Implantation
| Embryology - 26 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
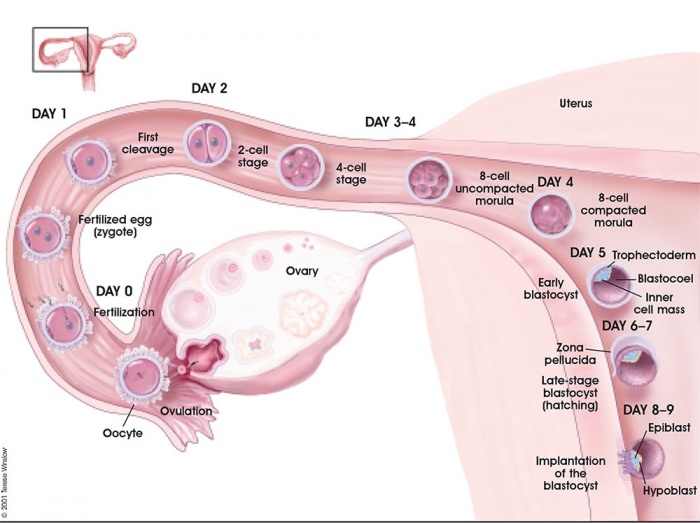
The term "implantation" is used to describe process of attachment and invasion of the uterus endometrium by the blastocyst (conceptus) in placental animals. In humans, this process begins at the end of week 1, with most successful human pregnancies the conceptus implants 8 to 10 days after ovulation, and early pregnancy loss increases with later implantation.[1] The implantation process continues through the second week of development.
The initial phase of the implantation process is "adplantation". This first phase requires the newly hatched blastocyst to loosely adhere to the endometrial epithelium, often "rolling" to the eventual site of implantation where it is firmly adhered. This process requires both the blastocyst adhesion interaction with the endometrium during the "receptive window".
Subsequent development of the placenta allows maternal support of embryonic and fetal development. If implantation has not proceeded sufficiently during the menstrual cycle to allow hormonal feedback to the ovary, then the next cycle may commence leading to conceptus loss. There is also evidence, from animal models, that a conceptus with major genetic does not develop or implant correctly leading to their loss during the first and second weeks of development.
In recent years with the development or Assisted Reproductive Technologies (ART or IVF) there is a growing interest in this process, with techniques that introduce the blastocyst into the uterus to allow normal implantation to occur.
Abnormal implantation is where this process does not occur in the body of the uterus (ectopic) or where the placenta forms incorrectly. In addition implantation can occur normally but with an abnormal conceptus, as in a hydatiform mole development.
Some Recent Findings
PNAS "In many mammalian species, embryo implantation and processes of early pregnancy occur in a hypoxic environment. However, the mechanisms underlying maternal adaptation to hypoxia during early pregnancy remain unclear. This work has uncovered an important mechanism in mammalian reproduction and development by identifying maternal secretory granules that mediate molecular dialogue between the maternal tissue compartments during early pregnancy. This dialogue, which is the molecular basis of adaptation to hypoxia, is critical for embryo implantation and establishment of pregnancy."
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Human+Embryo Implantation | Embryo Implantation | Ectopic Implantation | Uterine Implantation |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Week 1 and 2 Human Development Overview
Endometrial Receptivity
In humans, receptivity occurs 6 days after the post-ovulatory progesterone surge and lasts about 2 to 4 days (about days 20 to 24 of the menstrual cycle).
A similar "receptivity window" occurs following fertilization in other species: rat day 5 and mouse day 4.5. Many studies have looked into identifying markers for this receptivity period both to optimise and to block this process.
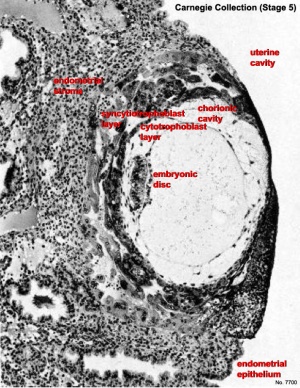
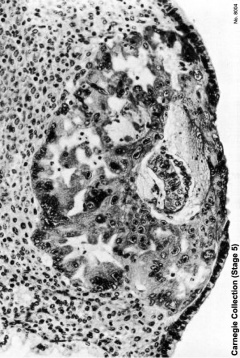
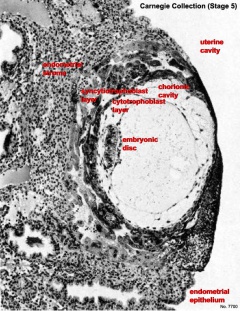
Carnegie Stage 5
The conceptus is shown fully implanted in the uterine wall. Uterine wall is shown on the left and the uterine cavity is shown on the right.
| Early Stage 5 | Late Stage 5 |
|---|---|
|
|
| Stage 5 Links: Week 2 | Implantation | Lecture | Practical | Carnegie Embryos | Category:Carnegie Stage 5 | Next Stage 6 | ||
| Historic Papers: 1941 | 1944 day 9-10 | 1945 day 7.5 | 1945 day 9-10 | ||
|
Implantation Animation
| <html5media height="290" width="250">File:Week2_001.mp4</html5media> | This animation shows the process of implantation, occurring during week 2 of development in humans.
The beginning of the animation shows: the uterus lining (endometrium epithelium), the hatched blastocyst with a flat outer layer of trophoblast cells (green), the inner cell mass which has formed into the bilaminar embryo (epiblast and hypoblast) and the large fluid-filled space (blastocoel). green cells - trophoblast layer of the conceptus blue cells - epiblast layer of the bilaminar embryo yellow cells - hypoblast layer of the bilaminar embryo white cells - uterine endometrium epithelium red - maternal blood vessel |

|
The second week of human development is concerned with the process of implantation and the differentiation of the blastocyst into early embryonic and placental forming structures.
- implantation commences about day 6 to 7
- Adplantation - begins with initial adhesion to the uterine epithelium
- blastocyst then slows in motility, "rolls" on surface, aligns with the inner cell mass closest to the epithelium and stops
- Implantation - migration of the blastocyst into the uterine epithelium, process complete by about day 9
- coagulation plug - left where the blastocyst has entered the uterine wall day 12
Normal Implantation Sites - in uterine wall superior, posterior, lateral
Uterine Epithelium
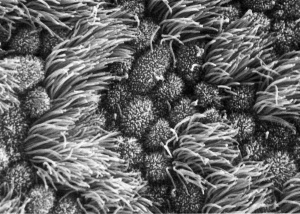
Uterine epithelial cilia are responsible for the initial movement of the ooycte and conceptus (zygote, morula, blastocyst). In humans, this is during the first week of development. Uterine epithelial microvilli are involved with the implantation process. Hormones (estrogen and progesterone) regulate both cilia and microvilli number and structure. The differences in size and shape of cilia and microvilli are shown by scanning micrographs of the lumenal surface of the epithelium lining the mammalian uterine tube.
| Cilia | Microvilli |
|---|---|
|
|
Uterodomes or Pinopods
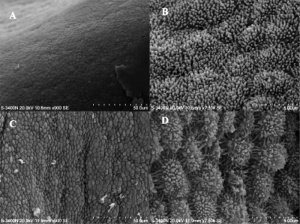
(uterodomes) Cellular feature seen on the apical uterine epithelium surface. The presence of these structures is thought in many species to be a marker for endometrial receptivity. In humans though, recent studies have shown pinopodes are also present throughout the luteal phase of the menstrual cycle.[12] It has also been suggested that their role is not primarily pinocytotic, hence the alternate suggested name "uterodomes" based upon their appearance when imaged by electron microscopy.[13]
These transient microprotrusions inter-digitate with microvilli on the apical syncytiotrophoblast surface of the blastocyst during initial adplantation and implantation process.
Decidua Secreted Factors
The decidual cells secrete many interleukins, growth factors and other factors that can be classified as either supporting implantation (pro-invasive) or inhibiting (anti-invasive). Note that both processes are required for the normal implantation station process to proceed correctly.
- Pro-invasive factors - IL-1β, IL-5, IL-6, IL-7, IL-8, IL-9, IL-13, IL-15, Eotaxin CCL11, IP-10 and RANTES.
- Anti-invasive factors - IL-10, IL-12 and VEGF.
Inhibitory Interactions
The epithelial surface has an associated glycocalyx, the cell surface formed by transmembrane and secreted glycoproteins. Mucins are a major components and mucin 1 (MUC1), a transmembrane protein appears to have dual roles. Firstly, acting as a barrier to both microbial infection and enzymatic attack. Secondly, its expression is altered by hormones and decreased MUC1 expression associated with receptivity. This suggests that high levels of mucin are inhibitory to implantation. Interestingly the same protein is highly expressed later in the placental amnion, where its anti-bacterial and anti-adhesive roles are again required.[14]
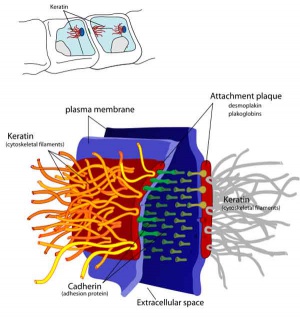
Adhesive Interactions
Cascade of endometrial/conceptus adhesive interactions[15]:
- Carbohydrate-mediated binding to the glycocalyx.
- Progressing to tighter binding involving osteopontin (OPN), members of the immunoglobulin superfamily (IgSF), integrin and cadherin families, trophinin and CD44.
- Activation of proteases including MMPs and ADAMs may well be important in these molecular assemblies.
- Lateral epithelial membrane components, including desmosomes, detach and reassemble as trophectoderm extends between maternal epithelial cells.
Implantation Factors
Molecular Implantation and Decidualization[16]
Trophinin
- Trophinin is a membrane protein expressed on blastocyst trophectoderm cells and on uterine endometrium epithelial cells.
- Adhesion is thought to occur through trophinin-trophinin binding.
- Adhesion also triggers two trophinin mediated effects:
- trophectoderm cells activate for implantation (proliferation, invasion)
- maternal endometrial epithelial cells induced programmed cell death (apoptosis).[17]
- Links: OMIM
Mucin 1
Mucin 1 is a transmembrane protein expressed on the apical borders of secretory epithelial cells including the placental amnion [14] and mammary cells.[18]
- Links: OMIM - Mucin 1
Cytokines
In mice, endometrial secretion of two IL-6 family cytokines, leukemia inhibitory factor (LIF) and Interleukin-11 (IL-11), are key requirements for implantation. A recent human study suggests that there is a similar requirement for human conceptus implantation.[19]
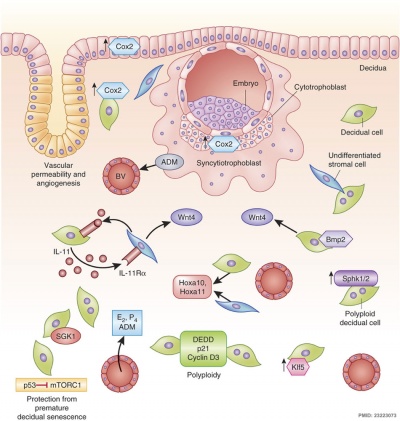
Uterine Leukemia Inhibitory Factor (LIF) Expression[20]
| a - At day 4 of pregnancy, oestrogen E2 induces LIF expression in the endometrial glands, leading to LIF secretion into the uterine lumen. There, LIF binds to its receptors on the surface of epithelial cells. | b - This makes the uterus receptive to the blastocyst, which implants by day 5 of pregnancy. Hu et al. find that LIF expression in the endometrial glands also depends on the regulatory activity of p53. In the absence of p53, insufficient LIF is produced, the uterus does not become adequately receptive, and fewer blastocysts implant. |
Galectin 9
Galectin 9, a protein that binds galactosides and has many different roles, has been identified as a marker for the mid- and late-secretory phases of human endometrium and decidua. The high expression at uterodomes during the period of implantation, suggests that it may also mark endometrial receptivity.[21]
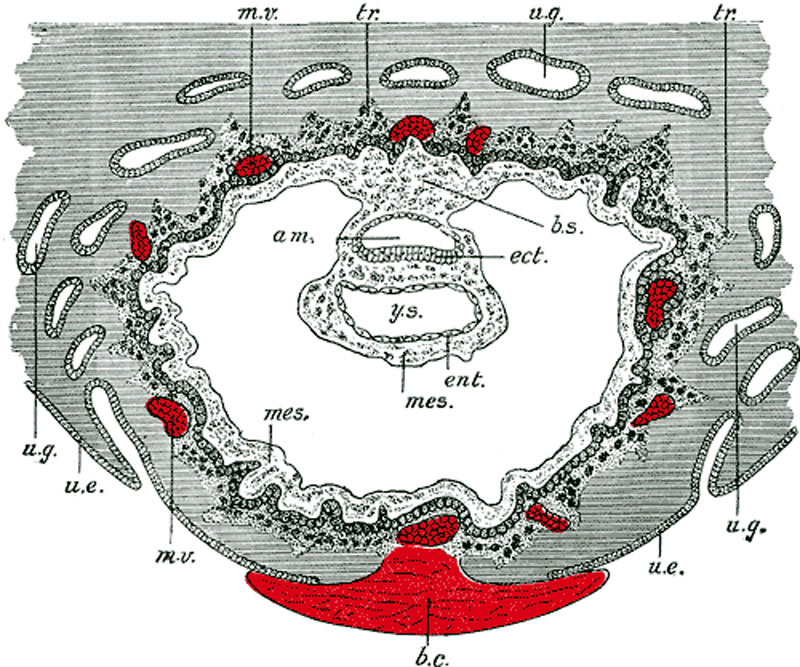
Day 8 to 9
- am. - amniotic cavity
- b.c. - blood clot, at the site of initial implantation
- b.s. - body-stalk, or connective stalk later forming the placental cord region with placental blood vessels
- ect. - embryonic ectoderm that will contribute to embryonic and placental membrane development
- ent. - entoderm (endoderm), this was the historic term for what we today call endoderm that will contribute to embryo development
- mes. - mesoderm, consisting of both embryonic mesoderm (in the trilaminar embryonic disc) and extraembryonic mesoderm (outside the trilaminar embryonic disc)
- m.v. - maternal vessels, spiral arteries that have been opened at their ends
- tr. - trophoblast, relative to the embryonic disc the outer syncitiotrophoblast and inner cytotrophoblast layers that will contribute to placental development
- u.e. - uterine epithelium, the epithelial layer that lines the unerus
- u.g. - uterine glands, the glands that secrete nutrients to support the initial growth both before and after implantation
- y.s. - yolk-sac, the endoderm lined and extraembryonic mesoderm covered cavity that will contribute to the gastrointestinal tract, blood and blood vessels
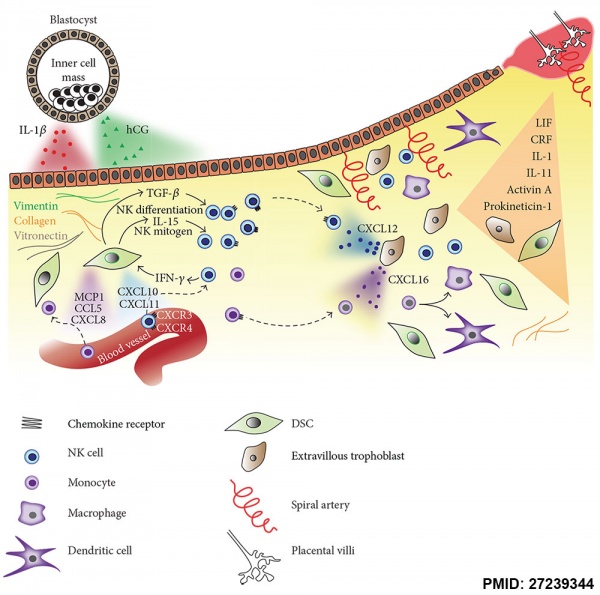
Maternal Immune
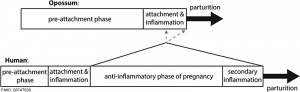
How does the implanting conceptus avoid immune rejection by the maternal immune system? There are a number of maternal and embryonic mechanisms that are thought to act to prevent immune rejection of the implanting conceptus.
Maternal Immune
How does the implanting conceptus avoid immune rejection by the maternal immune system? There are a number of maternal and embryonic mechanisms that are thought to act to prevent immune rejection of the implanting conceptus, though the complete mechanism(s) are unknown. This is particularly relevant to Assisted Reproductive Technologies involving donor eggs.
Below are some examples of research on this topic.
| Decidual Immune Cells | ||
|---|---|---|
| Decidual Macrophages (Mϕ) | Decidual T cells | Uterine Natural Killer cells |
|
|
|
Chemokine Gene Silencing
- Remove the attraction of maternal immune cells.
A mouse study[6] has shown that the normal immune response to inflammation, accumulation of effector T cells in response to chemokine secretion does not occur during implantation. This is prevented locally by epigenetic silencing of chemokine expression in the decidual stromal cells.
Corticotropin-Releasing Hormone
- Kill the maternal immune cells.
Both maternal and implanting conceptus release CRH at the embryo implantation site. This hormone then binds to receptors on the surface of trophoblast (extravillous trophoblast) cells leading to expression of a protein (Fas ligand, FasL) that activates the extrinsic cell death pathway on any local maternal immune cells ( T and B lymphocytes, natural killer cells, monocytes and macrophages).[22] This cannot be the only mechanism, as mice with dysfunctional FasL proteins are still fertile.
- Links: Immune System Development
Decidual Reaction
During pregnancy, at implantation the endometrium is altered by the maternal steroid hormones, estrogen and progesterone and in response to the implanting conceptus and renamed the "decidua". This process of signaling is called the decidual reaction or decidualization, and commences at the site of implantation and spreads throughout the uterine lining.
- occurs within the uterus wall
- initially at site of implantation and includes both cellular and matrix changes
- reaction spreads throughout entire uterus, not at cervix
- promoted by the maternal steroid hormones, estrogen and progesterone
- extensive proliferation and differentiation of uterine stromal cells
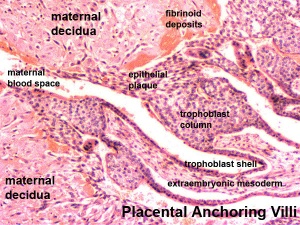
- deposition of fibrinoid and glycogen and epithelial plaque formation (at anchoring villi)
- presence of decidual cells are indicative of pregnancy
In uterine stromal decidualization, bone morphogenetic protein 2 (BMP2) nonactive precursor protein is cleaved by proprotein convertase 5/6 (PC6) to produce the active form. Deletion or knockdown of either BMP2 or PC6 inhibits decidualization leads to implantation failure and female infertility.[23]
A recent human histological study has shown that endometrial stromal cell decidualization leads to a loss of lymphatics particularly apparent around the uterine spiral arteries.[24]
Cervical Mucus Plug
Along with the decidualization, estrogen also stimulates the production of mucus from glands at the opening of the uterus, the cervix, where it joins the vagina. This secreted mucus then forms a plug/barrier (CMP) acting in a mechanical and antibacterial manner.
Abnormal Implantation
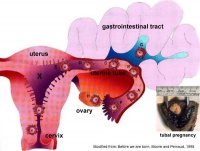
Tubal Pregnancy
Abnormal implantation sites or Ectopic Pregnancy occurs if implantation is in uterine tube or outside the uterus.
- sites - external surface of uterus, ovary, bowel, gastrointestinal tract, mesentry, peritoneal wall
- If not spontaneous then, embryo has to be removed surgically
Tubal pregnancy - 94% of ectopic pregnancies
- if uterine epithelium is damaged (scarring, pelvic inflammatory disease)
- if zona pellucida is lost too early, allows premature tubal implantation
- embryo may develop through early stages, can erode through the uterine horn and reattach within the peritoneal cavity
|
|
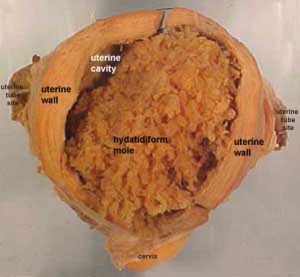
Hydatidiform Mole
Another type of abnormality is when only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole", which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus. The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo.
There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
This topic is also covered in Placenta - Abnormalities
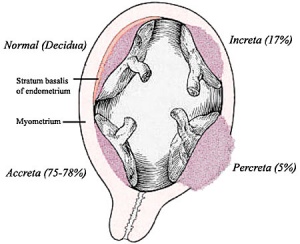
Abnormal Placentation
Abnormalities can range from anatomical associated with degree or site of inplantation, structure (as with twinning), to placental function, placento-maternal effects (pre-eclampsia, fetal erythroblastosis) and finally mechanical abnormalities associated with the placental (umbilical) cord.
This topic is also covered in Placenta - Abnormalities
References
- ↑ Wilcox AJ, Baird DD & Weinberg CR. (1999). Time of implantation of the conceptus and loss of pregnancy. N. Engl. J. Med. , 340, 1796-9. PMID: 10362823 DOI.
- ↑ 2.0 2.1 Griffith OW, Chavan AR, Protopapas S, Maziarz J, Romero R & Wagner GP. (2017). Embryo implantation evolved from an ancestral inflammatory attachment reaction. Proc. Natl. Acad. Sci. U.S.A. , 114, E6566-E6575. PMID: 28747528 DOI.
- ↑ Yu M, Qin H, Wang H, Liu J, Liu S & Yan Q. (2020). N-glycosylation of uterine endometrium determines its receptivity. J. Cell. Physiol. , 235, 1076-1089. PMID: 31276203 DOI.
- ↑ West RC, Ming H, Logsdon DM, Sun J, Rajput SK, Kile RA, Schoolcraft WB, Roberts RM, Krisher RL, Jiang Z & Yuan Y. (2019). Dynamics of trophoblast differentiation in peri-implantation-stage human embryos. Proc. Natl. Acad. Sci. U.S.A. , 116, 22635-22644. PMID: 31636193 DOI.
- ↑ Santillán I, Lozano I, Illán J, Verdú V, Coca S, Bajo-Arenas JM & Martinez F. (2015). Where and when should natural killer cells be tested in women with repeated implantation failure?. J. Reprod. Immunol. , 108, 142-8. PMID: 25708533 DOI.
- ↑ 6.0 6.1 Nancy P, Tagliani E, Tay CS, Asp P, Levy DE & Erlebacher A. (2012). Chemokine gene silencing in decidual stromal cells limits T cell access to the maternal-fetal interface. Science , 336, 1317-21. PMID: 22679098 DOI.
- ↑ Domínguez F, Simón C, Quiñonero A, Ramírez MÁ, González-Muñoz E, Burghardt H, Cervero A, Martínez S, Pellicer A, Palacín M, Sánchez-Madrid F & Yáñez-Mó M. (2010). Human endometrial CD98 is essential for blastocyst adhesion. PLoS ONE , 5, e13380. PMID: 20976164 DOI.
- ↑ Cakmak H & Taylor HS. (2011). Implantation failure: molecular mechanisms and clinical treatment. Hum. Reprod. Update , 17, 242-53. PMID: 20729534 DOI.
- ↑ Duzyj CM, Barnea ER, Li M, Huang SJ, Krikun G & Paidas MJ. (2010). Preimplantation factor promotes first trimester trophoblast invasion. Am. J. Obstet. Gynecol. , 203, 402.e1-4. PMID: 20708167 DOI.
- ↑ Fang LQ, Zhang H, Ding XY, Li DQ, Hou XL, Qiao H, Bai J & Wang ZB. (2010). Mouse trophoblastic cells exhibit a dominant invasiveness phenotype over cancer cells. Cancer Lett. , 299, 111-8. PMID: 20826050 DOI.
- ↑ Ren Q, Guan S, Fu J & Wang A. (2010). Temporal and spatial expression of Muc1 during implantation in sows. Int J Mol Sci , 11, 2322-35. PMID: 20640155 DOI.
- ↑ Quinn CE & Casper RF. (2009). Pinopodes: a questionable role in endometrial receptivity. Hum. Reprod. Update , 15, 229-36. PMID: 18997181 DOI.
- ↑ Murphy CR. (2000). Understanding the apical surface markers of uterine receptivity: pinopods-or uterodomes?. Hum. Reprod. , 15, 2451-4. PMID: 11098008
- ↑ 14.0 14.1 Sood R, Zehnder JL, Druzin ML & Brown PO. (2006). Gene expression patterns in human placenta. Proc. Natl. Acad. Sci. U.S.A. , 103, 5478-83. PMID: 16567644 DOI.
- ↑ Singh H & Aplin JD. (2009). Adhesion molecules in endometrial epithelium: tissue integrity and embryo implantation. J. Anat. , 215, 3-13. PMID: 19453302 DOI.
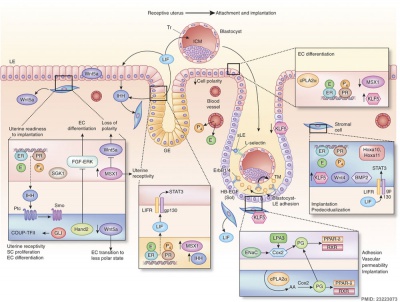
- ↑ Cha J, Sun X & Dey SK. (2012). Mechanisms of implantation: strategies for successful pregnancy. Nat. Med. , 18, 1754-67. PMID: 23223073 DOI.
- ↑ Tamura N, Sugihara K, Akama TO & Fukuda MN. (2011). Trophinin-mediated cell adhesion induces apoptosis of human endometrial epithelial cells through PKC-δ. Cell Cycle , 10, 135-43. PMID: 21191175 DOI.
- ↑ Gendler SJ, Lancaster CA, Taylor-Papadimitriou J, Duhig T, Peat N, Burchell J, Pemberton L, Lalani EN & Wilson D. (1990). Molecular cloning and expression of human tumor-associated polymorphic epithelial mucin. J. Biol. Chem. , 265, 15286-93. PMID: 1697589
- ↑ Marwood M, Visser K, Salamonsen LA & Dimitriadis E. (2009). Interleukin-11 and leukemia inhibitory factor regulate the adhesion of endometrial epithelial cells: implications in fertility regulation. Endocrinology , 150, 2915-23. PMID: 19213836 DOI.
- ↑ Hu W, Feng Z, Teresky AK & Levine AJ. (2007). p53 regulates maternal reproduction through LIF. Nature , 450, 721-4. PMID: 18046411 DOI.
- ↑ Shimizu Y, Kabir-Salmani M, Azadbakht M, Sugihara K, Sakai K & Iwashita M. (2008). Expression and localization of galectin-9 in the human uterodome. Endocr. J. , 55, 879-87. PMID: 18506087
- ↑ Makrigiannakis A, Zoumakis E, Kalantaridou S, Coutifaris C, Margioris AN, Coukos G, Rice KC, Gravanis A & Chrousos GP. (2001). Corticotropin-releasing hormone promotes blastocyst implantation and early maternal tolerance. Nat. Immunol. , 2, 1018-24. PMID: 11590404 DOI.
- ↑ Heng S, Paule S, Hardman B, Li Y, Singh H, Rainczuk A, Stephens AN & Nie G. (2010). Posttranslational activation of bone morphogenetic protein 2 is mediated by proprotein convertase 6 during decidualization for pregnancy establishment. Endocrinology , 151, 3909-17. PMID: 20555025 DOI.
- ↑ Volchek M, Girling JE, Lash GE, Cann L, Kumar B, Robson SC, Bulmer JN & Rogers PA. (2010). Lymphatics in the human endometrium disappear during decidualization. Hum. Reprod. , 25, 2455-64. PMID: 20729537 DOI.
Reviews
Fox C, Morin S, Jeong JW, Scott RT & Lessey BA. (2016). Local and systemic factors and implantation: what is the evidence?. Fertil. Steril. , 105, 873-84. PMID: 26945096 DOI.
Cha J, Sun X & Dey SK. (2012). Mechanisms of implantation: strategies for successful pregnancy. Nat. Med. , 18, 1754-67. PMID: 23223073 DOI.
Hannan NJ, Evans J & Salamonsen LA. (2011). Alternate roles for immune regulators: establishing endometrial receptivity for implantation. Expert Rev Clin Immunol , 7, 789-802. PMID: 22014020 DOI.
Paulson RJ. (2011). Hormonal induction of endometrial receptivity. Fertil. Steril. , 96, 530-5. PMID: 21880274 DOI.
Fukuda MN & Sugihara K. (2008). An integrated view of L-selectin and trophinin function in human embryo implantation. J. Obstet. Gynaecol. Res. , 34, 129-36. PMID: 18412772 DOI.
Aplin JD. (2006). Embryo implantation: the molecular mechanism remains elusive. Reprod. Biomed. Online , 13, 833-9. PMID: 17169205
Articles
Fukuda MN & Sugihara K. (2007). Signal transduction in human embryo implantation. Cell Cycle , 6, 1153-6. PMID: 17495530 DOI.
Search PubMed
Search Pubmed: Embryo Adplantation | Embryo Implantation | tubal pregnancy | Endometrial Receptivity | Placenta Abnormalities | Pinopods | decidualization
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
| Carnegie Collection - Stage 5 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Serial No. | Stage | Grade | Fixative | Embedding Medium | Thinness (µm) | Stain | Year | Notes | ||
| 8020 | 5a | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1942 | Hertig and Rock (1945a)[1] | ||
| 8155 | 5a | Exc. | Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1943 | Hertig and Rock (1949)[2] | ||
| 8225 | 5a | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1944 | Hertig and Rock (1945b)[3] | ||
| 8004 | 5b | Exc | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1942 | Hertig and Rock (1945a)[1] | ||
| 8171 | 5b | Exc | Alc. | C-P | 6 | (Stain - Haematoxylin Eosin) | 1943 | Hertig and Rock (1949)[2] | ||
| 8215 | 5b | Exc | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1944 | Hertig and Rock (1945c)[4] | ||
| 9350 | 5b | Exc | Bouin | ? | ? | (Stain - Haematoxylin Eosin) | 1955 | Heuser (1956)[5] | ||
| 4900 | 5c | Poor | p | P | 10 | p | 1925 | Incomplete. Streeter (1926)[6] | ||
| 7699 | 5c | Exc. | Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1939 | Hertig and Rock (1941)[7] | ||
| 7700 | 5c | Exc. | Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1938 | Hertig and Rock (1941)[7] | ||
| 7771 | 5c | Exc. | Bouin | C-P | 10 | (Stain - Haematoxylin Eosin) | 1940 | Abnormal | ||
| 7950 | 5c | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1941 | Hertig and Rock (1944)[8] | ||
| 8000 | 5c | Poor | Alc. & Bouin | C-P | 8 | (Stain - Haematoxylin Eosin) | 1942 | Abnormal | ||
| 8139 | 5c | Exc. | ? | C-P | 6 | (Stain - Haematoxylin Eosin) | 1943 | Incomplete. Marchetti (1945)[9] | ||
| 8299 | 5c | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin), phlox. | 1945 | Abnormal | ||
| 8329 | 5c | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin), phlox. | 1945 | Abnormal | ||
| 8330 | 5c | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin), phlox. | 1945 | |||
| 8370 | 5c | Poor | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin), phlox. | 1946 | Abnormal | ||
| 8558 | 5c | Exc. | Alc. & Bouin | C-P | 6 | (Stain - Haematoxylin Eosin) | 1947 | |||
| Stage 5 was originally subdivided into 3 sequential parts a, b, c.
Abbreviations
| ||||||||||
References
| ||||||||||
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 26) Embryology Implantation. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Implantation
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G