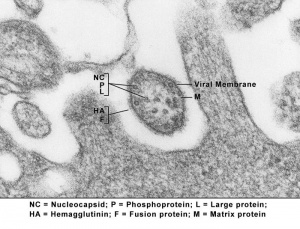
Abnormal Development - Viral Infection: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 39: | Line 39: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''The Association of H1N1 Pandemic Influenza with Congenital Anomaly Prevalence in Europe''' | * '''The Association of H1N1 Pandemic Influenza with Congenital Anomaly Prevalence in Europe'''{{#pmid:23381200|PMID23381200}} "In the context of the European Surveillance of Congenital Anomalies (EUROCAT) surveillance response to the 2009 influenza pandemic, we sought to establish whether there was a detectable increase of congenital anomaly prevalence among pregnancies exposed to influenza seasons in general, and whether any increase was greater during the 2009 pandemic than during other seasons. We performed an ecologic time series analysis based on 26,967 pregnancies with nonchromosomal congenital anomaly conceived from January 2007 to March 2011, reported by 15 EUROCAT registries. Analysis was performed for EUROCAT-defined anomaly subgroups, divided by whether there was a prior hypothesis of association with influenza. Influenza season exposure was based on World Health Organization data. Prevalence rate ratios were calculated comparing pregnancies exposed to influenza season during the congenital anomaly-specific critical period for embryo-fetal development to nonexposed pregnancies. Our data do not suggest an overall association of pandemic or seasonal influenza with congenital anomaly prevalence. One interpretation is that apparent influenza effects found in previous individual-based studies were confounded by or interacting with other risk factors. The associations of heart anomalies with pandemic influenza could be strain specific." | ||
* '''Influenza A/H1N1 MF59 adjuvanted vaccine in pregnant women and adverse perinatal outcomes: multicentre study''' | |||
* '''Update on childhood and adolescent immunizations: selected review of US recommendations and literature: part 2''' | * '''Influenza A/H1N1 MF59 adjuvanted vaccine in pregnant women and adverse perinatal outcomes: multicentre study'''{{#pmid:23381200|PMID23381200}} "This large study using primary data collection found that MF59 adjuvanted A/H1N1 influenza vaccine did not result in an increased risk of adverse perinatal events and suggested a lower risk among vaccinated women. These findings should contribute to inform stakeholders and decision makers on the prescription of vaccination against influenza A/H1N1 in pregnant women." | ||
* '''Fetal varicella - diagnosis, management, and outcome''' | |||
* '''ViralZone: a knowledge resource to understand virus diversity''' | * '''Update on childhood and adolescent immunizations: selected review of US recommendations and literature: part 2'''{{#pmid:21743328|PMID21743328}} "Vaccine coverage is relatively static or improving for the vaccines included in the 2010 annual harmonized immunization schedules. Providers should be reviewing patients' immunization records at each visit to take advantage of any opportunity to administer indicated, age-appropriate vaccines. There have been infectious disease outbreaks among highly immunized populations, although unvaccinated or undervaccinated individuals continue to play large roles in the spread of disease. Infants, many of whom are too young to be vaccinated, continue to bear a large disease burden, which underscores the importance of cocooning and, in some cases, vaccination of pregnant women. Influenza, measles, mumps, and rubella, varicella, hepatitis A, meningococcal conjugate, human papillomavirus, diphtheria and tetanus toxoids and acellular pertussis, and tetanus and diphtheria toxoids and acellular pertussis vaccines are reviewed in this second of two articles." | ||
* '''Fetal varicella - diagnosis, management, and outcome'''{{#pmid:22514124|PMID22514124}} "Fetal varicella syndrome (FVS) is due to transplacental infection by the Varicella zoster virus following maternal infection. The risks for the fetus and neonate depend on the timing. When varicella occurs around delivery, it often leads to disseminated neonatal varicella. When varicella occurs during pregnancy, transmission can occur, but is usually asymptomatic; some infants develop zoster postnatally and a few have FVS. Before 20 weeks' gestation, FVS can occur, with an incidence of about 1%. The lesions can affect the skin, limbs, central and autonomous nervous systems, eyes, cause calcifications, and growth retardation; mortality is high. Lesions typically follow one or several nerve territories, suggesting that damage results from in utero zoster following primary fetal infection." | |||
* '''ViralZone: a knowledge resource to understand virus diversity'''{{#pmid:20947564|PMID20947564}} "The molecular diversity of viruses complicates the interpretation of viral genomic and proteomic data. To make sense of viral gene functions, investigators must be familiar with the virus host range, replication cycle and virion structure. Our aim is to provide a comprehensive resource bridging together textbook knowledge with genomic and proteomic sequences. ViralZone web resource (www.expasy.org/viralzone/) provides fact sheets on all known virus families/genera with easy access to sequence data." | |||
* '''False-Positive Human Immunodeficiency Virus Enzyme Immunoassay Results in Pregnant Women''' <ref>False-Positive Human Immunodeficiency Virus Enzyme Immunoassay Results in Pregnant Women [http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3029371 PMC3029371]</ref> "False-positive HIV EIA results were rare and occurred less frequently among pregnant women than others." | * '''False-Positive Human Immunodeficiency Virus Enzyme Immunoassay Results in Pregnant Women''' <ref>False-Positive Human Immunodeficiency Virus Enzyme Immunoassay Results in Pregnant Women [http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3029371 PMC3029371]</ref> "False-positive HIV EIA results were rare and occurred less frequently among pregnant women than others." | ||
* '''Mother to child transmission of HIV among Zimbabwean women who seroconverted postnatally: prospective cohort study''' <ref>[http://www.bmj.com/content/341/bmj.c6580.abstract?etoc BMJ 2010; 341:c6580]</ref> "Breastfeeding associated transmission is high during primary maternal HIV infection and is mirrored by a high but transient peak in breast milk HIV load. Around two thirds of breastfeeding associated transmission by women who seroconvert postnatally may occur while the mother is still in the “window period” of an antibody based test, when she would test HIV negative using one of these tests." | * '''Mother to child transmission of HIV among Zimbabwean women who seroconverted postnatally: prospective cohort study''' <ref>[http://www.bmj.com/content/341/bmj.c6580.abstract?etoc BMJ 2010; 341:c6580]</ref> "Breastfeeding associated transmission is high during primary maternal HIV infection and is mirrored by a high but transient peak in breast milk HIV load. Around two thirds of breastfeeding associated transmission by women who seroconvert postnatally may occur while the mother is still in the “window period” of an antibody based test, when she would test HIV negative using one of these tests." | ||
*''' Transplacental transmission of Human Papillomavirus.''' | |||
* '''Infectious causes of stillbirth: a clinical perspective.''' | *''' Transplacental transmission of Human Papillomavirus.'''{{#pmid:18817577|PMID18817577}} "In conclusion the study suggests placental infection in 23.3% of the cases studied and transplacental transmission in 12.2%. It is suggested that in future HPV DNA be researched in the normal endometrium of women of reproductive age. The possible consequence of fetal exposure to HPV should be observed." | ||
* '''Infectious causes of stillbirth: a clinical perspective.'''{{#pmid:20661048|PMID20661048}}"In developed countries, up to 24% of stillbirths have been attributed to infection, although with increased availability of sophisticated diagnostics and rigorous screening, it appears likely that higher numbers may actually be associated with infection. In developed countries, ascending bacterial infection is usually the most common infectious cause of stillbirth, with a number of viral infections also an important factor. Screening, prevention, and treatment of maternal infections are important to reduce stillbirth risk." | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 76: | Line 84: | ||
(Image: CDC USA) | (Image: CDC USA) | ||
| Viral infection causes systemic infection and extensive brain damage and cell death by necrosis. Cytomegalovirus infection can also occur in the human placenta. | | Viral infection causes systemic infection and extensive brain damage and cell death by necrosis. Cytomegalovirus infection can also occur in the human placenta.{{#pmid:18637517|PMID18637517}} | ||
|} | |} | ||
| Line 87: | Line 95: | ||
Australia map - hendra virus outbreaks (1994-2008) | Australia map - hendra virus outbreaks (1994-2008) | ||
| Hendra virus is a paramyxoviridae (ssRNA negative-strand virus) that mainly infects large fruit bats (flying foxes) which can be passed on to horses. The infection has occasionally been passed onto people who have been in close contact with an infected horse. There is evidence of fetal and placental infection in flying fox | | Hendra virus is a paramyxoviridae (ssRNA negative-strand virus) that mainly infects large fruit bats (flying foxes) which can be passed on to horses. The infection has occasionally been passed onto people who have been in close contact with an infected horse. There is evidence of fetal and placental infection in flying fox{{#pmid:18198149|PMID18198149}} and animal models.{{#pmid:10684689|PMID10684689}} There is currently insufficient information to determine whether there are developmental effects in humans. | ||
10684689 | |||
Named after the Brisbane suburb where in 1994 the first outbreak was detected. Recently a vaccine for horses has been developed. | Named after the Brisbane suburb where in 1994 the first outbreak was detected. Recently a vaccine for horses has been developed. | ||
| Line 199: | Line 206: | ||
* The overall risk of transmission is approximately 2-6% with unknown maternal viral titers. | * The overall risk of transmission is approximately 2-6% with unknown maternal viral titers. | ||
* All pregnant women with HCV should have viral titers performed. | * All pregnant women with HCV should have viral titers performed. | ||
* The placenta appears to act as an immunological organ providing antiviral protection against hepatitis C viral transmission in the majority of cases. | * The placenta appears to act as an immunological organ providing antiviral protection against hepatitis C viral transmission in the majority of cases.{{#pmid:20814429|PMID20814429}} | ||
Data Sources | Data Sources{{#pmid:19007636|PMID19007636|}}{{#pmid:16480851|PMID16480851}}{{#pmid:16443611|PMID16443611}} | ||
| Line 216: | Line 223: | ||
Flu is the general term and abbreviation of influenzaviral infections and the possible teratogenic effect of influenza viruses (orthomyxoviruses, "flu") is unclear, with very little evidence directly linking the two events. A relatively recent study was unable to identify any placental transmission of influenza virus during the second and third trimester. Severe maternal infection though may lead to hyperthermia, which has been demonstrated to be teratogenic, particularly in early neural development. (More? [hyperthermia.htm Maternal Hyperthermia]) | Flu is the general term and abbreviation of influenzaviral infections and the possible teratogenic effect of influenza viruses (orthomyxoviruses, "flu") is unclear, with very little evidence directly linking the two events. A relatively recent study was unable to identify any placental transmission of influenza virus during the second and third trimester. Severe maternal infection though may lead to hyperthermia, which has been demonstrated to be teratogenic, particularly in early neural development. (More? [hyperthermia.htm Maternal Hyperthermia]) | ||
Influenza virus infection in the second and third trimesters of pregnancy: a clinical and seroepidemiological study. | Influenza virus infection in the second and third trimesters of pregnancy: a clinical and seroepidemiological study.{{#pmid:11028582|PMID11028582}} "We found no evidence for transplacental transmission of influenza virus or auto-antibody production in pregnancies complicated by influenza infections. There was an increase in the complications of pregnancy in our influenza cohort." | ||
Postnatally, the suggested initial limited neonatal immune system makes postnatal infection dangerous. | Postnatally, the suggested initial limited neonatal immune system makes postnatal infection dangerous. | ||
See also: Transmission of influenza A in human beings. | See also: Transmission of influenza A in human beings.{{#pmid:17376383|PMID17376383}} | ||
:'''Links:''' [http://www.nlm.nih.gov/medlineplus/flu.html Medline Plus - Flu] | Search Medical Microbiology [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Books&cmd=search&doptcmdl=DocSum&term=orthomyxovirus+AND+mmed%5Bbook%5D "orthomyxovirus"] | :'''Links:''' [http://www.nlm.nih.gov/medlineplus/flu.html Medline Plus - Flu] | Search Medical Microbiology [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Books&cmd=search&doptcmdl=DocSum&term=orthomyxovirus+AND+mmed%5Bbook%5D "orthomyxovirus"] | ||
| Line 228: | Line 235: | ||
Measles (rubeola) is a paramyxovirus appearing mainly as a respiratory viral infection, clinically different from Rubella. A single-stranded RNA virus which is highly contagious. Before measles vaccination (USA 1963) more than 90% of children had an infection before puberty and in developing countries it is still a common and often fatal childhood disease. Childhood immunisation and immunity persists in only about 80% of adults. | Measles (rubeola) is a paramyxovirus appearing mainly as a respiratory viral infection, clinically different from Rubella. A single-stranded RNA virus which is highly contagious. Before measles vaccination (USA 1963) more than 90% of children had an infection before puberty and in developing countries it is still a common and often fatal childhood disease. Childhood immunisation and immunity persists in only about 80% of adults. | ||
Pregnancy effects of measles results in a higher risk of premature labor, spontaneous abortion, low-birth-weight, and possibly rare cases of birth defects with no definable pattern of malformation. | Pregnancy effects of measles results in a higher risk of premature labor, spontaneous abortion, low-birth-weight, and possibly rare cases of birth defects with no definable pattern of malformation.{{#pmid:12850161|PMID12850161}} | ||
In terms of the immune response to post-natal infection, a monkey model has shown that the virus is initially regulated by T cells, but require an antibody response to eliminate the viral RNA. | In terms of the immune response to post-natal infection, a monkey model has shown that the virus is initially regulated by T cells, but require an antibody response to eliminate the viral RNA.{{#pmid:22872860|PMID22872860}} | ||
| Line 266: | Line 273: | ||
'''H1''' refers to one of the hemagglutinin subtypes (H1, H2 or H3) and '''N1''' refers to one of the two neuraminidase subtypes (N1 or N2). Over the last century there have been at least three different human viral pandemic circulating strains H1N1 (1918, 1977), H2N2 (1957) and H3N2 (1968). | '''H1''' refers to one of the hemagglutinin subtypes (H1, H2 or H3) and '''N1''' refers to one of the two neuraminidase subtypes (N1 or N2). Over the last century there have been at least three different human viral pandemic circulating strains H1N1 (1918, 1977), H2N2 (1957) and H3N2 (1968). | ||
A study of the perinatal outcomes after maternal 2009/H1N1 infection: national cohort study. | A study of the perinatal outcomes after maternal 2009/H1N1 infection: national cohort study.{{#pmid:221672992|PMID221672992}} | ||
:"Perinatal mortality was higher in infants born to infected women (10 deaths among 256 infants; rate 39 (95% confidence interval 19 to 71) per 1000 total births) than in infants of uninfected women (9 deaths among 1233 infants; rate 7 (3 to 13) per 1000 total births) (P < 0.001). This was principally explained by an increase in the rate of stillbirth (27 per 1000 total births v 6 per 1000 total births; P = 0.001). Infants of infected women were also more likely to be born prematurely than were infants of comparison women (adjusted odds ratio 4.0, 95% confidence interval 2.7 to 5.9). Infected women who delivered preterm were more likely to be infected in their third trimester (P = 0.046), to have been admitted to an intensive care unit (P < 0.001), and to have a secondary pneumonia (P = 0.001) than were those who delivered at term." | :"Perinatal mortality was higher in infants born to infected women (10 deaths among 256 infants; rate 39 (95% confidence interval 19 to 71) per 1000 total births) than in infants of uninfected women (9 deaths among 1233 infants; rate 7 (3 to 13) per 1000 total births) (P < 0.001). This was principally explained by an increase in the rate of stillbirth (27 per 1000 total births v 6 per 1000 total births; P = 0.001). Infants of infected women were also more likely to be born prematurely than were infants of comparison women (adjusted odds ratio 4.0, 95% confidence interval 2.7 to 5.9). Infected women who delivered preterm were more likely to be infected in their third trimester (P = 0.046), to have been admitted to an intensive care unit (P < 0.001), and to have a secondary pneumonia (P = 0.001) than were those who delivered at term." | ||
| Line 353: | Line 360: | ||
{| class="prettytable" | {| class="prettytable" | ||
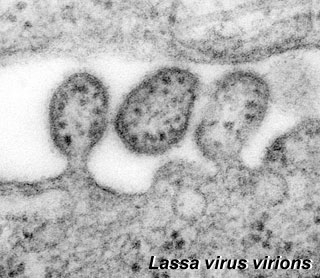
| <center>[[File:Lassa virus.jpg]]Lassa virions (Image: CDC USA)</center> | | <center>[[File:Lassa virus.jpg]]Lassa virions (Image: CDC USA)</center> | ||
| Lassa virus (LASV) of the arenaviridae family, a single-stranded RNA virus (see review | | Lassa virus (LASV) of the arenaviridae family, a single-stranded RNA virus (see review{{#pmid:23202452|PMID23202452}}). First isolated in 1969 from a missionary nurse who worked in a clinic the small town of Lassa, Nigeria. The virus is the causative agent of a hemorrhagic fever and can be transmitted between species (zoonotic). Death rates are high for women in the third trimester of pregnancy. Fetal death (95%) occurs in uterus of infected pregnant mothers. | ||
| Line 368: | Line 375: | ||
==Ljungan virus== | ==Ljungan virus== | ||
Zoonotic Ljungan virus associated with central nervous system malformations in terminated pregnancy. | Zoonotic Ljungan virus associated with central nervous system malformations in terminated pregnancy.{{#pmid:19180651|PMID19180651}} | ||
:"LV was diagnosed in 9 of 10 cases with hydrocephalus and in 1 of 18 trisomy 21 controls by immunohistochemistry. Five of nine cases with anencephaly had a positive PCR result, whereas none of the 12 trisomy 21 available for PCR testing had a positive result. The 47 newborn mice exposed to LV all developed encephalitis, with eight having hydrocephalus. None of the 52 control animals had encephalitis or hydrocephalus." | :"LV was diagnosed in 9 of 10 cases with hydrocephalus and in 1 of 18 trisomy 21 controls by immunohistochemistry. Five of nine cases with anencephaly had a positive PCR result, whereas none of the 12 trisomy 21 available for PCR testing had a positive result. The 47 newborn mice exposed to LV all developed encephalitis, with eight having hydrocephalus. None of the 52 control animals had encephalitis or hydrocephalus." | ||
| Line 382: | Line 389: | ||
Modified adenovirus is currently used as a research tool to introduce genes into cells in vitro and in animal systems. | Modified adenovirus is currently used as a research tool to introduce genes into cells in vitro and in animal systems.{{#pmid:12202428|PMID12202428}} | ||
| Line 419: | Line 426: | ||
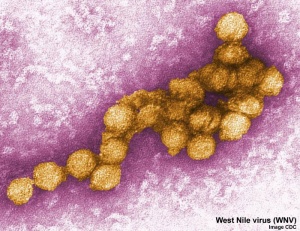
"Since 1999, more than 30,000 people in the United States have been reported as getting sick with West Nile virus. Infected mosquitoes spread West Nile virus (WNV) that can cause serious, life altering disease."<ref name="CDCSep2012">Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases (NCEZID), Division of Vector-Borne Diseases (DVBD)[http://www.cdc.gov/ncidod/dvbid/westnile/index.htm West Nile information page] viewed 11 September, 2012.</ref> | "Since 1999, more than 30,000 people in the United States have been reported as getting sick with West Nile virus. Infected mosquitoes spread West Nile virus (WNV) that can cause serious, life altering disease."<ref name="CDCSep2012">Centers for Disease Control and Prevention, National Center for Emerging and Zoonotic Infectious Diseases (NCEZID), Division of Vector-Borne Diseases (DVBD)[http://www.cdc.gov/ncidod/dvbid/westnile/index.htm West Nile information page] viewed 11 September, 2012.</ref> | ||
A 2006 study of 72 infected infants (2003 and 2004) "almost all seemed normal, and none had conclusive laboratory evidence of congenital WNV infection. ...Three infants had WNV infection that could have been congenitally acquired, seven infants had major malformations, but only 3 of these had defects that could have been caused by maternal WNV infection based on the timing of the infections and the sensitive developmental period for the specific malformations, and none had any conclusive evidence of WNV etiology." | A 2006 study of 72 infected infants (2003 and 2004) "almost all seemed normal, and none had conclusive laboratory evidence of congenital WNV infection. ...Three infants had WNV infection that could have been congenitally acquired, seven infants had major malformations, but only 3 of these had defects that could have been caused by maternal WNV infection based on the timing of the infections and the sensitive developmental period for the specific malformations, and none had any conclusive evidence of WNV etiology."{{#pmid:16510632|PMID16510632}} | ||
| Line 428: | Line 435: | ||
{| | {| | ||
| [[File:Zika virus TEM02.jpg|300px]] | | [[File:Zika virus TEM02.jpg|300px]] | ||
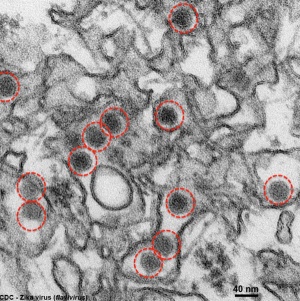
| Zika virus (ZIKV) is a mosquito-borne ''flavivirus'' related to dengue virus first isolated from a rhesus monkey in Zika forest, Uganda (1947). Transmitted by mosquitoes (''Aedes aegypti'') it was then identified in humans in Nigeria in 1954 and subsequently in South America, Asia and Pacific regions. More recently in Australia from returning travellers. | | Zika virus (ZIKV) is a mosquito-borne ''flavivirus'' related to dengue virus first isolated from a rhesus monkey in Zika forest, Uganda (1947). Transmitted by mosquitoes (''Aedes aegypti'') it was then identified in humans in Nigeria in 1954 and subsequently in South America, Asia and Pacific regions. More recently in Australia from returning travellers.{{#pmid:24944843|PMID24944843}} | ||
Virus particles are 40 nm in diameter, with an outer envelope, and an inner dense core. ZIKV is an RNA virus containing 10,794 nucleotides encoding 3,419 amino acids. | Virus particles are 40 nm in diameter, with an outer envelope, and an inner dense core. ZIKV is an RNA virus containing 10,794 nucleotides encoding 3,419 amino acids.{{#pmid:16223950|PMID16223950}} | ||
Mosquito bites lead to an initial infection of skin cells. | Mosquito bites lead to an initial infection of skin cells.{{#pmid:26085147|PMID26085147}} The virus may also be transmitted transplacentally or during delivery.{{#pmid:24721538|PMID24721538}} Some limited evidence, from Brazil, for association with microcephaly. | ||
|} | |} | ||
{| | {| | ||
| Line 478: | Line 485: | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:21480828}} | |||
{{#pmid:16480851}} | |||
{{#pmid:2189514}} | |||
{{#pmid:227524}} | |||
{{#pmid:8384522}} | |||
===Articles=== | ===Articles=== | ||
{{#pmid:19317971}} | |||
{{#pmid:4317516}} | |||
===Search Pubmed=== | ===Search Pubmed=== | ||
Revision as of 11:05, 24 February 2018
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Educational Use Only - Embryology is an educational resource for learning concepts in embryological development, no clinical information is provided and content should not be used for any other purpose. |
Introduction
Below is a list of some known maternal, then fetal and postnatal infections that impact upon development. Only a very brief overview is given for each virus for more specific details see the listed internal and external links.
Viruses cannot reproduce by themselves and therefore infect cells to use the cell machinery to produce more virus. Different viruses have genetic material as single- or double-stranded RNA or DNA. The infectious virus particle is called a "virion" and is the genetic material packed in a protein shell. Viruses come in many genetic sizes, as little as 4 proteins up to 200 proteins.
Viral infections can range in their effects from no discernable impact through to severe impacts on development. Furthermore this may occur either directly by the infection or indirectly by the associated maternal fever and other side-effects of the infection. (More? Abnormal Development - Maternal Hyperthermia)
Rubella virus "German Measles" infection during pregnancy is one of the most serious causing congenital rubella syndrome with serious malformations of the developing fetus.
Viral infections have also recently been in the news with the 2009 novel influenza A (H1N1) "Swine Flu" pandemic.
Some early postnatal viral infections can also impact upon development and have been the target for worldwide immunization and eradication.
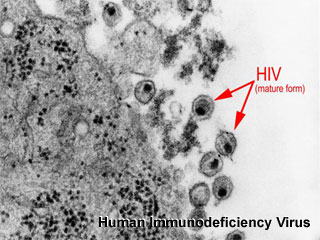
Human Immunodeficiency Virus (HIV) leads to AIDS and according to United Nations 2005 data about 38.6 million people had HIV. Of the 17.3 million women infected with HIV, 3.28 million gave birth each year (mostly in sub-Saharan Africa), leading to 700,000 new infections of HIV in children each year.
Note also that many cancers can be caused by viruses (papilloma viruses, hepatitis B and C viruses, Epstein-Barr virus and human T-cell lymphotropic virus). Virus-induced cancers account for about 20% of worldwide cancer incidence.
In a few developing countries, and mainly in Africa, at least 3 viruses induce a hemorrhagic fever: Ebola hemorrhagic fever, Marburg virus disease, and Lassa fever.
| Environmental Links: Introduction | low folic acid | iodine deficiency | Nutrition | Drugs | Australian Drug Categories | USA Drug Categories | thalidomide | herbal drugs | Illegal Drugs | smoking | Fetal Alcohol Syndrome | TORCH | viral infection | bacterial infection | fungal infection | zoonotic infection | toxoplasmosis | Malaria | maternal diabetes | maternal hypertension | maternal hyperthermia | Maternal Inflammation | Maternal Obesity | hypoxia | biological toxins | chemicals | heavy metals | air pollution | radiation | Prenatal Diagnosis | Neonatal Diagnosis | International Classification of Diseases | Fetal Origins Hypothesis |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Abnormal Development Viral Infection <pubmed limit=5>Abnormal Development Viral Infection</pubmed> |
Rubella Virus
Rubella virus (Latin, rubella = little red), also known as "German Measles" (due to early citation in German medical literature).
Infection during pregnancy can cause congenital rubella syndrome (CRS) with serious malformations of the developing fetus. The type and degree of abnormality relates to the time of maternal infection.
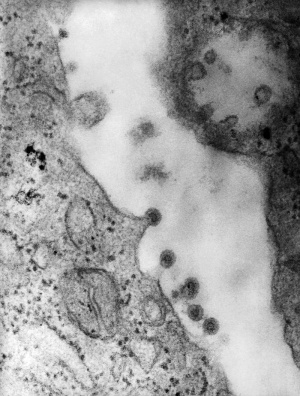
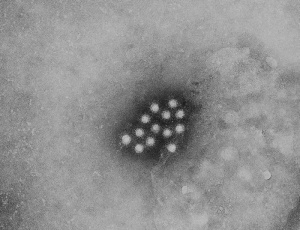
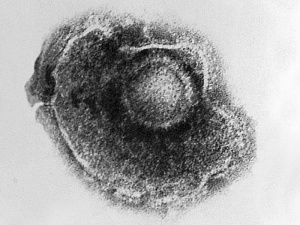
Cytomegalovirus
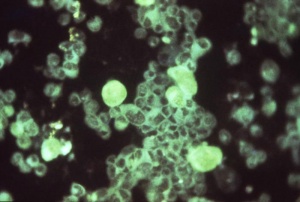
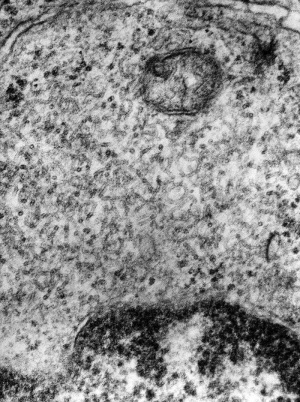
Immunoflourescent detection of Cytomegalovirus (Image: CDC USA) |
Viral infection causes systemic infection and extensive brain damage and cell death by necrosis. Cytomegalovirus infection can also occur in the human placenta.[9] |
- Links: Cytomegalovirus | Medical Microbiology- Cytomegalovirus | Search Medical Microbiology "Cytomegalovirus"
Hendra Virus
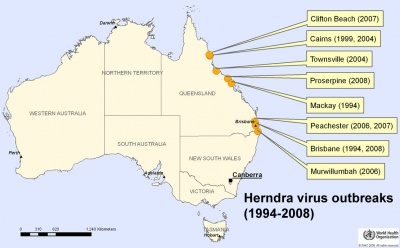
Australia map - hendra virus outbreaks (1994-2008) |
Hendra virus is a paramyxoviridae (ssRNA negative-strand virus) that mainly infects large fruit bats (flying foxes) which can be passed on to horses. The infection has occasionally been passed onto people who have been in close contact with an infected horse. There is evidence of fetal and placental infection in flying fox[10] and animal models.[11] There is currently insufficient information to determine whether there are developmental effects in humans.
Named after the Brisbane suburb where in 1994 the first outbreak was detected. Recently a vaccine for horses has been developed. |
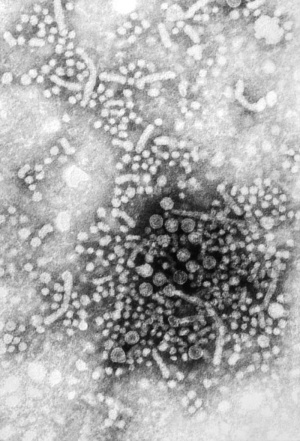
Herpes Simplex Virus
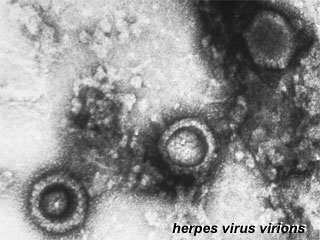
Herpes Virus, transmission electron micrograph (Image: CDC USA) |
Viral herpes simplex infection can cause a systemic infection and extensive brain damage and cell death by necrosis.
There is also some evidence which suggests that fetal exposure to herpesvirus infection is associated with pregnancy-induced hypertensive disorders and preterm birth. |
NCBI Bookshelf (external link) Search Medical Microbiology "Herpes Simplex Virus"
Search PubMed: term = Herpes Simplex Virus teratology | embryo infection | fetal infection | neonatal infection
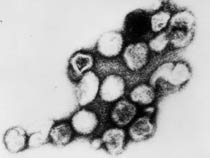
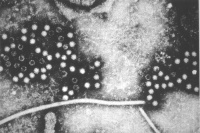
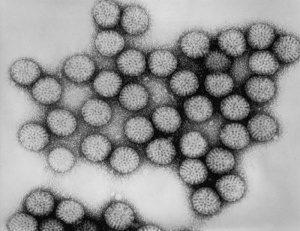
Human Papilloma Virus
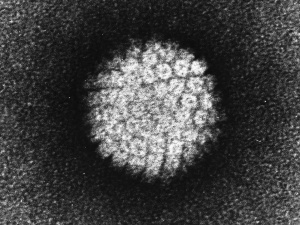
Human papilloma virus |
(HPV) A group of more that 118 identified viral strains about 40 infect the genital tract and 12 are known to be cancer-causing. Just eight HPV types 16, 18, 45, 33, 31, 52, 58, and 35 in descending order of frequency are responsible for more than 90 percent of cervical cancer cases. (Lancet 2010)
Infection can be detected by pap smear and at least 2 available vaccines, Cervarix (GSK) and Gardasil (Merck). Gardasil protects against HPV types 16 and 18, which cause 70% of cervical cancers in women and 90% of all HPV-related cancers in men. Also against two low-risk HPV types (types 6 and 11), which cause 90% of genital warts. through cross-protection also partially against HPV types 31 and 45.
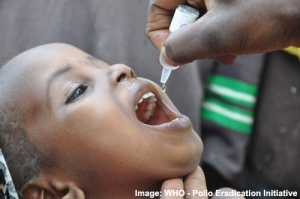
Polio VirusPoliomyelitis (polio) is a highly infectious viral disease, affecting only humans of any age, but mainly children under the age of 3 (> 50% cases) causing paralysis and death. There are three types (type 1, type 2, and type 3) of wild poliovirus which can invade the nervous system and can cause total paralysis in several hours. Polio cases have decreased by more than 99% since 1988.
A bivalent oral polio vaccine (BOPV) is presently being used in countries with high existing polio rates, India and Nigeria. A recent WHO release has shown the largest ever year-to-year drop in polio cases following the use of BOPV:
Human Immunodeficiency Virus
Hepatitis VirusHepatitis (inflammation of the liver) is caused in humans by one of 7 viruses (A, B, C, D, E) with the 2 additional F has not been confirmed as a distinct genotype; and G is a newly described flavivirus. "All of these viruses can cause an acute disease with symptoms lasting several weeks including yellowing of the skin and eyes (jaundice); dark urine; extreme fatigue; nausea; vomiting and abdominal pain. It can take several months to a year to feel fit again." (CDC text). Virus particles measure 42nm in overall diameter and contain a 27nm diameter DNA-based core. Hepatitis Transmission Risk to the FetusHepatitis A
Hepatitis B
Hepatitis C
Hepatitis E virions (CDC) Search PubMed: term = Hepatitis Virus teratology | embryo infection | fetal infection | neonatal infection Influenza VirusFlu is the general term and abbreviation of influenzaviral infections and the possible teratogenic effect of influenza viruses (orthomyxoviruses, "flu") is unclear, with very little evidence directly linking the two events. A relatively recent study was unable to identify any placental transmission of influenza virus during the second and third trimester. Severe maternal infection though may lead to hyperthermia, which has been demonstrated to be teratogenic, particularly in early neural development. (More? [hyperthermia.htm Maternal Hyperthermia]) Influenza virus infection in the second and third trimesters of pregnancy: a clinical and seroepidemiological study.[16] "We found no evidence for transplacental transmission of influenza virus or auto-antibody production in pregnancies complicated by influenza infections. There was an increase in the complications of pregnancy in our influenza cohort." Postnatally, the suggested initial limited neonatal immune system makes postnatal infection dangerous. See also: Transmission of influenza A in human beings.[17]
MeaslesMeasles (rubeola) is a paramyxovirus appearing mainly as a respiratory viral infection, clinically different from Rubella. A single-stranded RNA virus which is highly contagious. Before measles vaccination (USA 1963) more than 90% of children had an infection before puberty and in developing countries it is still a common and often fatal childhood disease. Childhood immunisation and immunity persists in only about 80% of adults. Pregnancy effects of measles results in a higher risk of premature labor, spontaneous abortion, low-birth-weight, and possibly rare cases of birth defects with no definable pattern of malformation.[18] In terms of the immune response to post-natal infection, a monkey model has shown that the virus is initially regulated by T cells, but require an antibody response to eliminate the viral RNA.[19]
RotavirusA type of virus that is a common cause of diarrhoea and vomiting (viral gastroenteritis) in infants and young children. The live attenuated rotavirus vaccine is contraindicated in pregnancy, but can be safely administered to household contacts of pregnant women. There is only a very small risk of transmission of the rotavirus vaccine virus to a susceptible pregnant woman and there is no evidence of risk to the fetus if pregnant women are in contact with recently vaccinated individuals.
(data based on: The Australian Immunisation Handbook 9th Edition 2008 2.3.2 Vaccination of women planning pregnancy, pregnant or breastfeeding women, and preterm infants - updated July 2009 )
Varicella Zoster Virus (chickenpox)Fetal varicella syndrome (FVS) is caused by transplacental infection by the varicella zoster (chickenpox) virus following maternal infection. Fetal and neonatal risks are dependent on infection timing.
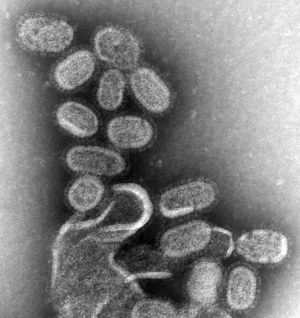
Swine Influenza VirusIn 2009 a global pandemic of the novel influenza A (H1N1) or Swine Flu has occurred. At this stage the possible teratogenic effect of this influenza virus should be considered the same as those described for the influenza virus above. The term "Pandemic" refers only to the spread of the virus, not the severity of the infection or potential developmental outcomes. This virus has also been spreading in an unseasonable pattern, that is in the northern hemisphere summer, when typically viral infections increase in the winter period. H1 refers to one of the hemagglutinin subtypes (H1, H2 or H3) and N1 refers to one of the two neuraminidase subtypes (N1 or N2). Over the last century there have been at least three different human viral pandemic circulating strains H1N1 (1918, 1977), H2N2 (1957) and H3N2 (1968). A study of the perinatal outcomes after maternal 2009/H1N1 infection: national cohort study.PubmedParser error: The PubmedParser extension received invalid XML data. ()
(Table data source: BMJ 2009;339:b2840) Confirmed infections per million population (July 2009)
Australia - H1N1 Influenza 09 (Human Swin Flu) Facts
Australian State Information
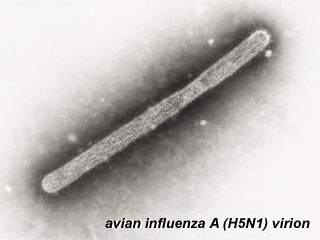
(Facts from: Australian Commonwealth Department of Health and Ageing H1N1 Influenza 09 (Human Swine influenza) – Summary Sheet for General Practitioners Current as of 24 May 2009) UK Information 20 July 2009: Chief Medical Officer's advice to pregnant women"Some pregnant women who catch the H1N1 (swine) ‘flu virus will develop complications of the infection (including pneumonia) that could put their own and their baby’s health at risk. The risks are greatest in the second and third trimesters of pregnancy. It is too early in the pandemic of influenza to be able to quantify these risks for the individual but most pregnant women who catch the disease are likely to make an uncomplicated recovery...." Links: WHO - H1N1 Pandemic | USA CDC - H1N1 Flu | Medline Plus - H1N1 Flu (Swine Flu) | GenBank sequences from pandemic (H1N1) 2009 viruses Avian Influenza Virus
Lassa Virus
Ebola VirusEbola virus disease (EVD, Ebola hemorrhagic fever, or EHF) is caused by three of the four species of “Ebola-like viruses” that appear to be maintained in a natural reservoir in the rain forests of Africa.
Ljungan virusZoonotic Ljungan virus associated with central nervous system malformations in terminated pregnancy.[21]
Adenovirus1953 - Human adenovirus was first isolated. This virus family is the causative agent of membrane infections in: respiratory tract, eyes, gastrointestinal tract, and urinary tract.
Modified adenovirus is currently used as a research tool to introduce genes into cells in vitro and in animal systems.[22]
Coxsackie
EchovirusesEchoviruses are RNA viruses found in the gastrointestinal tract that belongs to the genus Enterovirus of the Picornaviridae family. These viruses do not seem to damage the fetus. Mumps
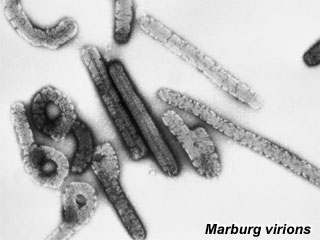
Marburg Virus
West Nile Virus
Zika Virus
Prevention
References
Textbooks
ReviewsFox KA & Theiler R. (2011). Vaccination in pregnancy. Curr Pharm Biotechnol , 12, 789-96. PMID: 21480828 Ornoy A & Tenenbaum A. (2006). Pregnancy outcome following infections by coxsackie, echo, measles, mumps, hepatitis, polio and encephalitis viruses. Reprod. Toxicol. , 21, 446-57. PMID: 16480851 DOI. Best JM & Banatvala JE. (1990). Congenital virus infections. BMJ , 300, 1151-2. PMID: 2189514 Waterson AP. (1979). Virus infections (other than rubella) during pregnancy. Br Med J , 2, 564-6. PMID: 227524 Oberst RD. (1993). Viruses as teratogens. Vet. Clin. North Am. Food Anim. Pract. , 9, 23-31. PMID: 8384522 ArticlesPandolfi E, Chiaradia G, Moncada M, Rava L & Tozzi AE. (2009). Prevention of congenital rubella and congenital varicella in Europe. Euro Surveill. , 14, 16-20. PMID: 19317971 Horstmann DM. (1969). Viral infections in pregnancy. Yale J Biol Med , 42, 99-112. PMID: 4317516 Search PubmedSearch Pubmed: embryo viral Infection | fetal viral Infection External LinksExternal Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
Cite this page: Hill, M.A. (2024, April 26) Embryology Abnormal Development - Viral Infection. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Viral_Infection
|