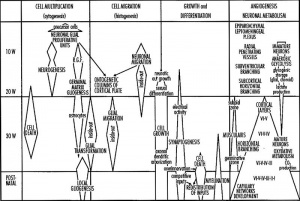
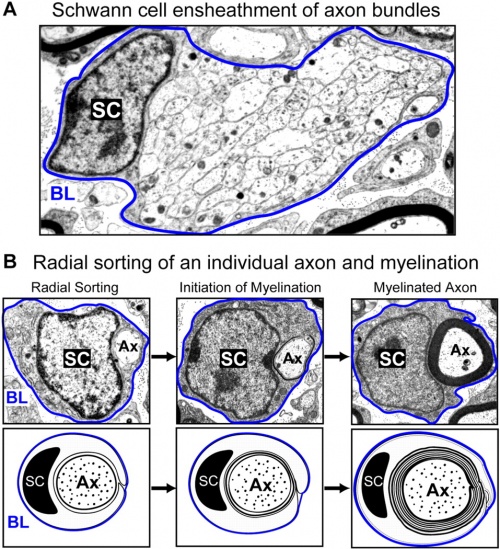
Neural System - Glial Development
| Embryology - 25 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The term gliogenesis refers to the development of the many different types of glial cells within the developing and adult central and peripheral nervous systems (CNS and PNS) including: radial glia, astrocytes, oligodendrocytes, Schwann cells, and microglia. Within the CNS the neural stem cells generate firstly neutrons and then most of the central gila cells. Note that PNS Schwann cells have a different embryonic origin (neural crest).
Glia (Greek, glia = "glue") and neurons have the same general embryonic origin, generated from neural tube ventricular layer stem cells and neural crest. The developmental process of glial cell development is described as gliogenesis. Glial cells have important roles in neural development and in the adult nervous system and have come a long way from their original description as "supportive cells".
Myelination is the process of close wrapping around a neural axon by a glial cell. This second process occurs as a late feature of glial and nervous system development in both the central and peripheral nervous system and has mainly been studied in relation to demyelinating diseases, such as multiple sclerosis.
Development of the neural crest and sensory systems (hearing/vision/smell) are only briefly introduced in these notes and are covered in detail in another notes sections. (More? neural crest | sensory).
| Neural Links: ectoderm | neural | neural crest | ventricular | sensory | Stage 22 | gliogenesis | neural fetal | Medicine Lecture - Neural | Lecture - Ectoderm | Lecture - Neural Crest | Lab - Early Neural | neural abnormalities | folic acid | iodine deficiency | Fetal Alcohol Syndrome | neural postnatal | neural examination | Histology | Historic Neural | Category:Neural |
Neural Crest Development | Sensory System Development
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Glial Embryology | Glial Development | Radial Glia | Astrocyte Development | Schwann Cell Development | Microglia Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Radial Glia
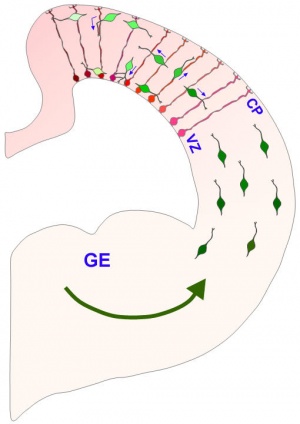
Radial glia were historically identified by Wilhelm His in 1880's studies of the human fetal brain using classical Golgi silver impregnation histology and were postulated to have a role in the developing central nervous system. It was not until Paso Rakic's much later studies in the 1970's[8][9] (see review[10]) that this roel was confirmed. These cells have an important role in nervous system development, guiding newly formed neurons from their birth zone (ventricular layer) outward to their final adult position. Radial glia generate initially neurons then astrocytes after neurogenesis has been completed.[11][12]
At about 19 weeks (GA 21 weeks) neuronal migration ends and the radial glial cells that aided the migration now become transformed into astrocytes and astrocytic precursors.[13]
- Links: PubMed - Pasko Rakic
Astroglia
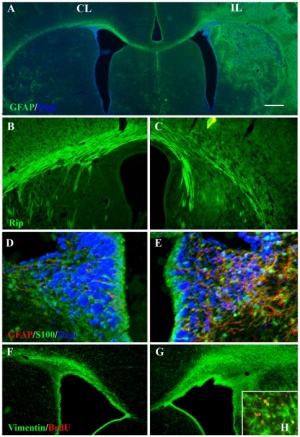
Astrocytes are glial cells named by their "star-like" branching appearance, and are the most abundant cells in the brain. They form a key component of the blood-brain barrier and the transfer of circulating nutrients to neurons.
Blood-Brain Barrier
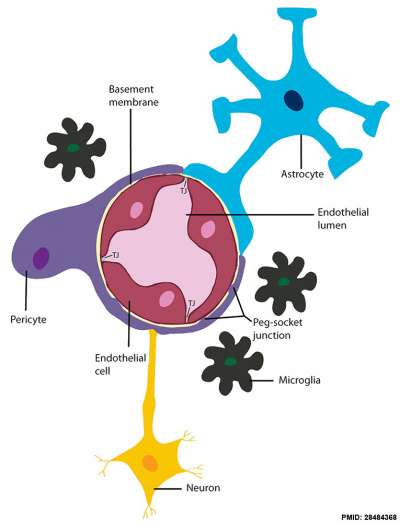
Blood-brain barrier cartoon[15] |
A layer of brain endothelial cells connected by tight junctions forms the blood-brain barrier (BBB). The intimate contact of these specialized endothelial cells with different cell types constitutes the neurovascular unit (NVU).
|
See reviews:
- Steindler DA, Laywell ED.] Astrocytes as stem cells: nomenclature, phenotype, and translation.[16]
- Scemes E, Giaume C. Astrocyte calcium waves: what they are and what they do. [17]
Oligodendroglia
In the early spinal cord, a ventral ventricular zone region generates oligodendrocyte precursors initially which then migrate both laterally and dorsally.
In the later spinal cord, the dorsal region provides a secondary source of oligodendrocyte precursors.
Oligodendrocyte wars. Richardson WD, Kessaris N, Pringle N. Nat Rev Neurosci. 2006 Jan;7(1):11-8. Review.
Baron W, Colognato H, ffrench-Constant C. Integrin-growth factor interactions as regulators of oligodendroglial development and function. Glia. 2005 Mar;49(4):467-79. Review.
Microglia
Glial cells that act within the central nervous system in the same role as macrophages (Mϕ) in other body tissues and act as an innate immune system. There other role is as the cellular mediators of neuroinflammatory processes[18] and cell death[19]. Microglia have also been suggested as having a role during brain developmental synaptogenesis.[20]
Human
In human development, microglia are present in the extracerebral mesenchyme at 4.5 weeks, invade the parenchyma during week 5, and can then be identified at several different neural locations.[21][22]
Microglia Developmental Location[23][21]
- Dorsal to the diencephalon–telencephalon fissure (behind cephalic flexure)
- At the level of the eminentia thalami, near the choroid plexus
- Edge of the ventricular zone (ganglionic eminence)
- Near or in the optic tract, ventral to the diencephalic–telencephalic fissure
- In the internal capsule
- At the genu of the internal capsule
- At the junction between the anterior limb of the internal capsule and the external capsule
- At the junction between the posterior limb of the internal capsule and the cerebral peduncle
- In the medial septal area
- Along the third ventricle in the hypothalamic area
Mouse
In mouse development, microglia derive only from you sac progenitor cells migrating and then proliferating within the CNS.[24][25][1] Microglia differentiation requires The hematopoietic transcription factor PU.1 and interferon regulatory factor IRF8 pathways.[26]
- Search PubMed: Microglia Development | Microglia Differentiation
Schwann cells
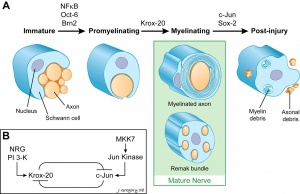
These cells are named after Theodor Schwann (1810 - 1882), a German physiologist and histologist, who along with Schleiden were early developers of the "cell theory". Neural crest cells differentiate to form the glial lineage, which in turn generate Schwann cell precursors. (More? Neural Crest Development).
Schwann cells formation is regulated by at least two signals, neuregulin-1 and endothelin. Neuregulins are a family (NRG1, NRG2, NRG3, and NRG4) of EGF-like signaling molecules that bind ErbB receptor tyrosine kinase receptors. Neuregulin-1 type III is expressed on axon surface and has been shown to also regulate Schwann cell membrane growth, adjusting myelin sheath thickness to match axonal calibre.
Mouse - sciatic nerve Schwann cell interaction[28]
- Links: Neural Crest Development
Development Overview
Human Neuralation - Early Stages
The stages below refer to specific Carneigie stages of development.
- stage 8 (about 18 postovulatory days) neural groove and folds are first seen
- stage 9 the three main divisions of the brain, which are not cerebral vesicles, can be distinguished while the neural groove is still completely open.
- stage 10 (two days later) neural folds begin to fuse near the junction between brain and spinal cord, when neural crest cells are arising mainly from the neural ectoderm
- stage 11 (about 24 days) the rostral (or cephalic) neuropore closes within a few hours
- closure is bidirectional
- it takes place from the dorsal and terminal lips and may occur in several areas simultaneously
- The two lips, however, behave differently.
- stage 12 (about 26 days) The caudal neuropore takes a day to close
- the level of final closure is approximately at future somitic pair 31
- corresponds to the level of sacral vertebra 2
- stage 13 (4 weeks) the neural tube is normally completely closed
- Secondary neurulation begins at stage 12
- is the differentiation of the caudal part of the neural tube from the caudal eminence (or end-bud) without the intermediate phase of a neural plate.
(Text modified from: Neurulation in the normal human embryo. O'Rahilly R, Muller F Ciba Found Symp 1994;181:70-82)
Spinal Cord Glia
Radial glia, astrocytes, oligodendrocytes and microglia have been reported in the human spinal cord from the end of the embryonic period (GA week 10).
Table data below from an imnuohistological study of the human fetal spinal cord.[29]
| week | GA week | Staining | Location |
|---|---|---|---|
| 10 | 12 | vimentin-positive radial glia | radial glia present at all three levels of the spinal cord |
| 13 | 15 | vimentin-positive processes radially arranged in the white matter | astrocytes (GFAP+) obvious in the anterior and anterolateral funiculi |
| 18 | 20 | gradients of glial fibrillary acidic protein and vimentin expression difficult to discern | microglia more abundant in white matter than grey matter |
Fetal - Third Trimester
Three-dimensional magnetic resonance imaging and image-processing algorithms have been used to quantitate between 29-41 weeks volumes of: total brain, cerebral gray matter, unmyelinated white matter, myelinated, and cerebrospinal fluid (grey matter- mainly neuronal cell bodies; white matter- mainly neural processes and glia). A study of 78 premature and mature newborns showed that total brain tissue volume increased linearly over this period at a rate of 22 ml/week. Total grey matter also showed a linear increase in relative intracranial volume of approximately 1.4% or 15 ml/week. The rapid increase in total grey matter is mainly due to a fourfold increase in cortical grey matter. Quantification of extracerebral and intraventricular CSF was found to change only minimally.[30]
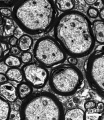
Myelination
The electron micrograph images below are selected neural tissues of adult mouse axon cross-sections surrounded by glial-derived myelin (black rings).
Links: http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=bnchm&part=A244
Postnatal Neural
Neural development continues after birth with substantial growth, death and reorganization occuring during the postnatally. (More? Postnatal Development - Neural) The references below give a sample of some recent findings and research methods.
Cortex Matures Faster in Youth with Highest IQ (More? NIH - Cortex Matures Faster in Youth with Highest IQ)
- Neural induction: old problem, new findings, yet more questions.[31] "During neural induction, the embryonic neural plate is specified and set aside from other parts of the ectoderm. A popular molecular explanation is the 'default model' of neural induction, which proposes that ectodermal cells give rise to neural plate if they receive no signals at all, while BMP activity directs them to become epidermis. However, neural induction now appears to be more complex than once thought, and can no longer be fully explained by the default model alone. This review summarizes neural induction events in different species and highlights some unanswered questions about this important developmental process."
- Diffusion tensor imaging of neurodevelopment in children and young adults.[32] "Diffusion tensor magnetic resonance imaging (DTI) was used to study regional changes in the brain's development from childhood (8-12 years, mean 11.1 +/- 1.3, N = 32) to young adulthood (21-27 years, mean 24.4 +/- 1.8, N = 28). ..... These findings suggest a continuation of the brain's microstructural development through adolescence."
Abnormalities
Multiple Sclerosis
Only humans spontaneously develop multiple sclerosis (MS)[33], a chronic demyelinating immune-mediated disease. This disease has an onset generally coinciding with the end of the long-term myelination process[34] and incidence has been recently increasing in female/male (F/M) ratio and occurring in women of childbearing age. [35]
Search Pubmed: Multiple Sclerosis
Experimental Autoimmune Encephalomyelitis
(EAE) This is an animal model of autoimmune demyelination, such as in multiple sclerosis (MS).[36]
Nogo (= Reticulon 4, RTN4, Neurite Growth Inhibitor 220) one of several myelin-associated proteins with inhibitory effects for neuronal neurite outgrowth. Nogo exists as 3 splice transcript variants (NOGO-A, NOGO-B and NOGO-C) which are differentially expressed in the developing central nervous system. Also associated with autoimmune demyelination, shown in models of multiple sclerosis (MS) such as experimental autoimmune encephalomyelitis (EAE).
Nogo-A myelin-associated protein which can inhibit neurite outgrowth and prevent regeneration in the adult central nervous system. Secreted by oligodendrocytes in the central nervous system, but not by Schwann cells in the peripheral nervous system. (More? OMIM - Reticulon 4)
Glioma
References
- ↑ 1.0 1.1 Grassivaro F, Menon R, Acquaviva M, Ottoboni L, Ruffini F, Bergamaschi A, Muzio L, Farina C & Martino G. (2019). Convergence between microglia and peripheral macrophages phenotype during development and neuroinflammation. J. Neurosci. , , . PMID: 31818979 DOI.
- ↑ Antonson AM, Lawson MA, Caputo MP, Matt SM, Leyshon BJ & Johnson RW. (2019). Maternal viral infection causes global alterations in porcine fetal microglia. Proc. Natl. Acad. Sci. U.S.A. , 116, 20190-20200. PMID: 31527230 DOI.
- ↑ Hashimoto H, Jiang W, Yoshimura T, Moon KH, Bok J & Ikenaka K. (2018). Strong sonic hedgehog signaling in the mouse ventral spinal cord is not required for oligodendrocyte precursor cell (OPC) generation but is necessary for correct timing of its generation. Neurochem. Int. , 119, 178-183. PMID: 29122585 DOI.
- ↑ Brancaccio M, Pivetta C, Granzotto M, Filippis C & Mallamaci A. (2010). Emx2 and Foxg1 inhibit gliogenesis and promote neuronogenesis. Stem Cells , 28, 1206-18. PMID: 20506244 DOI.
- ↑ Rompani SB & Cepko CL. (2010). A common progenitor for retinal astrocytes and oligodendrocytes. J. Neurosci. , 30, 4970-80. PMID: 20371817 DOI.
- ↑ Goetz AK, Scheffler B, Chen HX, Wang S, Suslov O, Xiang H, Brüstle O, Roper SN & Steindler DA. (2006). Temporally restricted substrate interactions direct fate and specification of neural precursors derived from embryonic stem cells. Proc. Natl. Acad. Sci. U.S.A. , 103, 11063-8. PMID: 16832065 DOI.
- ↑ Yokota Y, Gashghaei HT, Han C, Watson H, Campbell KJ & Anton ES. (2007). Radial glial dependent and independent dynamics of interneuronal migration in the developing cerebral cortex. PLoS ONE , 2, e794. PMID: 17726524 DOI.
- ↑ Rakic P. (1971). Guidance of neurons migrating to the fetal monkey neocortex. Brain Res. , 33, 471-6. PMID: 5002632
- ↑ Rakic P. (1972). Mode of cell migration to the superficial layers of fetal monkey neocortex. J. Comp. Neurol. , 145, 61-83. PMID: 4624784 DOI.
- ↑ Rakic P. (2003). Elusive radial glial cells: historical and evolutionary perspective. Glia , 43, 19-32. PMID: 12761862 DOI.
- ↑ Kriegstein AR & Götz M. (2003). Radial glia diversity: a matter of cell fate. Glia , 43, 37-43. PMID: 12761864 DOI.
- ↑ Nadarajah B. (2003). Radial glia and somal translocation of radial neurons in the developing cerebral cortex. Glia , 43, 33-6. PMID: 12761863 DOI.
- ↑ Kadhim HJ, Gadisseux JF & Evrard P. (1988). Topographical and cytological evolution of the glial phase during prenatal development of the human brain: histochemical and electron microscopic study. J. Neuropathol. Exp. Neurol. , 47, 166-88. PMID: 3339373
- ↑ Bain JM, Ziegler A, Yang Z, Levison SW & Sen E. (2010). TGFbeta1 stimulates the over-production of white matter astrocytes from precursors of the "brain marrow" in a rodent model of neonatal encephalopathy. PLoS ONE , 5, e9567. PMID: 20221422 DOI.
- ↑ Haddad-Tóvolli R, Dragano NRV, Ramalho AFS & Velloso LA. (2017). Development and Function of the Blood-Brain Barrier in the Context of Metabolic Control. Front Neurosci , 11, 224. PMID: 28484368 DOI.
- ↑ Steindler DA & Laywell ED. (2003). Astrocytes as stem cells: nomenclature, phenotype, and translation. Glia , 43, 62-9. PMID: 12761868 DOI.
- ↑ Scemes E & Giaume C. (2006). Astrocyte calcium waves: what they are and what they do. Glia , 54, 716-25. PMID: 17006900 DOI.
- ↑ Streit WJ, Mrak RE & Griffin WS. (2004). Microglia and neuroinflammation: a pathological perspective. J Neuroinflammation , 1, 14. PMID: 15285801 DOI.
- ↑ Bessis A, Béchade C, Bernard D & Roumier A. (2007). Microglial control of neuronal death and synaptic properties. Glia , 55, 233-8. PMID: 17106878 DOI.
- ↑ Michell-Robinson MA, Touil H, Healy LM, Owen DR, Durafourt BA, Bar-Or A, Antel JP & Moore CS. (2015). Roles of microglia in brain development, tissue maintenance and repair. Brain , 138, 1138-59. PMID: 25823474 DOI.
- ↑ 21.0 21.1 Monier A, Adle-Biassette H, Delezoide AL, Evrard P, Gressens P & Verney C. (2007). Entry and distribution of microglial cells in human embryonic and fetal cerebral cortex. J. Neuropathol. Exp. Neurol. , 66, 372-82. PMID: 17483694 DOI.
- ↑ Verney C, Monier A, Fallet-Bianco C & Gressens P. (2010). Early microglial colonization of the human forebrain and possible involvement in periventricular white-matter injury of preterm infants. J. Anat. , 217, 436-48. PMID: 20557401 DOI.
- ↑ Monier A, Evrard P, Gressens P & Verney C. (2006). Distribution and differentiation of microglia in the human encephalon during the first two trimesters of gestation. J. Comp. Neurol. , 499, 565-82. PMID: 17029271 DOI.
- ↑ Alliot F, Godin I & Pessac B. (1999). Microglia derive from progenitors, originating from the yolk sac, and which proliferate in the brain. Brain Res. Dev. Brain Res. , 117, 145-52. PMID: 10567732
- ↑ Pont-Lezica L, Béchade C, Belarif-Cantaut Y, Pascual O & Bessis A. (2011). Physiological roles of microglia during development. J. Neurochem. , 119, 901-8. PMID: 21951310 DOI.
- ↑ Kierdorf K, Erny D, Goldmann T, Sander V, Schulz C, Perdiguero EG, Wieghofer P, Heinrich A, Riemke P, Hölscher C, Müller DN, Luckow B, Brocker T, Debowski K, Fritz G, Opdenakker G, Diefenbach A, Biber K, Heikenwalder M, Geissmann F, Rosenbauer F & Prinz M. (2013). Microglia emerge from erythromyeloid precursors via Pu.1- and Irf8-dependent pathways. Nat. Neurosci. , 16, 273-80. PMID: 23334579 DOI.
- ↑ Salzer JL. (2008). Switching myelination on and off. J. Cell Biol. , 181, 575-7. PMID: 18490509 DOI.
- ↑ Chan JR. (2007). Myelination: all about Rac 'n' roll. J. Cell Biol. , 177, 953-5. PMID: 17576794 DOI.
- ↑ Weidenheim KM, Epshteyn I, Rashbaum WK & Lyman WD. (1994). Patterns of glial development in the human foetal spinal cord during the late first and second trimester. J. Neurocytol. , 23, 343-53. PMID: 7522270
- ↑ Hüppi PS, Warfield S, Kikinis R, Barnes PD, Zientara GP, Jolesz FA, Tsuji MK & Volpe JJ. (1998). Quantitative magnetic resonance imaging of brain development in premature and mature newborns. Ann. Neurol. , 43, 224-35. PMID: 9485064 DOI.
- ↑ Stern CD. (2005). Neural induction: old problem, new findings, yet more questions. Development , 132, 2007-21. PMID: 15829523 DOI.
- ↑ Matsuoka T, Ahlberg PE, Kessaris N, Iannarelli P, Dennehy U, Richardson WD, McMahon AP & Koentges G. (2005). Neural crest origins of the neck and shoulder. Nature , 436, 347-55. PMID: 16034409 DOI.
- ↑ Bove RM. (2018). Why monkeys do not get multiple sclerosis (spontaneously): An evolutionary approach. Evol Med Public Health , 2018, 43-59. PMID: 29492266 DOI.
- ↑ Mount CW & Monje M. (2017). Wrapped to Adapt: Experience-Dependent Myelination. Neuron , 95, 743-756. PMID: 28817797 DOI.
- ↑ Trojano M, Lucchese G, Graziano G, Taylor BV, Simpson S, Lepore V, Grand'maison F, Duquette P, Izquierdo G, Grammond P, Amato MP, Bergamaschi R, Giuliani G, Boz C, Hupperts R, Van Pesch V, Lechner-Scott J, Cristiano E, Fiol M, Oreja-Guevara C, Saladino ML, Verheul F, Slee M, Paolicelli D, Tortorella C, D'Onghia M, Iaffaldano P, Direnzo V & Butzkueven H. (2012). Geographical variations in sex ratio trends over time in multiple sclerosis. PLoS ONE , 7, e48078. PMID: 23133550 DOI.
- ↑ Gold R, Linington C & Lassmann H. (2006). Understanding pathogenesis and therapy of multiple sclerosis via animal models: 70 years of merits and culprits in experimental autoimmune encephalomyelitis research. Brain , 129, 1953-71. PMID: 16632554 DOI.
Journals
- Glia Content Listing
- Developmental Brain Research Content Listing
- Neural Development Welcome to Neural Development | Pubmed Central Volume 1 2006 | Pubmed Central Volume 2 2007 |
- International Journal for Developmental Neuroscience Official Journal of the International Society for Developmental Neuroscience |
- Developmental Neuroscience Journal Homepage | Hippocampal Development | Vol. 29, No. 3, 2007 |
- Neuroscience Official journal of The International Brain Research Organisation (IBRO)
- Neuron Neuroscience journal published by Cell press
Online Textbooks
Developmental Biology (6th ed) Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000. Formation of the Neural Tube | Differentiation of the Neural Tube | Tissue Architecture of the Central Nervous System | Neuronal Types | Snapshot Summary: Central Nervous System and Epidermis
Neuroscience Purves, Dale; Augustine, George J.; Fitzpatrick, David; Katz, Lawrence C.; LaMantia, Anthony-Samuel; McNamara, James O.; Williams, S. Mark. Sunderland (MA): Sinauer Associates, Inc. ; c2001 Early Brain Development | Construction of Neural Circuits | Modification of Brain Circuits as a Result of Experience
Molecular Biology of the Cell (4th Edn) Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter. New York: Garland Publishing; 2002. The three phases of neural development |
Search NLM Online Textbooks- "glial development" : Developmental Biology | Neuroscience | The Cell- A molecular Approach | Molecular Biology of the Cell | Endocrinology
Reviews
Thion MS, Ginhoux F & Garel S. (2018). Microglia and early brain development: An intimate journey. Science , 362, 185-189. PMID: 30309946 DOI.
Allen NJ & Lyons DA. (2018). Glia as architects of central nervous system formation and function. Science , 362, 181-185. PMID: 30309945 DOI.
Götz M & Huttner WB. (2005). The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. , 6, 777-88. PMID: 16314867 DOI.
Greene ND & Copp AJ. (2009). Development of the vertebrate central nervous system: formation of the neural tube. Prenat. Diagn. , 29, 303-11. PMID: 19206138 DOI.
Articles
Saitsu H & Shiota K. (2008). Involvement of the axially condensed tail bud mesenchyme in normal and abnormal human posterior neural tube development. Congenit Anom (Kyoto) , 48, 1-6. PMID: 18230116 DOI.
Ogata T, Yamamoto S, Nakamura K & Tanaka S. (2006). Signaling axis in schwann cell proliferation and differentiation. Mol. Neurobiol. , 33, 51-62. PMID: 16388110
Jessen KR & Mirsky R. (2002). Signals that determine Schwann cell identity. J. Anat. , 200, 367-76. PMID: 12090403
Weidenheim KM, Epshteyn I, Rashbaum WK & Lyman WD. (1994). Patterns of glial development in the human foetal spinal cord during the late first and second trimester. J. Neurocytol. , 23, 343-53. PMID: 7522270
Search PubMed
Search Pubmed: Gliogenesis | Glial Development | Myelination
Historic
Schwann T (1839) Mikroskopische Untersuchungen über die Uebereinstimmung in der Struktur und dem Wachsthum der Thiere und Pflanzen
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 25) Embryology Neural System - Glial Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_System_-_Glial_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G