Placenta Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (62 intermediate revisions by the same user not shown) | |||
| Line 3: | Line 3: | ||
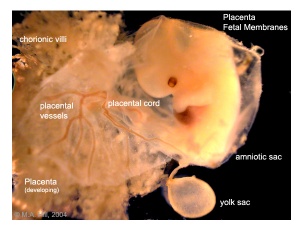
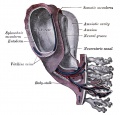
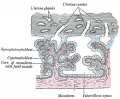
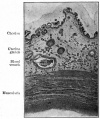
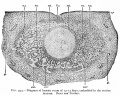
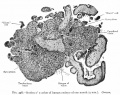
[[File:Placental_membranes.jpg|thumb|300px|Human Placental Membranes]] | [[File:Placental_membranes.jpg|thumb|300px|Human Placental Membranes]] | ||
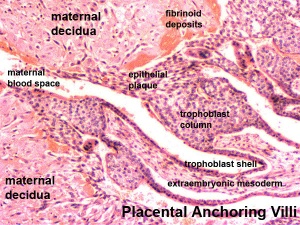
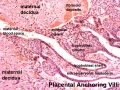
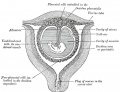
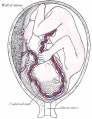
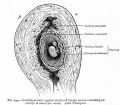
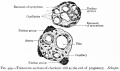
[[File:Placenta anchoring villi.jpg|thumb|Placenta anchoring villi and Maternal decidua]] | [[File:Placenta anchoring villi.jpg|thumb|Placenta anchoring villi and Maternal decidua]] | ||
The placenta (Greek, ''plakuos'' = flat cake) named on the basis of this organs gross anatomical appearance. The placenta a mateno-fetal organ which begins developing at implantation of the blastocyst and is delivered with the fetus at birth. During that 9 month period it provides nutrition, gas exchange, waste removal, endocrine and immune support for the developing fetus. | The {{placenta}} (Greek, ''plakuos'' = flat cake) named on the basis of this organs gross anatomical appearance. The placenta a mateno-fetal organ which begins developing at implantation of the blastocyst and is delivered with the fetus at birth. During that 9 month period it provides nutrition, gas exchange, waste removal, a source of hematopoietic stem cells, endocrine and immune support for the developing fetus. | ||
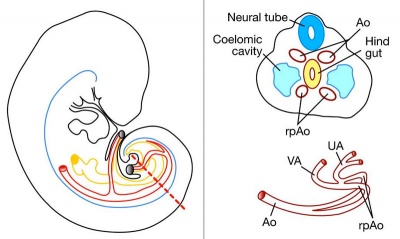
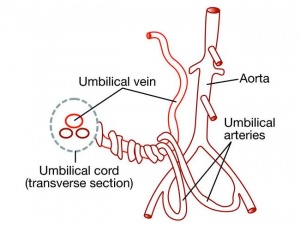
There are essentially 3 separate aortic/venous circulatory systems: umbilical, systemic and vitelline. The umbilical system is lost at birth, the vitelline contributes to the portal system and the systemic (embryonic) is extensively remodelled to form the mature cardiovascular system. | There are essentially 3 separate aortic/venous circulatory systems: umbilical, systemic and vitelline. The umbilical system is lost at birth, the vitelline contributes to the portal system and the systemic (embryonic) is extensively remodelled to form the mature cardiovascular system. | ||
{{ | Nutrition is derived from about 100–150 maternal uterine spiral arteries located in the basal plate and the human term placenta is about 9 cm in diameter. There appears to also be differences in placentation (function) between the sexes.{{#pmid:24720894|PMID24720894}}{{#pmid:26241064|PMID26241064}} | ||
<br> | |||
{{Placenta Links}} | |||
==Some Recent Findings== | ==Some Recent Findings== | ||
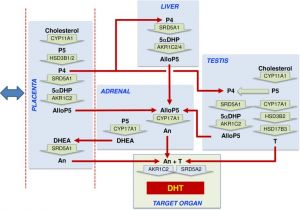
[[File:Model male androsterone synthesis.jpg|alt=Model male second trimester androsterone synthesis|thumb|Model male second trimester androsterone synthesis{{#pmid:30763313|PMID30763313}}]] | |||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | |||
* ''' | * '''Maternal platelets at the first trimester maternal-placental interface - Small players with great impact on placenta development'''{{#pmid:34920861|PMID34920861}} "In human pregnancy, maternal platelet counts decrease with each trimester, reaching a reduction by approximately ten percent at term in uncomplicated cases and recover to the levels of the non-pregnant state a few weeks postpartum. The time when maternal platelets start to occur in the early human placenta most likely coincides with the appearance of loosely cohesive endovascular trophoblast plugs showing capillary-sized channels by mid first trimester. At that time, platelets accumulate in intercellular gaps of anchoring parts of trophoblast columns and start to adhere to the surface of placental villi and the chorionic plate. This is considered as normal process that contributes to placenta development by acting on both the extravillous- and the villous trophoblast compartment. Release of platelet cargo into intercellular gaps of anchoring cell columns may affect partial epithelial-to-mesenchymal transition and invasiveness of extravillous trophoblasts as well as deposition of fibrinoid in the basal plate. Activation of maternal platelets on the villous surface leads to perivillous fibrin-type fibrinoid deposition, contributing to the shaping of the developing placental villi and the intervillous space. In contrast, excess platelet activation at the villous surface leads to deregulation of the endocrine activity, sterile inflammation and local apoptosis of the syncytiotrophoblast." | ||
* ''' | |||
* '''FcRn, but not FcγRs, drives maternal-fetal transplacental transport of human IgG antibodies''' [https://www.pnas.org/content/117/23/12943.abstract?etoc PNAS] "Transport of IgG antibodies from the maternal to the fetal circulation is a key process for neonatal immunity, as neonates cannot sufficiently generate IgG antibodies to reach protective levels during the first months after birth. In humans and other primates, maternal to fetal transport of IgG antibodies is largely mediated through the placental tissue. FcRn has been previously identified as the major driver of IgG transplacental transport. Here we examine whether other receptors, such as FcγRs, also contribute to the maternal-fetal IgG transfer. By characterizing the Fc domain structure of paired maternal-fetal IgG samples and modeling transplacental IgG transport in genetically engineered mouse strains, we determined that FcRn, but not FcγRs, is the major receptor that mediates transplacental IgG transport." | |||
* '''Review - Development of the human placenta'''{{#pmid:31776138|PMID31776138}} "The placenta is essential for normal in utero development in mammals. In humans, defective placental formation underpins common pregnancy disorders such as {{pre-eclampsia}} and fetal growth restriction. The great variation in placental types across mammals means that animal models have been of limited use in understanding human placental development. However, new tools for studying human placental development, including 3D {{organoids}}, stem cell culture systems and single cell RNA sequencing, have brought new insights into this field. Here, we review the morphological, molecular and functional aspects of human placental formation, with a focus on the defining cell of the placenta - the {{trophoblast}}." | |||
* '''Alternative (backdoor) androgen production and masculinization in the human fetus'''{{#pmid:30763313|PMID30763313}} "Masculinization of the external genitalia in humans is dependent on formation of 5α-dihydrotestosterone (DHT) through both the canonical androgenic pathway and an alternative (backdoor) pathway. The fetal testes are essential for canonical androgen production, but little is known about the synthesis of backdoor androgens, despite their known critical role in masculinization. ...Instead, the data indicate that {{placenta}}l progesterone acts as substrate for synthesis of backdoor androgens, which occurs across several tissues. Masculinization of the human fetus depends, therefore, on testosterone and androsterone synthesis by both the fetal testes and nongonadal tissues, leading to DHT formation at the genital tubercle. Our findings also provide a solid basis to explain why placental insufficiency is associated with disorders of sex development in humans." {{male}} {{genital}} {{genital abnormalities}} | |||
* '''Associations Between the Features of Gross Placental Morphology and Birthweight'''{{#pmid:30012074|PMID30012074}} "The {{placenta}} plays a critical role in regulating fetal growth. Recent studies suggest that there may be sex-specific differences in placental development. The purpose of our study was to evaluate the associations between {{birthweight}} and placental morphology in models adjusted for covariates and to assess sex-specific differences in these associations. We analyzed data from the Stillbirth Collaborative Research Network's population-based case-control study conducted between 2006 and 2008, which recruited cases of stillbirth and population-based controls in 5 states. Our analysis was restricted to singleton live births with a placental examination (n = 1229). Characteristics of placental morphology evaluated include thickness, surface area, difference in diameters, shape, and umbilical cord insertion site. We used linear regression to model birthweight as a function of placental morphology and covariates. Surface area had the greatest association with birthweight; a reduction in surface area of 83 cm2, which reflects the interquartile range, is associated with a 260.2-g reduction in birthweight (95% confidence interval, -299.9 to -220.6), after adjustment for other features of placental morphology and covariates. Reduced placental thickness was also associated with lower birthweight. These associations did not differ between males and females. Our results suggest that reduced placental thickness and surface area are independently associated with lower birthweight and that these relationships are not related to sex." {{Birthweight}} | |||
* '''The human placental proteome secreted into the maternal and fetal circulations in normal pregnancy based on 4-vessel sampling'''{{#pmid:30335547|PMID30335547}} "Blood samples from the maternal radial artery and uterine vein and umbilical artery and vein were obtained during {{cesarean section}} in 35 healthy women with term pregnancy. Slow off-rate modified aptamer (SOMA) protein-binding technology was used to quantify 1310 known proteins. The uteroplacental and umbilical venoarterial concentration differences were calculated. Thirty-four proteins were significantly secreted by the placenta into the maternal circulation, including placental growth factor, growth/differentiation factor 15, and matrix metalloproteinase 12. There were 341 proteins significantly secreted by the placenta into the fetal circulation. Only 7 proteins were secreted into both the fetal and maternal circulations, suggesting a distinct directionality in placental protein release. We examined changes across gestation in the proteins found to be significantly secreted by the placenta into the maternal circulation using serial blood samples from healthy women. Among the 34 proteins secreted into the maternal circulation, 8 changed significantly across gestation. The identified profiles of secreted placental proteins will allow us to identify novel minimally invasive biomarkers for human placental function across gestation and discover previously unknown proteins secreted by the human placenta that regulate maternal physiology and fetal development." | |||
|} | |} | ||
{| class="wikitable collapsible collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
! More recent papers | ! More recent papers | ||
|- | |- | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: ''Placenta Embryology'' | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Placenta+Embryology ''Placenta Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Placenta+Development ''Placenta Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Placentation ''Placentation''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=decidualization ''decidualization''] | ||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''Murine transcription factor Math6 is a regulator of placenta development'''{{#pmid:30301918|PMID30301918}} "The murine basic helix-loop-helix transcription (bHLH) factor mouse atonal homolog 6 (Math6) is expressed in numerous organs and supposed to be involved in several developmental processes. Characterization of the endogenous spatiotemporal Math6 expression during placenta development reveals that Math6 is essential for an ordered decidualization and an important regulator of the maternal-fetal endocrine crosstalk regulating endometrial trophoblast invasion and differentiation. The strongly disturbed vascularization observed in the maternal placenta appears as an additional consequence of the altered endocrine status and as the main cause for the general hemorrhagic crisis." | |||
* '''S100P enhances the motility and invasion of human trophoblast cell lines'''{{#pmid:30065265|PMID30065265}} "S100P has been shown to be a marker for carcinogenesis where its expression in solid tumours correlates with metastasis and a poor patient prognosis. This protein's role in any physiological process is, however, unknown. Here we first show that S100P is expressed both in trophoblasts in vivo as well as in some corresponding cell lines in culture. We demonstrate that S100P is predominantly expressed during the early stage of placental formation with its highest expression levels occurring during the first trimester of gestation, particularly in the invading columns and anchoring villi. Using gain or loss of function studies through overexpression or knockdown of S100P expression respectively, our work shows that S100P stimulates both cell motility and cellular invasion in different trophoblastic and first trimester EVT cell lines. Interestingly, cell invasion was seen to be more dramatically affected than cell migration. Our results suggest that S100P may be acting as an important regulator of trophoblast invasion during placentation." | |||
* '''Development of the Human Placenta and Fetal {{Heart}}: Synergic or Independent?'''{{#pmid:29706899|PMID29706899}}The placenta is the largest fetal organ, and toward the end of pregnancy the umbilical circulation receives at least 40% of the biventricular cardiac output. It is not surprising, therefore, that there are likely to be close haemodynamic links between the development of the placenta and the fetal heart. Development of the placenta is precocious, and in advance of that of the fetus. The placenta undergoes considerable remodeling at the end of the first trimester of pregnancy, and its vasculature is capable of adapting to environmental conditions and to variations in the blood supply received from the mother. ... In pregnancies complicated by preeclampsia and/or fetal growth restriction, utero-placental malperfusion induces smooth muscle cells surrounding the placental arteries to dedifferentiate and adopt a proliferative phenotype. This change is associated with increased umbilical resistance measured by Doppler ultrasound, and is likely to exert a major effect on the developing heart through the afterload. Thus, both the umbilical and maternal placental circulations may impact on development of the heart." | |||
* '''{{Hox}} genes in the allantois essential for proper extra-embryonic function in placental mammals'''{{#pmid:22219351|PMID22219351}} " Here, we provide evidence that this functional novelty is not exclusive to Hoxa13 but is shared with its neighboring Hoxa11 and Hoxa10 genes. We show that the extra-embryonic function of these three Hoxa genes stems from their specific expression in the allantois, an extra-embryonic hallmark of amniote vertebrates. Interestingly, Hoxa10-13 expression in the allantois is conserved in chick embryos, which are non-placental amniotes, suggesting that the extra-embryonic recruitment of Hoxa10, Hoxa11 and Hoxa13 most likely arose in amniotes, i.e. prior to the emergence of placental mammals." | |||
* '''A transient placental source of serotonin for the fetal forebrain'''{{#pmid:21512572|PMID21512572}} "Serotonin (5-hydroxytryptamine or 5-HT) is thought to regulate neurodevelopmental processes through maternal-fetal interactions that have long-term mental health implications. It is thought that beyond fetal 5-HT neurons there are significant maternal contributions to fetal 5-HT during pregnancy but this has not been tested empirically. ...We uncovered a placental 5-HT synthetic pathway from a maternal tryptophan precursor in both mice and humans. This study reveals a new, direct role for placental metabolic pathways in modulating fetal brain development and indicates that maternal-placental-fetal interactions could underlie the pronounced impact of 5-HT on long-lasting mental health outcomes." | |||
* '''Comparative systems biology of human and mouse'''{{#pmid:19536202|PMID19536202}} "We collected protein and mRNA expression data through shot-gun proteomics and microarray expression analysis of the highly vascular exchange region, microdissected from the human and mouse near-term placenta. Over 7000 ortholog genes were detected with 70% co-expressed in both species. Close to 90% agreement was found between our human proteomic results and 1649 genes assayed by immunohistochemistry for expression in the human placenta in the Human Protein Atlas. Interestingly, over 80% of genes known to cause placental phenotypes in mouse are co-expressed in human." | |||
|} | |} | ||
==Reading== | ==Reading== | ||
[[File:Spiegel1626_table07.jpg|thumb|Spiegel and Casseri: De formato foetu liber singularis (Dedication dated 1626).]] | [[File:Spiegel1626_table07.jpg|thumb|Spiegel and Casseri: De formato foetu liber singularis (Dedication dated 1626).]] | ||
| Line 42: | Line 64: | ||
==Placental Classification== | ==Placental Classification== | ||
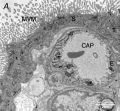
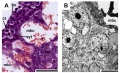
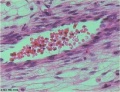
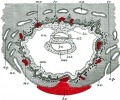
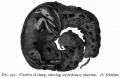
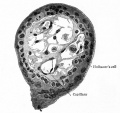
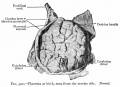
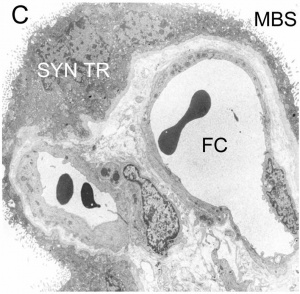
[[File:Haemomonochorial human placenta EM01.jpg|thumb|Haemomonochorial human placenta villi (EM) showing syncitiotrophoblast cells. | [[File:Haemomonochorial human placenta EM01.jpg|thumb|Haemomonochorial human placenta villi (EM) showing syncitiotrophoblast cells.{{#pmid:15236656|PMID15236656}}]] | ||
Classification of placenta is on the basis of histological (microscopic) structural organization and layers between fetal and maternal circulation. | Classification of placenta is on the basis of histological (microscopic) structural organization and layers between fetal and maternal circulation. | ||
| Line 48: | Line 70: | ||
# '''Haemochorial''' - placenta where the chorion comes in direct contact with maternal blood (human). | # '''Haemochorial''' - placenta where the chorion comes in direct contact with maternal blood (human). | ||
# '''Endotheliochorial''' - maternal endometrial blood vessels are bare to their endothelium and these comes in contact with the chorion ( | # '''Endotheliochorial''' - maternal endometrial blood vessels are bare to their endothelium and these comes in contact with the chorion ({{dog}}s, {{cat}}s). | ||
# '''Epitheliochorial''' - maternal epithelium of the uterus comes in contact with the chorion, considered as primitive ( | # '''Epitheliochorial''' - maternal epithelium of the uterus comes in contact with the chorion, considered as primitive ({{pig}}s, {{Bovine}}). | ||
The presence of these three differing types of placenta have also been used to describe the pattern mammalian evolution. | The presence of these three differing types of placenta have also been used to describe the pattern mammalian evolution. | ||
{| class="wikitable collapsible collapsed" | {{Species Placenta collapsetable1}} | ||
! Male Placenta? | |||
{{Mouse Rat Pig table}} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Male Placenta? | |||
|- | |- | ||
| [[File:Mark_Hill.jpg|left|50px]] Well not exactly, but close. In vertebra and in placental mammals embryonic development is associated with the mother (maternal). The exception to this rule occurs in seahorses (''syngnathidae'') where the male has a "uterus", a special pouch (brood pouch), where deposited eggs embed in depressions of the pouch interior lining. This pouch also has "placental" functions serving as aeration, protection, osmoregulation and nutrition for the egg development. (More? PMID 23213429) | | [[File:Mark_Hill.jpg|left|50px]] Well not exactly, but close. In vertebra and in placental mammals embryonic development is associated with the mother (maternal). The exception to this rule occurs in seahorses (''syngnathidae'') where the male has a "uterus", a special pouch (brood pouch), where deposited eggs embed in depressions of the pouch interior lining. This pouch also has "placental" functions serving as aeration, protection, osmoregulation and nutrition for the egg development. (More? [https://www.ncbi.nlm.nih.gov/pubmed/23213429 PMID 23213429]) | ||
|} | |} | ||
| Line 63: | Line 90: | ||
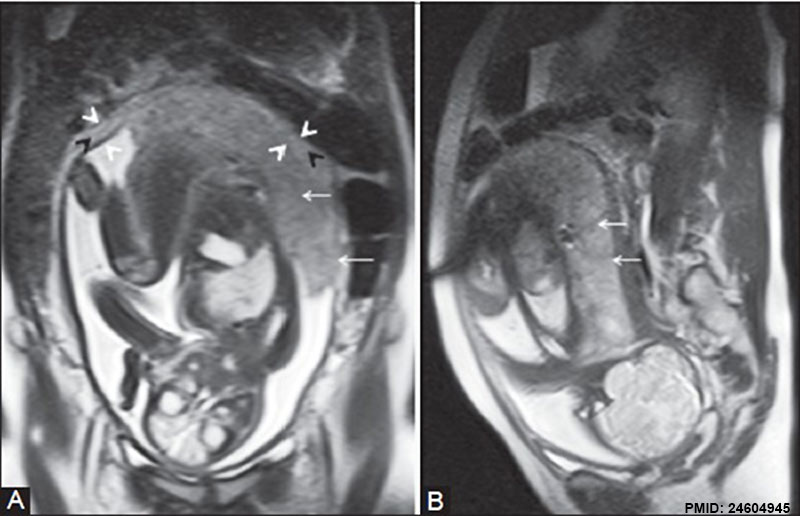
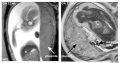
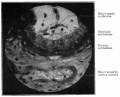
[[File:Placenta_MRI_01.jpg]] | [[File:Placenta_MRI_01.jpg]] | ||
Placenta MRI (37 weeks) | Placenta MRI (37 weeks){{#pmid:24604945|PMID24604945}} | ||
Placenta located at the upper segment of uterus. | |||
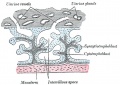
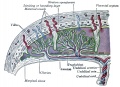
==Placental Villi Blood Vessels== | ==Placental Villi Blood Vessels== | ||
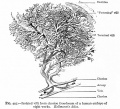
[[File:Implanting human conceptus 01.jpg|thumb|Implanting Conceptus | [[File:Implanting human conceptus 01.jpg|thumb|Implanting Conceptus{{#pmid:15265238|PMID15265238}}]] | ||
Some of the following data is from a histological study of human placental villi.{{#pmid:2481376|PMID2481376}} | |||
Some of the following data is from a histological study of human placental villi. | |||
* macrophage-like cells first cells to differentiate at day 21 (post-conception) from mesenchymal precursors. | * macrophage-like cells first cells to differentiate at day 21 (post-conception) from mesenchymal precursors. | ||
* haemangioblastic cell cords (angiogenic cell cords, ACC) at day 21 (post-conception) also from mesenchymal cells, are the precursors of the capillary endothelium and haematopoietic stem cells | * haemangioblastic cell cords (angiogenic cell cords, ACC) at day 21 (post-conception) also from mesenchymal cells, are the precursors of the capillary endothelium and haematopoietic stem cells | ||
| Line 81: | Line 105: | ||
* third trimester - fetal villous angiogenesis occurs by proliferation of the existing endothelium and pericytes rather than through haemangioblastic cells. | * third trimester - fetal villous angiogenesis occurs by proliferation of the existing endothelium and pericytes rather than through haemangioblastic cells. | ||
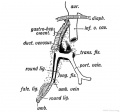
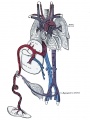
== | ==Fetal Circulation== | ||
{| | {| | ||
! Placental | |||
! Systematic | |||
|- | |- | ||
| | | [[File:Gray0039.jpg|500px]] | ||
| | | [[File:Gray0502.jpg|300px]] | ||
| | |||
| | |||
|} | |} | ||
==Human Villi Timeline== | |||
The placental vill development data below is based upon a recent immunochemistry confocal laser scanning microscope (CLSM) study.{{#pmid:17545656|PMID17545656}} Note that the paper uses clinical gestational age (GA) from last menstrual period (LMP) and has been corrected for post-conception (fertilization) age, approximately 14 days later. | |||
{{Placenta Villi Timeline table}} | |||
| Line 167: | Line 137: | ||
Uteroplacental blood flow (UPBF) was historically measured by a number of different mathematical calculations and probe methods, currently the method involves transvaginal doppler ultrasonography. | Uteroplacental blood flow (UPBF) was historically measured by a number of different mathematical calculations and probe methods, currently the method involves transvaginal doppler ultrasonography. | ||
In human singleton pregnancies, uteroplacental blood flow (UPBF) begins at 20–50 ml/min and increases (linearly) to 450–800 ml/min, with twin pregnancy values in excess of 1 l/min. | In human singleton pregnancies, uteroplacental blood flow (UPBF) begins at 20–50 ml/min and increases (linearly) to 450–800 ml/min, with twin pregnancy values in excess of 1 l/min.{{#pmid:19196652|PMID19196652}} | ||
===Uterine Artery Diameter=== | ===Uterine Artery Diameter=== | ||
The following data is from a study of 18 pregnant women using ultrasound and doppler analysis of the uterine artery. | The following data is from a study of 18 pregnant women using ultrasound and doppler analysis of the uterine artery.{{#pmid:1448242|PMID1448242}} | ||
* {{GA}} week 21 doubled (from 1.4 to 2.8 mm). | * {{GA}} week 21 doubled (from 1.4 to 2.8 mm). | ||
| Line 184: | Line 154: | ||
:'''Links:''' [[ | :'''Links:''' {{ultrasound}} | {{cardiovascular}} | ||
===Blood Oxygen Levels=== | |||
Maternal and Fetal Placental Circulations{{#pmid:29377190|PMID29377190}} | |||
Showing the major compartments and published attributed in vivo oxygen values. | |||
[[File:Placental imaging 01.jpg|alt=maternal and fetal placental circulations, showing the major compartments and published attributed in vivo oxygen values|650px]] | |||
==Term Placenta Measurements== | ==Term Placenta Measurements== | ||
| Line 196: | Line 173: | ||
* '''placental volume''' - is measured by a range of different methods and calculations, more recently with three-dimensional ultrasound. | * '''placental volume''' - is measured by a range of different methods and calculations, more recently with three-dimensional ultrasound. | ||
Detailed morphometric indices at term of placental composition, villous capillarization and the mean cross-sectional areas of peripheral villi and capillaries, data from a study sample of 15 normal placenta (mean placental volume, 652 ml). | Detailed morphometric indices at term of placental composition, villous capillarization and the mean cross-sectional areas of peripheral villi and capillaries, data from a study sample of 15 normal placenta (mean placental volume, 652 ml).{{#pmid:18328557|PMID18328557}}{{#pmid:19141109|PMID19141109}} | ||
{{Term Placenta Measurements table}} | |||
===Placental Weight=== | |||
[[File:Placental mean weight graph01.jpg|600px]] | |||
Fetal Placental weight growth{{#pmid:17516963|PMID17516963}} | |||
==Placental Factors== | ==Placental Factors== | ||
| Line 263: | Line 189: | ||
===Placental Growth Factor=== | ===Placental Growth Factor=== | ||
(PGF, PLGF) A growth factor of the vascular endothelial growth factor (VEGF) family, released from the placental [[T#trophoblast|trophoblast]] cells and other sources that stimulates blood vessel growth. See review. | (PGF, PLGF) A growth factor of the vascular endothelial growth factor (VEGF) family, released from the placental [[T#trophoblast|trophoblast]] cells and other sources that stimulates blood vessel growth. See review.{{#pmid:22228176|PMID22228176}} | ||
| Line 293: | Line 219: | ||
[[File:Galletti1770_placenta previa.jpg|thumb|Historic model of placenta previa]] | [[File:Galletti1770_placenta previa.jpg|thumb|Historic model of placenta previa]] | ||
[[File:Hydatidiform_mole.jpg|thumb|Hydatidiform mole pathology]] | [[File:Hydatidiform_mole.jpg|thumb|Hydatidiform mole pathology]] | ||
Note there is also a specific page covering the topic of [[Placenta - Abnormalities|placental abnormalities]]. | |||
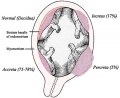
* '''Placenta Accreta''' - abnormal adherence, with absence of decidua basalis. The incidence of placenta accreta also significantly increases in women with previous cesarean section compared to those without a prior surgical delivery. | * '''Placenta Accreta''' - abnormal adherence, with absence of decidua basalis. The incidence of placenta accreta also significantly increases in women with previous cesarean section compared to those without a prior surgical delivery. | ||
* '''Placenta Increta''' - occurs when the placenta attaches deep into the uterine wall and penetrates into the uterine muscle, but does not penetrate the uterine serosa. Placenta increta accounts for approximately 15-17% of all cases. | * '''Placenta Increta''' - occurs when the placenta attaches deep into the uterine wall and penetrates into the uterine muscle, but does not penetrate the uterine serosa. Placenta increta accounts for approximately 15-17% of all cases. | ||
| Line 324: | Line 253: | ||
:'''Links:''' | :'''Links:''' {{malaria}} | [[Abnormal_Development_-_Viral_Infection|Viral]] | [[Abnormal_Development_-_Bacterial_Infection|Bacterial]] | ||
===Placental Pathology=== | ===Placental Pathology=== | ||
| Line 333: | Line 262: | ||
* '''Neuroblastoma''' - a fetal malignancy that leads to an enlarged placenta, with tumor cells in the fetal circulation and rarely in the chorionic villi. | * '''Neuroblastoma''' - a fetal malignancy that leads to an enlarged placenta, with tumor cells in the fetal circulation and rarely in the chorionic villi. | ||
* '''Thrombophilias''' - (protein C or S deficiency, factor V Leiden, sickle cell disease, antiphospholipid antibody) can generate an increased fibrin/fibrinoid deposition in the maternal or intervillous space, this can trap and kill villi. | * '''Thrombophilias''' - (protein C or S deficiency, factor V Leiden, sickle cell disease, antiphospholipid antibody) can generate an increased fibrin/fibrinoid deposition in the maternal or intervillous space, this can trap and kill villi. | ||
[[File:Kurt Benirschke.jpg|thumb|150px|Dr Kurt Benirschke]] | |||
==Comparative Placentation== | |||
The usual {{mouse}} model does not reflect human placentation in structure or function. Mouse does not synthesise estrogen not (Hyperglycosylated) chorionic gonadotropin. | |||
The [http://placentation.ucsd.edu/homefs.html Comparative Placentation] website has been available online for many years providing valuable information about placentation in a wide range of species. The site was developed by Dr Kurt Benirschke, a longstanding expert in the field.{{#pmid:9579593|PMID9579593}}{{#pmid:12068178|PMID12068178}} Dr Kurt Benirschke retired in 1994 and continued to develop and maintain the website (that is still available) until 2012, but it has not been updated since that time. | |||
:'''Links:''' [http://placentation.ucsd.edu/homefs.html homepage] | [http://placentation.ucsd.edu/homefs.html index] | [http://placentation.ucsd.edu/homefs.html glossary] | |||
==References== | ==References== | ||
<references/> | <references/> | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:28955183}} | |||
{{#pmid:21470579}} | |||
{{#pmid:21272934}} | |||
{{#pmid:20837731}} | |||
{{#pmid:21470579}} | |||
{{#pmid:21272934}} | |||
{{#pmid:20837731}} | |||
{{#pmid:18755720}} | |||
Wang Y, Zhao S. '''Vascular Biology of the Placenta.''' San Rafael (CA): Morgan & Claypool Life Sciences; 2010. [http://www.ncbi.nlm.nih.gov/books/NBK53247 Bookshelf NBK53247] | Wang Y, Zhao S. '''Vascular Biology of the Placenta.''' San Rafael (CA): Morgan & Claypool Life Sciences; 2010. [http://www.ncbi.nlm.nih.gov/books/NBK53247 Bookshelf NBK53247] | ||
===Articles=== | ===Articles=== | ||
{{#pmid:32900950}} | |||
{{#pmid:32905973}} | |||
{{#pmid:20933281}} | |||
{{#pmid:9518951}} | |||
===Books=== | ===Books=== | ||
| Line 358: | Line 316: | ||
* [[Book_-_A_Laboratory_Manual_and_Text-book_of_Embryology_4|Manual of Human Embryology (1917)]] (unedited) | * [[Book_-_A_Laboratory_Manual_and_Text-book_of_Embryology_4|Manual of Human Embryology (1917)]] (unedited) | ||
* [[Book_-_Text-Book_of_Embryology_19|Text-Book of Embryology (1921)]] | [[Book_-_Text-Book_of_Embryology_19#The_Foetal_Membranes_in_Man|The Foetal Membranes in Man]] | * [[Book_-_Text-Book_of_Embryology_19|Text-Book of Embryology (1921)]] | [[Book_-_Text-Book_of_Embryology_19#The_Foetal_Membranes_in_Man|The Foetal Membranes in Man]] | ||
* | |||
* {{Ref-Bloxam1840}} | |||
* {{Ref-Herzog1909}} | |||
* {{Ref-Grosser1910}} | |||
* {{Ref-Bremer1918}} | |||
* {{Ref-Fujimura1921}} | |||
* {{Ref-Corner1921}} | |||
* {{Ref-Dawson1922}} | |||
* {{Ref-PMID5965440}} | |||
* {{Ref-Strachan1923}} | |||
* {{Ref-WislockiBennett1943}} | |||
* {{Ref-BakerHookSeveringhaus1944}} | |||
* {{Ref-Hertig1946b}} | |||
* {{Ref-Ramsey1960}} | |||
* {{Ref-Ramsey1972}} | |||
| Line 375: | Line 347: | ||
<gallery> | <gallery> | ||
File:Placental imaging 03.jpg|Placental imaging PMID 29377190 | |||
File:Placental imaging 03A.jpg|Placental villi (EM) PMID 29377190 | |||
File:Placenta MRI 02.jpg|Placenta MRI 22 and 32 week | |||
File:Placenta_anchoring_villi.jpg|Placenta anchoring villi | File:Placenta_anchoring_villi.jpg|Placenta anchoring villi | ||
File:Placenta_humans_and_guinea-pig_cartoon.jpg|Placenta human and guinea-pig cartoon | File:Placenta_humans_and_guinea-pig_cartoon.jpg|Placenta human and guinea-pig cartoon | ||
| Line 384: | Line 359: | ||
File:Placental_cord_cross-section.jpg|Placental cord cross-section | File:Placental_cord_cross-section.jpg|Placental cord cross-section | ||
File:Placenta_abnormalities.jpg|Placenta_abnormalities | File:Placenta_abnormalities.jpg|Placenta_abnormalities | ||
File:Mouse_placenta_01.jpg|Mouse placenta | File:Mouse_placenta_01.jpg|Mouse placenta {{ME16.5}} | ||
File:Mouse_placenta_02.jpg|Mouse placenta | File:Mouse_placenta_02.jpg|Mouse placenta {{ME16.5}} | ||
File:Human placenta vascular 01.jpg|Human placenta viewed from the fetal side | File:Human placenta vascular 01.jpg|Human placenta viewed from the fetal side | ||
File:Placental_cord_ultrasound_02.jpg|Cord with one artery and one vein | File:Placental_cord_ultrasound_02.jpg|Cord with one artery and one vein | ||
| Line 392: | Line 367: | ||
===Historic=== | ===Historic=== | ||
{{Historic Disclaimer}} | |||
<gallery> | <gallery> | ||
File:Spiegel1626_table07.jpg|Placenta and Fetus | File:Spiegel1626_table07.jpg|Placenta and Fetus | ||
File:Spiegel1626_table05fig1.jpg|Placenta Fetal Side | File:Spiegel1626_table05fig1.jpg|Placenta Fetal Side | ||
File:Spiegel1626_table05fig2.jpg|Placenta Maternal Side | File:Spiegel1626_table05fig2.jpg|Placenta Maternal Side | ||
</gallery> | |||
<gallery> | |||
File:Keith1902_fig189.jpg|Keith 1902 Fig. 189. Diagram of the Remnants of the Umbilical Vein in the Adult | |||
</gallery> | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Gray 1918 | |||
|- | |||
| {{Ref-Gray1918}} | |||
<gallery> | |||
File:Gray0031.jpg|Historic drawing of early implantation (stage 8-9) | File:Gray0031.jpg|Historic drawing of early implantation (stage 8-9) | ||
File:Gray0036.jpg | File:Gray0036.jpg | ||
| Line 413: | Line 391: | ||
File:Gray0038.jpg | File:Gray0038.jpg | ||
File:Gray0039.jpg | File:Gray0039.jpg | ||
File: | File:Gray0502.jpg|Fetal circulation overview | ||
</gallery> | </gallery> | ||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Bailey1921 | |||
|- | |||
| {{Ref-Bailey1921}} | |||
<gallery> | |||
Bailey489.jpg | |||
Bailey490.jpg | |||
Bailey491.jpg | |||
Bailey492.jpg | |||
Bailey493.jpg | |||
Bailey494.jpg | |||
Bailey495.jpg | |||
Bailey496.jpg | |||
Bailey497.jpg | |||
Bailey498.jpg | |||
Bailey499.jpg | |||
Bailey500.jpg | |||
Bailey501.jpg | |||
Bailey502.jpg | |||
</gallery> | |||
|} | |||
==Terms== | ==Terms== | ||
{{Placenta terms}} | |||
==External Links== | |||
{{External Links}} | |||
* ''' | * '''Comparative Placentation''' [http://placentation.ucsd.edu/homefs.html homepage] | [http://placentation.ucsd.edu/homefs.html index] | [http://placentation.ucsd.edu/homefs.html glossary] | ||
* ''' | * '''The Human Placenta Project''' - a collaborative research effort, launched by the NICHD to understand the role of the placenta in health and disease.[https://www.nichd.nih.gov/research/HPP/Pages/default.aspx Human Placenta Project] | [http://youtu.be/MGhGkMM9-B0 Video] | ||
* ''' | * '''International Federation of Placenta Associations''' [http://www.ifpafederation.org/meetings/IFPA_meetings.htm IFPA Meetings] | [http://tsforum.event123.no/IFPA/EPG2011 2011 IFPA Meeting, Geilo, Norway] | ||
* | * USA NICHD - [https://www.nichd.nih.gov/research/supported/HPP/default Human Placenta Project] (HPP) is a collaborative research effort to understand the role of the placenta in health and disease. [https://www1.nichd.nih.gov/publications/pubs/Documents/HumanPlacentaProject.pdf Factsheet PDF] | ||
{{Systems}} | {{Systems}} | ||
Latest revision as of 19:46, 9 February 2022
| Embryology - 26 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The placenta (Greek, plakuos = flat cake) named on the basis of this organs gross anatomical appearance. The placenta a mateno-fetal organ which begins developing at implantation of the blastocyst and is delivered with the fetus at birth. During that 9 month period it provides nutrition, gas exchange, waste removal, a source of hematopoietic stem cells, endocrine and immune support for the developing fetus.
There are essentially 3 separate aortic/venous circulatory systems: umbilical, systemic and vitelline. The umbilical system is lost at birth, the vitelline contributes to the portal system and the systemic (embryonic) is extensively remodelled to form the mature cardiovascular system.
Nutrition is derived from about 100–150 maternal uterine spiral arteries located in the basal plate and the human term placenta is about 9 cm in diameter. There appears to also be differences in placentation (function) between the sexes.[1][2]
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Placenta Embryology | Placenta Development | Placentation | decidualization |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Reading
- Human Embryology (2nd ed.) Larson Chapter 7 p151-188 Heart, Chapter 8 p189-228 Vasculature
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 14: p304-349
- Before we Are Born (5th ed.) Moore and Persaud Chapter 12; p241-254
- Essentials of Human Embryology Larson Chapter 7 p97-122 Heart, Chapter 8 p123-146 Vasculature
- Human Embryology Fitzgerald and Fitzgerald Chapter 13-17: p77-111
Placental Classification
Classification of placenta is on the basis of histological (microscopic) structural organization and layers between fetal and maternal circulation.
Three main groups:
- Haemochorial - placenta where the chorion comes in direct contact with maternal blood (human).
- Endotheliochorial - maternal endometrial blood vessels are bare to their endothelium and these comes in contact with the chorion (dogs, cats).
- Epitheliochorial - maternal epithelium of the uterus comes in contact with the chorion, considered as primitive (pigs, bovine).
The presence of these three differing types of placenta have also been used to describe the pattern mammalian evolution.
| Species Placental Comparison | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Postnatal Animal Models | mouse | rat | pig |
|---|---|---|---|
| Pregnancy period (days) | 18 – 21 | 21 – 23 | 110 – 118 |
| Placenta type | Discoidal, decidual hemoendothelial choroidea |
Discoidal, decidual hemoendothelial choroidea |
Epitheliochorial |
| Litter size | 6 – 12 | 6 – 15 | 11 – 16 |
| Birth weight (g) | 0.5 – 1.5 | 3 – 5 | 900 – 1600 |
| Weaning weight male/female (g) | 18 – 25/16 – 25 | 55 – 90/45 – 80 | 6000 – 8000 |
| Suckling period (days) | 21–28 | 21 | 28–49 |
| Solid diet beginning (days) | 10 | 12 | 12 – 15 |
| Puberty male/female (week) | 4 – 6/5 | 6/6 – 8 | 20 – 28 |
| Life expectancy (years) | 1 - 2 | 2 - 3 | 14 – 18 |
| Table data - Otis and Brent (1954)[15] Links: timeline | |||
| Male Placenta? |
|---|
| Well not exactly, but close. In vertebra and in placental mammals embryonic development is associated with the mother (maternal). The exception to this rule occurs in seahorses (syngnathidae) where the male has a "uterus", a special pouch (brood pouch), where deposited eggs embed in depressions of the pouch interior lining. This pouch also has "placental" functions serving as aeration, protection, osmoregulation and nutrition for the egg development. (More? PMID 23213429) |
Placenta MRI (37 weeks)[16]
Placenta located at the upper segment of uterus.
Placental Villi Blood Vessels
Some of the following data is from a histological study of human placental villi.[18]
- macrophage-like cells first cells to differentiate at day 21 (post-conception) from mesenchymal precursors.
- haemangioblastic cell cords (angiogenic cell cords, ACC) at day 21 (post-conception) also from mesenchymal cells, are the precursors of the capillary endothelium and haematopoietic stem cells
- pericytes form later and are a third cell population derived from the mesenchymal cells
- first main vascular patterns grow towards the longitudinal axis of the developing villi
- capillary basal lamina cannot be detected earlier than in the third trimester
- third trimester - fetal villous angiogenesis occurs by proliferation of the existing endothelium and pericytes rather than through haemangioblastic cells.
Fetal Circulation
| Placental | Systematic |
|---|---|
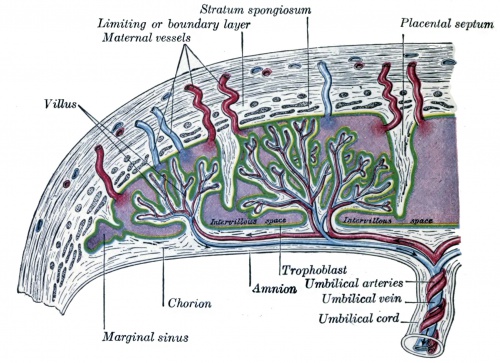
|
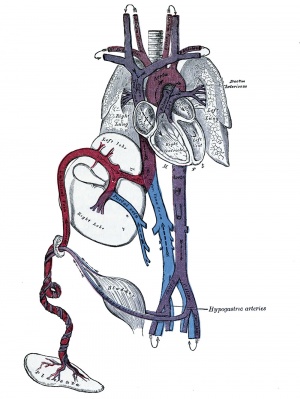
|
Human Villi Timeline
The placental vill development data below is based upon a recent immunochemistry confocal laser scanning microscope (CLSM) study.[19] Note that the paper uses clinical gestational age (GA) from last menstrual period (LMP) and has been corrected for post-conception (fertilization) age, approximately 14 days later.
| Fertilization Age
(weeks) |
Gestational Age
(weeks) |
Vessel Lumen Diameter
(range in microns) |
|
| 3 to 4 | 5 and 6 | 10 - 15 |
|
| 5 to 6 | 7 and 8 | 10 - 26 |
|
| 7 to 8 | 9 and 10 | 60 - 75 two central vessels
26 - 34 capillary network |
|
| 9 to 10 | 11 and 12 | 70 - 90 two central vessels
26 - 34 capillary network |
|
| Term | Terminal villi
| ||
| Table data[19] Paper uses clinical gestational age (GA) table corrected also for post-conception (fertilization) age. | |||
CD31 - (PECAM-1, Platelet Endothelial Cell Adhesion Molecule) is a cluster of differentiation molecule found on endothelial and other blood cells.
Trophoblast Cells
Following implantation the initial trophoblast cells can differentiate into 2 pathways:
- Extravillous - cytotrophoblastic cells proliferate and differentiate into an invasive phenotype that invade (interstitial trophoblast) the maternal decidual stroma and the spiral arteries (endovascular trophoblast) of the myometrium.
- Villous - cytotrophoblastic cells proliferate and fuse to form the multi-nucleated syncytiotrophoblast cells that form the outer surface of the fetal placental villi.
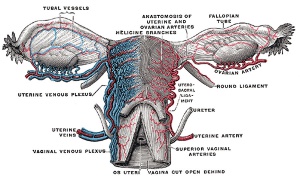
Maternal Blood Flow
Maternal blood pressure normally decreases or remains unchanged during pregnancy while both cardiac output and vascular volume are increased. Uterine blood flow changes are principally due to a decrease in uterine vascular resistance. There is also an associated structural enlargement of both the uterine arterial and venous trees, reduced vascular tone (vasodilation) and placenta development.
Uteroplacental blood flow (UPBF) was historically measured by a number of different mathematical calculations and probe methods, currently the method involves transvaginal doppler ultrasonography.
In human singleton pregnancies, uteroplacental blood flow (UPBF) begins at 20–50 ml/min and increases (linearly) to 450–800 ml/min, with twin pregnancy values in excess of 1 l/min.[20]
Uterine Artery Diameter
The following data is from a study of 18 pregnant women using ultrasound and doppler analysis of the uterine artery.[21]
- GA week 21 doubled (from 1.4 to 2.8 mm).
- GA week 21 to 30 remained constant (2.9mm).
- GA week 30 to 36 increased (to 3.4 mm).
Uterine artery mean flow velocity also increased nearly eight times from non-pregnant (8.4 cm/second) to GA week 36 (61.4 cm/second).
Arcuate and Radial Arteries
These branches from the paired uterine arteries also remodel enlarging in lumen diameter between 25 to 220% with either no change or an increase in wall thickness. Arcuate arteries also elongate either by longitudinal growth or by the progressive straightening of these coiled vessels.
- Links: ultrasound | cardiovascular
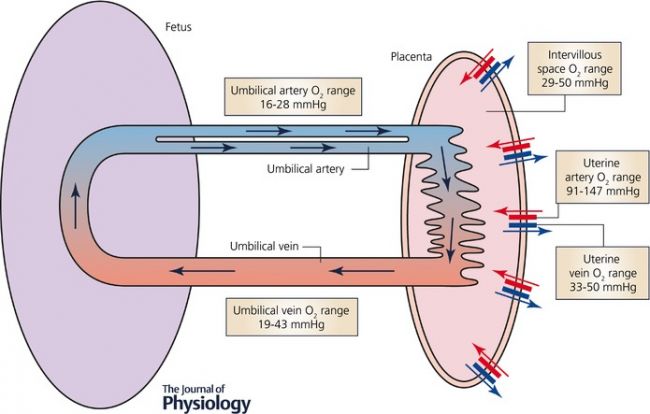
Blood Oxygen Levels
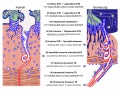
Maternal and Fetal Placental Circulations[22]
Showing the major compartments and published attributed in vivo oxygen values.
Term Placenta Measurements
There are a variety of diagnostic and morphological measurements that can be made of the placenta during pregnancy and at term.
Simple measurements of overall placental diameter, thickness and volume:
- placental diameter - is measured in the transverse section by calculating the maximum dimensions of the chorionic surface.
- placental thickness - is measured at its mid-portion from the chorionic plate to the basilar plate, on a longitudinal plane (less than 4 cm at term). Excludes any abnormalities (fibroids, myometrial contractions, or venous lakes). The placental thickness approximates in millimeters to the weeks of gestation.
- placental volume - is measured by a range of different methods and calculations, more recently with three-dimensional ultrasound.
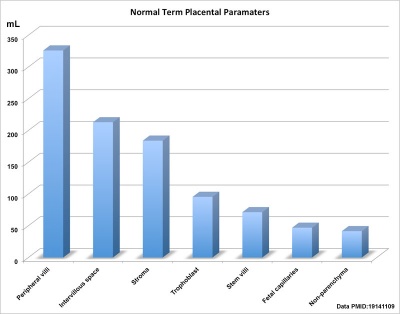
Detailed morphometric indices at term of placental composition, villous capillarization and the mean cross-sectional areas of peripheral villi and capillaries, data from a study sample of 15 normal placenta (mean placental volume, 652 ml).[23][24]
| Variable | Unit | Placenta (mean, n = 15) |
| Intervillous space | mL | 213 |
| Stem villi | mL | 71.4 |
| Peripheral villi | mL | 326 |
| Trophoblast | mL | 95.5 |
| Stroma | mL | 184 |
| Fetal capillaries | mL | 46.9 |
| Non-parenchyma | mL | 41.5 |
| Peripheral villi | km | 89.2 |
| Fetal capillaries | km | 310 |
| total surface area villi | µm2 | 3700 |
| total surface area capillary | µm2 | 150 |
| Capillaries | mL mL-1 | 0.147 |
| Length ratio | km km-1 | 3.6 |
| Table data[24] |
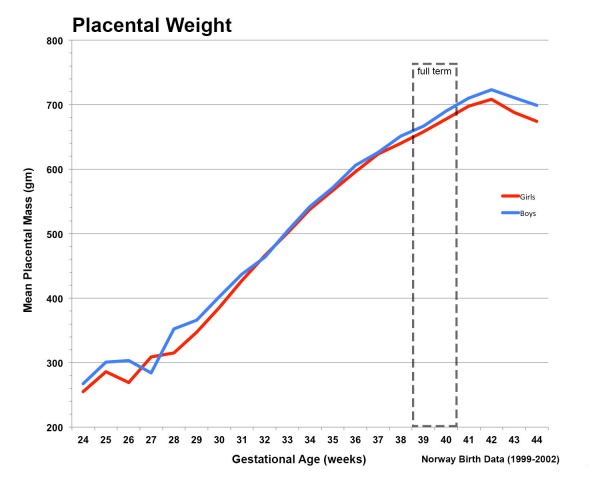
Placental Weight
Fetal Placental weight growth[25]
Placental Factors
As the placenta develops it becomes the source of many different factors (hormones, growth factors) and also has the ability to metabolise both maternal and fetal factors.
Placental Growth Factor
(PGF, PLGF) A growth factor of the vascular endothelial growth factor (VEGF) family, released from the placental trophoblast cells and other sources that stimulates blood vessel growth. See review.[26]
- Links: OMIM601121 | Search PubMed
Insulin/insulin-like Growth Factor
- IGF1 and IGF2 are both synthesized in the placenta and the fetus.
- IGF1 present in syncytiotrophoblast and cytotrophoblast at all stages.
- IGF2 not found in the syncytiotrophoblasts.
- IGF2 expression in the villous and extravillous cytotrophoblasts in the first trimester becomes undetectable at term.
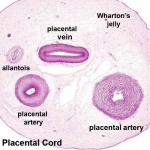
Placental Arteries and Vein
Placental Cord Histology
- Links: Placenta - Cord | Placenta - Histology
Placental Abnormalities
Note there is also a specific page covering the topic of placental abnormalities.
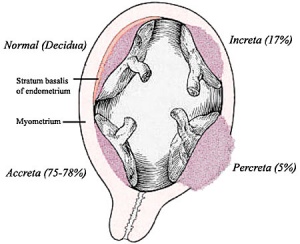
- Placenta Accreta - abnormal adherence, with absence of decidua basalis. The incidence of placenta accreta also significantly increases in women with previous cesarean section compared to those without a prior surgical delivery.
- Placenta Increta - occurs when the placenta attaches deep into the uterine wall and penetrates into the uterine muscle, but does not penetrate the uterine serosa. Placenta increta accounts for approximately 15-17% of all cases.
- Placenta Percreta - placental villi penetrate myometrium and through to uterine serosa.
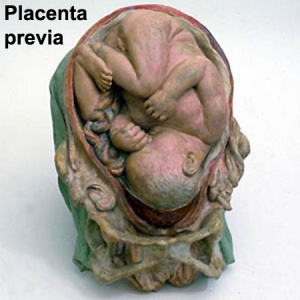
- Placenta Previa - In this placenatal abnormality, the placenta overlies internal os of uterus, essentially covering the birth canal. This condition occurs in approximately 1 in 200 to 250 pregnancies. In the third trimester and at term, abnormal bleeding can require cesarian delivery and can also lead to Abruptio Placenta. Ultrasound screening programs during 1st and early 2nd trimester pregnancies now include placental localization. Diagnosis can also be made by transvaginal ultrasound.
- Vasa Previa - (vasa praevia) placental abnormality where the fetal vessels lie within the membranes close too or crossing the inner cervical os (opening). This occurs normally in 1:2500-5000 pregnancies and leads to complications similar too those for Placenta Previa.Type II is defined as the condition where the fetal vessels are found crossing over the internal os connecting either a bilobed placenta or a succenturiate lobe with the main placental mass. Some recent evidence of successful in utero laser ablation of type II vasa previa at 22.5 weeks of gestation.
- Abruptio Placenta - a retroplacental blood clot formation, abnormal hemorrhage prior to delivery.
- Chronic Intervillositis - (massive chronic intervillositis, chronic histiocytic intervillositis) Rare placental abnormality and pathology defined by inflammatory placental lesions, mainly in the intervillous space (IVS), with a maternal infiltrate of mononuclear cells (monocytes, lymphocytes, histiocytes) and intervillous fibrinoid deposition.
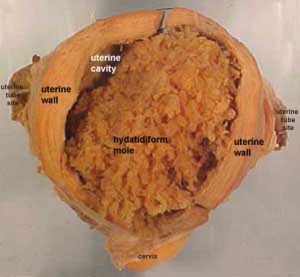
- Hydatidiform mole - placental tumor with no embryo development. Several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
- Links: Placenta - Abnormalities
Placental Cord Abnormalities
There are few abnormalities associated with umbilical cord development, other that abnormally short or long cords, which in most cases do not cause difficulties. In some cases though, long cords can wrap around limbs or the fetus neck, which can then restrict blood flow or lead to tissue or nerve damage, and therefore effect develoment.
- Cord knotting - can also occur (1%) in most cases these knots have no effect, in some cases of severe knotting this can prevents the passage of placental blood.
- Cord torsion - Rare event where even without knot formation can also affect placental blood flow, even leading to fetal demise.
- Links: Placenta Cord Development | Placenta - Abnormalities | WebPath - umbilical cord knot 1 | WebPath - umbilical cord knot 2 | WebPath - Pseudoknot of umbilical cord, gross | WebPath - Torsion of umbilical cord, gross | WebPath - Torsion of umbilical cord, with fetal demise, gross
Placental Infections
- Several infective agents may cross into the placenta from the maternal circulation, as well as enter the embry/fetal circulation. The variety of bacterial infections that can occur during pregnancy is as variable as the potential developmental effects, from virtually insignificant to a major developmental, abortive or fatal in outcome.
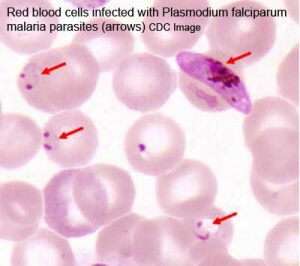
- Pregnant women have an increased susceptibility to malaria infection. Malarial infection of the placenta by sequestration of the infected red blood cells leading to low birth weight and other effects. There are four types of malaria caused by the protozoan parasite Plasmodium falciparum (main), Plasmodium vivax, Plasmodium ovale, Plasmodium malariae). This condition is common in regions where malaria is endemic with women carrying their first pregnancy (primigravida).
Placental Pathology
- Chronic Villitis - can occur following placental infection leading to maternal inflammation of the villous stroma, often with associated intervillositis. The inflammation can lead to disruption of blood flow and necrotic cell death.
- Massive Chronic Intervillositis (MCI) - maternal blood-filled space is filled with CD68-positive histiocytes and an increase in fibrin, occuring more commonly in the first trimester.
- Meconium Myonecrosis - prolonged meconium exposure leads to toxic death of myocytes of placental vessels (umbilical cord or chorionic plate).
- Neuroblastoma - a fetal malignancy that leads to an enlarged placenta, with tumor cells in the fetal circulation and rarely in the chorionic villi.
- Thrombophilias - (protein C or S deficiency, factor V Leiden, sickle cell disease, antiphospholipid antibody) can generate an increased fibrin/fibrinoid deposition in the maternal or intervillous space, this can trap and kill villi.
Comparative Placentation
The usual mouse model does not reflect human placentation in structure or function. Mouse does not synthesise estrogen not (Hyperglycosylated) chorionic gonadotropin.
The Comparative Placentation website has been available online for many years providing valuable information about placentation in a wide range of species. The site was developed by Dr Kurt Benirschke, a longstanding expert in the field.[27][28] Dr Kurt Benirschke retired in 1994 and continued to develop and maintain the website (that is still available) until 2012, but it has not been updated since that time.
References
- ↑ Brown ZA, Schalekamp-Timmermans S, Tiemeier HW, Hofman A, Jaddoe VW & Steegers EA. (2014). Fetal sex specific differences in human placentation: a prospective cohort study. Placenta , 35, 359-64. PMID: 24720894 DOI.
- ↑ Rosenfeld CS. (2015). Sex-Specific Placental Responses in Fetal Development. Endocrinology , 156, 3422-34. PMID: 26241064 DOI.
- ↑ 3.0 3.1 O'Shaughnessy PJ, Antignac JP, Le Bizec B, Morvan ML, Svechnikov K, Söder O, Savchuk I, Monteiro A, Soffientini U, Johnston ZC, Bellingham M, Hough D, Walker N, Filis P & Fowler PA. (2019). Alternative (backdoor) androgen production and masculinization in the human fetus. PLoS Biol. , 17, e3000002. PMID: 30763313 DOI.
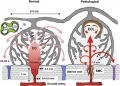
- ↑ Guettler J, Forstner D & Gauster M. (2021). Maternal platelets at the first trimester maternal-placental interface - Small players with great impact on placenta development. Placenta , , . PMID: 34920861 DOI.
- ↑ Turco MY & Moffett A. (2019). Development of the human placenta. Development , 146, . PMID: 31776138 DOI.
- ↑ Freedman AA, Hogue CJ, Marsit CJ, Rajakumar A, Smith AK, Goldenberg RL, Dudley DJ, Saade GR, Silver RM, Gibbins KJ, Stoll BJ, Bukowski R & Drews-Botsch C. (2019). Associations Between the Features of Gross Placental Morphology and Birthweight. Pediatr. Dev. Pathol. , 22, 194-204. PMID: 30012074 DOI.
- ↑ Michelsen TM, Henriksen T, Reinhold D, Powell TL & Jansson T. (2018). The human placental proteome secreted into the maternal and fetal circulations in normal pregnancy based on 4-vessel sampling. FASEB J. , , fj201801193R. PMID: 30335547 DOI.
- ↑ Böing M, Brand-Saberi B & Napirei M. (2018). Murine transcription factor Math6 is a regulator of placenta development. Sci Rep , 8, 14997. PMID: 30301918 DOI.
- ↑ Tabrizi MEA, Lancaster TL, Ismail TM, Georgiadou A, Ganguly A, Mistry JJ, Wang K, Rudland PS, Ahmad S & Gross SR. (2018). S100P enhances the motility and invasion of human trophoblast cell lines. Sci Rep , 8, 11488. PMID: 30065265 DOI.
- ↑ Burton GJ & Jauniaux E. (2018). Development of the Human Placenta and Fetal Heart: Synergic or Independent?. Front Physiol , 9, 373. PMID: 29706899 DOI.
- ↑ Scotti M & Kmita M. (2012). Recruitment of 5' Hoxa genes in the allantois is essential for proper extra-embryonic function in placental mammals. Development , 139, 731-9. PMID: 22219351 DOI.
- ↑ Bonnin A, Goeden N, Chen K, Wilson ML, King J, Shih JC, Blakely RD, Deneris ES & Levitt P. (2011). A transient placental source of serotonin for the fetal forebrain. Nature , 472, 347-50. PMID: 21512572 DOI.
- ↑ Cox B, Kotlyar M, Evangelou AI, Ignatchenko V, Ignatchenko A, Whiteley K, Jurisica I, Adamson SL, Rossant J & Kislinger T. (2009). Comparative systems biology of human and mouse as a tool to guide the modeling of human placental pathology. Mol. Syst. Biol. , 5, 279. PMID: 19536202 DOI.
- ↑ Carter AM & Enders AC. (2004). Comparative aspects of trophoblast development and placentation. Reprod. Biol. Endocrinol. , 2, 46. PMID: 15236656 DOI.
- ↑ Otis EM and Brent R. Equivalent ages in mouse and human embryos. (1954) Anat Rec. 120(1):33-63. PMID 13207763
- ↑ Varghese B, Singh N, George RA & Gilvaz S. (2013). Magnetic resonance imaging of placenta accreta. Indian J Radiol Imaging , 23, 379-85. PMID: 24604945 DOI.
- ↑ Hempstock J, Cindrova-Davies T, Jauniaux E & Burton GJ. (2004). Endometrial glands as a source of nutrients, growth factors and cytokines during the first trimester of human pregnancy: a morphological and immunohistochemical study. Reprod. Biol. Endocrinol. , 2, 58. PMID: 15265238 DOI.
- ↑ Demir R, Kaufmann P, Castellucci M, Erbengi T & Kotowski A. (1989). Fetal vasculogenesis and angiogenesis in human placental villi. Acta Anat (Basel) , 136, 190-203. PMID: 2481376
- ↑ 19.0 19.1 Lisman BA, van den Hoff MJ, Boer K, Bleker OP, van Groningen K & Exalto N. (2007). The architecture of first trimester chorionic villous vascularization: a confocal laser scanning microscopical study. Hum. Reprod. , 22, 2254-60. PMID: 17545656 DOI.
- ↑ Osol G & Mandala M. (2009). Maternal uterine vascular remodeling during pregnancy. Physiology (Bethesda) , 24, 58-71. PMID: 19196652 DOI.
- ↑ Palmer SK, Zamudio S, Coffin C, Parker S, Stamm E & Moore LG. (1992). Quantitative estimation of human uterine artery blood flow and pelvic blood flow redistribution in pregnancy. Obstet Gynecol , 80, 1000-6. PMID: 1448242
- ↑ Nye GA, Ingram E, Johnstone ED, Jensen OE, Schneider H, Lewis RM, Chernyavsky IL & Brownbill P. (2018). Human placental oxygenation in late gestation: experimental and theoretical approaches. J. Physiol. (Lond.) , 596, 5523-5534. PMID: 29377190 DOI.
- ↑ Mayhew TM, Jenkins H, Todd B & Clifton VL. (2008). Maternal asthma and placental morphometry: effects of severity, treatment and fetal sex. Placenta , 29, 366-73. PMID: 18328557 DOI.
- ↑ 24.0 24.1 Mayhew TM. (2009). A stereological perspective on placental morphology in normal and complicated pregnancies. J. Anat. , 215, 77-90. PMID: 19141109 DOI.
- ↑ Thompson JM, Irgens LM, Skjaerven R & Rasmussen S. (2007). Placenta weight percentile curves for singleton deliveries. BJOG , 114, 715-20. PMID: 17516963 DOI.
- ↑ De Falco S. (2012). The discovery of placenta growth factor and its biological activity. Exp. Mol. Med. , 44, 1-9. PMID: 22228176 DOI.
- ↑ Benirschke K. (1998). Remarkable placenta. Clin Anat , 11, 194-205. PMID: 9579593 <194::AID-CA8>3.0.CO;2-T DOI.
- ↑ Benirschke K. (2002). Placentas, peccaries, and pathologists: reminiscences of Kurt Benirschke on his career: an interview with Rebecca N. Baergen. Int. J. Gynecol. Pathol. , 21, 289-300. PMID: 12068178
Reviews
Raeside JI. (2017). A Brief Account of the Discovery of the Fetal/Placental Unit for Estrogen Production in Equine and Human Pregnancies: Relation to Human Medicine. Yale J Biol Med , 90, 449-461. PMID: 28955183
Ferner K & Mess A. (2011). Evolution and development of fetal membranes and placentation in amniote vertebrates. Respir Physiol Neurobiol , 178, 39-50. PMID: 21470579 DOI.
Riquelme G. (2011). Review: Placental syncytiotrophoblast membranes--domains, subdomains and microdomains. Placenta , 32 Suppl 2, S196-202. PMID: 21272934 DOI.
Cartwright JE, Fraser R, Leslie K, Wallace AE & James JL. (2010). Remodelling at the maternal-fetal interface: relevance to human pregnancy disorders. Reproduction , 140, 803-13. PMID: 20837731 DOI.
Ferner K & Mess A. (2011). Evolution and development of fetal membranes and placentation in amniote vertebrates. Respir Physiol Neurobiol , 178, 39-50. PMID: 21470579 DOI.
Riquelme G. (2011). Review: Placental syncytiotrophoblast membranes--domains, subdomains and microdomains. Placenta , 32 Suppl 2, S196-202. PMID: 21272934 DOI.
Cartwright JE, Fraser R, Leslie K, Wallace AE & James JL. (2010). Remodelling at the maternal-fetal interface: relevance to human pregnancy disorders. Reproduction , 140, 803-13. PMID: 20837731 DOI.
Huppertz B. (2008). The anatomy of the normal placenta. J. Clin. Pathol. , 61, 1296-302. PMID: 18755720 DOI.
Wang Y, Zhao S. Vascular Biology of the Placenta. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. Bookshelf NBK53247
Articles
Dhakal P, Kelleher AM, Behura SK & Spencer TE. (2020). Sexually dimorphic effects of forkhead box a2 (FOXA2) and uterine glands on decidualization and fetoplacental development. Proc. Natl. Acad. Sci. U.S.A. , , . PMID: 32900950 DOI.
Porto L, Aviram A, Jackson R, Carson M, Melamed N, Barrett J & Mei-Dan E. (2020). Lateral placentation and adverse perinatal outcomes. Placenta , 101, 1-3. PMID: 32905973 DOI.
Salafia CM, Yampolsky M, Misra DP, Shlakhter O, Haas D, Eucker B & Thorp J. (2010). Placental surface shape, function, and effects of maternal and fetal vascular pathology. Placenta , 31, 958-62. PMID: 20933281 DOI.
Yetter JF. (1998). Examination of the placenta. Am Fam Physician , 57, 1045-54. PMID: 9518951
Books
- Wang Y, Zhao S. Vascular Biology of the Placenta. San Rafael (CA): Morgan & Claypool Life Sciences; 2010. Available from: http://www.ncbi.nlm.nih.gov/books/NBK53247
- Boyd, J.D., Hamilton, W.J. 1970. The utero-placental circulatory system. In: The Human Placenta. W Heffer and Sons Ltd, London.
Historic Textbooks and Research
- The Elements of Embryology (1883)
- Atlas of the Development of Man Volume 1 (1907)
- Manual of Human Embryology (1910)
- Manual of Human Embryology (1917) (unedited)
- Text-Book of Embryology (1921) | The Foetal Membranes in Man
- Bloxam W. On the structure of the human placenta, and its connexion with the uterus. (1840)
- Herzog MA. A contribution to our knowledge of the earliest known stages of placentation and embryonic development in man. (1909) Amer. J Anat., 9(3): 361-400.
- Grosser O. VII. The development of the egg membranes and the placenta; menstruation in Keibel F. and Mall FP. Manual of Human Embryology I. (1910) J. B. Lippincott Company, Philadelphia.
- Bremer JL. The interrelations of the mesonephros, kidney and placenta in different classes of animals. (1918) Amer. J Anat. 19(2): 179-209.
- Fujimura G. Cytological studies on the internal secretory functions in the human placenta and decidua. (1921) J Morphol. 35(3): 486-576.
- Corner GW. Cyclic changes in the ovaries and uterus of swine, and their relations to the mechanism of implantation. (1921) Contrib. Embryol., Carnegie Inst. Wash. Publ. 394, :117-146.
- Dawson AB. The origin and occurrence of the single umbilical artery in normal and abnormal human fetuses. (1922) Anat. Rec. 24(5): 321-342.
- Boyd JD. Hamilton WJ. Electron microscopic observations on the cytotrophoblast contribution to the syncytium in the human placenta. (1966) J. Anat., 100(Pt 3): 535-48. PMID 5965440
- Strachan GI. The development and structure of the human placenta. (1923) J Obstet. and Gynaecol. (1923)
- Wislocki GE. and Bennett SB. The histology and cytology of the human and monkey placenta, with special reference to the trophoblast. (1943) Amer. J Anat. 337-448.
- Baker BL. Hook SJ. and Severinghaus AE. The cytological structure of the human chorionic villus and decidua parietalis. (1944) Amer. J Anat. 73(3): 291-325.
- Hertig AT. lnvolution of tissues in fetal life: a review. (1946) Anat. Rec. 94: 96-116.
- Ramsey EM. Corner GW. Jr. Donner MW. and Stran HM. Radioangiographic studies of circulation in the maternal placenta of the rhesus monkey: preliminary report. (1960) Proc. Natl. Acad. Sci. U.S.A., 46(7): 1003-8 PMID 16590693
- Ramsey EM. Placental Circulation. (1972) MCV Quarterly, 8(1): 61-68.
Search PubMed
Search April 2010
- Placenta Development - All (10032) Review (1896) Free Full Text (2435)
- Placental Development - All (9103) Review (1762) Free Full Text (2219)
- Placental Membranes - All (10083) Review (748) Free Full Text (1728)
Search Pubmed: Placenta Development | Placental Development | Placental Membranes
Additional Images
see all online Placental materials
Historic
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
| Gray 1918 |
|---|
| Gray H. Anatomy of the human body. (1918) Philadelphia: Lea & Febiger.
|
| Bailey1921 |
|---|
| Bailey FR. and Miller AM. Text-Book of Embryology (1921) New York: William Wood and Co.
|
Terms
| Placenta Terms (expand to view) |
|---|
with an incidence of about 2.8 per 1,000 pregnancies, there is also a rarer form of extra-abdominal varices.PMID 24883288
with an incidence of about 2.8 per 1,000 pregnancies, there is also a rarer form of extra-abdominal varices. PMID 24883288
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- The Human Placenta Project - a collaborative research effort, launched by the NICHD to understand the role of the placenta in health and disease.Human Placenta Project | Video
- International Federation of Placenta Associations IFPA Meetings | 2011 IFPA Meeting, Geilo, Norway
- USA NICHD - Human Placenta Project (HPP) is a collaborative research effort to understand the role of the placenta in health and disease. Factsheet PDF
| System Links: Introduction | Cardiovascular | Coelomic Cavity | Endocrine | Gastrointestinal Tract | Genital | Head | Immune | Integumentary | Musculoskeletal | Neural | Neural Crest | Placenta | Renal | Respiratory | Sensory | Birth |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 26) Embryology Placenta Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Placenta_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G