Gastrointestinal Tract - Liver Development: Difference between revisions
mNo edit summary |
|||
| (135 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
{{Header}} | |||
==Introduction== | |||
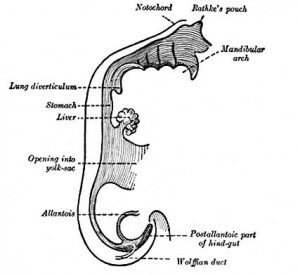
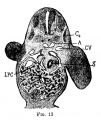
[[File:Gray0982a.jpg|thumb|Embryonic hepatic bud formation.]] | |||
This section of notes gives an overview of how the {{liver}} develops. Initially, the transverse septum (septum transversum) arises at an embryonic junctional site. The junctional region externally is where the ectoderm of the amnion meets the endoderm of the {{yolk sac}}. The junctional region internally is where the {{foregut}} meets the {{midgut}}. The mesenchymal structure of the transverse septum provides a support within which both blood vessels and the liver begin to form. Arises at embryonic junction (septum transversum): externally is where ectoderm of amnion meets endoderm of yolk sac and internally is where the foregut meets the midgut. Mesenchymal structure of transverse septum provides a support within which both blood vessels and liver begin to form in the underlying splanchnic {{mesoderm}}. | |||
In the early embryo, the liver and heart grow rapidly forming obvious external swellings on the ventral embryo surface. The liver's initial embryonic function is mainly {{cardiovascular}}. Firstly, as a vascular connection between the developing placental vessels to the heart. Secondly, as a haemopoietic tissue where blood stem cells reside before bone marrow development. | |||
The fetal liver also has an endocrine role by 16-hydroxylation, that results in estriol being the major estrogen type produced in late human pregnancy. | |||
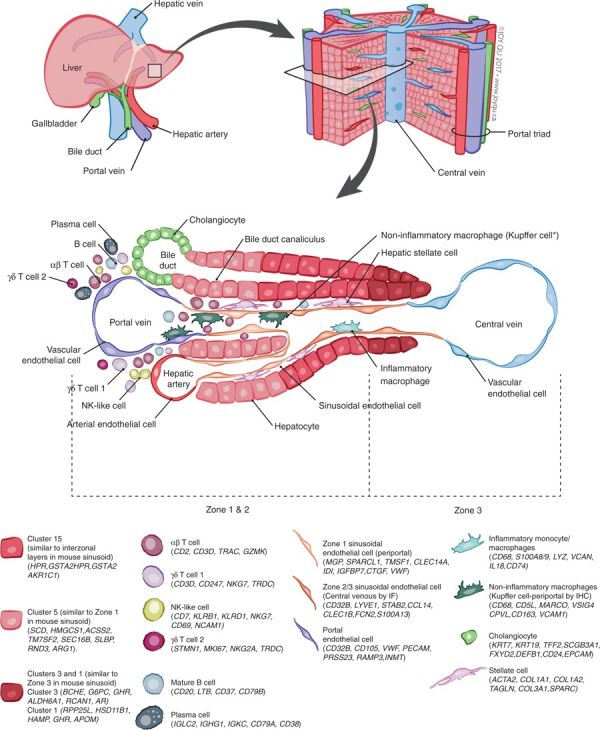
A recent molecular study has shown that within the [[Gastrointestinal Tract - Liver Development#Adult Liver Cells|adult liver]] at least 20 discrete cell populations exist these include: hepatocytes, endothelial cells, cholangiocytes, hepatic stellate cells, B cells, conventional and non-conventional T cells, NK-like cells, and distinct intrahepatic monocyte/macrophage populations.{{#pmid:30348985|PMID30348985}} | |||
{ | See also {{liver histology}} showing both developmental and adult histology. | ||
{{Liver Vignette}} | |||
<br> | |||
{{Gastrointestinal Tract Links}} | |||
|- | ::[[Embryology_History|'''Historic Embryology''']]: [[Paper - A Study of the Structural Unit of the Liver|1906 Liver Structural Unit]] | [[Book_-_Manual_of_Human_Embryology_17-7|1912 Liver]] | [[Paper - Functions of the liver in the embryo|1920 Embryonic Function]] | [[Book_-_Text-Book_of_Embryology_12#The_Development_of_the_Liver|1921 Liver]] | [[Paper - The embryogenesis of human bile capillaries and ducts|1926 Bile Capillaries and Ducts]] | [[Book - Liver development|2008 Liver]] | ||
| | |||
| | |||
== Some Recent Findings == | |||
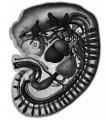
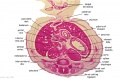
[[File:Stage 22 image 084.jpg|thumb|Human Liver (week 8, GA week 10)]] | |||
{| | |||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''Decoding human fetal liver {{haematopoiesis}}'''{{#pmid:31597962|PMID31597962}} "Definitive {{haematopoiesis}} in the fetal {{liver}} supports self-renewal and differentiation of haematopoietic stem cells and multipotent progenitors (HSC/MPPs) but remains poorly defined in humans. Here, using single-cell transcriptome profiling of approximately 140,000 liver and 74,000 skin, kidney and {{yolk sac}} cells, we identify the repertoire of human {{blood}} and {{immune}} cells during development. We infer differentiation trajectories from HSC/MPPs and evaluate the influence of the tissue microenvironment on blood and immune cell development. We reveal physiological erythropoiesis in fetal skin and the presence of mast cells, natural killer and innate lymphoid cell precursors in the {{yolk sac}}. We demonstrate a shift in the haemopoietic composition of fetal liver during gestation away from being predominantly erythroid, accompanied by a parallel change in differentiation potential of HSC/MPPs, which we functionally validate. Our integrated map of fetal liver haematopoiesis provides a blueprint for the study of paediatric blood and immune disorders, and a reference for harnessing the therapeutic potential of HSC/MPPs." {{blood}} | |||
* '''A human liver cell atlas reveals heterogeneity and epithelial progenitors'''{{#pmid:31292543|PMID31292543}} "The human liver is an essential multifunctional organ. The incidence of liver diseases is rising and there are limited treatment options. However, the cellular composition of the liver remains poorly understood. Here we performed single-cell RNA sequencing of about 10,000 cells from normal liver tissue from nine human donors to construct a human liver cell atlas. Our analysis identified previously unknown subtypes of endothelial cells, {{Kupffer cell}}s, and {{hepatocyte}}s, with transcriptome-wide zonation of some of these populations. We show that the EPCAM+ population is heterogeneous, comprising hepatocyte-biased and cholangiocyte populations as well as a TROP2int progenitor population with strong potential to form bipotent liver organoids. As a proof-of-principle, we used our atlas to unravel the phenotypic changes that occur in hepatocellular carcinoma cells and in human hepatocytes and liver endothelial cells engrafted into a mouse liver. Our human liver cell atlas provides a powerful resource to enable the discovery of previously unknown cell types in normal and diseased livers." | |||
hepatic | * '''The contributions of mesoderm-derived cells in liver development'''{{#pmid:30193996|PMID30193996}} "The liver is an indispensable organ for metabolism and drug detoxification. The liver consists of endoderm-derived hepatobiliary lineages and various mesoderm-derived cells, and interacts with the surrounding tissues and organs through the ventral mesentery. Liver development, from hepatic specification to liver maturation, requires close interactions with mesoderm-derived cells, such as mesothelial cells, hepatic stellate cells, mesenchymal cells, liver sinusoidal endothelial cells and hematopoietic cells. These cells affect liver development through precise signaling events and even direct physical contact. Through the use of new techniques, emerging studies have recently led to a deeper understanding of liver development and its related mechanisms, especially the roles of mesodermal cells in liver development. Based on these developments, the current protocols for in vitro hepatocyte-like cell induction and liver-like tissue construction have been optimized and are of great importance for the treatment of liver diseases. Here, we review the roles of mesoderm-derived cells in the processes of liver development, hepatocyte-like cell induction and liver-like tissue construction." | ||
* '''Molecular regulation of mammalian hepatic architecture'''{{#pmid:30797519|PMID30797519}} "The essential liver exocrine and endocrine functions require a precise spatial arrangement of the hepatic lobule consisting of the central vein, portal vein, hepatic artery, intrahepatic bile duct system, and hepatocyte zonation. This allows blood to be carried through the liver parenchyma sampled by all hepatocytes and bile produced by the hepatocytes to be carried out of the liver through the intrahepatic bile duct system composed of cholangiocytes. The molecular orchestration of multiple signaling pathways and epigenetic factors is required to set up lineage restriction of the bipotential hepatoblast progenitor into the hepatocyte and cholangiocyte cell lineages, and to further refine cell fate heterogeneity within each cell lineage reflected in the functional heterogeneity of hepatocytes and cholangiocytes. In addition to the complex molecular regulation, there is a complicated morphogenetic choreography observed in building the refined hepatic epithelial architecture. Given the multifaceted molecular and cellular regulation, it is not surprising that impairment of any of these processes can result in acute and chronic hepatobiliary diseases. To enlighten the development of potential molecular and cellular targets for therapeutic options, an understanding of how the intricate hepatic molecular and cellular interactions are regulated is imperative. Here, we review the signaling pathways and epigenetic factors regulating hepatic cell lineages, fates, and epithelial architecture." | |||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! More recent papers | |||
|- | |- | ||
| < | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Liver+Embryology ''Liver Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Liver+Development ''Liver Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Hepatic+Embryology ''Hepatic Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Fetal+Hepatocytes ''Fetal Hepatocytes''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Fetal+Liver+Function ''Fetal Liver Function''] | |||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''The fate of the vitelline and umbilical veins during the development of the human liver'''{{#pmid:28786203|PMID28786203}} "Differentiation of endodermal cells into hepatoblasts is well studied, but the remodeling of the vitelline and umbilical veins during liver development is less well understood. We compared human embryos between 3 and 10 weeks of development with pig and mouse embryos at comparable stages, . ...We found no evidence for large-scale fragmentation of embryonic veins as claimed by the 'vestigial' theory. Instead and in agreement with the 'lineage' theory, the vitelline and umbilical veins remained temporally identifiable inside the liver after being engulfed by hepatoblasts. In agreement with the 'hemodynamic' theory, the left-right shunts develop de novo." | |||
* '''A single-cell transcriptomic analysis reveals precise pathways and regulatory mechanisms underlying hepatoblast differentiation'''{{#pmid:28681484|PMID28681484}} "How bipotential hepatoblasts differentiate into hepatocytes and cholangiocytes remains unclear. Here, using single-cell transcriptomic analysis of hepatoblasts, hepatocytes, and cholangiocytes sorted from embryonic day 10.5 (E10.5) to E17.5 mouse embryos, we found that hepatoblast-to-hepatocyte differentiation occurred gradually and followed a linear default pathway. As more cells became fully differentiated hepatocytes, the number of proliferating cells decreased. Surprisingly, proliferating and quiescent hepatoblasts exhibited homogeneous differentiation states at a given developmental stage. This unique feature enabled us to combine single-cell and bulk-cell analyses to define the precise timing of the hepatoblast-to-hepatocyte transition, which occurs between E13.5 and E15.5. In contrast to hepatocyte development at almost all levels, hepatoblast-to-cholangiocyte differentiation underwent a sharp detour from the default pathway. New cholangiocyte generation occurred continuously between E11.5 and E14.5, but their maturation states at a given developmental stage were heterogeneous. Even more surprising, the number of proliferating cells increased as more progenitor cells differentiated into mature cholangiocytes. Based on an observation from the single-cell analysis, we also discovered that the protein kinase C/mitogen-activated protein kinase signaling pathway promoted cholangiocyte maturation. CONCLUSION: Our studies have defined distinct pathways for hepatocyte and cholangiocyte development in vivo, which are critically important for understanding basic liver biology and developing effective strategies to induce stem cells to differentiate toward specific hepatic cell fates in vitro. | |||
* ''' Three-dimensional reconstructions of intrahepatic bile duct tubulogenesis in human liver'''{{#pmid:21943389|PMID21943389}} "Samples from human prenatal livers ranging from 7 weeks + 2 days to 15½ weeks post conception as well as adult normal and acetaminophen intoxicated liver were used. ....In the developing human liver, three-dimensional reconstructions using multiple marker proteins confirmed that the human intrahepatic biliary tree forms through several developmental stages involving an initial transition of primitive hepatocytes into cholangiocytes shaping the ductal plate followed by a process of maturation and remodeling where the intrahepatic biliary tree develops through an asymmetrical form of cholangiocyte tubulogenesis. | |||
* '''Dynamic signaling network for the specification of embryonic pancreas and liver progenitors'''{{#pmid:19556507|PMID19556507}} Studies of the formation of pancreas and liver progenitors have focused on individual inductive signals and cellular responses. Here, we investigated how bone morphogenetic protein, transforming growth factor-beta (TGFbeta), and fibroblast growth factor signaling pathways converge on the earliest genes that elicit pancreas and liver induction in mouse embryos. The inductive network was found to be dynamic; it changed within hours. Different signals functioned in parallel to induce different early genes, and two permutations of signals induced liver progenitor domains, which revealed flexibility in cell programming. Also, the specification of pancreas and liver progenitors was restricted by the TGFbeta pathway." | |||
|} | |||
==Movies== | |||
{| border='0px' | |||
| valign="bottom"|{{Lesser sac movie}} | |||
| valign="bottom"|{{Greater omentum movie}} | |||
|} | |||
== Liver Development Stages == | |||
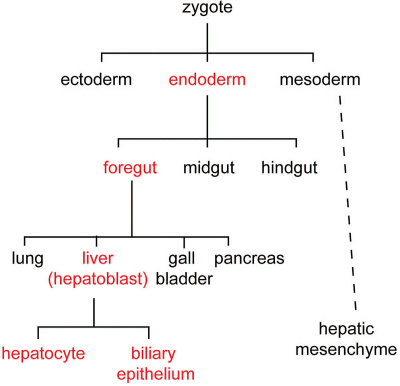
[[File:Zorn2008 fig01.jpg|400px]] | |||
Cell lineage during hepatic development (red) from uncommitted endoderm to functional adult hepatocytes and biliary epithelium.<ref name=Zorn2008>{{Ref-Zorn2008}}</ref> | |||
{{Liver stages simple table01}} | |||
{{Embryonic Liver Timeline Table}} | |||
* '''Size''' - the liver initially occupies the entire anterior body area. | |||
* '''Hepatoblast''' - endoderm the bipotential progenitor for both hepatocytes and cholangiocytes. | |||
* '''Vascular''' - mesoderm blood vessels enter the liver (3 systems: systemic, placental, vitelline) | |||
* '''Sinusoids''' - first blood vessels from vessels in septum transversum mesenchyme. Initially continuous endothelium, become fenestrated in fetal period and reticular development ongoing. | |||
==Adult Liver Cells== | |||
{| | |||
|- | |- | ||
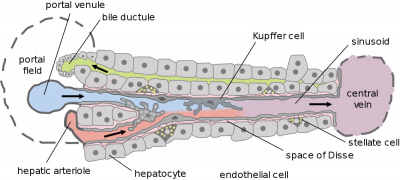
| | | [[File:Liver structure cartoon.jpg|400px]] | ||
| | | | ||
# hepatocytes - form 80% of liver, functional cells | |||
# cholangiocytes - epithelial cells that line the bile ducts | |||
# stellate cells - mesenchymal cells in the space of Disse | |||
# Kupffer cells - liver macrophage in the sinusoids | |||
|} | |||
{| | |||
! Summary Cell Map of the Adult Human Liver | |||
|- | |||
| This study{{#pmid:30348985|PMID30348985}} has shown in the adult human liver at least 20 discrete cell populations exist. These include: hepatocytes, endothelial cells, cholangiocytes, hepatic stellate cells, B cells, conventional and non-conventional T cells, NK-like cells, and distinct intrahepatic monocyte/macrophage populations. | |||
<br><br> | |||
* Parenchymal cells - hepatocytes | |||
* Non-parenchymal cells - endothelial cells, cholangiocytes, macrophages, hepatic stellate cells, and liver infiltrating lymphocytes- including B cells, αβ and γδ, T cells, and NK cells | |||
<br> | |||
Non-inflammatory macrophages are labeled ∗Kupffer cells based on their transcriptional similarity to mouse KC. The location of B cells, plasma cells, T cells, and NK cells has yet to be confirmed by immunohistochemical staining of these populations in situ so their location in this schematic is not representative of their zonated distribution.(text modified from original figure legend) | |||
| [[File:Adult human liver cells.jpg|600px]] | |||
|} | |||
| | ==Liver Growth== | ||
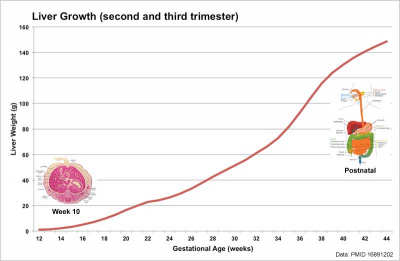
[[File:Fetal_liver_weight_growth_graph.jpg|400px]] | |||
Human Liver Growth (weight grams){{#pmid:16891202|PMID16891202}} | |||
==Liver Buds== | |||
* Differentiates to form the hepatic diverticulum and hepatic primordium, generates the {{gallbladder}} then divides into right and left hepatic (liver) buds. | |||
* Three connecting stalks (cystic duct, hepatic ducts) which fuse to form bile duct. | |||
===Left Hepatic Bud=== | |||
* left lobe, quadrate, caudate (both q and c anatomically Left) | |||
* caudate lobe of human liver consists of 3 anatomical parts: Spiegel's lobe, caudate process, and paracaval portion. | |||
===Right Hepatic Bud=== | |||
* right lobe | |||
==Liver Structural Origins== | |||
[[File:Liver animated cartoon.gif|thumb|300px|The Liver Lobule]] | |||
* Hepatic Buds - form hepatocytes, produce bile from week 13 (forms meconium of newborn) | |||
* Vitelline Veins - form sinusoids | |||
* Mesenchyme - form connective tissue and Kupffer cells | |||
===Function - Haemopoiesis=== | |||
Embryonic liver also involved in blood formation, after the yolk sac and blood islands acting as a primary site. | |||
== Components of Liver Formation == | == Components of Liver Formation == | ||
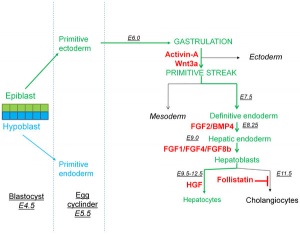
[[File:Liver development signaling.jpg|thumb|Mouse liver development signaling.{{#pmid:21829697|PMID21829697}}]] | |||
'''Primitive Endoderm''' | '''Primitive Endoderm''' | ||
{| class="prettytable" | {| class="prettytable" | ||
| Line 69: | Line 142: | ||
*** hepatic diverticulum | *** hepatic diverticulum | ||
**** cystic primordium | **** cystic primordium | ||
***** | ***** {{gallbladder}} | ||
****** common bile duct | ****** common bile duct | ||
******* hepatic ducts | ******* hepatic ducts | ||
******** '''liver/ | ******** '''liver/{{gallbladder}}''' | ||
*** hepatic primordium | *** hepatic primordium | ||
**** hepatic parenchyma | **** hepatic parenchyma | ||
***** hepatic sinusoids | ***** hepatic sinusoids | ||
****** lobes of liver | ****** lobes of liver | ||
******* '''liver/ | ******* '''liver/{{gallbladder}}''' | ||
|- | |- | ||
| | | | ||
| Line 85: | Line 158: | ||
Data from mouse <ref>Kaufman and Bard, '''The Anatomical Basis of Mouse Development''' 1999 Academic Press</ref> | Data from mouse <ref>Kaufman and Bard, '''The Anatomical Basis of Mouse Development''' 1999 Academic Press</ref> | ||
'''Hepatoblasts''' - endoderm-derived cells can differentiate into either: | |||
# hepatocytes - populate the bulk of the liver parenchyma. | |||
# cholangiocytes - line the intrahepatic bile ducts. | |||
:'''Links:''' [[Endoderm]] | [[Mouse Development]] | :'''Links:''' [[Endoderm]] | [[Mouse Development]] | ||
==Stage 13== | ==Development== | ||
===Stage 13=== | |||
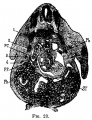
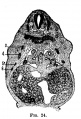
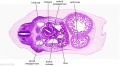
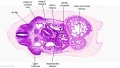
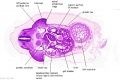
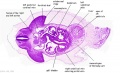
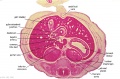
[[File:Stage_13_image_073.jpg|thumb|[[Carnegie stage 13|stage 13]] embryo]] | [[File:Stage_13_image_073.jpg|thumb|[[Carnegie stage 13|stage 13]] embryo]] | ||
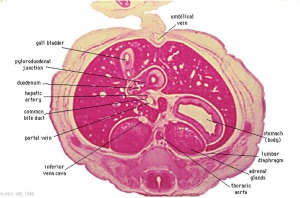
The images below link to larger cross-sections of the mid-embryonic period (end week 4) [[Carnegie stage 13|stage 13]] embryo starting just above the level of the liver and then in sequence through the liver to the level of the stomach. Note the relative position of the liver with respect to the abdominal cavity, the | The images below link to larger cross-sections of the mid-embryonic period (end week 4) [[Carnegie stage 13|stage 13]] embryo starting just above the level of the liver and then in sequence through the liver to the level of the stomach. Note the relative position of the liver with respect to the abdominal cavity, the {{gallbladder}} and the heart. | ||
The [[T#transverse septum|transverse septum]] differentiates to form the hepatic diverticulum and the hepatic primordium, these two structures together will go on to form different components of the mature liver and | The [[T#transverse septum|transverse septum]] differentiates to form the hepatic diverticulum and the hepatic primordium, these two structures together will go on to form different components of the mature liver and {{gallbladder}}. At this stage large vascular channels can be seen coursing through the liver primordium. | ||
{| | {| | ||
| Line 118: | Line 199: | ||
|} | |} | ||
:'''Links:''' [[Carnegie stage 13 - serial sections]] | [[Embryo Serial Sections]] | [[ | :'''Links:''' [[Carnegie stage 13 - serial sections]] | [[Embryo Serial Sections]] | [[Embryo Carnegie stage 13 Movies]] | ||
===Stage 22=== | |||
{| | |||
! colspan=6|Virtual Slide Features - Stage 22 Liver | |||
|- | |||
| [[File:Stage 22 image 034.jpg|150px|link=https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html]] | |||
| colspan=3|[https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html '''Virtual Slide''' - Stage 22 Liver and Ductus Venosus] [[Embryo Virtual Slides|All Virtual Slides]] | |||
The links shown in the table below are to specific features shown on the Human embryo (stage 22) Liver and Ductus Venosus virtual slide. See also notes on [[Gastrointestinal Tract - Liver Development|Liver Development]] | |||
==Stage 22 | Clicking the text will open the slide at a detailed view with the structure generally located in the centre of the view. The slide then can also be zoomed out from the set magnification using the controls in the upper left or the mouse. | ||
Use your browser back button to return to this table. | |||
|colspan=4|You can also make your own selected feature view. | |||
# Set the virtual slide to the region and zoom of interest. | |||
# Click the Permalink (lower righthand corner). | |||
# Then bookmark in your browser, or copy the web address. | |||
See also [[Help:Virtual Slides Permalink|Permalink help]] | |||
|- | |||
| width=150px valign=top|Cardiovascular | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=4&lat=-2765&lon=4922&layers=B ductus venosus] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-5474&lon=5220&layers=B dorsal aorta] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=5&lat=-4674&lon=4196&layers=B inferior vena cava] | |||
| width=150px valign=top|[[Gastrointestinal Tract - Liver Development|Liver]] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=4&lat=-2765&lon=4922&layers=B ductus venosus] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-4127&lon=3336&layers=B hepatocytes] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-3569&lon=1837&layers=B sinusoids] | |||
| width=150px valign=top|Endocrine | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=5&lat=-5416&lon=3951&layers=B R adrenal (fetal cortex)] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=5&lat=-5314&lon=5949&layers=B L adrenal (fetal cortex)] | |||
| width=150px valign=top|Musculoskeletal | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=5&lat=-6218&lon=4901&layers=B vertebral body (cartilage)] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-6454&lon=3223&layers=B rib (cartilage)] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-2746&lon=1171&layers=B body wall (sk. muscle)] | |||
| width=150px valign=top|Neural | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-7220&lon=5075&layers=B spinal cord] | |||
* [https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-6990&lon=4552&layers=B dorsal root ganglia] | |||
| width=150px valign=top|Gastrointestinal | |||
[https://embryology.med.unsw.edu.au/embryology/Slides/Embryo_Stages/Stage22/34/Stage22-34.html?zoom=6&lat=-4497&lon=5779&layers=B stomach (pylorus)] | |||
|} | |||
{{Virtual Slide Features - Stage 22 Liver}} | |||
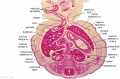
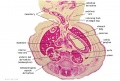
The images below link to larger cross-sections of the end of the embryonic period (week 8) [[Carnegie stage 22|stage 22]] embryo starting just above the level of the liver and then in sequence through the entire liver. (Note the sections are viewed from below, LR axis is reversed) | The images below link to larger cross-sections of the end of the embryonic period (week 8) [[Carnegie stage 22|stage 22]] embryo starting just above the level of the liver and then in sequence through the entire liver. (Note the sections are viewed from below, LR axis is reversed) | ||
| Line 156: | Line 277: | ||
|} | |} | ||
:'''Links:''' [[Carnegie stage 22 - serial sections]] | [[Embryo Serial Sections]] | [[ | :'''Links:''' [[Carnegie stage 22 - serial sections]] | [[Embryo Serial Sections]] | [[Embryo Carnegie stage 22 Movies]] | ||
===Selected Stage 22 Images=== | ===Selected Stage 22 Images=== | ||
| Line 170: | Line 291: | ||
|} | |} | ||
===Week 9=== | |||
{| | |||
| [[File:Human liver week 9.jpg|600px]] | |||
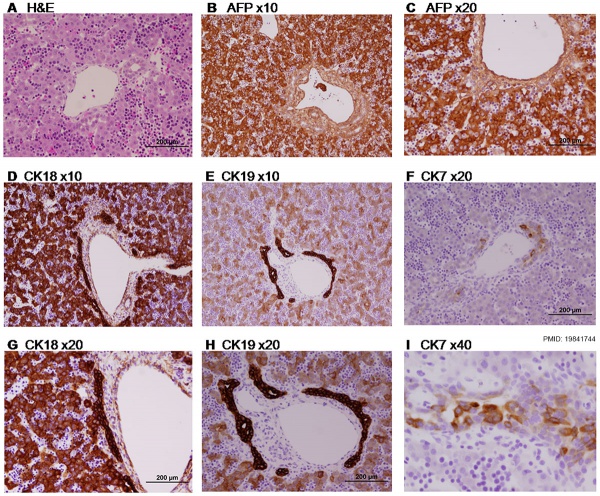
Paraffin-embedded sections of human embryonic liver at 9 weeks ({{GA}} 11 weeks).{{#pmid:19841744|PMID19841744}} | |||
| valign=middle| | |||
* '''A''' - {{HE}} | |||
* '''B, C''' - Alpha-Fetoprotein (AFP) | |||
* '''D, G''' - Cytokeratin 18 (CK18) | |||
* '''E, H''' - Cytokeratin 19 (CK19) | |||
* '''F, I''' - Cytokeratin 7 (CK7) | |||
|} | |||
== Ductal Plate == | == Ductal Plate == | ||
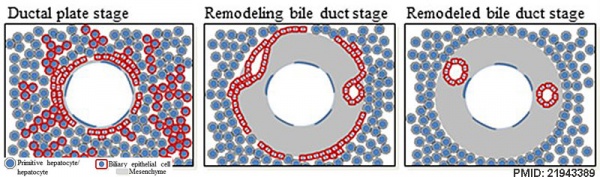
The ductal plate is a primitive biliary epithelium which develops in mesenchyme adjacent to portal vein branches (periportal hepatoblasts). During liver development it is extensively reorganised (ductal plate remodelling) within the developing liver to form the intrahepatic bile ducts (IHBD). If remodelling does not occur, leading to excess of embryonic bile duct structures in the portal tract, these developmental abnormalities are described as "ductal plate malformation" (DPM). | The ductal plate is a primitive biliary epithelium which develops in mesenchyme adjacent to portal vein branches (periportal hepatoblasts). During liver development it is extensively reorganised (ductal plate remodelling) within the developing liver to form the intrahepatic bile ducts (IHBD). If remodelling does not occur, leading to excess of embryonic bile duct structures in the portal tract, these developmental abnormalities are described as "ductal plate malformation" (DPM). | ||
Cholangiocyte tubulogenesis: "ductal plate" stages -> "remodeling bile duct" stage -> "remodeled bile duct" | |||
[[File:Liver cholangiocyte tubulogenesis 01.jpg|600px]] | |||
Cartoon model of bile duct formation.{{#pmid:21943389|PMID21943389}} | |||
See also [[Gastrointestinal Tract - Liver Development#Ductal Plate Malformations|Ductal Plate Malformations]] | |||
== Bile Secretion == | == Bile Secretion == | ||
| Line 188: | Line 324: | ||
# connected to '''intrahepatic bile ducts''' | # connected to '''intrahepatic bile ducts''' | ||
# intrahepatic bile ducts connect to the '''hepatic duct''' | # intrahepatic bile ducts connect to the '''hepatic duct''' | ||
# then the '''cystic duct''' for storage in the '''gallbladder''' | # then the '''cystic duct''' for storage in the '''{{gallbladder}}''' | ||
# then the '''common bile duct''' into the duodenum | # then the '''common bile duct''' into the duodenum | ||
The term '''extrahepatic bile ducts''' (EHBDs) is used to describe the hepatic, cystic, and common bile ducts. | The term '''extrahepatic bile ducts''' (EHBDs) is used to describe the hepatic, cystic, and common bile ducts. | ||
The developing bile ducts express VEGF while hepatoblasts express angiopoietin-1, these two signals are thought to regulate arterial vasculogenesis and remodeling of the hepatic artery respectively.< | The developing bile ducts express VEGF while hepatoblasts express angiopoietin-1, these two signals are thought to regulate arterial vasculogenesis and remodeling of the hepatic artery respectively.{{#pmid:12360416|PMID12360416}} | ||
==Liver Vascular== | |||
Vascular development data below from{{#pmid:18484606|PMID18484606}} | |||
'''Venous''' | |||
* week 4 - hepatic primordium in contact with vitelline veins and the umbilical veins. | |||
* week 4 to 6 - efferent venous vessels form from the vitelline veins. Afferent venous liver circulation present. | |||
* Week 6 onward - portal vein formed from segments of the vitelline veins | |||
** portal sinus (from subhepatic intervitelline anastomosis) connects umbilical vein to portal system. | |||
** ductus venosus connects portal sinus to vena cava inferior. | |||
* birth - both umbilical vein and ductus venosus collapse. | |||
** portal vein becomes the only afferent vein of the liver. | |||
'''Arterial''' | |||
* week 8 - hepatic artery forms. | |||
* week 10 to 15 - intrahepatic arterial branches progressively extend from the central to the peripheral areas of the liver. | |||
Hepatic sinusoids | |||
* week 4 - hepatic cords invade the septum transversum | |||
** progressively acquire structural and functional characters, through a multistage process. | |||
===Liver Blood Flow=== | |||
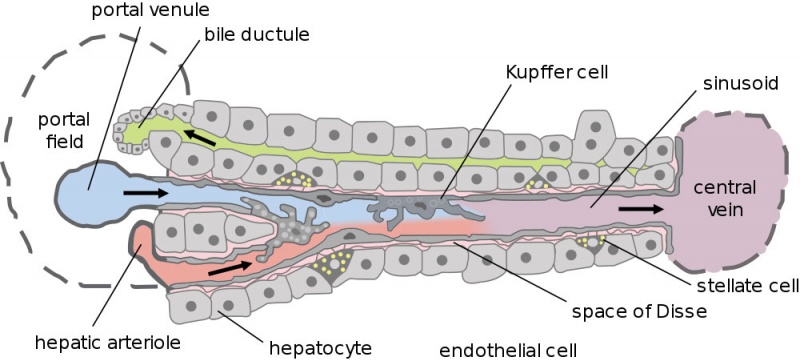
[[File:Liver_structure_cartoon.jpg|800px]] | |||
Dual blood supply of the liver merges upon entry into the liver lobule at the portal field. The blood flows along the sinusoid and exits at the central vein. | |||
# branches of the portal vein | |||
# branches of the hepatic artery | |||
'''Portal Vein''' - is the sole supplier to the liver until about human 20 mm CRL stage. Portal vein primary branches extend around the periphery of each primitive liver lobule. This branching process continues, from primary to secondary, with each development supplying all newly forming liver lobules. Primary branches also lie parallel to the branches of hepatic veins, that drain the blood from the centre of each early lobule. | |||
'''Hepatic Artery''' - from the coeliac axis, initially contact the hepatic duct and {{gallbladder}}, later grows into the connective tissue about the larger bile ducts and branches of the portal vein. The hepatic artery will also supply the capsule of the liver. | |||
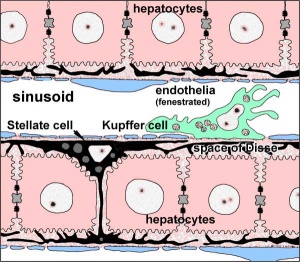
== Hepatocytes == | |||
[[File:Liver-sinusoid colour cartoon.jpg|thumb|Adult Liver sinusoid structure]] | |||
These are the adult functional cells forming the majority of the liver (80% of the cells). | |||
Many different functions including: | |||
* '''Storage''' of substances including glucose (as glycogen), vitamin A (possibly in specialized adipocytes), vitamin B12, folic acid and iron. | |||
* '''Lipid Turnover''' synthesis of plasmalipoproteins | |||
* '''Plasma Protein Synthesis''' albumin, alpha and beta globulins, prothrombin, fibrinogen | |||
* '''Metabolism''' fat soluble compounds (drugs, insecticides), steroid hormones turnover | |||
* '''Secretion''' bile (about 1 litre/day) | |||
== Kupffer Cells == | |||
Kupffer Cells are a population of tissue macrophages found in the lumen of hepatic sinusoids, their role is endocytic acting against blood-borne materials entering the liver. | |||
Primordial (primitive) macrophages arise in the yolk sac and then differentiate into fetal macrophages, either of these enter the blood and migrate into the developing liver.{{#pmid:15057601|PMID15057601}} | |||
Tissue macrophages are a family of cells found in many organs{{#pmid:28662662|PMID28662662}}: {{liver}} Kupffer cells, {{neural}} microglia, {{respiratory}} alveolar macrophages, and {{integumentary}} epidermal Langerhans cells. In the embryo, they have a common embryonic origin from yolk sac (YS) erythro-myeloid progenitors (EMPs). In the adult, they are self-maintained in tissues independently of hematopoietic stem cells (HSCs).{{#pmid:27492475|PMID27492475}} | |||
:'''Search PubMed:''' [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=pubmed&cmd=search&term=Kupffer%20cell%20development Kupffer cell development] | |||
==Liver Associated Vessels== | |||
{| | |||
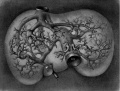
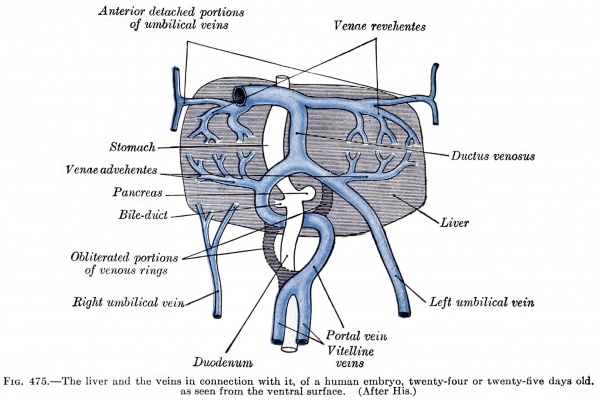
| [[File:Gray0475.jpg|600px]] | |||
Liver ventral surface and associated veins (human embryo, 24-25 days, after His.) | |||
| | |||
* '''ductus venosus''' - shunts approximately half the umbilical vein blood flow directly to the inferior vena cava. | |||
* '''portal vein''' - carries blood from the gastrointestinal tract and spleen to the liver. | |||
* '''left umbilical vein''' - carries oxygenated blood from the placenta to the embryo/fetus. | |||
* '''right umbilical vein''' - vessel degenerates leaving a single (left) umbilical vein. | |||
* '''vena revehens''' - veins from the sinusoid vessels in the liver to the inferior vena cava, that later develop into the hepatic veins. | |||
|} | |||
==Adult Liver Transplants== | |||
* About 6,000 liver transplant operations are performed in the United States (http://www.liverfoundation.org/education/info/transplant/) | |||
* About 600–700 in the UK every year (http://www.britishlivertrust.org.uk/home/the-liver/liver-transplantation/a-history-of-liver-transplantation-and-current-statistics.aspx). | |||
* The main limitation on numbers are the availability of donor organs.{{#pmid:20169088|PMID20169088}} | |||
==Histology== | |||
[[File:Liver animated cartoon.gif]] | |||
The Liver Lobule | |||
<gallery> | |||
File:Liver histology 009.jpg|Histology-fetal liver HEx40 | |||
File:Liver histology 008.jpg|Histology-fetal liver x100 | |||
</gallery> | |||
Adult liver Portal Triad | |||
[[File:Liver_histology_005.jpg|600px]] | |||
:'''Links:''' {{liver histology}} | |||
==Abnormalities== | |||
{| | |||
|-bgcolor="FFCC00" | |||
! {{ICD-11}} {{ICD11weblink}}1737370752 '''LB20''' Structural developmental anomalies of gallbladder, bile ducts or liver] | |||
|-bgcolor="FEF9E7" | |||
| | |||
{{ICD11weblink}}557775192 LB20.00 Fibropolycystic liver disease] | |||
{{ICD11weblink}}645741117 LB20.21 Biliary atresia] - ''Biliary atresia is a rare disease characterised by an inflammatory biliary obstruction of unknown origin that presents in the neonatal period. It is the most frequent surgical cause of cholestatic jaundice in this age group. Untreated, this condition leads to cirrhosis and death within the first years of life.'' | |||
{{ICD11weblink}}682625970 LA90.21 Anomalous portal venous connection] | |||
|} | |||
===Congenital Absence of Portal Vein=== | |||
Congenital Absence of the Portal Vein (CAPV) is a rare abnormality where the intestinal and splenic venous drainage bypass the liver and drain directly into the systemic veins through various porto-systemic shunts. | |||
:'''Links:''' [[Gastrointestinal Tract - Abnormalities]] | |||
===Ductal Plate Malformations=== | |||
* '''Interlobular bile ducts''' - autosomal recessive polycystic kidney disease | |||
* '''Smaller interlobular ducts''' - von Meyenburg complexes | |||
* '''Larger intrahepatic bile ducts''' - Caroli's disease | |||
===Hepatobiliary cysts=== | |||
Fetal hepatic cysts are generally benign, with a low likelihood of associated anomalies of the hepatobiliary tract, abnormal liver function or clinical symptoms.{{#pmid:27553859|PMID27553859}} | |||
===Maternal Liver=== | |||
There are several maternal pregnancy-associated direct and indirect liver diseases, see recent review.{{#pmid:30046419|PMID30046419}} | |||
* Intrahepatic cholestasis of pregnancy (ICP) - most common liver disease in pregnancy, presenting mainly in the second and third trimesters, with pruritis, elevated serum bile acids, and abnormal liver function tests.{{#pmid:30396107|PMID30396107}}{{#pmid:29480924|PMID29480924}} Many countries have developed guidelines to address the risks, diagnosis and management of ICP. (see [https://www.kemh.health.wa.gov.au/~/media/Files/Hospitals/WNHS/For%20health%20professionals/Clinical%20guidelines/OG/WNHS.OG.CholestasisInPregnancy.pdf Western Australia information]) | |||
** Bile Acids levels greater than 10 μmol/L are a common diagnostic marker. | |||
** Liver Function Tests - significant aminotransferase increase, serum bilirubin not usually raised. | |||
* Hyperemesis gravidarum - the clinical term for severe form of nausea and vomiting, which are common symptoms of early pregnancy (4 - 16 weeks). Causal factors include increased human chorionic gonadotropin (hCG) and steroids, multiple pregnancy and vitamin deficiency. The condition can lead to dehydration, ketonuria, catabolism and may require hospitalisation. | |||
* Acute fatty liver disease of pregnancy (AFLP) - fatty infiltration of the liver, occurring usually in the third trimester though can also occur postnatally. | |||
* Pre-eclampsia | |||
* HELLP - haemolysis (with a micoangiopathic blood smear), elevated liver enzymes (LFTs) and low platelet count. | |||
:'''Links:''' [https://www.kemh.health.wa.gov.au/~/media/Files/Hospitals/WNHS/For%20health%20professionals/Clinical%20guidelines/OG/WNHS.OG.CholestasisInPregnancy.pdf PDF - Australia WA Department of Health. Cholestasis in Pregnancy. April 2016]. | |||
==Animal Models== | |||
===Mouse=== | |||
See molecular review by Zorn.<ref name=Zorn2008>{{Ref-Zorn2008}}</ref> | |||
* {{ME9.5}} to {{ME15}} - liver bud undergoes growth and is the major site of haematopoiesis. | |||
* {{ME13}} - bi-potential hepatoblasts differentiate into hepatocytes or biliary epithelial cells. | |||
* {{ME16.5}} - ductal plate partially becomes bi-layered | |||
* {{ME17.5}} - ductal plate remodelling, focal dilations appear between the two cell layers. | |||
==References== | ==References== | ||
<references/> | <references/> | ||
===Books=== | |||
{{Ref-Zorn2008}} | |||
===Reviews=== | ===Reviews=== | ||
{{#pmid:31201265}} | |||
{{#pmid:30046419}} | |||
{{#pmid:26970006}} | |||
{{#pmid:26081571}} | |||
{{#pmid:23799566}} | |||
{{#pmid:23720329}} | |||
{{#pmid:21199531}} | |||
{{#pmid:20551648}} | |||
{{#pmid:20169172}} | |||
{{#pmid:20159590}} | |||
{{#pmid:20086072}} | |||
{{#pmid:18484608}} | |||
===Articles=== | |||
{{#pmid:26413551}} | |||
{{#pmid:22371217}} | |||
{{#pmid:22105167}} | |||
{{#pmid:21708104}} | |||
{{#pmid:18484606}} | |||
{{#pmid:6832717}} | |||
===Historic=== | |||
{{Ref-Mall1906liver}} | |||
{{Ref-Severn1971}} | |||
{{Ref-Severn1972}} | |||
===Search Pubmed=== | ===Search Pubmed=== | ||
| Line 207: | Line 543: | ||
'''Search Pubmed Now:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=Liver%20Development Liver Development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=embryonic%20Liver%20Development Embryonic Liver Development] | '''Search Pubmed Now:''' [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=Liver%20Development Liver Development] | [http://www.ncbi.nlm.nih.gov/sites/entrez?db=pubmed&cmd=search&term=embryonic%20Liver%20Development Embryonic Liver Development] | ||
==Additional Images== | |||
===Historic=== | |||
{{Ref-Mall1906liver}} | |||
<gallery> | |||
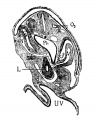
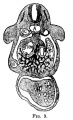
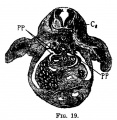
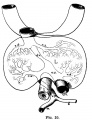
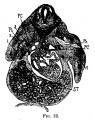
File:Mall1906-fig06.jpg|Fig 6 human embryo 2.1 mm | |||
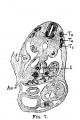
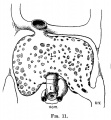
File:Mall1906-fig07.jpg|Fig 7 human embryo 4.3 mm | |||
File:Mall1906-fig08.jpg|Fig 8 human embryo 4.3 mm | |||
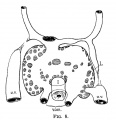
File:Mall1906-fig09.jpg|Fig 9 human embryo 4.5 mm | |||
File:Mall1906-fig10.jpg|Fig 10 human embryo 4.5 mm | |||
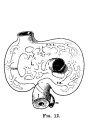
File:Mall1906-fig11.jpg|Fig 11 human embryo 4 mm | |||
File:Mall1906-fig12.jpg|Fig 12 human embryo 6.6 mm | |||
File:Mall1906-fig13.jpg|Fig 13 human embryo 5 mm | |||
File:Mall1906-fig14.jpg|Fig 14 human embryo 5 mm | |||
File:Mall1906-fig15.jpg|Fig 15 human embryo 7 mm | |||
File:Mall1906-fig16.jpg|Fig 16 human embryo 7 mm | |||
File:Mall1906-fig17.jpg|Fig 17 human embryo 7 mm | |||
File:Mall1906-fig18.jpg|Fig 18 human embryo 9 mm | |||
File:Mall1906-fig19.jpg|Fig 19 human embryo 9 mm | |||
File:Mall1906-fig20.jpg|Fig 20 human embryo 9 mm | |||
File:Mall1906-fig21.jpg|Fig 21 human embryo 11 mm | |||
File:Mall1906-fig22.jpg|Fig 22 human embryo 11 mm | |||
File:Mall1906-fig23.jpg|Fig 23 human embryo 11 mm | |||
File:Mall1906-fig24.jpg|Fig 24 human embryo 11 mm | |||
File:Mall1906-fig25.jpg|Fig 25 human embryo 11 mm | |||
File:Mall1906-fig26.jpg|Fig 26 human embryo 20 mm | |||
File:Mall1906-fig27.jpg|Fig 27 human embryo 20 mm | |||
File:Mall1906-fig28.jpg|Fig 28 human embryo 24 mm | |||
File:Mall1906-fig29.jpg|Fig 29 model human embryo 24 mm | |||
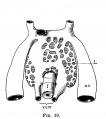
File:Mall1906-fig30.jpg|Fig 30 model human embryo 24 mm | |||
</gallery> | |||
==Terms== | |||
{{GIT terms}} | |||
{{Glossary}} | |||
{{ | {{Footer}} | ||
[[Category:Liver]] | |||
Latest revision as of 13:49, 23 January 2020
| Embryology - 10 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
This section of notes gives an overview of how the liver develops. Initially, the transverse septum (septum transversum) arises at an embryonic junctional site. The junctional region externally is where the ectoderm of the amnion meets the endoderm of the yolk sac. The junctional region internally is where the foregut meets the Midgut. The mesenchymal structure of the transverse septum provides a support within which both blood vessels and the liver begin to form. Arises at embryonic junction (septum transversum): externally is where ectoderm of amnion meets endoderm of yolk sac and internally is where the foregut meets the midgut. Mesenchymal structure of transverse septum provides a support within which both blood vessels and liver begin to form in the underlying splanchnic mesoderm.
In the early embryo, the liver and heart grow rapidly forming obvious external swellings on the ventral embryo surface. The liver's initial embryonic function is mainly cardiovascular. Firstly, as a vascular connection between the developing placental vessels to the heart. Secondly, as a haemopoietic tissue where blood stem cells reside before bone marrow development.
The fetal liver also has an endocrine role by 16-hydroxylation, that results in estriol being the major estrogen type produced in late human pregnancy.
A recent molecular study has shown that within the adult liver at least 20 discrete cell populations exist these include: hepatocytes, endothelial cells, cholangiocytes, hepatic stellate cells, B cells, conventional and non-conventional T cells, NK-like cells, and distinct intrahepatic monocyte/macrophage populations.[1]
See also liver histology showing both developmental and adult histology.
| Historic Embryology |
|
Marcello Malpighi (1628 – 1694) was an Italian biologist and physician who in 1666 first named the liver lobules - "the livers of all vertebrates are conglomerate glands, being composed of lobules which in turn contain acini". He is also known for structures that bear his name in the spleen Malpighian bodies (white pulp) and renal Malpighian corpuscle (renal corpuscle). See also Mall's 1906 Liver Historical Note |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Liver Embryology | Liver Development | Hepatic Embryology | Fetal Hepatocytes | Fetal Liver Function |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Movies
|
|
Liver Development Stages
Cell lineage during hepatic development (red) from uncommitted endoderm to functional adult hepatocytes and biliary epithelium.[10]
| Feature | ||
|---|---|---|
| hepatic diverticulum development (ductal plate) | ||
| cell differentiation
septum transversum forming liver stroma hepatic diverticulum forming hepatic trabeculae | ||
| epithelial cord proliferation enmeshing stromal capillaries | ||
| hepatic gland and its vascular channels enlarge
hematopoietic function appeared | ||
| obturation due to epithelial proliferation
bile ducts became reorganized (continuity between liver cells and gut) | ||
| biliary ductules developed in periportal connective tissue
produces ductal plates that receive biliary capillaries | ||
| Human data[11], see also liver development in the rat embryonic period (Carnegie stages 15-23).[12] (More? Detailed Timeline | Timeline human development) | ||
| Carnegie Stage | Age (days) | CRL (mm) | Biliary system | Vascular | Hepatic parenchyma |
|---|---|---|---|---|---|
| 14 | 33 | 7 |
|
|
|
| 18 | 46 | 15 |
|
|
|
| 21 | 53 | 22.5 | Bile duct morphology as earlier stage. Common bile duct empties at the level of the proximal duodenum. |
|
Hepatic parenchyma a large rounded mass. |
| 23 | 58 | 27 | Bile duct morphology as earlier stage. |
|
|
| Data from a recent human study[13] Links: liver | Carnegie stage 14 | 18 | 21 | 23 | simple embryonic timeline | Timeline human development | |||||
- Size - the liver initially occupies the entire anterior body area.
- Hepatoblast - endoderm the bipotential progenitor for both hepatocytes and cholangiocytes.
- Vascular - mesoderm blood vessels enter the liver (3 systems: systemic, placental, vitelline)
- Sinusoids - first blood vessels from vessels in septum transversum mesenchyme. Initially continuous endothelium, become fenestrated in fetal period and reticular development ongoing.
Adult Liver Cells
|
|
| Summary Cell Map of the Adult Human Liver | |
|---|---|
| This study[1] has shown in the adult human liver at least 20 discrete cell populations exist. These include: hepatocytes, endothelial cells, cholangiocytes, hepatic stellate cells, B cells, conventional and non-conventional T cells, NK-like cells, and distinct intrahepatic monocyte/macrophage populations.
|
|
Liver Growth
Human Liver Growth (weight grams)[14]
Liver Buds
- Differentiates to form the hepatic diverticulum and hepatic primordium, generates the gallbladder then divides into right and left hepatic (liver) buds.
- Three connecting stalks (cystic duct, hepatic ducts) which fuse to form bile duct.
Left Hepatic Bud
- left lobe, quadrate, caudate (both q and c anatomically Left)
- caudate lobe of human liver consists of 3 anatomical parts: Spiegel's lobe, caudate process, and paracaval portion.
Right Hepatic Bud
- right lobe
Liver Structural Origins
- Hepatic Buds - form hepatocytes, produce bile from week 13 (forms meconium of newborn)
- Vitelline Veins - form sinusoids
- Mesenchyme - form connective tissue and Kupffer cells
Function - Haemopoiesis
Embryonic liver also involved in blood formation, after the yolk sac and blood islands acting as a primary site.
Components of Liver Formation
Primitive Endoderm
|
|
|
Data from mouse [16]
Hepatoblasts - endoderm-derived cells can differentiate into either:
- hepatocytes - populate the bulk of the liver parenchyma.
- cholangiocytes - line the intrahepatic bile ducts.
- Links: Endoderm | Mouse Development
Development
Stage 13
The images below link to larger cross-sections of the mid-embryonic period (end week 4) stage 13 embryo starting just above the level of the liver and then in sequence through the liver to the level of the stomach. Note the relative position of the liver with respect to the abdominal cavity, the gallbladder and the heart.
The transverse septum differentiates to form the hepatic diverticulum and the hepatic primordium, these two structures together will go on to form different components of the mature liver and gallbladder. At this stage large vascular channels can be seen coursing through the liver primordium.
|
|
|
|
|
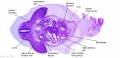
|
| D3L | D4L | D5L | D6L | D7L | E1L |
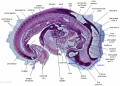
|
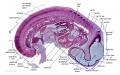
| ||||
| G6L | G7L |
- Links: Carnegie stage 13 - serial sections | Embryo Serial Sections | Embryo Carnegie stage 13 Movies
Stage 22
| Virtual Slide Features - Stage 22 Liver | |||||||
|---|---|---|---|---|---|---|---|
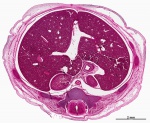
|
Virtual Slide - Stage 22 Liver and Ductus Venosus All Virtual Slides
The links shown in the table below are to specific features shown on the Human embryo (stage 22) Liver and Ductus Venosus virtual slide. See also notes on Liver Development Clicking the text will open the slide at a detailed view with the structure generally located in the centre of the view. The slide then can also be zoomed out from the set magnification using the controls in the upper left or the mouse. Use your browser back button to return to this table. |
You can also make your own selected feature view.
See also Permalink help | |||||
| Cardiovascular | Liver | Endocrine | Musculoskeletal | Neural | Gastrointestinal | ||
| Virtual Slide Features - Stage 22 Liver | |||||||
|---|---|---|---|---|---|---|---|

|
Virtual Slide - Stage 22 Liver and Ductus Venosus All Virtual Slides
The links shown in the table below are to specific features shown on the Human embryo (stage 22) Liver and Ductus Venosus virtual slide. See also notes on Liver Development Clicking the text will open the slide at a detailed view with the structure generally located in the centre of the view. The slide then can also be zoomed out from the set magnification using the controls in the upper left or the mouse. Use your browser back button to return to this table. |
You can also make your own selected feature view.
See also Permalink help | |||||
| Cardiovascular | Liver | Endocrine | Musculoskeletal | Neural | Gastrointestinal | ||
The images below link to larger cross-sections of the end of the embryonic period (week 8) stage 22 embryo starting just above the level of the liver and then in sequence through the entire liver. (Note the sections are viewed from below, LR axis is reversed)
The rapidly developing liver also forms a visible surface bulge on the embryo directly under the heart bulge. The liver now occupies the entire ventral body cavity with parts of the gastrointestinal tract and urinary system "embedded" within its structure. Note in this image the large central ductus venosus.
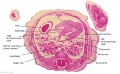
|
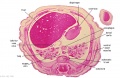
|
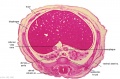
|
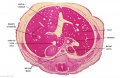
|
|
| E3L | E4L | E5L | E6L | E7L |
|
|
|
|
|
| F1L | F2L | F3L | F4L | F5L |
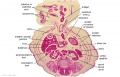
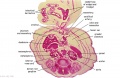
- Links: Carnegie stage 22 - serial sections | Embryo Serial Sections | Embryo Carnegie stage 22 Movies
Selected Stage 22 Images
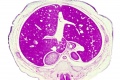
|
E3 Overview of liver region for selected high power views shown below. Note the position and size of the developing liver spanning the entire abdomen and within the liver the large central ductus venosus. |
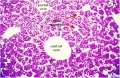
|
E4 Central veins of liver. Radiating appearance of hepatic sinusoids. unlabeled version |
|
E5 Central vein with endothelial lining, containing nucleated erythrocytes, fetal red blood cells. The fetal liver has an important haemopoietic role. unlabeled version |
Week 9
Paraffin-embedded sections of human embryonic liver at 9 weeks (GA 11 weeks).[17] |
|
Ductal Plate
The ductal plate is a primitive biliary epithelium which develops in mesenchyme adjacent to portal vein branches (periportal hepatoblasts). During liver development it is extensively reorganised (ductal plate remodelling) within the developing liver to form the intrahepatic bile ducts (IHBD). If remodelling does not occur, leading to excess of embryonic bile duct structures in the portal tract, these developmental abnormalities are described as "ductal plate malformation" (DPM).
Cholangiocyte tubulogenesis: "ductal plate" stages -> "remodeling bile duct" stage -> "remodeled bile duct"
Cartoon model of bile duct formation.[8]
See also Ductal Plate Malformations
Bile Secretion
The epithelial cells that line the bile ducts are called cholangiocytes.
The pathway below describes the production and passage of bile for final excretion into the duodenum:
- hepatocytes produce bile
- secreted into bile canaliculi
- connected to intrahepatic bile ducts
- intrahepatic bile ducts connect to the hepatic duct
- then the cystic duct for storage in the gallbladder
- then the common bile duct into the duodenum
The term extrahepatic bile ducts (EHBDs) is used to describe the hepatic, cystic, and common bile ducts.
The developing bile ducts express VEGF while hepatoblasts express angiopoietin-1, these two signals are thought to regulate arterial vasculogenesis and remodeling of the hepatic artery respectively.[18]
Liver Vascular
Vascular development data below from[19]
Venous
- week 4 - hepatic primordium in contact with vitelline veins and the umbilical veins.
- week 4 to 6 - efferent venous vessels form from the vitelline veins. Afferent venous liver circulation present.
- Week 6 onward - portal vein formed from segments of the vitelline veins
- portal sinus (from subhepatic intervitelline anastomosis) connects umbilical vein to portal system.
- ductus venosus connects portal sinus to vena cava inferior.
- birth - both umbilical vein and ductus venosus collapse.
- portal vein becomes the only afferent vein of the liver.
Arterial
- week 8 - hepatic artery forms.
- week 10 to 15 - intrahepatic arterial branches progressively extend from the central to the peripheral areas of the liver.
Hepatic sinusoids
- week 4 - hepatic cords invade the septum transversum
- progressively acquire structural and functional characters, through a multistage process.
Liver Blood Flow
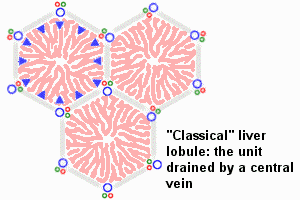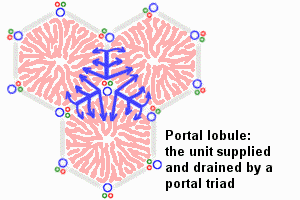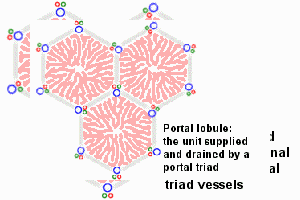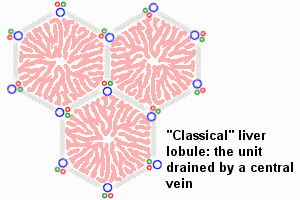
Dual blood supply of the liver merges upon entry into the liver lobule at the portal field. The blood flows along the sinusoid and exits at the central vein.
- branches of the portal vein
- branches of the hepatic artery
Portal Vein - is the sole supplier to the liver until about human 20 mm CRL stage. Portal vein primary branches extend around the periphery of each primitive liver lobule. This branching process continues, from primary to secondary, with each development supplying all newly forming liver lobules. Primary branches also lie parallel to the branches of hepatic veins, that drain the blood from the centre of each early lobule.
Hepatic Artery - from the coeliac axis, initially contact the hepatic duct and gallbladder, later grows into the connective tissue about the larger bile ducts and branches of the portal vein. The hepatic artery will also supply the capsule of the liver.
Hepatocytes
These are the adult functional cells forming the majority of the liver (80% of the cells).
Many different functions including:
- Storage of substances including glucose (as glycogen), vitamin A (possibly in specialized adipocytes), vitamin B12, folic acid and iron.
- Lipid Turnover synthesis of plasmalipoproteins
- Plasma Protein Synthesis albumin, alpha and beta globulins, prothrombin, fibrinogen
- Metabolism fat soluble compounds (drugs, insecticides), steroid hormones turnover
- Secretion bile (about 1 litre/day)
Kupffer Cells
Kupffer Cells are a population of tissue macrophages found in the lumen of hepatic sinusoids, their role is endocytic acting against blood-borne materials entering the liver.
Primordial (primitive) macrophages arise in the yolk sac and then differentiate into fetal macrophages, either of these enter the blood and migrate into the developing liver.[20]
Tissue macrophages are a family of cells found in many organs[21]: liver Kupffer cells, neural microglia, respiratory alveolar macrophages, and integumentary epidermal Langerhans cells. In the embryo, they have a common embryonic origin from yolk sac (YS) erythro-myeloid progenitors (EMPs). In the adult, they are self-maintained in tissues independently of hematopoietic stem cells (HSCs).[22]
- Search PubMed: Kupffer cell development
Liver Associated Vessels
Liver ventral surface and associated veins (human embryo, 24-25 days, after His.) |
|
Adult Liver Transplants
- About 6,000 liver transplant operations are performed in the United States (http://www.liverfoundation.org/education/info/transplant/)
- About 600–700 in the UK every year (http://www.britishlivertrust.org.uk/home/the-liver/liver-transplantation/a-history-of-liver-transplantation-and-current-statistics.aspx).
- The main limitation on numbers are the availability of donor organs.[23]
Histology
The Liver Lobule
Adult liver Portal Triad
- Links: liver histology
Abnormalities
| ICD-11 LB20 Structural developmental anomalies of gallbladder, bile ducts or liver |
|---|
|
LB20.00 Fibropolycystic liver disease LB20.21 Biliary atresia - Biliary atresia is a rare disease characterised by an inflammatory biliary obstruction of unknown origin that presents in the neonatal period. It is the most frequent surgical cause of cholestatic jaundice in this age group. Untreated, this condition leads to cirrhosis and death within the first years of life. |
Congenital Absence of Portal Vein
Congenital Absence of the Portal Vein (CAPV) is a rare abnormality where the intestinal and splenic venous drainage bypass the liver and drain directly into the systemic veins through various porto-systemic shunts.
Ductal Plate Malformations
- Interlobular bile ducts - autosomal recessive polycystic kidney disease
- Smaller interlobular ducts - von Meyenburg complexes
- Larger intrahepatic bile ducts - Caroli's disease
Hepatobiliary cysts
Fetal hepatic cysts are generally benign, with a low likelihood of associated anomalies of the hepatobiliary tract, abnormal liver function or clinical symptoms.[24]
Maternal Liver
There are several maternal pregnancy-associated direct and indirect liver diseases, see recent review.[25]
- Intrahepatic cholestasis of pregnancy (ICP) - most common liver disease in pregnancy, presenting mainly in the second and third trimesters, with pruritis, elevated serum bile acids, and abnormal liver function tests.[26][27] Many countries have developed guidelines to address the risks, diagnosis and management of ICP. (see Western Australia information)
- Bile Acids levels greater than 10 μmol/L are a common diagnostic marker.
- Liver Function Tests - significant aminotransferase increase, serum bilirubin not usually raised.
- Hyperemesis gravidarum - the clinical term for severe form of nausea and vomiting, which are common symptoms of early pregnancy (4 - 16 weeks). Causal factors include increased human chorionic gonadotropin (hCG) and steroids, multiple pregnancy and vitamin deficiency. The condition can lead to dehydration, ketonuria, catabolism and may require hospitalisation.
- Acute fatty liver disease of pregnancy (AFLP) - fatty infiltration of the liver, occurring usually in the third trimester though can also occur postnatally.
- Pre-eclampsia
- HELLP - haemolysis (with a micoangiopathic blood smear), elevated liver enzymes (LFTs) and low platelet count.
Animal Models
Mouse
See molecular review by Zorn.[10]
- E9.5 to E15 - liver bud undergoes growth and is the major site of haematopoiesis.
- E13 - bi-potential hepatoblasts differentiate into hepatocytes or biliary epithelial cells.
- E16.5 - ductal plate partially becomes bi-layered
- E17.5 - ductal plate remodelling, focal dilations appear between the two cell layers.
References
- ↑ 1.0 1.1 MacParland SA, Liu JC, Ma XZ, Innes BT, Bartczak AM, Gage BK, Manuel J, Khuu N, Echeverri J, Linares I, Gupta R, Cheng ML, Liu LY, Camat D, Chung SW, Seliga RK, Shao Z, Lee E, Ogawa S, Ogawa M, Wilson MD, Fish JE, Selzner M, Ghanekar A, Grant D, Greig P, Sapisochin G, Selzner N, Winegarden N, Adeyi O, Keller G, Bader GD & McGilvray ID. (2018). Single cell RNA sequencing of human liver reveals distinct intrahepatic macrophage populations. Nat Commun , 9, 4383. PMID: 30348985 DOI.
- ↑ Popescu DM, Botting RA, Stephenson E, Green K, Webb S, Jardine L, Calderbank EF, Polanski K, Goh I, Efremova M, Acres M, Maunder D, Vegh P, Gitton Y, Park JE, Vento-Tormo R, Miao Z, Dixon D, Rowell R, McDonald D, Fletcher J, Poyner E, Reynolds G, Mather M, Moldovan C, Mamanova L, Greig F, Young MD, Meyer KB, Lisgo S, Bacardit J, Fuller A, Millar B, Innes B, Lindsay S, Stubbington MJT, Kowalczyk MS, Li B, Ashenberg O, Tabaka M, Dionne D, Tickle TL, Slyper M, Rozenblatt-Rosen O, Filby A, Carey P, Villani AC, Roy A, Regev A, Chédotal A, Roberts I, Göttgens B, Behjati S, Laurenti E, Teichmann SA & Haniffa M. (2019). Decoding human fetal liver haematopoiesis. Nature , 574, 365-371. PMID: 31597962 DOI.
- ↑ Aizarani N, Saviano A, Sagar L, Mailly S, Durand JS, Herman P, Pessaux TF, Baumert D & Grün. (2019). A human liver cell atlas reveals heterogeneity and epithelial progenitors. Nature , , . PMID: 31292543 DOI.
- ↑ Yang L, Li LC, Lamaoqiezhong X, Wang WH, Wang YC, Wang CR & Xu. (2019). The contributions of mesoderm-derived cells in liver development. Semin. Cell Dev. Biol. , 92, 63-76. PMID: 30193996 DOI.
- ↑ Huppert SS & Iwafuchi-Doi M. (2019). Molecular regulation of mammalian hepatic architecture. Curr. Top. Dev. Biol. , 132, 91-136. PMID: 30797519 DOI.
- ↑ Hikspoors JPJM, Peeters MMJP, Mekonen HK, Kruepunga N, Mommen GMC, Cornillie P, Köhler SE & Lamers WH. (2017). The fate of the vitelline and umbilical veins during the development of the human liver. J. Anat. , 231, 718-735. PMID: 28786203 DOI.
- ↑ Yang L, Wang WH, Qiu WL, Guo Z, Bi E & Xu CR. (2017). A single-cell transcriptomic analysis reveals precise pathways and regulatory mechanisms underlying hepatoblast differentiation. Hepatology , 66, 1387-1401. PMID: 28681484 DOI.
- ↑ 8.0 8.1 Vestentoft PS, Jelnes P, Hopkinson BM, Vainer B, Møllgård K, Quistorff B & Bisgaard HC. (2011). Three-dimensional reconstructions of intrahepatic bile duct tubulogenesis in human liver. BMC Dev. Biol. , 11, 56. PMID: 21943389 DOI.
- ↑ Wandzioch E & Zaret KS. (2009). Dynamic signaling network for the specification of embryonic pancreas and liver progenitors. Science , 324, 1707-10. PMID: 19556507 DOI.
- ↑ 10.0 10.1 Zorn AM. (2008). Liver development. , , . PMID: 20614624 DOI. | online extract | PDF
- ↑ Godlewski G, Gaubert-Cristol R, Rouy S & Prudhomme M. (1997). Liver development in the rat and in man during the embryonic period (Carnegie stages 11-23). Microsc. Res. Tech. , 39, 314-27. PMID: 9407542 <314::AID-JEMT2>3.0.CO;2-H DOI.
- ↑ Godlewski G, Gaubert-Cristol R, Rouy S & Prudhomme M. (1997). Liver development in the rat during the embryonic period (Carnegie stages 15-23). Acta Anat (Basel) , 160, 172-8. PMID: 9718390
- ↑ Lhuaire M, Tonnelet R, Renard Y, Piardi T, Sommacale D, Duparc F, Braun M & Labrousse M. (2015). Developmental anatomy of the liver from computerized three-dimensional reconstructions of four human embryos (from Carnegie stage 14 to 23). Ann. Anat. , 200, 105-13. PMID: 25866917 DOI.
- ↑ Archie JG, Collins JS & Lebel RR. (2006). Quantitative standards for fetal and neonatal autopsy. Am. J. Clin. Pathol. , 126, 256-65. PMID: 16891202 DOI.
- ↑ Pauwelyn K, Roelandt P, Notelaers T, Sancho-Bru P, Fevery J & Verfaillie CM. (2011). Culture of mouse embryonic stem cells with serum but without exogenous growth factors is sufficient to generate functional hepatocyte-like cells. PLoS ONE , 6, e23096. PMID: 21829697 DOI.
- ↑ Kaufman and Bard, The Anatomical Basis of Mouse Development 1999 Academic Press
- ↑ Tzur G, Israel A, Levy A, Benjamin H, Meiri E, Shufaro Y, Meir K, Khvalevsky E, Spector Y, Rojansky N, Bentwich Z, Reubinoff BE & Galun E. (2009). Comprehensive gene and microRNA expression profiling reveals a role for microRNAs in human liver development. PLoS ONE , 4, e7511. PMID: 19841744 DOI.
- ↑ Crawford JM. (2002). Development of the intrahepatic biliary tree. Semin. Liver Dis. , 22, 213-26. PMID: 12360416 DOI.
- ↑ Collardeau-Frachon S & Scoazec JY. (2008). Vascular development and differentiation during human liver organogenesis. Anat Rec (Hoboken) , 291, 614-27. PMID: 18484606 DOI.
- ↑ Naito M, Hasegawa G, Ebe Y & Yamamoto T. (2004). Differentiation and function of Kupffer cells. Med Electron Microsc , 37, 16-28. PMID: 15057601 DOI.
- ↑ Gordon S & Plüddemann A. (2017). Tissue macrophages: heterogeneity and functions. BMC Biol. , 15, 53. PMID: 28662662 DOI.
- ↑ Mass E, Ballesteros I, Farlik M, Halbritter F, Günther P, Crozet L, Jacome-Galarza CE, Händler K, Klughammer J, Kobayashi Y, Gomez-Perdiguero E, Schultze JL, Beyer M, Bock C & Geissmann F. (2016). Specification of tissue-resident macrophages during organogenesis. Science , 353, . PMID: 27492475 DOI.
- ↑ Sharma R, Greenhough S, Medine CN & Hay DC. (2010). Three-dimensional culture of human embryonic stem cell derived hepatic endoderm and its role in bioartificial liver construction. J. Biomed. Biotechnol. , 2010, 236147. PMID: 20169088 DOI.
- ↑ Leombroni M, Buca D, Celentano C, Liberati M, Bascietto F, Gustapane S, Marrone L, Manzoli L, Rizzo G & D'Antonio F. (2017). Outcomes associated with fetal hepatobiliary cysts: systematic review and meta-analysis. Ultrasound Obstet Gynecol , 50, 167-174. PMID: 27553859 DOI.
- ↑ Kelly C & Pericleous M. (2018). Pregnancy-associated liver disease: a curriculum-based review. Frontline Gastroenterol , 9, 170-174. PMID: 30046419 DOI.
- ↑ Bicocca MJ, Sperling JD & Chauhan SP. (2018). Intrahepatic cholestasis of pregnancy: Review of six national and regional guidelines. Eur. J. Obstet. Gynecol. Reprod. Biol. , 231, 180-187. PMID: 30396107 DOI.
- ↑ Wood AM, Livingston EG, Hughes BL & Kuller JA. (2018). Intrahepatic Cholestasis of Pregnancy: A Review of Diagnosis and Management. Obstet Gynecol Surv , 73, 103-109. PMID: 29480924 DOI.
Books
Zorn AM. (2008). Liver development. , , . PMID: 20614624 DOI. | online extract | PDF
Reviews
Schulze RJ, Schott MB, Casey CA, Tuma PL & McNiven MA. (2019). The cell biology of the hepatocyte: A membrane trafficking machine. J. Cell Biol. , 218, 2096-2112. PMID: 31201265 DOI.
Kelly C & Pericleous M. (2018). Pregnancy-associated liver disease: a curriculum-based review. Frontline Gastroenterol , 9, 170-174. PMID: 30046419 DOI.
Zaret KS. (2016). From Endoderm to Liver Bud: Paradigms of Cell Type Specification and Tissue Morphogenesis. Curr. Top. Dev. Biol. , 117, 647-69. PMID: 26970006 DOI.
Gordillo M, Evans T & Gouon-Evans V. (2015). Orchestrating liver development. Development , 142, 2094-108. PMID: 26081571 DOI.
Zong Y & Stanger BZ. (2012). Molecular mechanisms of liver and bile duct development. Wiley Interdiscip Rev Dev Biol , 1, 643-55. PMID: 23799566 DOI.
Dixon LJ, Barnes M, Tang H, Pritchard MT & Nagy LE. (2013). Kupffer cells in the liver. Compr Physiol , 3, 785-97. PMID: 23720329 DOI.
Nakamura T, Sakai K, Nakamura T & Matsumoto K. (2011). Hepatocyte growth factor twenty years on: Much more than a growth factor. J. Gastroenterol. Hepatol. , 26 Suppl 1, 188-202. PMID: 21199531 DOI.
Ando H. (2010). Embryology of the biliary tract. Dig Surg , 27, 87-9. PMID: 20551648 DOI.
Kung JW, Currie IS, Forbes SJ & Ross JA. (2010). Liver development, regeneration, and carcinogenesis. J. Biomed. Biotechnol. , 2010, 984248. PMID: 20169172 DOI.
Si-Tayeb K, Lemaigre FP & Duncan SA. (2010). Organogenesis and development of the liver. Dev. Cell , 18, 175-89. PMID: 20159590 DOI.
Le Lay J & Kaestner KH. (2010). The Fox genes in the liver: from organogenesis to functional integration. Physiol. Rev. , 90, 1-22. PMID: 20086072 DOI.
Roskams T & Desmet V. (2008). Embryology of extra- and intrahepatic bile ducts, the ductal plate. Anat Rec (Hoboken) , 291, 628-35. PMID: 18484608 DOI.
Articles
Szpinda M, Paruszewska-Achtel M, Woźniak A, Mila-Kierzenkowska C, Elminowska-Wenda G, Dombek M, Szpinda A & Badura M. (2015). Volumetric Growth of the Liver in the Human Fetus: An Anatomical, Hydrostatic, and Statistical Study. Biomed Res Int , 2015, 858162. PMID: 26413551 DOI.
Cardinale V, Wang Y, Carpino G, Mendel G, Alpini G, Gaudio E, Reid LM & Alvaro D. (2012). The biliary tree--a reservoir of multipotent stem cells. Nat Rev Gastroenterol Hepatol , 9, 231-40. PMID: 22371217 DOI.
Friedman JR & Kaestner KH. (2011). On the origin of the liver. J. Clin. Invest. , 121, 4630-3. PMID: 22105167 DOI.
Carpentier R, Suñer RE, van Hul N, Kopp JL, Beaudry JB, Cordi S, Antoniou A, Raynaud P, Lepreux S, Jacquemin P, Leclercq IA, Sander M & Lemaigre FP. (2011). Embryonic ductal plate cells give rise to cholangiocytes, periportal hepatocytes, and adult liver progenitor cells. Gastroenterology , 141, 1432-8, 1438.e1-4. PMID: 21708104 DOI.
Collardeau-Frachon S & Scoazec JY. (2008). Vascular development and differentiation during human liver organogenesis. Anat Rec (Hoboken) , 291, 614-27. PMID: 18484606 DOI.
Rudolph AM. (1983). Hepatic and ductus venosus blood flows during fetal life. Hepatology , 3, 254-8. PMID: 6832717
Historic
Mall FP. A study of the structural unit of the liver. (1906) Amer. J Anat. 5:227-308.
Severn CB. A morphological study of the development of the human liver. I. Development of the hepatic diverticulum. (1971) Amer. J Anat. 131: 133-158. PMID 5575887
Severn CB. A morphological study of the development of the human liver. II. Establishment of liver parenchyma, extrahepatic ducts and associated venous channels. (1972) Amer. J Anat. 133: 85-107. PMID 5008885
Search Pubmed
Search Bookshelf Liver Development
Search Pubmed Now: Liver Development | Embryonic Liver Development
Additional Images
Historic
Mall FP. A study of the structural unit of the liver. (1906) Amer. J Anat. 5:227-308.
Terms
| Gastrointestinal Tract Terms | ||
|---|---|---|
| ||
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 10) Embryology Gastrointestinal Tract - Liver Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Gastrointestinal_Tract_-_Liver_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G