors. These studies advance our knowledge on the molecular mechanisms guiding the generation of specific neocortical neuronal lineages, their implications in neurodevelopmental diseases, and may guide future studies on how progenitor cells may be utilized for brain repair." | ||
* '''Sonic hedgehog promotes chondrogenesis of rabbit bone marrow stem cells in a rotary cell culture system'''{{#pmid:31401976|PMID31401976}} "Sonic hedgehog (Shh) is an important signalling protein involved in the induction of early cartilaginous differentiation. Herein, we demonstrate that Shh markedly induces {{chondrogenesis}} of {{rabbit}} {{bone marrow}} stromal cells (BMSCs) under microgravity conditions, and promotes cartilage regeneration. In the rotary cell culture system (RCCS), chondrogenic differentiation was revealed by stronger Toluidine Blue and collagen II immunohistochemical staining in the Shh transfection group, and chondroinductive activity of Shh was equivalent to that of TGF-β. Western blotting and qRT-PCR analysis results verified the stronger expression of Sox9, aggrecan (ACAN), and collagen II in rabbit BMSCs treated with Shh or TGF-β in a microgravity environment. Low levels of chondrogenic hypertrophy, osteogenesis, and adipogenesis-related factors were detected in all groups. After transplantation in vivo, histological analysis revealed a significant improvement in cartilage and subchondral repair in the Shh transfection group. These results suggested that Shh signalling promoted chondrogenesis in rabbit BMSCs under microgravity conditions equivalent to TGF-β, and improved the early stages of the repair of cartilage and subchondral defects. Furthermore, RCCS provided a dynamic culture microenvironment conducive for cell proliferation, aggregation and differentiation." {{cartilage}} | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! More recent papers | ! More recent papers | ||
|- | |- | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Sonic hedgehog ''Sonic hedgehog''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Sonic+hedgehog ''Sonic hedgehog''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=SHH ''SHH''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Patched ''Patched''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=BOC/CDON ''BOC/CDON''] | ||
Search all databases: [http://www.ncbi.nlm.nih.gov/sites/gquery?itool=toolbar&cmd=search&term=sonic+hedgehog sonic hedgehog] | |||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''SHH signaling mediated by a prechordal and brain enhancer controls forebrain organization'''{{#pmid:31685615|PMID31685615}} "Sonic hedgehog (SHH) signaling plays a pivotal role in 2 different phases during brain development. Early SHH signaling derived from the prechordal plate (PrCP) triggers secondary Shh induction in the forebrain, which overlies the PrCP, and the induced SHH signaling, in turn, directs late neuronal differentiation of the forebrain. Consequently, Shh regulation in the PrCP is crucial for initiation of forebrain development. However, no enhancer that regulates prechordal Shh expression has yet been found. Here, we identified a prechordal enhancer, named SBE7, in the vicinity of a cluster of known forebrain enhancers for Shh This enhancer also directs Shh expression in the ventral midline of the forebrain, which receives the prechordal SHH signal. Thus, the identified enhancer acts not only for the initiation of Shh regulation in the PrCP but also for subsequent Shh induction in the forebrain. Indeed, removal of the enhancer from the mouse genome markedly down-regulated the expression of Shh in the rostral domains of the axial mesoderm and in the ventral midline of the {{forebrain}} and {{hypothalamus}} in the mouse embryo, and caused a craniofacial abnormality similar to human {{holoprosencephaly}} (HPE). These findings demonstrate that SHH signaling mediated by the newly identified enhancer is essential for development and growth of the ventral midline of the forebrain and hypothalamus. Understanding of the Shh regulation governed by this prechordal and brain enhancer provides an insight into the mechanism underlying craniofacial morphogenesis and the etiology of HPE. | |||
* '''Region-specific endodermal signals direct {{neural crest}} cells to form the three {{middle ear}} ossicles'''{{#pmid:30630826|PMID30630826}} "Defects in the middle ear ossicles - {{malleus}}, {{incus}} and {{stapes}} - can lead to conductive hearing loss. During development, {{neural crest}} cells (NCCs) migrate from the dorsal hindbrain to specific locations in {{pharyngeal arch}} (PA) 1 and 2, to form the malleus-incus and stapes, respectively. It is unclear how migratory NCCs reach their proper destination in the PA and initiate mesenchymal condensation to form specific ossicles. We show that secreted molecules sonic hedgehog ({{SHH}}) and bone morphogenetic protein 4 ({{BMP}}4) emanating from the pharyngeal {{endoderm}} are important in instructing region-specific NCC condensation to form malleus-incus and stapes, respectively, in mouse. Tissue-specific knockout of {{Shh}} in the pharyngeal endoderm or Smo (a transducer of SHH signaling) in NCCs causes the loss of malleus-incus condensation in PA1 but only affects the maintenance of stapes condensation in PA2. By contrast, knockout of {{Bmp}}4 in the pharyngeal endoderm or Smad4 (a transducer of TGFβ/BMP signaling) in the NCCs disrupts NCC migration into the stapes region in PA2, affecting stapes formation. These results indicate that region-specific endodermal signals direct formation of specific middle ear ossicles." [http://dev.biologists.org/cgi/pmidlookup?view=long&pmid=30630826 Development] | |||
* '''Constitutive activation of hedgehog signaling adversely affects epithelial cell fate during palatal fusion'''{{#pmid:29981310|PMID29981310}} "{{Cleft palate}} is one of the most common craniofacial congenital defects in humans. It is associated with multiple genetic and environmental risk factors, including mutations in the genes encoding signaling molecules in the {{sonic hedgehog}} ({{SHH}}) pathway, which are risk factors for cleft palate in both humans and mice. However, the function of Shh signaling in the palatal epithelium during palatal fusion remains largely unknown. Although components of the Shh pathway are localized in the palatal epithelium, specific inhibition of Shh signaling in palatal epithelium does not affect palatogenesis. ...In this study, we discovered that constitutive activation of Hh signaling in the palatal epithelium results in submucous cleft palate and persistence of the medial edge epithelium (MEE). Further investigation revealed that precise downregulation of Shh signaling is required at a specific time point in the MEE during palatal fusion. Upregulation of Hh signaling in the palatal epithelium maintains the proliferation of MEE cells. This may be due to a dysfunctional p63/Irf6 regulatory loop. The resistance of MEE cells to apoptosis is likely conferred by enhancement of a cell adhesion network through the maintenance of p63 expression." | |||
* '''Structures of human Patched and its complex with native palmitoylated sonic hedgehog'''{{#pmid:29995851|PMID29995851}} "Hedgehog (HH) signalling governs embryogenesis and adult tissue homeostasis in mammals and other multicellular organisms1-3. Whereas deficient HH signalling leads to birth defects, unrestrained HH signalling is implicated in human cancers2,4-6. N-terminally palmitoylated HH releases the repression of Patched to the oncoprotein smoothened (SMO); however, the mechanism by which HH recognizes Patched is unclear. Here we report cryo-electron microscopy structures of human patched 1 (PTCH1) alone and in complex with the N-terminal domain of 'native' sonic hedgehog (native SHH-N has both a C-terminal cholesterol and an N-terminal fatty-acid modification), at resolutions of 3.5 Å and 3.8 Å, respectively. The structure of PTCH1 has internal two-fold pseudosymmetry in the transmembrane core, which features a sterol-sensing domain and two homologous extracellular domains, resembling the architecture of Niemann-Pick C1 (NPC1) protein7. The palmitoylated N terminus of SHH-N inserts into a cavity between the extracellular domains of PTCH1 and dominates the PTCH1-SHH-N interface, which is distinct from that reported for SHH-N co-receptors8. Our biochemical assays show that SHH-N may use another interface, one that is required for its co-receptor binding, to recruit PTCH1 in the absence of a covalently attached palmitate." | |||
* '''Bergmann {{glia}}l Sonic hedgehog signaling activity is required for proper cerebellar cortical expansion and architecture'''{{#pmid:29792854|PMID29792854}} "Neuronal-glial relationships play a critical role in the maintenance of central nervous system architecture and neuronal specification. A deeper understanding of these relationships can elucidate cellular cross-talk capable of sustaining proper development of neural tissues. In the cerebellum, cerebellar granule neuron precursors (CGNPs) proliferate in response to Purkinje neuron-derived Sonic hedgehog (Shh) before ultimately exiting the cell cycle and migrating radially along Bergmann glial fibers. However, the function of Bergmann glia in CGNP proliferation remains not well defined. Interestingly, the Hh pathway is also activated in Bergmann glia, but the role of Shh signaling in these cells is unknown. In this study, we show that specific ablation of Shh signaling using the tamoxifen-inducible TNCYFP-CreER line to eliminate Shh pathway activator Smoothened in Bergmann glia is sufficient to cause severe cerebellar hypoplasia and a significant reduction in CGNP proliferation. TNCYFP-CreER; SmoF/- (SmoCKO) mice demonstrate an obvious reduction in cerebellar size within two days of ablation of Shh signaling. Mutant cerebella have severely reduced proliferation and increased differentiation of CGNPs due to a significant decrease in Shh activity and concomitant activation of Wnt signaling in SmoCKO CGNPs, suggesting that this pathway is involved in cross-talk with the Shh pathway in regulating CGNP proliferation. In addition, Purkinje cells are ectopically located, their dendrites stunted, and the Bergmann glial network disorganized. Collectively, these data demonstrate a previously unappreciated role for Bergmann glial Shh signaling activity in the proliferation of CGNPs and proper maintenance of cerebellar architecture." {{SHH}} | |||
* '''Divergent axial morphogenesis and early shh expression in vertebrate prospective floor plate'''{{#pmid:29423139|PMID29423139}} "The notochord has organizer properties and is required for floor plate induction and dorsoventral patterning of the neural tube. This activity has been attributed to sonic hedgehog (shh) signaling, which originates in the notochord, forms a gradient, and autoinduces shh expression in the floor plate. However, reported data are inconsistent and the spatiotemporal development of the relevant shh expression domains has not been studied in detail. We therefore studied the expression dynamics of shh in rabbit, chicken and Xenopus laevis embryos (as well as indian hedgehog and desert hedgehog as possible alternative functional candidates in the chicken). ...While shh expression patterns in rabbit and X. laevis embryos are roughly compatible with the classical view of "ventral to dorsal induction" of the floor plate, the early shh expression in the chick floor plate challenges this model. Intriguingly, this alternative sequence of domain induction is related to the asymmetrical morphogenesis of the primitive node and other axial organs in the chick. Our results indicate that the floor plate in X. laevis and chick embryos may be initially induced by planar interaction within the ectoderm or epiblast. Furthermore, we propose that the mode of the floor plate induction adapts to the variant topography of interacting tissues during gastrulation and notochord formation and thereby reveals evolutionary plasticity of early embryonic induction." {{Chicken}} | |||
* '''Disruption of Scube2 impairs endochondral bone formation'''{{#pmid:25639508|PMID25639508}} "SCUBE2 (signal peptide-CUB-EGF domain-containing protein 2) belongs to a secreted and membrane-tethered multi-domain SCUBE protein family composed of 3 members found in vertebrates and mammals. Recent reports suggested that zebrafish scube2 could facilitate sonic hedgehog (Shh) signaling for proper development of slow muscle. However, whether SCUBE2 can regulate the signaling activity of two other hedgehog ligands (Ihh and Dhh), and the developmental relevance of the SCUBE2-induced hedgehog signaling in mammals remain poorly understood. In this study, we first showed that as compared with SCUBE1 or 3, SCUBE2 is the most potent modulator of IHH signaling in vitro. In addition, gain and loss-of-function studies demonstrated that SCUBE2 exerted an osteogenic function by enhancing Ihh-stimulated osteoblast differentiation in the mouse mesenchymal progenitor cells." (More? [[Musculoskeletal System - Bone Development|Bone Development]] | [http://omim.org/entry/611747 OMIM611747]) | |||
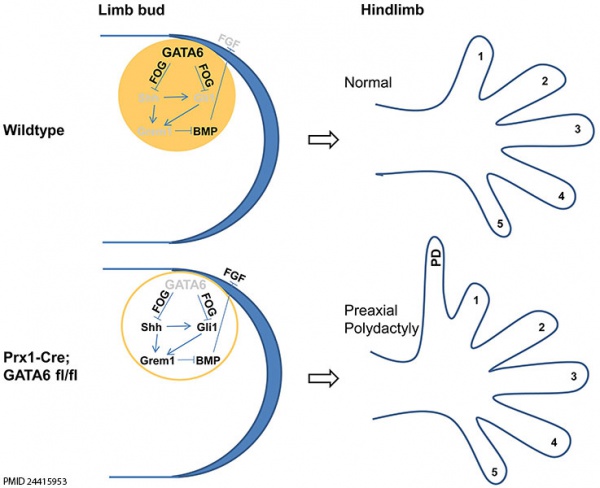
* '''GATA6 Is a Crucial Regulator of Shh in the Limb Bud'''{{#pmid:24415953|PMID24415953}} "In the limb bud, patterning along the anterior-posterior (A-P) axis is controlled by Sonic Hedgehog (Shh), a signaling molecule secreted by the "Zone of Polarizing Activity", an organizer tissue located in the posterior margin of the limb bud. We have found that the transcription factors GATA4 and GATA6, which are key regulators of cell identity, are expressed in an anterior to posterior gradient in the early limb bud, raising the possibility that GATA transcription factors may play an additional role in patterning this tissue. While both GATA4 and GATA6 are expressed in an A-P gradient in the forelimb buds, the hindlimb buds principally express GATA6 in an A-P gradient." [[Musculoskeletal_System_-_Limb_Development|Limb Development]] | |||
* '''Dynamic interpretation of hedgehog signaling in the Drosophila wing disc'''{{#pmid:19787036|PMID19787036}} | |||
* '''Patched 1 is a crucial determinant of asymmetry and digit number in the vertebrate limb'''{{#pmid:19783740|PMID19783740}} | |||
* '''Uncoupling Sonic hedgehog control of pattern and expansion of the developing limb bud'''{{#pmid:18410737|PMID18410737}} "One of the first changes we noted was that the dorsoventral polarity of the forebrain was disturbed, which manifested as a loss of Shh in the ventral telencephalon, a reduction in expression of the ventral markers Nkx2.1 and Dlx2, and a concomitant expansion of the dorsal marker Pax6. In addition to changes in the forebrain neuroectoderm, we observed altered gene expression patterns in the facial ectoderm. For example, Shh was not induced in the frontonasal ectoderm, and Ptc and Gli1 were reduced in both the ectoderm and adjacent mesenchyme." | |||
* THM1 negatively modulates mouse sonic hedgehog signal transduction and affects retrograde intraflagellar transport in cilia.{{#pmid:18327258|PMID18327258}} | |||
* Triphalangeal thumb-polysyndactyly syndrome and syndactyly type IV are caused by genomic duplications involving the long-range, limb-specific SHH enhancer.{{#pmid:18417549|PMID18417549}} | |||
* Notochord-derived Shh concentrates in close association with the apically positioned basal body in neural target cells and forms a dynamic gradient during neural patterning.{{#pmid:18272593|PMID18272593}} | |||
|} | |} | ||
==Hedgehog Family== | ==Human Hedgehog Family== | ||
[[File:Hedgehog signaling pathway.jpg|thumb|300px|alt=Hedgehog signaling pathway cartoon|Hedgehog signaling pathway | |||
{{Hedgehog family table}} | |||
{{Hedgehog family collapsetable}} | |||
[[File:Hedgehog signaling pathway.jpg|thumb|300px|alt=Hedgehog signaling pathway cartoon|Hedgehog signaling pathway{{#pmid:19040769|PMID19040769}}]] | |||
Vertebrates have a number of signaling proteins and receptors that belong to the hedgehog (Hh) family. | Vertebrates have a number of signaling proteins and receptors that belong to the hedgehog (Hh) family. | ||
| Line 63: | Line 100: | ||
===Neural=== | ===Neural=== | ||
[[File:Shh frog notochord 1.jpg|thumb|Shh frog notochord]] | [[File:Shh frog notochord 1.jpg|thumb|Shh frog notochord]] | ||
[[File:Neural tube SHH patterning cartoon.jpg|thumb|Neural tube SHH patterning | [[File:Neural tube SHH patterning cartoon.jpg|thumb|Neural tube SHH patterning{{#pmid:9197245|PMID9197245}}]] | ||
* SHH is secreted by the notochord, ventralizes the neural tube, inducing the floor plate and motor neurons. | * SHH is secreted by the notochord, ventralizes the neural tube, inducing the floor plate and motor neurons. | ||
* Regulation of patched by sonic hedgehog in the developing neural tube. | * Regulation of patched by sonic hedgehog in the developing neural tube.{{#pmid:8790332|PMID8790332}} "The pattern of PTC expression suggests that Sonic hedgehog may play an inductive role in more dorsal regions of the neural tube than have been previously demonstrated. Examination of the pattern of PTC expression also suggests that PTC may act in a negative feedback loop to attenuate hedgehog signaling." | ||
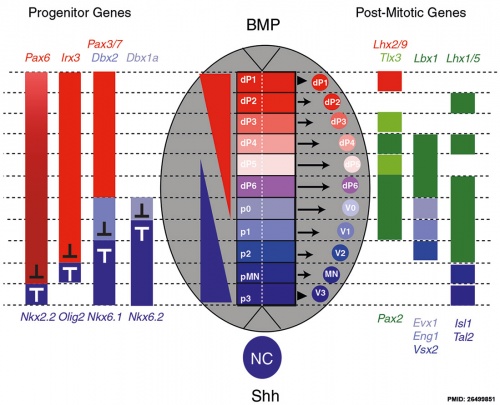
[[File:Neural tube dorsoventral patterning SHH BMP.jpg|500px]] | [[File:Neural tube dorsoventral patterning SHH BMP.jpg|500px]] | ||
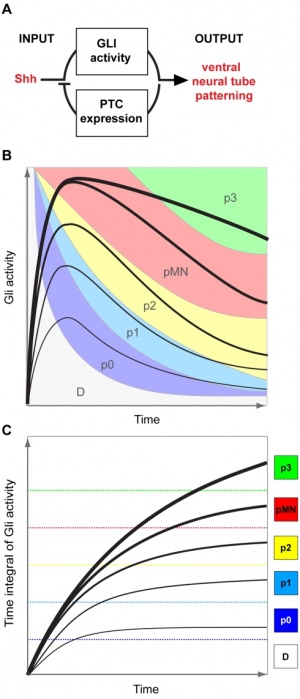
Neural tube cross section dorsoventral domains are established by opposing concentration gradients of Sonic Hedgehog (Shh) and Bone Morphogenic Protein (BMP). | Neural tube cross section dorsoventral domains are established by opposing concentration gradients of Sonic Hedgehog (Shh) and Bone Morphogenic Protein (BMP).{{#pmid:26499851|PMID26499851}} | ||
* left - These regulate progenitor gene expression. The progenitor genes cross-repress each other to establish domain boundaries. | * left - These regulate progenitor gene expression. The progenitor genes cross-repress each other to establish domain boundaries. | ||
| Line 81: | Line 118: | ||
| [[File:Neural tube - SHH model.jpg|300px|alt=Neural tube - SHH model]] | | [[File:Neural tube - SHH model.jpg|300px|alt=Neural tube - SHH model]] | ||
|- | |- | ||
| Neural tube - SHH model | | Neural tube - SHH model{{#pmid:20532235|PMID20532235}} | ||
|} | |} | ||
===Neural Crest=== | ===Neural Crest=== | ||
{| | |||
| [[File:Mouse cranial nerve model SHH.jpg|400px]] | |||
| A model of mouse cranial nerve development{{#pmid:25799573|PMID25799573}} includes SHH action during neural crest cell migrating from rhombomere 2 (r2) or 4 (r4) interaction with placodal cells to develop the cranial nerves. | |||
|} | |||
* Neural crest cell survival in the foregut endoderm of jaw development | |||
* Sonic hedgehog is required for cardiac outflow tract and neural crest cell development. | * Neural crest cell survival in the foregut endoderm of jaw development{{#pmid:16868080|PMID16868080}} | ||
* Sonic hedgehog is required for cardiac outflow tract and neural crest cell development.{{#pmid:15936751|PMID15936751}} | |||
===Hearing=== | ===Hearing=== | ||
During mouse cochlea development the adjacent auditory (spiral) ganglion acts as a source of Sonic hedgehog. Without SHH the cochlear duct is shortened, sensory hair cell precursors prematurely leave the mitotic cell cycle, and hair cell differentiation closely follows cell cycle exit in a similar apical-to-basal direction. | During mouse cochlea development the adjacent auditory (spiral) ganglion acts as a source of Sonic hedgehog. Without SHH the cochlear duct is shortened, sensory hair cell precursors prematurely leave the mitotic cell cycle, and hair cell differentiation closely follows cell cycle exit in a similar apical-to-basal direction.{{#pmid:23918393|PMID23918393}} | ||
<gallery> | <gallery> | ||
| Line 98: | Line 140: | ||
===Somite=== | ===Somite=== | ||
A study has shown an essential role for Shh signalling in the control of laminin-111 synthesis and in the initiation of basement membrane assembly in the myotome. | A study has shown an essential role for Shh signalling in the control of laminin-111 synthesis and in the initiation of basement membrane assembly in the myotome.{{#pmid:19783738|PMID19783738}} | ||
===Limb=== | ===Limb=== | ||
[[File:Mouse-_hindlimb_buds_Shh_expression.jpg|thumb|Mouse hindlimb budsZPA Shh expression. | [[File:Mouse-_hindlimb_buds_Shh_expression.jpg|thumb|Mouse hindlimb budsZPA Shh expression.{{#pmid:20386744|PMID20386744}}]] | ||
* SHH is secreted by the zone of polarizing activity (ZPA) organizing limb axis formation. | * SHH is secreted by the zone of polarizing activity (ZPA) organizing limb axis formation. | ||
* Extended exposure to Sonic hedgehog is required for patterning the posterior digits of the vertebrate limb. | * Extended exposure to Sonic hedgehog is required for patterning the posterior digits of the vertebrate limb.{{#pmid:17610861|PMID17610861}} | ||
{| | {| | ||
| Line 109: | Line 151: | ||
| [[File:Limb patterning factors 07.jpg|300px]] | | [[File:Limb patterning factors 07.jpg|300px]] | ||
|- | |- | ||
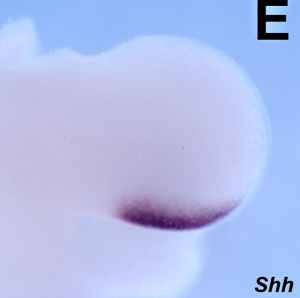
| Shh mouse forelimb | | Shh mouse forelimb {{ME11.5}}{{#pmid:17194222|PMID17194222}} | ||
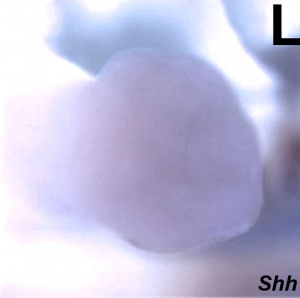
| Shh mouse hindlimb | | Shh mouse hindlimb {{ME12.5}}<ref name="PMID17194222" /> | ||
|} | |} | ||
| Line 118: | Line 160: | ||
| [[File:Limb_bud_geometry_and_patterning.jpg|600px]] | | [[File:Limb_bud_geometry_and_patterning.jpg|600px]] | ||
|- | |- | ||
| Geometry and patterning of the developing limb bud | | Geometry and patterning of the developing limb bud{{#pmid:20644713|PMID20644713}} | ||
|} | |} | ||
| Line 124: | Line 166: | ||
[[File:Model GATA6 hindlimb.jpg|600px]] | [[File:Model GATA6 hindlimb.jpg|600px]] | ||
Model for GATA6 action Patterning developing Hindlimb Buds | Model for GATA6 action Patterning developing Hindlimb Buds{{#pmid:24415953|PMID4415953}} | ||
===Cartilage=== | |||
'''Sonic hedgehog promotes chondrogenesis of rabbit bone marrow stem cells in a rotary cell culture system'''{{#pmid:31401976|PMID31401976}} "Sonic hedgehog (Shh) is an important signalling protein involved in the induction of early cartilaginous differentiation. Herein, we demonstrate that Shh markedly induces {{chondrogenesis}} of {{rabbit}} bone marrow stromal cells (BMSCs) under microgravity conditions, and promotes cartilage regeneration. In the rotary cell culture system (RCCS), chondrogenic differentiation was revealed by stronger Toluidine Blue and collagen II immunohistochemical staining in the Shh transfection group, and chondroinductive activity of Shh was equivalent to that of TGF-β. Western blotting and qRT-PCR analysis results verified the stronger expression of Sox9, aggrecan (ACAN), and collagen II in rabbit BMSCs treated with Shh or TGF-β in a microgravity environment. Low levels of chondrogenic hypertrophy, osteogenesis, and adipogenesis-related factors were detected in all groups. After transplantation in vivo, histological analysis revealed a significant improvement in cartilage and subchondral repair in the Shh transfection group. These results suggested that Shh signalling promoted chondrogenesis in rabbit BMSCs under microgravity conditions equivalent to TGF-β, and improved the early stages of the repair of cartilage and subchondral defects. Furthermore, RCCS provided a dynamic culture microenvironment conducive for cell proliferation, aggregation and differentiation." | |||
:'''Links:''' {{cartilage}} | |||
===Face=== | ===Face=== | ||
SHH is secreted by the forebrain neuroectoderm and the facial ectoderm for upper face and nasofrontal bud development. | SHH is secreted by the forebrain neuroectoderm and the facial ectoderm for upper face and nasofrontal bud development.{{#pmid:15979605|PMID15979605}} | ||
SHH from {{pharyngeal arch}} 1 epithelium is necessary for early mandibular arch cell survival and later cartilage condensation differentiation.{{#pmid:25626636|PMID25626636}} | |||
===Palate=== | |||
Components of the Shh pathway are localized in the palatal epithelium{{#pmid:19304890|PMID19304890}}{{#pmid:24590292|PMID24590292}}, though inhibition of Shh signaling in palatal epithelium does not affect palatogenesis. A recent mouse study with constitutive activation of hedgehog signaling results in submucous cleft palate and persistence of the medial edge epithelium (MEE).{{#pmid:29981310|PMID29981310}} Precise down-regulation of Shh signaling is required in the MEE during palatal fusion, persistence may prevent MEE cells {{apoptosis}}. | |||
:'''Links:''' {{palate}} | {{cleft palate}} | |||
===Integumentary=== | |||
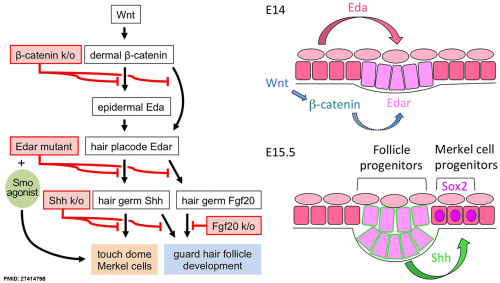
SHH is involved with the development of the Merkel cell from progenitor cells in epidermal placodes. | |||
[[File:Integumentary touch dome model 01.jpg|500px]] | |||
A molecular signalling cascade from Wnt, Eda, and Shh is required for touch dome Merkel cell development.{{#pmid:27414798|PMID27414798}} Keratin 8 (K8) and Keratin 18 (K18) are Merkel cell markers identified in late fetal and adult skin. | |||
===Other=== | ===Other=== | ||
| Line 140: | Line 206: | ||
===Non-Canonical Pathway=== | ===Non-Canonical Pathway=== | ||
Suggested that there are at least 3 other potential pathways | Suggested that there are at least 3 other potential pathways{{#pmid:19399989|PMID9399989}}: | ||
# Involves hedgehog pathway components but which is independent of GLI-mediated transcription. | # Involves hedgehog pathway components but which is independent of GLI-mediated transcription. | ||
# Direct interaction of hedgehog signaling components with components of other molecular pathways. | # Direct interaction of hedgehog signaling components with components of other molecular pathways. | ||
| Line 168: | Line 234: | ||
(HHIP) A surface receptor antagonist that is equipotent against all three mammalian Hh homologs. The structure of human HHIP is comprised of two EGF domains and a six-bladed beta-propeller domain. | (HHIP) A surface receptor antagonist that is equipotent against all three mammalian Hh homologs. The structure of human HHIP is comprised of two EGF domains and a six-bladed beta-propeller domain. | ||
HHIP1 is secreted by cells during embryo development and binds heparan sulfate (HS) extracellularly to antagonise the HH pathway and therefore restricts the HH ligand function. | HHIP1 is secreted by cells during embryo development and binds heparan sulfate (HS) extracellularly to antagonise the HH pathway and therefore restricts the HH ligand function.{{#pmid:26056142|PMID26056142}} | ||
| Line 191: | Line 257: | ||
<references/> | <references/> | ||
===Reviews=== | |||
{{#pmid:29615588}} | |||
{{#pmid:26499980}} | |||
{{#pmid:28293554}} | |||
===Articles=== | |||
{{#pmid:32829689}} | |||
{{#pmid:32826873}} | |||
{{#pmid:32612978}} | |||
{{#pmid:30638444}} | |||
{{#pmid:25626636}} | |||
{{#pmid:16492970}} | |||
===Search Pubmed=== | ===Search Pubmed=== | ||
| Line 215: | Line 303: | ||
[[Category:Sonic Hedgehog]] [[Category:Molecular]] [[Category:Pattern]] | [[Category:Sonic Hedgehog]] [[Category:Molecular]] [[Category:Pattern]] | ||
[[Category:Neural]] [[Category:Neural Crest]] [[Category:Notochord]] | [[Category:Neural]] [[Category:Neural Crest]] [[Category:Notochord]][[Category:Limb]] | ||
Latest revision as of 09:01, 2 December 2021
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The secreted growth factor sonic hedgehog (SHH, Shh) binds the patched (ptc) receptor on cell membrane. This protein has many different functions in different tissues of the developing embryo, the main role would be of establishing "pattern". SHH name derives from homology to the original fruitfly (drosophila) "hedgehog" mutation, where these flies have hairs located in regions (ventral) which are normally hair-free, and therefore have a disrupted body pattern.
Sonic Hedgehog was isolated by homology to the Hedgehog (Hh) gene. A single Hh homolog is present in Drosophila, but three Hh homologs, Sonic Hh, Indian Hh, and Desert Hh, are present in mammals.
SHH is a true signaling switch used in differentiating subpopulations of cells throughout the embryo. Depending on where the signal is being secreted, how far away the responsive cell population is and how SHH is proteolytically cleaved, will determine SHH function. SHH binds to the membrane receptors Patched (ptc) and BOC/CDON.
Nomenclature note - upper case SHH in humans, lower case shh used for other species.
| Factor Links: AMH | hCG | BMP | sonic hedgehog | bHLH | HOX | FGF | FOX | Hippo | LIM | Nanog | NGF | Nodal | Notch | PAX | retinoic acid | SIX | Slit2/Robo1 | SOX | TBX | TGF-beta | VEGF | WNT | Category:Molecular |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Sonic hedgehog | SHH | Patched | BOC/CDON Search all databases: sonic hedgehog |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Human Hedgehog Family
| Table - Human Hedgehog Family | ||||
| Approved Symbol |
Approved Name | Previous Symbols |
Synonyms | Chromosome |
|---|---|---|---|---|
| DHH | desert hedgehog | "HHG-3, MGC35145" | 12q13.12 | |
| IHH | indian hedgehog | "HHG2, BDA1" | 2q35 | |
| SHH | sonic hedgehog | "HPE3, HLP3" | "HHG1, SMMCI, TPT, TPTPS, MCOPCB5" | 7q36.3 |
| Links: sonic hedgehog | OMIM Fgf1 | HGNC | Bmp Family | Fgf Family | Hedgehog Family | Sox Family | Tbx Family | ||||
| Human Hedgehog Family | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||
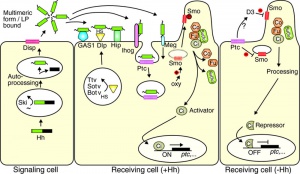
Vertebrates have a number of signaling proteins and receptors that belong to the hedgehog (Hh) family.
Signaling proteins
- Desert hedgehog (Dhh)
- Indian hedgehog (Ihh)
- Sonic hedgehog (Shh)
Receptors
- Patched homologs - Patched-1 (Ptc1) and Patched-2 (Ptc2)
- Cubitus interruptus (Ci) homologs - Gli1, Gli2 and Gli3
Functions
Developmental patterning signal.
Neural
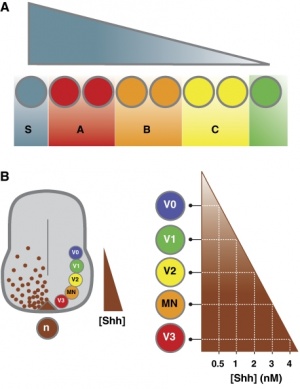
- SHH is secreted by the notochord, ventralizes the neural tube, inducing the floor plate and motor neurons.
- Regulation of patched by sonic hedgehog in the developing neural tube.[21] "The pattern of PTC expression suggests that Sonic hedgehog may play an inductive role in more dorsal regions of the neural tube than have been previously demonstrated. Examination of the pattern of PTC expression also suggests that PTC may act in a negative feedback loop to attenuate hedgehog signaling."
Neural tube cross section dorsoventral domains are established by opposing concentration gradients of Sonic Hedgehog (Shh) and Bone Morphogenic Protein (BMP).[22]
- left - These regulate progenitor gene expression. The progenitor genes cross-repress each other to establish domain boundaries.
- right - Each domain will give rise to a specific cell type that expresses various post-mitotic differentiation genes.
|
| Neural tube - SHH model[23] |
Neural Crest
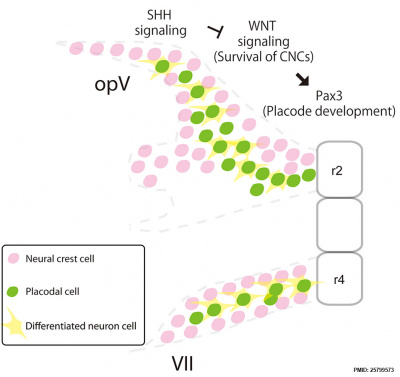
|
A model of mouse cranial nerve development[24] includes SHH action during neural crest cell migrating from rhombomere 2 (r2) or 4 (r4) interaction with placodal cells to develop the cranial nerves. |
- Neural crest cell survival in the foregut endoderm of jaw development[25]
- Sonic hedgehog is required for cardiac outflow tract and neural crest cell development.[26]
Hearing
During mouse cochlea development the adjacent auditory (spiral) ganglion acts as a source of Sonic hedgehog. Without SHH the cochlear duct is shortened, sensory hair cell precursors prematurely leave the mitotic cell cycle, and hair cell differentiation closely follows cell cycle exit in a similar apical-to-basal direction.[27]
Somite
A study has shown an essential role for Shh signalling in the control of laminin-111 synthesis and in the initiation of basement membrane assembly in the myotome.[28]
Limb

- SHH is secreted by the zone of polarizing activity (ZPA) organizing limb axis formation.
- Extended exposure to Sonic hedgehog is required for patterning the posterior digits of the vertebrate limb.[30]
|
|
| Shh mouse forelimb E11.5[31] | Shh mouse hindlimb E12.5[31] |
- Mouse Limb Images: Tbx3 and Tbx2 forelimb E10 | Alx3 and Gli3 forelimb E10 | Fgf and Hox forelimb E10.5 | Bmp4 forelimb E11.5 | Bmp4 hindlimb E11.5 | Shh forelimb E11.5 | Fgf8 hindlimb E11.5 | Sox9 forelimb E12.5 | Msx2 forelimb E12.5 | Shh hindlimb E12.5
- Links: Fgf | Hox | Shh | Sox | Limb Development | Mouse Development
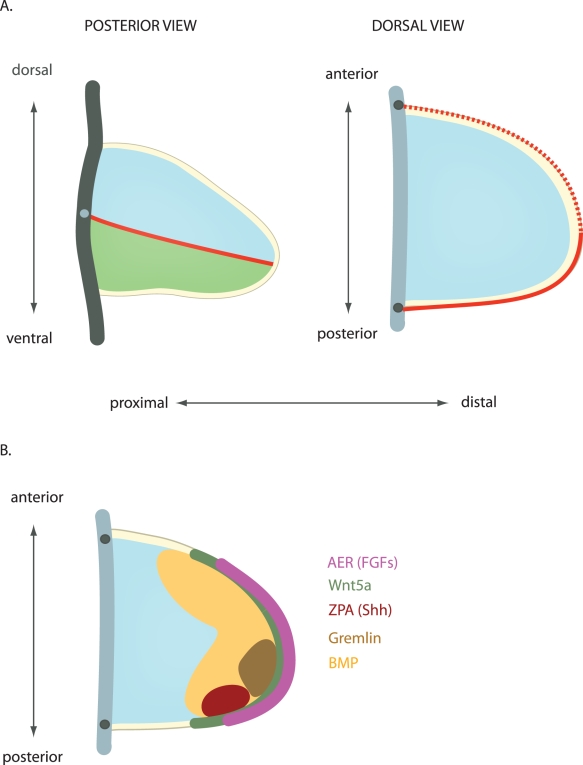
|
| Geometry and patterning of the developing limb bud[32] |
Model for GATA6 action Patterning developing Hindlimb Buds[33]
Cartilage
Sonic hedgehog promotes chondrogenesis of rabbit bone marrow stem cells in a rotary cell culture system[4] "Sonic hedgehog (Shh) is an important signalling protein involved in the induction of early cartilaginous differentiation. Herein, we demonstrate that Shh markedly induces chondrogenesis of rabbit bone marrow stromal cells (BMSCs) under microgravity conditions, and promotes cartilage regeneration. In the rotary cell culture system (RCCS), chondrogenic differentiation was revealed by stronger Toluidine Blue and collagen II immunohistochemical staining in the Shh transfection group, and chondroinductive activity of Shh was equivalent to that of TGF-β. Western blotting and qRT-PCR analysis results verified the stronger expression of Sox9, aggrecan (ACAN), and collagen II in rabbit BMSCs treated with Shh or TGF-β in a microgravity environment. Low levels of chondrogenic hypertrophy, osteogenesis, and adipogenesis-related factors were detected in all groups. After transplantation in vivo, histological analysis revealed a significant improvement in cartilage and subchondral repair in the Shh transfection group. These results suggested that Shh signalling promoted chondrogenesis in rabbit BMSCs under microgravity conditions equivalent to TGF-β, and improved the early stages of the repair of cartilage and subchondral defects. Furthermore, RCCS provided a dynamic culture microenvironment conducive for cell proliferation, aggregation and differentiation."
- Links: cartilage
Face
SHH is secreted by the forebrain neuroectoderm and the facial ectoderm for upper face and nasofrontal bud development.[34]
SHH from pharyngeal arch 1 epithelium is necessary for early mandibular arch cell survival and later cartilage condensation differentiation.[35]
Palate
Components of the Shh pathway are localized in the palatal epithelium[36][37], though inhibition of Shh signaling in palatal epithelium does not affect palatogenesis. A recent mouse study with constitutive activation of hedgehog signaling results in submucous cleft palate and persistence of the medial edge epithelium (MEE).[7] Precise down-regulation of Shh signaling is required in the MEE during palatal fusion, persistence may prevent MEE cells apoptosis.
- Links: palate | cleft palate
Integumentary
SHH is involved with the development of the Merkel cell from progenitor cells in epidermal placodes.
A molecular signalling cascade from Wnt, Eda, and Shh is required for touch dome Merkel cell development.[38] Keratin 8 (K8) and Keratin 18 (K18) are Merkel cell markers identified in late fetal and adult skin.
Other
SHH has still others roles in organ development in lung, pancreas.
Signaling Pathway
The secreted protein binds to the cell surface membrane protein the patched receptor (ptc).
Canonical Pathway
Series of repressive interactions which culminates in GLI-mediated transcriptional regulation of a variety of cellular processes.
Non-Canonical Pathway
Suggested that there are at least 3 other potential pathways[39]:
- Involves hedgehog pathway components but which is independent of GLI-mediated transcription.
- Direct interaction of hedgehog signaling components with components of other molecular pathways.
- "Non-contiguous" or "atypical" interaction of core hedgehog pathway components with one another.
SHH Receptors
Patched Receptor
In the fly, D. melanogaster, patched (ptc) is a protein ligand receptor involved in the smo receptor signalling pathway which is a component of the integral plasma membrane protein. There are homologues in Homo sapiens , Mus , Caenorhabditis elegans , Drosophila sp. , Mus musculus and Saccharomyces cerevisiae.
D. melanogaster expression in the embryo (Malpighian tubule , analia , embryonic/larval hindgut , head and 2 other listed tissues). Protein interacts genetically with fu , rho , hh , ci , gsb , B , kn , N , l(1)sc , smo , Su(fu) and vn . There are 62 recorded mutant alleles , of which at least 16 are available from the public stock centers. Amorphic mutations have been isolated which affect the anterior wing , the costal cell , the wing vein and 8 other listed tissues and are embryonic lethal, visible and tissue polarity. ptc is discussed in 325 published references , dated between 1948 and 1999. These include at least 58 studies of mutant phenotypes , one study of wild-type function and 4 molecular studies . Among findings on ptc mutants, ptc mutant analysis and stage-specific laser inactivation of ptc protein indicates that ptc activity is functionally redeployed after the segmentation phenocritical period to discriminate between neural and epithelial cell fates. (Some text modified from Flybase entry for Hedgehog Gene and refers to the fruitfly hh gene)
BOC/CDON Receptors
Two recently identified related receptors for SHH, Boc and Cdon are cell surface receptors of the immunoglobulin (Ig)/fibronectin type III that interact with each other and are coexpressed in development.
- Brother of Cdon (BOC) OMIM 608708
- Cell adhesion molecule-regulated/downregulated by oncogenes (CDON) OMIM 608707
Megalin
In development, it has been shown that sonic hedgehog can also bind megalin (lipoprotein receptor-related protein-2, LRP2, gp330) a transmembrane protein which acts as an endocytic receptor on the apical surface of polarised epithelial cells. It requires interaction with another protein, cubulin, for the endocytosis of ligands.
- Megalin OMIM 600073
- Cubulin OMIM 602997
Hedgehog-Interacting Protein
(HHIP) A surface receptor antagonist that is equipotent against all three mammalian Hh homologs. The structure of human HHIP is comprised of two EGF domains and a six-bladed beta-propeller domain.
HHIP1 is secreted by cells during embryo development and binds heparan sulfate (HS) extracellularly to antagonise the HH pathway and therefore restricts the HH ligand function.[40]
Abnormalities
Holoprosencephaly
Defects in the SHH protein or its signalling pathway are a cause of Holoprosencephaly (International Classification of Diseases - Q04 Other congenital malformations of brain - Q04.2 Holoprosencephaly).
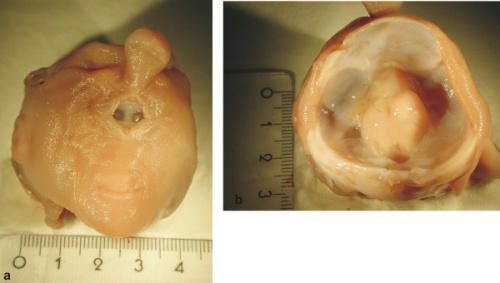
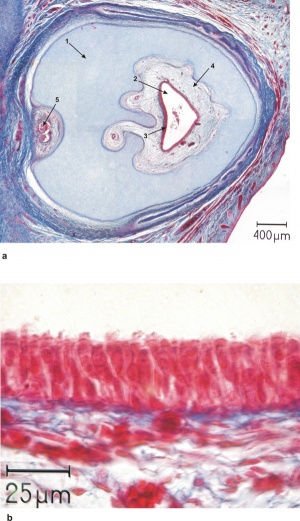
Below are shown images of human fetal holoprosencephaly and associated cyclopia.[41]
|
|
| Human holoprosencephaly cyclopean dissection | Proboscis histology |
- Links: Human holoprosencephaly cyclopia dissection | Proboscis histology | Holoprosencephaly | Neural Abnormalities | Sonic hedgehog
References
- ↑ Sanders TA, Llagostera E & Barna M. (2013). Specialized filopodia direct long-range transport of SHH during vertebrate tissue patterning. Nature , 497, 628-32. PMID: 23624372 DOI.
- ↑ Fujii K, Zhulyn O, Byeon GW, Genuth NR, Kerr CH, Walsh EM & Barna M. (2021). Controlling tissue patterning by translational regulation of signaling transcripts through the core translation factor eIF3c. Dev Cell , 56, 2928-2937.e9. PMID: 34752747 DOI.
- ↑ Yabut OR, Ng HX, Yoon K, Arela JC, Ngo T & Pleasure SJ. (2020). The neocortical progenitor specification program is established through combined modulation of SHH and FGF signaling. J. Neurosci. , , . PMID: 32737167 DOI.
- ↑ 4.0 4.1 Chen L, Liu G, Li W & Wu X. (2019). Sonic hedgehog promotes chondrogenesis of rabbit bone marrow stem cells in a rotary cell culture system. BMC Dev. Biol. , 19, 18. PMID: 31401976 DOI.
- ↑ Sagai T, Amano T, Maeno A, Ajima R & Shiroishi T. (2019). SHH signaling mediated by a prechordal and brain enhancer controls forebrain organization. Proc. Natl. Acad. Sci. U.S.A. , , . PMID: 31685615 DOI.
- ↑ Ankamreddy H, Min H, Kim JY, Yang X, Cho ES, Kim UK & Bok J. (2019). Region-specific endodermal signals direct neural crest cells to form the three middle ear ossicles. Development , 146, . PMID: 30630826 DOI.
- ↑ 7.0 7.1 Li J, Yuan Y, He J, Feng J, Han X, Jing J, Ho TV, Xu J & Chai Y. (2018). Constitutive activation of hedgehog signaling adversely affects epithelial cell fate during palatal fusion. Dev. Biol. , 441, 191-203. PMID: 29981310 DOI.
- ↑ Qi X, Schmiege P, Coutavas E, Wang J & Li X. (2018). Structures of human Patched and its complex with native palmitoylated sonic hedgehog. Nature , , . PMID: 29995851 DOI.
- ↑ Cheng FY, Fleming JT & Chiang C. (2018). Bergmann glial Sonic hedgehog signaling activity is required for proper cerebellar cortical expansion and architecture. Dev. Biol. , 440, 152-166. PMID: 29792854 DOI.
- ↑ Kremnyov S, Henningfeld K, Viebahn C & Tsikolia N. (2018). Divergent axial morphogenesis and earlyshhexpression in vertebrate prospective floor plate. Evodevo , 9, 4. PMID: 29423139 DOI.
- ↑ Lin YC, Roffler SR, Yan YT & Yang RB. (2015). Disruption of Scube2 Impairs Endochondral Bone Formation. J. Bone Miner. Res. , 30, 1255-67. PMID: 25639508 DOI.
- ↑ Kozhemyakina E, Ionescu A & Lassar AB. (2014). GATA6 is a crucial regulator of Shh in the limb bud. PLoS Genet. , 10, e1004072. PMID: 24415953 DOI.
- ↑ Nahmad M & Stathopoulos A. (2009). Dynamic interpretation of hedgehog signaling in the Drosophila wing disc. PLoS Biol. , 7, e1000202. PMID: 19787036 DOI.
- ↑ Butterfield NC, Metzis V, McGlinn E, Bruce SJ, Wainwright BJ & Wicking C. (2009). Patched 1 is a crucial determinant of asymmetry and digit number in the vertebrate limb. Development , 136, 3515-24. PMID: 19783740 DOI.
- ↑ Zhu J, Nakamura E, Nguyen MT, Bao X, Akiyama H & Mackem S. (2008). Uncoupling Sonic hedgehog control of pattern and expansion of the developing limb bud. Dev. Cell , 14, 624-32. PMID: 18410737 DOI.
- ↑ Tran PV, Haycraft CJ, Besschetnova TY, Turbe-Doan A, Stottmann RW, Herron BJ, Chesebro AL, Qiu H, Scherz PJ, Shah JV, Yoder BK & Beier DR. (2008). THM1 negatively modulates mouse sonic hedgehog signal transduction and affects retrograde intraflagellar transport in cilia. Nat. Genet. , 40, 403-410. PMID: 18327258 DOI.
- ↑ Sun M, Ma F, Zeng X, Liu Q, Zhao XL, Wu FX, Wu GP, Zhang ZF, Gu B, Zhao YF, Tian SH, Lin B, Kong XY, Zhang XL, Yang W, Lo WH & Zhang X. (2008). Triphalangeal thumb-polysyndactyly syndrome and syndactyly type IV are caused by genomic duplications involving the long range, limb-specific SHH enhancer. J. Med. Genet. , 45, 589-95. PMID: 18417549 DOI.
- ↑ Chamberlain CE, Jeong J, Guo C, Allen BL & McMahon AP. (2008). Notochord-derived Shh concentrates in close association with the apically positioned basal body in neural target cells and forms a dynamic gradient during neural patterning. Development , 135, 1097-106. PMID: 18272593 DOI.
- ↑ Bürglin TR. (2008). The Hedgehog protein family. Genome Biol. , 9, 241. PMID: 19040769 DOI.
- ↑ Fogarty P, Campbell SD, Abu-Shumays R, Phalle BS, Yu KR, Uy GL, Goldberg ML & Sullivan W. (1997). The Drosophila grapes gene is related to checkpoint gene chk1/rad27 and is required for late syncytial division fidelity. Curr. Biol. , 7, 418-26. PMID: 9197245
- ↑ Marigo V & Tabin CJ. (1996). Regulation of patched by sonic hedgehog in the developing neural tube. Proc. Natl. Acad. Sci. U.S.A. , 93, 9346-51. PMID: 8790332
- ↑ Zannino DA & Sagerström CG. (2015). An emerging role for prdm family genes in dorsoventral patterning of the vertebrate nervous system. Neural Dev , 10, 24. PMID: 26499851 DOI.
- ↑ Dessaud E, Ribes V, Balaskas N, Yang LL, Pierani A, Kicheva A, Novitch BG, Briscoe J & Sasai N. (2010). Dynamic assignment and maintenance of positional identity in the ventral neural tube by the morphogen sonic hedgehog. PLoS Biol. , 8, e1000382. PMID: 20532235 DOI.
- ↑ Kurosaka H, Trainor PA, Leroux-Berger M & Iulianella A. (2015). Cranial nerve development requires co-ordinated Shh and canonical Wnt signaling. PLoS ONE , 10, e0120821. PMID: 25799573 DOI.
- ↑ Brito JM, Teillet MA & Le Douarin NM. (2006). An early role for sonic hedgehog from foregut endoderm in jaw development: ensuring neural crest cell survival. Proc. Natl. Acad. Sci. U.S.A. , 103, 11607-12. PMID: 16868080 DOI.
- ↑ Washington Smoak I, Byrd NA, Abu-Issa R, Goddeeris MM, Anderson R, Morris J, Yamamura K, Klingensmith J & Meyers EN. (2005). Sonic hedgehog is required for cardiac outflow tract and neural crest cell development. Dev. Biol. , 283, 357-72. PMID: 15936751 DOI.
- ↑ Bok J, Zenczak C, Hwang CH & Wu DK. (2013). Auditory ganglion source of Sonic hedgehog regulates timing of cell cycle exit and differentiation of mammalian cochlear hair cells. Proc. Natl. Acad. Sci. U.S.A. , 110, 13869-74. PMID: 23918393 DOI.
- ↑ Anderson C, Thorsteinsdóttir S & Borycki AG. (2009). Sonic hedgehog-dependent synthesis of laminin alpha1 controls basement membrane assembly in the myotome. Development , 136, 3495-504. PMID: 19783738 DOI.
- ↑ Galli A, Robay D, Osterwalder M, Bao X, Bénazet JD, Tariq M, Paro R, Mackem S & Zeller R. (2010). Distinct roles of Hand2 in initiating polarity and posterior Shh expression during the onset of mouse limb bud development. PLoS Genet. , 6, e1000901. PMID: 20386744 DOI.
- ↑ Scherz PJ, McGlinn E, Nissim S & Tabin CJ. (2007). Extended exposure to Sonic hedgehog is required for patterning the posterior digits of the vertebrate limb. Dev. Biol. , 308, 343-54. PMID: 17610861 DOI.
- ↑ 31.0 31.1 Bandyopadhyay A, Tsuji K, Cox K, Harfe BD, Rosen V & Tabin CJ. (2006). Genetic analysis of the roles of BMP2, BMP4, and BMP7 in limb patterning and skeletogenesis. PLoS Genet. , 2, e216. PMID: 17194222 DOI.
- ↑ Kicheva A & Briscoe J. (2010). Limbs made to measure. PLoS Biol. , 8, e1000421. PMID: 20644713 DOI.
- ↑ Kozhemyakina E, Ionescu A & Lassar AB. (2014). GATA6 is a crucial regulator of Shh in the limb bud. PLoS Genet. , 10, e1004072. PMID: 24415953 DOI.
- ↑ Marcucio RS, Cordero DR, Hu D & Helms JA. (2005). Molecular interactions coordinating the development of the forebrain and face. Dev. Biol. , 284, 48-61. PMID: 15979605 DOI.
- ↑ Billmyre KK & Klingensmith J. (2015). Sonic hedgehog from pharyngeal arch 1 epithelium is necessary for early mandibular arch cell survival and later cartilage condensation differentiation. Dev. Dyn. , 244, 564-76. PMID: 25626636 DOI.
- ↑ Lan Y & Jiang R. (2009). Sonic hedgehog signaling regulates reciprocal epithelial-mesenchymal interactions controlling palatal outgrowth. Development , 136, 1387-96. PMID: 19304890 DOI.
- ↑ Kurosaka H, Iulianella A, Williams T & Trainor PA. (2014). Disrupting hedgehog and WNT signaling interactions promotes cleft lip pathogenesis. J. Clin. Invest. , 124, 1660-71. PMID: 24590292 DOI.
- ↑ Xiao Y, Thoresen DT, Miao L, Williams JS, Wang C, Atit RP, Wong SY & Brownell I. (2016). A Cascade of Wnt, Eda, and Shh Signaling Is Essential for Touch Dome Merkel Cell Development. PLoS Genet. , 12, e1006150. PMID: 27414798 DOI.
- ↑ Jenkins D. (2009). Hedgehog signalling: emerging evidence for non-canonical pathways. Cell. Signal. , 21, 1023-34. PMID: 19399989
- ↑ Holtz AM, Griffiths SC, Davis SJ, Bishop B, Siebold C & Allen BL. (2015). Secreted HHIP1 interacts with heparan sulfate and regulates Hedgehog ligand localization and function. J. Cell Biol. , 209, 739-57. PMID: 26056142 DOI.
- ↑ <pubmed>19563629</pubmed>| PMC2709107
Reviews
Dworkin S, Boglev Y, Owens H & Goldie SJ. (2016). The Role of Sonic Hedgehog in Craniofacial Patterning, Morphogenesis and Cranial Neural Crest Survival. J Dev Biol , 4, . PMID: 29615588 DOI.
De Luca A, Cerrato V, Fucà E, Parmigiani E, Buffo A & Leto K. (2016). Sonic hedgehog patterning during cerebellar development. Cell. Mol. Life Sci. , 73, 291-303. PMID: 26499980 DOI.
Tickle C & Towers M. (2017). Sonic Hedgehog Signaling in Limb Development. Front Cell Dev Biol , 5, 14. PMID: 28293554 DOI.
Articles
Groves I, Placzek M & Fletcher AG. (2020). Of mitogens and morphogens: modelling Sonic Hedgehog mechanisms in vertebrate development. Philos. Trans. R. Soc. Lond., B, Biol. Sci. , 375, 20190660. PMID: 32829689 DOI.
Liao H, Cai J, Liu C, Shen L, Pu X, Yao Y, Han B, Yu T, Cheng SY & Yue S. (2020). Protein phosphatase 4 promotes Hedgehog signaling through dephosphorylation of Suppressor of fused. Cell Death Dis , 11, 686. PMID: 32826873 DOI.
Yun T, Wang J, Yang J, Huang W, Lai L, Tan W & Liu Y. (2020). Discovery of Small Molecule Inhibitors Targeting the Sonic Hedgehog. Front Chem , 8, 498. PMID: 32612978 DOI.
Xu J, Liu H, Lan Y, Adam M, Clouthier DE, Potter S & Jiang R. (2019). Hedgehog signaling patterns the oral-aboral axis of the mandibular arch. Elife , 8, . PMID: 30638444 DOI.
Billmyre KK & Klingensmith J. (2015). Sonic hedgehog from pharyngeal arch 1 epithelium is necessary for early mandibular arch cell survival and later cartilage condensation differentiation. Dev. Dyn. , 244, 564-76. PMID: 25626636 DOI.
Yamagishi C, Yamagishi H, Maeda J, Tsuchihashi T, Ivey K, Hu T & Srivastava D. (2006). Sonic hedgehog is essential for first pharyngeal arch development. Pediatr. Res. , 59, 349-54. PMID: 16492970 DOI.
Search Pubmed
Search Bookshelf sonic hedgehog
Search Term: Sonic hedgehog | SHH | Patched Receptor
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- OMIM - Sonic hedgehog
- EMBL UniProt - Sonic hedgehog protein
- PubMed - Crystal Structure Of Human Sonic Hedgehog N-Terminal Domain
- Genetics Home Reference. - SHH
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Developmental Signals - Sonic hedgehog. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Signals_-_Sonic_hedgehog
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G