Paper - The Development of the Atrio-Ventricular Valves in Man
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Odgers PNB. The development of the atrio-ventricular valves in man. (1939) J Anat. 73: 643-57. PMID 17104787
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The Development of the Atrio-Ventricular Valves in Man
By P. N. B. Odgers
From the Department of Human Anatomy, University of Oxford
THE fact that in every text-book of embryology the details of the formation of the atrio-ventricular valves are scanty and that no particular account of their development has appeared since 187 6 when Bernays, who used chiefly calves’ hearts for his material, published his classical paper, suggested a reinvestigation of this matter.
For this purpose I have had at my disposal human embryos of 7.1, 11.2, 11.4, 12.5, 14.5, 15.5, 17, 17.5, 23, 28.5, 46 and 61 mm. crown-rump length. In addition to these the hearts from foetuses of 85, 105, 130 and 210 mm., from two full-time stillborn infants and from a baby of 11 months were specially sectioned for this purpose.
In the detailed description which follows it will perhaps make for clarity to explain that in the tricuspid valve the right cusp in the embryo becomes eventually the inferior or posterior cusp, while the septal cusp becomes the medial one: at the mitral orifice the right cusp of the embryonic heart corresponds to the anterior or aortic cusp, the left one to the posterior cusp of the fully developed valve. Further, it is sometimes convenient to speak of lateral and central cusps. In the former group are included the right cusp of the tricuspid and the left cusp of the mitral valve, while the septal cusp of the tricuspid and the right cusp of the mitral constitute the central cusps.
7.1 mm Embryo
While the superior (dorsal) and inferior (ventral) auriculo-ventricular cushions (hereafter abbreviated to A.-V. cushions) are well marked here, there is no evidence yet of any right or left lateral cushions. There is, however, marked trabeculation of the Ventricular wall.
11.2 mm Embryo
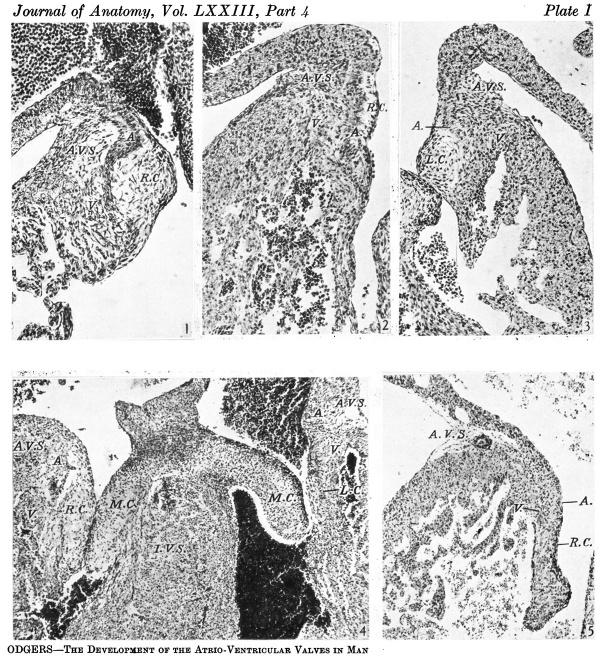
When the right bulbar ridge (or proximal bulbar swelling) is traced in a caudal direction, its left border is seen to join the right side of the superior A.-V. cushion above the level of the right A.-V. orifice. At the level of that orifice the bulbar ridge swells out slightly to become the right lateral cushion (Pl. I, fig. 1). The left lateral cushion also has made its appearance at this stage. It forms a plump projection beneath the endocardium. The positions of these lateral cushions seem to be slightly different. The dorsal half of the right one, where it is at its broadest, is bounded laterally by auricular muscle which separates it from the epicardium occupying the right A.-V. sulcus, while its ventral half, thinning out in a tail-like fashion, is bordered laterally by the muscular trabeculae of the ventricle. In contrast to this, in the case of the left one, which is roughly quadrangular in shape, its lateral border is in relation to the ventricular wall only, while its upper border is in contact with auricular muscle. In conformity with this the left A.-V. sulcus, which is shallower and much less extensive in all its diameters than the right, is just above the level of the cushion. Both the lateral cushions are already connected with the lateral ventricular walls by muscular trabeculae, the chordae musculares. While the right cusp of the tricuspid valve and the left cusp of the mitral are thus clearly to be distinguished at this stage, there is no evidence of an anterior cusp for the tricuspid valve. The right and left superior and inferior tubercles of the fused A.-V. cushions project prominently on the medial margins of the A.-V. orifices, the sulcus between the tubercles on the left side giving this border a definitely bifid appearance, but there is no other indication of the formation of the central cusps.
11.4 mm Embryo
This specimen is, in my opinion, older than its C.R. length would indicate. The latter was computed by counting the sections and there is no record of its original length.
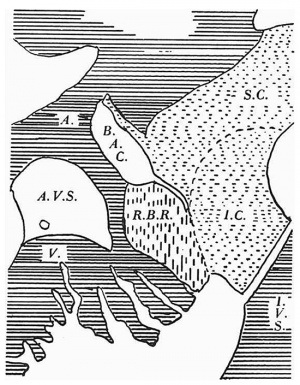
The left border of the right bulbar ridge (R.B.R.) is here seen to have grown across the upper part of the right A.-V. orifice to form the bulbo-auricular channel (B.A.C.) and thus to have become contiguous with the right side of the fused A.-V. cushions, first with the superior and then more caudally with the inferior cushion (I .C.) (Text-fig. 1). At the same time the ventral border of the bulbar ridge projects above the remainder of the original right A.-V. opening as the anlage of the anterior cusp of the tricuspid valve in which it forms all the cushion tissue. This latter is perforce continuous caudally with the right lateral cushion, both cushions being derivatives of the right bulbar ridge. The right and left lateral cushions still maintain their rather different situations which were noted above in the 11.2 mm. embryo. The right cusp of the mitral valve can now be recognized as a well-marked structure. It is obviously formed by the left extremities of the superior and inferior moieties of the fused A.-V. cushions which project as superior and inferior tubercles.
Each of the latter is connected with the lateral wall of the left ventricle by quite a stout band of muscle. At first sight it seems a little difficult to imagine exactly how this occurs, as obviously the trabeculae cannot cross the A.-V. orifice to make contact with the cushion tissue. Each band, however, derived from trabeculae of the ventricular wall, reaches the cushion above and below the left A.-V. orifice and spreads over it, thus forming the anterior and posterior papillary muscles with their chordae musculares. There is no evidence yet of a septal cusp for the tricuspid valve.
12.5, 14.5 and 15.5 mm Embryos
These three specimens may be grouped together. In all of them there is little change in the condition of the lateral cusps.
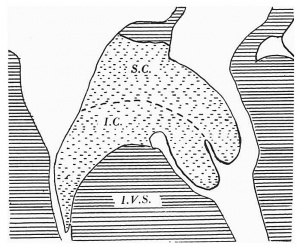
All three, however, show a curiously bilobed appearance in the right cusp of the mitral valve (Text-fig. 2). At 12-5 mm., as in the earlier 11-4 mm. embryo, this is marked only by a notch, but in the later specimens it is much more accentuated. This bilobulation corresponds to the superior and inferior tubercles of the fused cushions. The first indication of the septal cusp of the tricuspid valve occurs in my 14.5 mm. embryo (Text fig- 2) as a projection from the ventral extremity of the right margin of the inferior A.-V. cushion. This is much more pronounced in the later 15-5 mm. specimen.
17 and 17.5 mm Embryos
The tricuspid valve. In the anterior cusp the base of the flap, which is still for the most part composed of cushion tissue, is being invaded by muscle from the ventricular wall. Strands of muscle from both the anterior and the right Ventricular wall appear to meet in a V, which separates the cushion tissue from the epicardium in the A.-V. sulcus. In the right cusp (Pl. 1, fig. 2) the right lateral cushion, which is much less plump than in the earlier specimens, is in contact with auricular muscle for the greater part of its extent. This separates it from the epicardium of the A.-V. sulcus completely, although at the junction of auricular and ventricular muscle the connexion between the two latter becomes tenuous in some sections. Only the ventral extremity of the cushion borders the ventricular muscle which is forming the valve flap. This muscular flap is much better formed than in the earlier embryos. In contrast to its appearance in the 11.2 mm. specimen the A.-V. sulcus has now become definitely angulated on the right side. Whether this is caused by an increasing growth of the ventricular wall dorsalwards or no, it must indicate an invagination of the sulcus here. It may be noted that the auricular and ventricular muscle meet each other just dorsal to the attachment of the valve cusp which thus contains no auricular muscle. The differentiation of the septal cusp proceeds slowly. In these embryos there is, however, just ventral to the right ventral border of the A.-V. cushion, commencing trabeculation of the muscular interventricular septum, and some muscle fibres are growing upwards on either side of the projecting spur of cushion tissue. The papillary muscles of this valve can now be distinguished in great part. The anterior one springs from the right wall of the ventricle and is joined at its lower border to the septal wall by a stout moderator band. The anterior papillary muscle is connected with both the anterior and lateral cusps. The posterior papillary muscle comes from the septal wall and joins by its chordae musculares the lateral cusp. The septal papillary muscle is continuous with the posterior one and is very small. It is represented merely by the trabeculation of the septal wall noted above, just ventral to the A.-V. cushion.
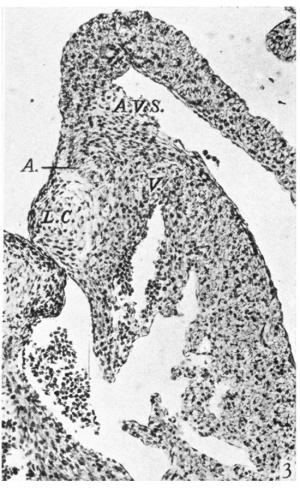
The mitral valve. ‘While in the greater part of its extent the right cusp is formed solely by the fused A.-V. cushions, at its cranial and caudal borders it is much more muscular. This is seen best near its caudal end where the chordae musculares of the posterior papillary muscle are continuous on the ventral border of the cushion tissue with the left side of the muscular interventricular septum. There is now no bilobulation of this cusp as was noted in the earlier specimens. The left lateral cushion is rather smaller and somewhat more oval in shape than it was in the earlier embryos. Its relations to the auricular muscle and to that of the ventricular wall show little change. In contrast, however, to the right side, there is here no angulation of the A.-V. sulcus (Pl. I, fig. 3).
23 mm Embryo
(Pl. I, fig. 4)
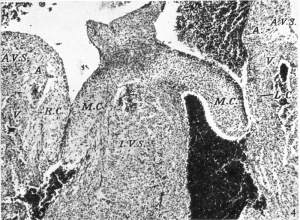
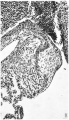
In this specimen the cushion tissue in the valves with the exception of the left lateral one appears to be at its maximum. It is more marked than in the previous embryo or in any of the later ones. The central cusps (M .C.) straddling the dorsal border of the muscular interventricular septum are strikingly prominent. The lateral ones again may be contrasted. On the right side, as in the two previous embryos, the angulation of the A.-V. sulcus is marked and, while the contracted state of the ventricle makes it difficult to define precisely the base of the cusp, I think the auricular muscle joins that of the ventricle just ventral to the line of its attachment. No such angulation is noticed on the left side. In both lateral cusps the cushion tissue seems to have become more ventral. In the case of the right cushion (R.C.), its lower two-thirds are in contact with ventricular muscle which is invading its lateral border, while in the left lateral cusp (L.C.), which is almost completely muscular now, the much smaller cushion mass is bordered entirely by it. In both cusps the cushion tissue thus enters much more into the composition of the actual valve flap than it has done previously. Further, it may be noted, that the dorsal portion of the left lateral cushion has altered its character: it has become more cellular and stains more intensely than the rest of it.
28.5 mm Embryo
In contrast to the last specimen the heart of this embryo shows a marked diminution of cushion tissue as a distinct constituent of the A.-V. valves and its replacement by muscle. Further, the cushion tissue itself, as was first noted in the left lateral cusp of the previous embryo, is more cellular and in the case of the two lateral cusps appears as elongated thickenings of the endocardium.
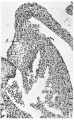
The tricuspid valve. The anterior cusp is almost completely muscularized by muscle which has grown into it from the ventricular wall. There is only a little cushion tissue still remaining on its left margin. The right cusp (Pl. I, fig. 5) is composed for the most part of ventricular muscle with the much attenuated cushion bordering it medially throughout its extent. This cushion tissue, in its lower four-fifths, is in contact with ventricular muscle while in its upper fifth it meets the muscle of the auricle. The junction of the latter with the ventricular muscle is seen to be just ventral to the attachments of the valve cusp. This is much more obvious now than in the previous 23 mm. specimen and is due apparently to an increasing invagination of the right A.-V. sulcus, which was first noted at the 17-5 mm. stage. The base of this cusp at this stage thus consists from within outwards of cushion tissue, auricular muscle, epicardium and ventricular muscle. The septal cusp is better differentiated. There is a small mass of cushion tissue dorsally and laterally but the flap is essentially muscular, muscle strands from the right side of the muscular interventricular septum meeting those which have grown upwards from the chordae musculares.
The mitral valve. The right cusp still contains a good deal of cushion tissue, but it is being muscularized on its ventral border by the muscular chordae which appear to split and to grow along both its ventricular and auricular aspects, while muscle fibres from the left border of the muscular interventricular septum are investing the ventricular surface of the cusp in its dorsal portion. In the left cusp very little cushion tissue remains. It is composed almost entirely of muscle derived from the lateral wall of the ventricle which is continuous with that of the chordae musculares. Neither auricular muscle nor epicardium from the left A.-V. sulcus enter into its composition, for there is still no invagination of the sulcus here as is seen on the right side.
Comparing the different valve cusps at this stage, while there is still much cushion tissue to be seen in the right cusp of the mitral valve and in the septal cusp of the tricuspid, there is little in the right cusp of the tricuspid, less in the left cusp of the mitral, and the anterior cusp of the tricuspid valve is almost completely muscularized.
46 and 61 mm Embryos
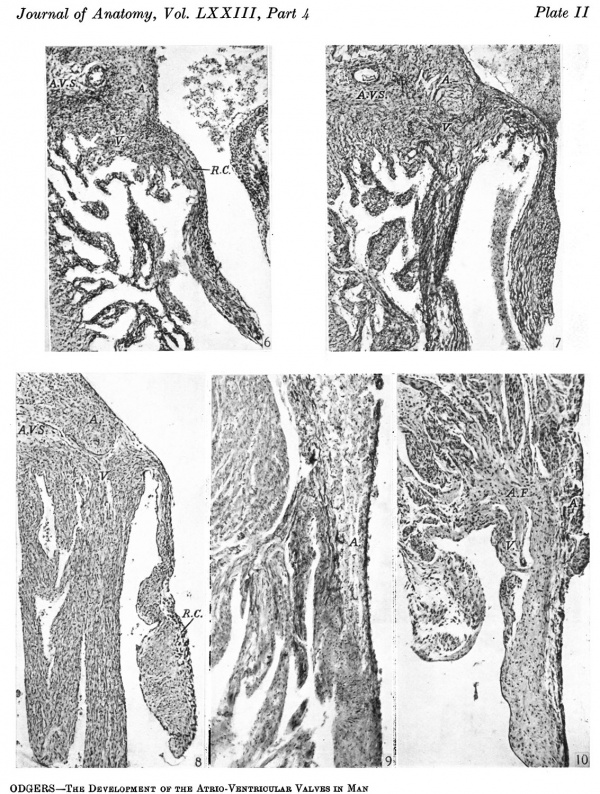
(Pl. II, figs. 6, 7)
In both of these all the valve cusps are increasingly muscular. The remains of the cushion tissue have become more difficult to define accurately and appear as elongated strands of cells, which are often broken up into two parts by portions of the cusp entirely muscular, but which are always lining the auricular surface of the flaps immediately beneath the endocardium. As has been noted in the 28-5 mm. embryo, however, this attenuated cushion tissue reaches almost to the free border of the cusps in the 46 mm. specimen, but in the 61 mm. stage it is much less extensive. The angulation of the A.-V. sulcus seen on the right side in the 28-5 mm. embryo can still be found in the 46 mm. one. Here the vertical limb of the sulcus appears as a narrow line separating the lateral border of the most ventral part of the auricular muscle from that of the ventricle (Pl. II, fig. 6), but it differs from the earlier specimen in that it is less extensive and does not enter into the base of the cusp, from which it is always excluded by the junction of auricular with ventricular muscle. As a corollary to this, auricular muscle now takes no part in the composition of the cusp.
At 61 mm. (Pl. II, fig. 7) even the remains of this angulation has gone and the sulcus appears to be straightened out. With this, whether it be propter hoe or not, the auricular canal can now be said to have disappeared. In neither of these embryos does the muscle of the auricle extend on to any of the other valve cusps.
85, 105 and 130 mm Foetuses
All the hearts so far examined came from embryos already serially sectioned and stained with haemotoxylin and eosin. In the case of the foetuses now to be considered, the hearts were removed and appropriately sectioned, the sections being stained either by van Gieson or by Masson’s trichrome method, which latter was recommended to me by Dr H. M. Carleton. Owing to the plane of the sections the anterior cusp of the tricuspid valve was not examined in these three specimens. All three may be considered together, and the valve cusps in all of them show two remarkable changes when compared with those of the earlier embryos. In the first place, instead of being almost entirely muscular they have become almost completely collagenous, while, secondly, in the case of the lateral cusps this collagenous plate is directly continuous with the epicardium filling the A.-V. sulcus. Actually the collagenous change is most marked in the 85 mm. foetus (Pl. II, fig. 8) where this alteration in character is almost complete in all the cusps except the left lateral one. With it of course the chordae musculares of the earlier hearts have now become chordae tendineae. What may be cushion remains appear in the more ventral half of the cusps as looser, more cellular, tissue beneath the endocardium. In some of the cusps in the 105 and 130 mm. foetuses there are still some muscle fibres on their auricular aspect: this muscle, as must be concluded from the earlier specimens, is ventricular in origin. There are, however, in both my 85 and 105 mm. foetuses instances of auricular muscle straying on to the valve flap in both cusps of the mitral valve in the former and in both the lateral cusps in the latter specimen. There are sections through the right cusp of the tricuspid valve in the 105 mm. foetus which show auricular muscle extending some distance on to the flap and being continuous with the degenerating ventricular muscle on this aspect of it: in other sections this continuity is interrupted opposite the A.-V. sulcus by collagenous fibres derived from the latter. Sections through the left cusp of the mitral valve in the same heart show quite unequivocally auricular muscle lining this surface of the flap in its upper two-fifths. In all these three hearts, in the case of the lateral cusps, the continuity of the collagenous fibres of the cusp with those of the epicardium in the A.-V.. sulcus separates the muscle of the auricle from that of the ventricle. This is in direct contrast to what was constantly found in this situation in all the previous embryos.
210 mm Foetuses
The valve cusps of this heart are all entirely collagenous, and I do not see any evidence of the remains of cushion tissue. There is a little muscle continuous with that of the ventricular wall to be seen on the ventricular aspect of the upper sixth of the anterior cusp of the tricuspid valve, while in the more central sections of the right cusp auricular muscle strays on to the cusp just beyond its attached border (Pl. II, fig. 9). Nothing is seen of this in the more rostral and in the more caudal sections. In all the other cusps of either the tricuspid or the mitral valves auricular muscle is entirely absent. A comparison of the left lateral cusp of this heart with the same cusp at the 130 mm. stage provides a remarkable contrast.
Two Full-Time Stillborn Infants
There is very little difference to record in the appearance of the valve cusps in these hearts in comparison with my 210 mm. foetus. The annulus fibrosus is, however, thicker and better defined. The right cusp of the tricuspid valve in either heart shows some small broken-up bundles of auricular muscle just beyond its attached border but these are always very trifling (Pl. II, fig. 10), while similarly on the ventricular aspect of its base avery small amount of ventricular muscle can be distinguished. On the other hand, none of the other cusps of any valve in these hearts shows any evidence of auricular muscle in its composition. One must mention in this connexion the right or anterior cusp of the mitral valve. Some of my sections of one of these hearts show a picture similar to that illustrated in more than one text-book from adult hearts where the muscle of the left auricle is said to be clothing the base of this cusp. I am sure this is not the case in my specimen. There is nothing to indicate accurately the line of attachment of the anterior cusp here, which appears to be continuous with the wall of the aortic vestibule right up to the semilunar valves, except that opposite the base of the left (posterior) mitral cusp the wall of the vestibule tapers to the much thinner valve cusp. If the two mitral cusps, however, are compared (and this is possible here as both cusps appear in the same section), the auricular muscle is found not to extend so far anteriorly as it does posteriorly, i.e. the left A.-V. sulcus is on a line caudal to the limit of the auricular muscle on the posterior aspect of the aortic vestibule. Therefore, to include auricular muscle in the anterior cusp one must make the line of the latter’s attachment considerably rostral to that of the posterior cusp and this, so far as I know, has never been suggested.
Discussion
From the above detailed description one can distinguish three definite periods i11 the formation of these valves. I (from 11-2 to 23 mm.). Here the valve cusps have two distinct components, the cushion tissue and the muscular trabeculae of the ventricular wall, of which the former is the more prominent. II (from 28-5 to 61 mm.). In these hearts the cusps are apparently essentially muscular with only the remains of the cushion tissue on their auricular aspect. III (from 85 mm. to full term). In this period the muscle in the cusps is replaced entirely by collagenous tissue. These different stages are shown in Pls. I and II in the case of the right lateral cusp.
I. From 11-2 to 23 mm. While the dorsal and ventral A.-V. cushions are well-marked structures in my 5 mm. specimen, the lateral cushions do not appear in my series until the 11-2 mm. stage (Pl. I, fig. 1). Waterston (1918) found both right and left lateral cushions in his 8 mm. embryo, while Mall (1912) described the presence of the left lateral one at 9 mm., “but both are not well pronounced until the embryo is 11 mm. long”. Such a diagram as Walmsley (1929) draws of a transverse section through the atrial canal, which shows it surrounded completely by a continuous ring of cushion tissue with the A.-V. cushions unfused, is clearly misleading. The right bulbar ridge (Frazer, 1931) or proximal bulbar swelling (Tandler, 1913) is certainly continuous in my 11-2 mm. embryo, as Waterston noted, with the right lateral cushion. Whether, as he interpreted it, the latter should be regarded as entirely formed by a downward prolongation of this ridge or whether there is an independent subendocardial proliferation with which the ridge gains continuity, as Mall suggested, is a difficult question to decide. Although in my 11-2 mm. specimen the greatest transverse diameter of the cushion slightly exceeds that of the ridge above it, I am inclined to agree with Waterston and to regard it as formed wholly from the bulbar ridge. Certainly, too, the right bulbar ridge, as is seen in my series from the 11-4« to the 17-5 mm. stage, forms the cushion tissue of the anterior cusp of the tricuspid valve. As Frazer (1916-17) first showed clearly, although both Mall and VVaterston suggested it, the right bulbar ridge grows across the tricuspid orifice (Text-fig. 1) and joins by its left border the fused A.-V. cushions, while its free margin bulges ventrally and overhangs the caudal remaining portion of the tricuspid opening (of which the rostral part is forming the bulbo-auricular channel) to persist as the cushion basis of the anterior cusp of this valve. The right bulbar ridge is thus seen to be responsible for the formation of both the anterior and the right, or posterior, cusps of this valve and their common origin explains the continuity between them which is seen in the adult heart. In the stages I am now considering, this continuity is such that one can only distinguish between the two cusps in serial sections by the patency or otherwise of the A.-V. canal. Further, the primitive fusion of the bulbar ridge with the A.-V. cushions and subsequently with the proliferation from the latter, which fills the caudal ventral portion of the interventricular foramen (0(lgers, 1937~8), accounts for the attachment of the anterior cusp medially in the adult heart to the membranous part of the septum where it meets and becomes continuous with the anterior border of the septal cusp.
The valve cusps appear in the following sequence: While both the lateral cusps are beginning to be well formed at the 11-2 mm. stage, the right, or anterior, cusp of the mitral valve is rather later in its appearance: it is a wellmarked structure in my 11-4« mm. embryo. The anterior cusp of the tricuspid can just be recognized in the same embryo, but is not well formed until the 14-5 mm. stage is reached. The last one to appear is the septal cusp of the tricuspid: this is first indicated clearly in the 15-5 mm. specimen in my series. While both superior (dorsal) and inferior (ventral) A.-V. cushions enter into the composition of the right mitral cusp as is indicated by its bilobulation noted in the 12-5, 14-5 and 15-5 n1m. embryos (Text-fig. 2), the cushion tissue of the septal cusp of the tricuspid valve is formed only from the inferior one. Granted that its accurate definition is always diflicult, it is probably correct to say that the right cusp of the mitral valve has the largest amount of cushion tissue in its composition and the left one the least of all the valve cusps.
I should again insist on the position and relations of the lateral cushions in this stage of their development. They are subendocardial and are entirely separated on their lateral side from the epicardium filling the A.-V. sulcus by auricular muscle joining that of the ventricle, while their ventral halves are in contact with the muscular trabeculae of the ventricular wall.
II. 28-5, 46, and 61 mm. embryos (Pl. I, fig. 5; Pl. II, figs. 6, 7). In the three embryos included in this group one finds the muscular element in the valve cusps becoming more and more predominant. The muscle gradually invades and replaces the cushion tissue. This invasion appears to proceed at the base of the cusp from ventricular muscle derived in the case of the lateral cusps from the lateral wall and in that of the central ones from the muscular interventricular septum. (In this connexion it is interesting to note that the A.-V. cushions themselves are largely invaded by this same muscle in these later embryos and it is possible, although I have not been able to confirm this with my present material, that they go through a muscular stage like the valves and like them later become collagenous. At all events this proliferation would account for the presence of muscle fibres in the pars membranacea septi in the adult heart (Walmsley).) At the same time, on the ventral borders of the central cusps the chordae musculares grow up on either side of the cushion tissue: this is perhaps seen better in the case of the right cusp of the mitral valve. The remains of the cushion tissue appear to migrate ventralwards. This migration is difficult to explain. It is clearly seen to occur if the microphotographs of the right lateral cusp of the 17-5 and the 46 mm. embryos are compared (Pl. I, fig. 2 and P1. II, fig. 6). In the former the upper end of the cushion is in contact with auricular muscle, while in the latter it is all ventral to this, and instead of being confined to the base its remains extend almost to the free border of the cusp. I think this migration must be due to the active proliferation of the cushion tissue, and this would account for its more cellular appearance.
Is there, at this stage, an increasing invagination at the A.-V. sulcus? Waterston found in the hearts of his 28-6 and 30 mm. embryos that the right lateral cusp showed an increasing infolding at the A.-V. sulcus so that at this stage the cusp consists, near its base, of a core of fibrous tissue continuous with the epicardium of the sulcus with auricular muscle on its medial and with ventricular muscle on its lateral surface. Tandler, whose description is not easy to follow, would seem to give a like account. I should first perhaps define as precisely as possible what I take to be the upper limit of the cusp. This is not always easy, as it varies in different sections with the presence or absence in any particular section of the most medial row of chordae musculares. I take, however, for this purpose a line at right angles to the long axis of the cusp at the most ventral point of its basal attachment to the ventricular wall. This corresponds in most cases to that given by Gross & Kugel (1931), although their method of determining it was a different one. If with this definition in mind the microphotograph from my 17-5 mm. embryo (Pl. I, fig. 2) is compared with that from my 28-5 mm. one (Pl. I, fig. 5), there does seem to be a commencing invagination, marked by its angulation, at the right A.-V. sulcus in the former embryo, which has so far increased in the later specimen that the junction of auricular and ventricular muscle is seen to be ventral to the line of the attachment of the cusp. The result of this is that epicardium does invade the uppermost quarter of the right lateral cusp, and this portion of it is thus precisely like Waterston’s description. Thereeis nothing like this in the case of the corresponding left lateral cusp, of the composition of which Waterston makes no mention at this stage. In my embryos this consists solely of ventricular muscle with a small amount of cushion tissue on its medial aspect in its upper portion. Here one may note that the left A.-V. sulcus is much less deep than the right one throughout my series. From my 11-2 mm. embryo up to my 61 mm. one, the proportion is roughly 63: 100 as average figures. The microphotograph of the 23 mm. embryo shows this contrast (Pl. I, fig. 4-) which must be correlated with the thicker wall of the right ventricle. In the later embryos of this same stage, 46 and 61 mm., the anatomy of the right lateral cusp is again different. Here the angulation of the A.-V. sulcus gradually disappears and the auricular muscle stops abruptly well above the level of the cusp attachment so that its junction with the ventricular muscle prevents any epicardium from entering the base of the valve, which consists essentially of ventricular muscle with some remains of cushion tissue on its medial aspect (Pl. II, figs. 6, 7). The condition seen in my 28-5 mm. embryo must be a transitory phase. That it is not exceptional is proved by Waterston finding it the same in his 28~6 and 30 mm. specimens. It is, too, this temporary condition of the right lateral cusp which must account for such a description of the development of these valves as Keith (1933) gives. “The auricular canal with an attenuated envelopment derived from the ventricle thus comes to hang within the ventricular chambers and forms the lateral cusps of the tricuspid and mitral valves. The morphological demarcation between auricle and ventricle lies at the free edge of the A.-V. valves.” Even for this stage such an account would seem to be a great exaggeration, and, while it may be substantially true for the pig’s heart at 10 mm., it finds no confirmation, in my opinion, in either the earlier or the later stages of the development of these valves in the heart of man.
III. Foetuses of 85, 105 and 210 mm., and at full time. As has been seen above, two changes occur simultaneously at this stage. The muscular flap becomes increasingly collagenous. As Benninghoff (1930) remarks, it is uncertain if one should speak of the degeneration and replacement of the muscle or of its transformation into fibrous tissue. Probably the former would be more in accord with accepted views. With this change the collagenous cusp in the case of the lateral ones becomes continuous with the epicardium filling the A.-V. sulcus. This confirms Bernays’ (1876) original statement: “Die Vereinigungsstelle zwischen Klappen und Pericardialfortsatz wird also sehr spat gebildet, sie entspricht dem ‘annulus fibrocartilagineus’ der Autoren.” The effect of this is that, while up to this time, i.e. up to about 4- months, auricular muscle has met that of the ventricle, this junction is now for the first time interrupted. At 6;» months, which corresponds to my 210 mm. stage, the cusps are all completely collagenous and there is no further change in their essential anatomy.
In view of the differences of opinion about the condition of the adult valves, it is interesting to note here the presence or absence of muscle in the later stages of their development now being considered. There is, in some sections of the lateral cusps at difl'erent ages, a small amount of Ventricular muscle just arching over their attached borders to reach the uppermost portion of the cusp on its ventricular aspect. This seems to occur quite capriciously. In my 85 mm. foetus it is present in both lateral cusps, in my 210 mm. specimen it is absent in both, but it does occur in the anterior cusp of the tricuspid valve, while at full term it is present in the right lateral cusp but is absent in the left one. With regard to the presence of auricular muscle in the cusps, again in this series of foetal hearts its appearance is quite irregular. I have found it just overlapping the base of the cusp, according to the definition of the latter I have enunciated already, in the following instances: at 85 mm. both the mitral cusps show that auricular muscle just overlaps their attached borders in certain sections; in my 105 mm. specimen auricular muscle occurs in both lateral cusps and is particularly well marked in the left one; in the heart of the 210 mm. embryo some sections of the right lateral cusp show its presence, while at full term in many sections of the same cusp it is still seen but is less extensive (Pl. II, figs. 9, 10). Lastly, in the heart of an 11 months old baby it is found in some sections of the left lateral cusp. In no other Valve cusp in this series is there any evidence of auricular muscle taking any part in its constitution, so that it is found only in some sections of seven out of a possible thirty—two cusps. I agree with Bernays that it is not found much before the fifth month, but I have found no evidence that it spreads more and more on to the surface of the valves in the later months of foetal life as he insisted. He correlated the presence of auricular muscle in the cusps with their increasing growth. The valves grow, he wrote, from the periphery and draw with them in their growth auricular muscle and the endocardium covering it; the extension of this muscular layer must go hand in hand with the growth of the cusp. The question of an increasing infolding at the A.-V. sulcus in these later foetal stages is a difficult one to answer, as the sulcus naturally deepens with the increasing size of the heart. Certainly in the case of the right lateral cusp in my 210 mm. specimen there is not only an increase in the depth of the sulcus but it is angulated, and this is more marked again at full term. I believe that this alteration could be simply explained as being wholly due to the growth of the ventricular wall in both a dorsal and a lateral direction. Further, I do not see that the presence of auricular muscle need be now compulsorily associated with increasing infolding or cusp growth (in contrast to the much earlier 28-5 mm. stage) as it is now divorced from that of the ventricle by the continuity of the collagenous matrix of the cusp with the A.-V. sulcus. This view is confirmed if the corresponding left lateral cusp is considered. Here there is no change either in the depth or in the direction of the A.-V. sulcus like that seen on the right side until full term is reached, and yet at 105 mm. there is a remarkable extension of auricular muscle on to it. I have already referred fully to the exceptional condition found in the right lateral cusp in my 28-5 mm. embryo. Waterston did not examine any later stage than that seen at 30 mm., so that it is hardly correct for him to say in his final discussion of this question that “the structure of the infolded cusp is a centrally wedge-shaped mass of connective tissue derived from the external surface of the A.-V. junction with a layer of muscle tissue continuous with the atrial muscle coat on the atrial surface and of ventricular muscle substance on the ventricular aspect”. Benninghofl‘ described the composition of a lateral cusp in a 12-4 cm. embryo as consisting of auricular muscle, epicardium from the primitive coronary sulcus and the cortical and trabecular muscle of the ventricle. The illustration of a reconstruction of this cusp, which he gives and which is presumably the left lateral one, differs very much from any of my sections, while his definition of the cusp includes the medial two-thirds of the A.-V. sulcus. (Probably 12-4 cm. should read 12-4 mm., but even so I cannot agree with his description.) From this he concludes, as Tandler (1913) does, that “die Entwicklung der Atrioventrikularklappen lasst es verstandlich erscheinen dass ausser den Sehnenfaden auch die Klappenmembran zu beiden Seiten Reste von Herzmuskulatur nahe der Klappenwurzel enthalten Kann”.
My own observations would not support this view. The appearance of auricular muscle in the valve cusps in the series that I have examined is so infrequent and so irregular that I cannot correlate it with any increasing infolding at the A.-V. sulcus. Nor would this method account for its occurrence in the central cusps, where no such invagination can take place. I would suggest rather that its presence in the adult heart valves should be looked upon as an irregular proliferation which is as capricious as it is in the foetal heart and for which there is no constant embryological foundation.
The fate of the endocardial cushions
In the earlier stages (Pl. I, figs. 1-3) only the more ventral portion of the cushions can be said to be actually forming part of the cusps. Later, as is seen in my 28-5, 46 and 61 mm. embryos (Pl. I, fig. 5; Pl. II, figs. 6, 7), the remains of the cushion tissue appear to have proliferated as elongated strands of cells beneath the endocardium which extend almost to the free border of the cusps in the case of the first two, but are much less well marked in the third member of this group. At 85 mm. (Pl. II, fig. 8) and at 130 mm. the greater part of the cusps are collagenous, but there is still in their ventral halves a looser, more cellular tissue beneath the endocardium. I think one should regard these areas as cushion remains. Bernays suggested that the nodules of Albini represent their last survival, but in my 210 mm. specimen the whole cusp appears to be homogeneously collagenous. There does seem thus to be evidence that some remains of the endocardial cushions do survive their invasion by muscle and do not disappear until the whole valve cusp is entirely collagenous.
Conclusions
1. The development of the atrio-ventricular valve cusps falls into three stages: (a) From 11-2 to 23 mm. they are composed of endocardial cushion tissue and muscle derived from the ventricular trabeculae. The right bulbar ridge provides the basis of cushion tissue for the anterior and right cusps of the tricuspid valve. (b) From 28-5 to 61 mm. they are becoming increasingly muscular, while the remains of cushion tissue can be still distinguished on their auricular surfaces. (c) From 85 to 210 mm. they gradually become entirely collagenous and there is no further change in their composition.
2. Except for the temporary appearance presented by the right cusp.of the tricuspid valve near its base at 28-5 mm., in no stage in this series are the cusps regularly composed of auricular and ventricular muscle separated by a wedge of connective tissue. The occasional presence of auricular muscle seems to be entirely capricious.
3. There is therefore no constant embryological foundation for the occurrence of auricular muscle in the valves of the adult heart.
I have to thank Prof. E. Fawcett and Prof. D. M. Blair for the loan of embryos from their collections which amplified our own series here, and Mr W. Chesterman of this department for the preparation of the microphotographs.
References
Bamunonorr, A. (1930). Hamdb. milcr. Armt. M erwchen, Bd. v1, Teil 1, S. 179. W. v. Miillendorfll
BERNAYS, A. C. (1876). Morplwl. Jb. Bd. 11, S. 478.
Fnazaa, J. E. (1916-17). J. Anat., Lcmd., vol. LI, p. 19.
a (1931). Manual of Embryology, p. 317.
GROSS, L. & KUGEL, M. A. (1931). Amer. J. Path. vol. VII, p. 445.
KEITH, Sir A. (1933). Human Embryology and Morphology, p. 370.
MALL, F. P. (1912). Amer. J. Anat. vol. xm, p. 249.
Onamzs, P. N. B. (l937—8). J. Anat, Lomi., vol. Lxxn, p. 247.
TANDLER, J. (1913). Anatomic dea Herzena. Bd. 111, Handb. Anat. M enachen. K. V. Bardeleben.
Wanmsmw, T. (1929). Quain’s Elements of Anatomy, vol. IV, pt. 3, p. 59.
Warrmsrou, D. (1918). Trans. R. Soc. Edinb. vol. 1.11, p. 257.
Explanation of Plates I And II
Plate I
Fig. 1. A section through the right A.-V. orifice in an 11-2 mm. embryo ( x 109). It shows the right lateral cushion, 13.0., separated from the A.-V. sulcus, A. V.S., by auricular muscle, A., joining that of the ventricle, V.
Fig. 2. A section of the right lateral cusp in a 17-5 mm. embryo ( x 109) to show the angulation of the right A.-V. sulcus, A. V.S., which is still separated from the right lateral cushion, R.0., by the junction of auricular, A., with ventricular muscle, V.
Fig. 3. A section through the left lateral cusp in the same embryo as fig. 2 ( x 109) to show in contrast the absence of any angulation of the sulcus, A.V.S., while‘ the left lateral cushion, L.0., maintains its original shape and relations to A., auricular muscle and to V., ventricular muscle.
Fig. 4. A section through the heart in a 23 mm. embryo ( x 60). It shows the cushions of the central cusps, M .0., capping the muscular interventricular septum, I .V.S., and those of the lateral cusps, 12.0’. and L0. Note the angulation of the right A.-V. sulcus, A.V.S. A. auricular, V. ventricular muscle.
Fig. 5. A section through the right lateral cusp in a 28-5 mm. embryo ( x 60) to show the effect of the invagination which has now occurred at the right A.-V. sulcus, A. V.S. Auricular muscle, A., joins that of the ventricle, V., ventral to the line of attachment of the cusp, so that its base is now composed of auricular muscle, A., and ventricular muscle, 17., with a core of connective tissue continuous with that of the sulcus. The cushion tissue, R.C., extends almost to the tip of the cusp.
Plate II
6. A section through the right lateral cusp in a 46 mm. embryo ( x 60). Here the cusp consists entirely of ventricular muscle, V., with the remains of the right cushion, R.C'. The junction of auricular muscle, A., with that of the ventricle, V., is now dorsal to the line of attachment of the cusp and the angulation of the sulcus, A.V.S., is much less marked.
7. A section through the right lateral cusp in a 61 mm. embryo ( x 60). The angulation of the sulcus, A.V.S., has now disappeared and, as before, auricular muscle, .11., joins that of the ventricle, V., dorsal to the base of the cusp.
8. A section through the right lateral cusp in an 85 mm. foetus ( x 60). The cusp is now collagenous and is continuous with the A.-V. sulcus, A.V.S., this continuity separating auricrular muscle, A., from that of the ventricle, V. Ventricular muscle just comes over on to the base of the cusp. R.C'. marks the apparent remains of cushion tissue.
9. A section through the right lateral cusp in a 210 mm. foetus ( x 60) to show that auricular muscle, A., does extend on to the base of the cusp in some sections at this stage, the rest of which is entirely collagenous.
10. A section through the right lateral cusp of a full time foetus ( x 60). Note the well-marked annulus fibrosus AR, and that both ventricular muscle, V., and auricular muscle, A., stray on to the base of the cusp to a very small extent.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - The Development of the Atrio-Ventricular Valves in Man. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_The_Development_of_the_Atrio-Ventricular_Valves_in_Man
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G