Abnormal Development - Teratogens
Introduction
How and why do things go wrong in development? Embryological development is a robust biological system able to cope with many stresses without long-term consequences. When development does go wrong there are generally 3 major types groups: Genetic (inherited), Environmental (maternal) derived and Unknown (not determined or known) abnormalities. Also often not considered, is that pregnancy itself can also expose abnormalities in the mother (congenital heart disease, diabetes, reproductive disorders) that until the pregnancy had gone undetected.
- Infections, collectively grouped under the acronym TORCH for Toxoplasmosis, Other organisms (parvovirus, HIV, Epstein-Barr, herpes 6 and 8, varicella, syphilis, enterovirus) , Rubella, Cytomegalovirus and Hepatitis. See also the related topics on maternal hyperthermia and bacterial infections.
- Maternal diet the best characterised is the role of low folic acid and Neural Tube Defects (NTDs) see also abnormal neural development and Neural Tube Defects (NTDs). More recently the focus has been on dietary iodine levels and the role they also play on neural development.
- Maternal drugs effects either prescription drugs (therapeutic chemicals/agents, thalidomide limb development), non-prescription drugs (smoking), and illegal drugs (Cannabis/Marijuana, Methamphetamine/Amphetamine, Cocaine, Heroin, Lysergic Acid Diethylamide)
- Environment (smoking, chemicals, heavy metals, radiation) and maternal endocrine function (maternal diabetes, thyroid development) and maternal stress.
- Teratogen synergism, different environmental effects can act individually or in combination on the same developing system. For example, neural development can be impacted upon by alcohol (fetal alcohol syndrome), viral infection (rubella) and/or inadequate dietry folate intake (neural tube defects). These effects may also not be seen as a direct effect on a system or systems but result in a reduced birth weight and the potential postnatal developmental effects. Consider also this in relation to the increasing support to the fetal origins hypothesis.
Use the page links below to explore specific teratogens.
Some Recent Findings
|
Birth Defects Res B Dev Reprod Toxicol. 2011 Feb;92(1):69-76. doi: 10.1002/bdrb.20283. Epub 2011 Jan 19.
- Maternal exposure to multi-wall carbon nanotubes does not induce embryo-fetal developmental toxicity in rats.
Lim JH, Kim SH, Shin IS, Park NH, Moon C, Kang SS, Kim SH, Park SC, Kim JC.
College of Veterinary Medicine, Chonnam National University, Gwangju, Republic of Korea. Abstract BACKGROUND: Although the potential risk of carbon nanotubes (CNTs) to humans has recently increased due to expanding production and widespread use, the potential adverse effects of CNTs on embryo-fetal development have not yet been determined.
METHODS: This study investigated the potential effects of multi-wall CNTs (MWCNTs) on pregnant dams and embryo-fetal development in rats. MWCNTs were administered to pregnant rats by gavage at 0, 40, 200, and 1,000 mg/kg/day. All dams were subjected to Cesarean section on day 20 of gestation, and the fetuses were examined for any morphological abnormalities.
RESULTS: All animals survived to the end of the study. A decrease in thymus weight was observed in the high dose group in a dose-dependent manner. However, maternal body weight, food consumption, and oxidant-antioxidant balance in the liver were not affected by treatment with MWCNTs. No treatment-related differences in gestation index, fetal deaths, fetal and placental weights, or sex ratio were observed between the groups. Morphological examinations of the fetuses demonstrated no significant difference in incidences of abnormalities between the groups.
CONCLUSIONS: The results show that repeated oral doses of MWCNTs during pregnancy induces minimal maternal toxicity and no embryo-fetal toxicity at 1,000 mg/kg/day in rats. The no-observed-adverse-effect level of MWCNTs is considered to be 200 mg/kg/day for dams and 1,000 mg/kg/day for embryo-fetal development. In this study, the dosing formulation was not analyzed to determine the degree of reaggregation (or not), nor were blood levels of CNT's measured in the dosed animals to verify or characterize absorption. Birth Defects Res (Part B) 92:69-76, 2011. © 2011 Wiley-Liss, Inc.
© 2011 Wiley-Liss, Inc. PMID: 21254368
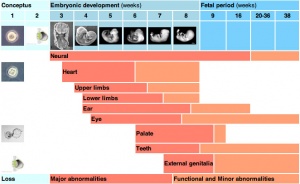
Critical Periods of Development
When studying this topic remember the concept of "critical periods of development" that will affect the overall impact of the above listed factors, as outlined in the table below. This can be extended to the potential differences between prenatal and postnatal effects, for example with infections and outcomes.
| Conceptus | Embryonic development (weeks) | Fetal period (weeks) | |||||||||||||||||
|
|||||||||||||||||||
| Neural | |||||||||||||||||||
| Heart | |||||||||||||||||||
| Upper limbs | |||||||||||||||||||
| Lower limbs | |||||||||||||||||||
| Ear | |||||||||||||||||||
| Eye | |||||||||||||||||||
| Palate | |||||||||||||||||||
| Teeth | |||||||||||||||||||
| External genitalia | |||||||||||||||||||
| Loss | Major abnormalities | Functional and Minor abnormalities | |||||||||||||||||
'
References
- ↑ <pubmed>20561110</pubmed>
Journals
- Birth Defects Research Part A: Clinical and Molecular Teratology
- Birth Defects Research Part B: Developmental and Reproductive Toxicology
- Part C: Embryo Today: Reviews
Reviews
Articles
Search Pubmed
Search Pubmed: teratogen | teratogenesis
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 14) Embryology Abnormal Development - Teratogens. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Teratogens
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G