Abnormal Development - Hepatitis Virus
Introduction
Hepatitis (inflammation of the liver) is caused in humans by one of 7 viruses (A, B, C, D, E) with the 2 additional F has not been confirmed as a distinct genotype; and G is a newly described flavivirus.
"All of these viruses can cause an acute disease with symptoms lasting several weeks including yellowing of the skin and eyes (jaundice); dark urine; extreme fatigue; nausea; vomiting and abdominal pain. It can take several months to a year to feel fit again." (CDC text).
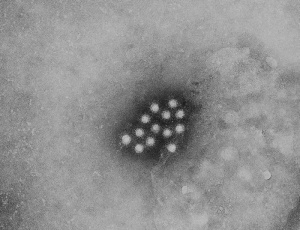
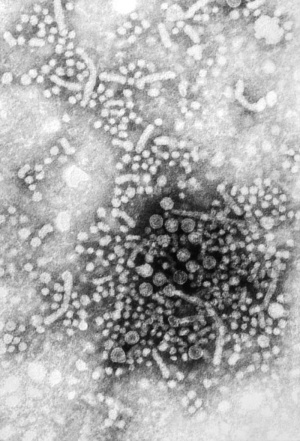
Virus particles measure 42nm in overall diameter and contain a 27nm diameter DNA-based core.
| Environmental Links: Introduction | low folic acid | iodine deficiency | Nutrition | Drugs | Australian Drug Categories | USA Drug Categories | thalidomide | herbal drugs | Illegal Drugs | smoking | Fetal Alcohol Syndrome | TORCH | viral infection | bacterial infection | fungal infection | zoonotic infection | toxoplasmosis | Malaria | maternal diabetes | maternal hypertension | maternal hyperthermia | Maternal Inflammation | Maternal Obesity | hypoxia | biological toxins | chemicals | heavy metals | air pollution | radiation | Prenatal Diagnosis | Neonatal Diagnosis | International Classification of Diseases | Fetal Origins Hypothesis |
Some Recent Findings
|
Hepatitis Transmission Risk to the Fetus
Hepatitis A
- Fetal transmission of virus occurs with extreme rarity.
Hepatitis B
- Can occur as a consequence of intrapartum exposure, transplacental transmission, and breastfeeding.
- 20% - 30% of HBsAg-positive/HbeAg-negative women will transmit virus to their infants.
- 90% of HBsAg- and HBeAg-positive women will transmit virus to their infants.
- Immunoprophylaxis at birth with both HBIG and Hepatitis B vaccine within 12 hours of birth decreases the risk of transmission.
- Passive (HBIG) and active immunization is 85-95% effective in preventing neonatal HBV infection.
Hepatitis C
- The overall risk of transmission is approximately 2-6% with unknown maternal viral titers.
- All pregnant women with HCV should have viral titers performed.
- The placenta appears to act as an immunological organ providing antiviral protection against hepatitis C viral transmission in the majority of cases.[2]
Hepatitis E virions (CDC)
References
Textbooks
- Medical Microbiology. 4th edition. Baron S, editor. Galveston (TX): University of Texas Medical Branch at Galveston; 1996.
- Molecular Biology of the Cell. 4th edition. Alberts B, Johnson A, Lewis J, et al. New York: Garland Science; 2002.
Reviews
Articles
Search Pubmed
Search PubMed: term = Hepatitis Virus teratology | embryo infection | fetal infection | neonatal infection
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- International Committee on Taxonomy of Viruses
- Australian Government The Australian Immunisation Handbook 9th Edition 2008
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 16) Embryology Abnormal Development - Hepatitis Virus. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Hepatitis_Virus
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G