Paper - A Human Embryo of Twenty-five Somites
| Embryology - 17 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
West CM. A human embryo of twenty-five somites. (1937) J. Anat., 71(2): 169-200.1. PMID 17104635
| Online Editor Note | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| This historic 1937 paper by West describes an embryo at 25 somite stage, would be similar to a Carnegie stage 12 (26 - 30 days), caudal neuropore closes, Somite Number 21-29.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
A Human Embryo of Twenty-five Somites
Cecil M. West
University College, Cardiff
Introduction
THE specimen about to be described was kindly given to me by Dr Jethro Gough, of the Pathological Department, Welsh National School of Medicine. It was obtained at a post-mortem examination of a woman, aged 39, who died of a strangulated hernia. The post-mortem examination was made 47 hours afterdeath; thereis,however, little probability that the material had suffered much from this lapse of time, for the body was kept in an efficient refrigerator, which fact, combined with the season of the year, January, lessens the likelihood of decomposition having set in,and Dr Goughtelsme that he noticed no evidence of this when making the autopsy.
Technique Employed on Material Received
The material received from Dr Gough consisted of the uterus, ovaries and tubes. The uterus had been opened in front along the middle line, and there was thus exposed on the anterior wall and near the left cornu a chorionic vesicle, which was unopened.
There was a corpus luteum in the left ovary, which had been opened, and a hydatid cyst was present near the ovarian fimbria of the left side. The whole specimen was placed in 4 per cent formalin for a few days, and then the chorionic vesicle was removed from the uterus and opened, and an apparently normal embryo was exposed.
The embryo, enclosed in the amnion, was lying on its left side; the yolk stalk passed out towards the right and the body stalk towards the left; the tail of the embryo was twisted towards the right.
The amnion was then removed and the embryo, with the body stalk and the adjacent part of the chorion, was embedded in paraffin and cut into sections of 10,um thickness, which were stained on the slides with Ehrlich's haematoxylin and orange G.
Every section was then photographed on to bromide paper at a magnifica- tion of x 100, and many reconstructions, both graphic and of wax, were made of the embryo as a whole and of its various regions.
In order to obtain the correct orientation for the reconstructions the following method was adopted: the sections had been fixed to the slides with a solution of chromatized gelatin, and after the removal of the paraffin with xylol, the sections were stained and it was found that the edges of the paraffin block could be easily identified as they had slightly taken up the haematoxylin stain; provided the paraffin block has been trimmed square, a satisfactory method of orientation is thus easily accessible; in addition to this method use was made of photographs which had been taken of the embryo before, and after, its removal from the chorionic sac.
Age of the Specimen
There was no menstrual history, so that in estimating the age comparison must be made with embryos for which more definite data are available. Recent work, in Baltimore, by Streeter (1932, 1933), Hartman and their colleagues, has made itpossible to determine the true ovulation age and rate of development in embryos of the monkey, Macacus rhesus; and while the two genera-man and monkey-differ in form and in rate of differentiation and growth, it has nevertheless been found that such differences do not become appreciable until the end of the second month; so that, for the first 6 weeks, the known ages of Macacus rhesus may be transferred to human embryos of corresponding developmental stages. Streeter has thus estimated the ovulation ages of certain well-known human embryos, and has found that the first somites make their appearance on the 22nd day and that differentiation of the somites occurs rapidly, 1, 5 and 8 somites being found on the same day; a 6.5mm. embryo,with triangular fin-like arm buds and beginning leg buds was obtained on the 27th day.
The present specimen has 25 somites and very early limb buds and may thus be between the 22nd and 27th days; taking into account the rapidity of development at this stage, an ovulation age of 24 days would, I believe, be a fair estimate for this specimen.
From published data on menstrual history it is possible to work out the ovulation age of certain other embryos which resemble, in general, the present specimen; for example, embryos described by Atwell, 17 somites (1930), Watt, 17-19 somites (1915), Davis, 20 somites (1923), Girgis, 22 somites (1926), Johnson, 24 somites (1917), Waterston, 27 somites (1914). Basing the calculation of the ovulation age on the assumption that ovulation has occurred on the 14th day after the commencement of the last menstrual period, I find that this series of embryos covers a range of ages from 19 to 82 days, and of de- velopmental stages from 17 to 27 somites; such ages do not fit in exactly with Streeter's findings, but they are based on menstrual histories and on the as- sumption that menstruation has been regular and that ovulation has occurred on the 14th day after the commencement of the last period. Whilst some observers would fix a more or less hard and fast date for ovulation, others would consider the date to be more variable; for example, Hain (1934) has shown in an individual in whom intermenstrual bleeding was recorded for a period of over 2 years that,if such intermenstrual bleeding may beconsidered indicative of ovulation, then ovulation may occur as late as, and after, the 20th day from the beginning of the previous period, and that there may be more than one ovulation in a cycle; there may thus be great variation in ovulation ages as estimated from menstrual histories, and this emphasizes the importance of Streeter's and Hartman's work.
The Uterus and Embryonic Membranes
The uterus, after fixation, measured 96 mm. from fundus to external os, and 67 mm. from side to side at the widest part of the body; the walls were 12mm. thick.
The chorionic vesicle was removed intact from the uterus and was covered completely with vili; itwas rather flattened and measured 18 x 15 mm.
The part of the chorion, at the attachment of the body stalk, that was cut out and sectioned with the embryo was carefully examined; the general appearance is very like that shown in Grosser's Fig. 107, p. 133, in Keibel & Mall (1910), of a section through the chorion of an aborted ovum of 1 month. Each villus and its branches are usually covered with two layers of cells: an outer layer in which cel boundaries are not well defined, and in which the nuclei are flattened, loosely spaced and often faintly stained; and an inner layer in which celboundaries are defined, and in which the nuclei are round, closely packed and deeply stained; these two layers of the chorionic epithelium are the syncytiotrophoblast and cytotrophoblast respectively. Here and there it is quite impossible to make out any cellular structure whatever in the syncytiotrophoblast, whilst in some places the nuclei of this layer can be faintly discerned.
When the final ramifications of a villus are reached it is found that the syncytiotrophoblast fades away or disintegrates, and the cytotrophoblast becomes spread out in a sheet so that the cells of which it is composed are seen in surface view instead of in section; the cels, too, appear to increase in size and their boundaries become every sharp,and the sheet of cells forms a beautiful mosaic. When the cytotrophoblast oft wo adjacent vili thus becomes spread out into sheets the syncytiotrophoblast dips in between the sheets and gives the appearance of being an intrusion (PI.I,fig.4); similarly a part of the intervillous space comes to be included in the sheet and contains maternal blood and sometimes some fibrinous material. These intrusions into the sheet are composed of masses of protoplasm without any cell boundaries and with small, numerous and deeply stained nuclei which stand out very prominently front the lightly stained and well-defined cells of the trophoblast sheet; these sheets are the "cell islands" of Grosser. But there are to be found in the sheets and in the intervillous spaces other protoplasmic masses of an appearance quite different from that of the intrusions mentioned above; these are small masses of protoplasm, of any shape, homogeneous in appearance, rather opalescent and stained slightly pink, with crowds of brightly stained nuclei arranged usually in a circle to rinarosette (PI.I,fig.4); these are the "giant cells" of Grosser, and they are either protrusions or actually detached portions of syncytiotrophoblast, and they seem to be very similar to the "proliferation nodes" to
which also Grosser refers.
The mesodermal core of the vili has undergone some shrinkage from the overlying trophoblast; it contains the subdivisions of the umbilical vessels, and is composed of a loose tissue, the cells of which are of an irregular polygonal or stellate shape with round, oval or spindle-shaped nuclei; there are a few ofthe large celsofHofbauer, but they are not at all numerous.
Teacher (1924), in his account of the embryo T.B. 2, describes a condition which is just like what I find in this specimen; he writes, p. 172, that the syncytium "ends a little beyond the tip of the villus leaving the cytotrophoblast uncovered, and the latterfuses with that ofneighbouring viliforming a complex structure tunnelled here and there by passages for the maternal blood". Coventry (1923), in an account of the placenta of the Guinea baboon at the 4th month, writes of trophodermic cel islands being completely covered with a layer of syncytium continuous with that of the villus to which they are attached; comparing this with the condition in the human subject, he states that the trophoblastic masses in man are not covered with a continuous layer of syncytium, but that it is usual to find syncytial masses and buds scattered over the surface of the masses, and he suggests that the syncytium is at first stretched out over the developing cell mass and then, its elasticity failing, it becomes broken up into patches; this is just the impression that I get from a study of this specimen, and it seems to correspond with the condition described by Johnson (1917) in an embryo of 24 somites. The line of attachment of the amnion to the body wall follows closely the ventral borders of the two umbilical veins, and where these enter the heart the lines of reflection join just caudal to the heart and cranial to the yolk stalk, with the result that the heart is included within the amniotic cavity and the yolk stalk is not; the condition is very similar to that shown by Politzer & Sternberg (1930) in an embryo of 2-7mm. in length and with 25 somites.The amnion covers the caudal aspect of the body stalk and is just beginning to encroach on the ventral aspect; it is most closely applied to the body of the embryo in the forebrain and tail regions, whereas it stands out at some distance from the rest of the brain and from the dorsal surface of the body, though it does follow to a slight extent the dorsal concavity of the body.
Filmy strands passed from the outer surface of the amnion and of the yolk sac to the inner surface of the chorion, but they could be removed from both structures without causing any damage. I am unable to describe the structure of these strands, but they seem to be a fairly common feature in young chorionic vesicles and are, presumably, remains of the extra-embryonic mesoderm which have persisted after the formation of the extra-embryonic coelom.
The amnion is a very thin bilaminar structure and, since most of it was removed before the embryo was sectioned, it can be studied microscopically only at the cut edges of the lines of reflection; at such places the two layers of which it is composed can be seen well; near the line of reflection between heart and yolk stalk the amnion is a little thicker than elsewhere, as was described by Johnson (1917), and here it is possible to recognize a small amount of mesenchyme between the true mesodermal and ectodermal layers.
The Embryo
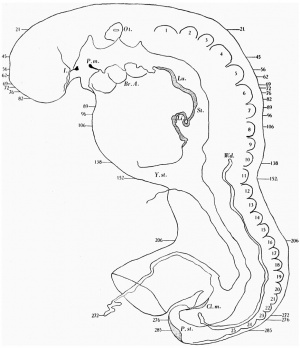
External form. The embryo was cut into 297 sections, each 10 ,u thick, and since the sections are almost horizontal, the embryo measures nearly 3mm. in length.
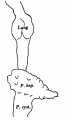
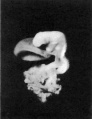
The general form of the embryo can be estimated best from the photographs (PI. I, figs. 1 and 2) and a lengthy description will therefore not be given, but the following points may be noticed: the heart is prominent and causes a swelling larger on the right than on the left, as has been noted by other observers in specimens of about this stage; the roof of the hindbrain is already thin and was sufficiently transparent to allow the neuromeres to be seen through it; there is a concavity of the dorsal body wall just caudal to the attachment of the yolk stalk, and herein lies the main difference between this specimen and van den Broek's (1911) embryo A, and an embryo with 25 somites referred to by Politzer & Sternberg (1930), both of which are bent in a gentle and continuous curve; the ventral body wall is widely deficient from the caudal margin of the heart to the cranial border of the body stalk, and the gut, suspended by its mesentery and with the yolk stalk opening from it, is thus clearly visible between the rounded elevations of the umbilical veins, just as described by Politzer & Sternberg; a very slight rounded elevation marks the position of the forelimb bud, and there is just a suspicion of a hindlimb bud. Various theories have been given to account for the dorsal concavity of the body; Bartelmez & Evans (1926) suggest collapse of the yolk sac during dehydration as a possible cause; Atwell (1930) suggests factors in operation during life, such as turgidity of the yolk sac, rapid increase in growth of the two ends of the body and the difficulty in accommodating both heart and yolk sac on the ventral side of the body without there being some bending. There is no doubt that these curves do appear opposite the attachment of the yolk sac, and they do tend to be more marked in the earlier stages when the yolk sac is relatively large and the body wall frail and slender; a dorsal concavity is present, for example, in nearly al the embryos, of from 1 to 16 somites, described by Bartelmez & Evans (1926), in Wilson's (1914) embryo[1] H 98, of 1 -27 mm., in Ingalls' (1920) embryo of 1 - 38 mm., in Dandy's (1910) embryo of 2 mm.; there is a slight concavity in Atwell's (1930) embryo of 2 - 81 mm., in Watt's (1915) embryo of 3.35 mm.; it is very slight in an embryo of 5 mm. described by Politzer & Sternberg (1930), while it is absent in embryos of 2-7, 4,6,7mm. described by the same authors, and in Thompson's (1907) embryo of 2-5mm. I believe that in the present specimen the concavity was developed as a result of factors in operation during life, for it could be seen as soon as the chorionic sac was opened and when the embryo had been subjected to no interference other than fixation in 4 per cent formalin.
I believe that such dorsal curvatures must be regarded as a normal variation in early developmental stages.
The specimens which this embryo resembles most closely are Thompson's (1907) embryo of 2-5 mm. length and with 23 somites, Politzer & Sternberg's (1930) embryo of 2-7 mm. length and with 25 somites, van den Broek's (1911) embryo A with 28 somites, Broman's (1896) embryo Lf of 3 mm. length and with 30 somites (this last specimen is described as No. 11, in Keibel and Elze's Normentafeln, 1908). The present specimen differs from most others of about the same stage in the greater development of the head, the greater length of the hindbrain, and the more definite neck bend.
Somites
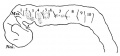
There are 25 somites on the left side and 24 on the right, and beyond these, two additional somites are in process of differentiation on each side of the body, but are not isolated from each other (Text-fig. 1).
The 1st somite on each side is indefinite and merges into the general mesenchyme of the head, and it is situated at the level of the summit of the 8th neuromere of the hindbrain. The division of the somites into occipital, cervical, thoracic, etc., has been a matter of some difficulty to me, as itappears to have been to others, for in looking through the literature one finds the number of somites, described as occipital, varying from 2 to 5, and the allocation of certain somites to certain regions seems to have been made without any very obvious criteria having been employed. As long ago as 1901, Bardeen & Lewis pointed out (p. 10) that "the divisions of the myotomes into occipital, cervical, thoracic, lumbar, sacral and coccygeal groups depends upon the nerves and skeletal structures related to the body segments in which the myotomes lie", and "before the spinal nerves have appeared they (the occipital myotomes) cannot with certainty be distinguished"; and (p. 11) "the most caudal myotome lying opposite the arm bud may, therefore, be taken to represent the first thoracic segment". Bartelmez & Evans (1926) have shown that the relative distance between the first pair of somites and the otic plate gradually increases as a result of growth of the intervening region.That there appears to be a caudal migration of the first somite relative to the neuromeres is evident if one notices the position of the first somite in embryos of gradually increasing age, thus:
| Embryo | Somites | Neuromeres | Level of first somite |
| Corner (1929) | 10 | 5 | Slightly caudal to 5 |
| Heuser (1930) | 14 | 5 | Just caudal to vagus, or 7 |
| Atwell (1930) | 17 | 7 | At level of 7 |
| Watt, VI (1915) | 17-19 | 9 | Between 7 and 8 |
| Davis (1923) | 20 | 0 | Between 7 and 8 |
| Girgis (1926) | 22 | 6 | Well caudal of 6 |
| Thompson (1907) | 23 | 7 | Just caudal to 7 |
| Johnson (1917) | 24 | 9? | Crest of 8 |
| Present specimen | 25 | 10 | Crest of 8 |
| Waterston (1914) | 27 | 7 | Well caudal of 7 |
Johnson (1917) describes the position of the 1st somite, at the crest of the 8th rhombomere, as being that which he would expect to find occupied by the 2nd somite from his studies of young embryos of pig, sheep, cat, and of a slightly older human embryo; and from the descriptions of Butcher (1929) for therat, Atwell (1930), Davis (1923), Heuser (1930), Johnson (1917), Low (1908), Politzer (1928a), Thompson (1907) and Watt (1915), for man, it appears that (a) the 1st somite is always small; (b) its cells are sometimes radially arranged; (c) it is often rudimentary; (d) it may show signs of degeneration; (e)itmay show evidence of a reversal in development; (f) there is a progressive development of the somites from the tail up to the 4th somite and from there a decrease to the 1st.
In the present specimen the 1st somite is small, its cells are radially arranged, it is rudimentary, it shows signs of degeneration, and the 4th somite is the largest; there is, moreover, a tendency towards continuity and similarity of development between the first three somites which is not shared by the remainder, a fact which might be urged in favour of including these three as occipital; the swelling for the forelimb bud is just recognizable, and is found to extend over somites 8-12.
Here, then, is strong evidence in favour of considering, at least in this specimen, the first three somites as being occipital; their common characteristics, their small size, and the relation which they bear to the developing fore-limb; the 4th somite in the present specimen is, therefore, considered as being the 1st cervical.
The fullest account of the structure of the somites at about this period has been given by Watt (1915), and, with certain modifications, the present specimen conforms with his description.
The dorsal edge of the cortex of the somite is turned in, to form the myo- tome, more than the ventral edge; differentiation between sclerotome and myotome is seen best in the more caudal somites, where the sclerotome cells stream out from the somite in a ventromedial direction; one would expect those sclerotomes which are destined to form vertebrae to have an appearance rather different from those which are to contribute towards the formation of the basis cranii, and one finds that in the more cranial somites the ventral edge of the somite and the dorsal edge have become so folded over that the myocoele is reduced to a mere slit (an appearance due partly to the plane of section), which communicates only through a narrow cleft with the main cavity of the somite, whilst ventromedial to the somite cavity are the celswhich form the sclerotome, though there is nothing except their position to lead to their identification. It is in the 4th somite that one begins to see a clear indication of the differentiation of the sclerotome, owing, I believe, to the fact that this is the first somite of which the sclerotome comes to embrace the spinal cord, and of which the sclerotome cells spread medialwards as well as ventralwards,whereas in the case of the more cranial somites the presence of the hind brain precludes such a medial extension; but since a single vertebra is derived from parts of two adjacent sclerotomes, the 4th presumably takes a share in the formation of the basis cranii as well as of the atlas vertebra, which is perhaps what Thompson (1907) had in mind when he stated (p. 164) that the first three, or three and a half, somites might be considered as occipital.
As in Gage's (1905) embryo of 3 weeks, intersomitic grooves on the side of the head are well marked and help in the identification of the first three somites.
An external limiting membrane to the cells of the dermatome is not generally recognizable; but in some sections a separation of the ectoderm from the underlying tissue has occurred and in these sections the outer edge of the dermatome has a very definite and clean cut border which looks very like a limiting membrane.
There is not the same intimate association between the medial surface of the somite and the neural tube as isdescribed by Watt, but the two structures are separated, especially as regards the more anterior somites, by loose mesenchyme, by the migrating celsofthe neural crest and by a longitudinal vessel which is specially large in the 10th, 11th and 12th intersegmental regions and which is formed by a union of the dorsal divisions of lateral intersegmental branches of the aorta; more caudally the association between somite and neural tube is closer, since the cels of the neural crest are here interposed between the neural tube and the dorsal angle, rather than the medial surface, of the somite, and the vessel referred to above cannot be traced beyond the 15th somite.
Limb Buds
The stage of development of the limb buds resembles closely that of Mall's embryo XII, of which, however, Bardeen & Lewis (1901) state,"There are no external visible signs of limb buds", which was, indeed, my first conclusion in the present case. Politzer & Sternberg (1930) give a figure(9d) of a section The forelimb bud is in a very early stage of development, and it shows as a small rounded elevation caudal and dorsal to the prominence of the heart. In the sections, where I was able to recognize it only after prolonged and careful examination, it shows as a thickening of the somatic mesoderm, opposite about the 8th to the 12th somites and just to the lateral side of the dorsal angle of the embryonic coelom, but no definite boundaries can be given for its extent. It is recognizable by the following characteristics: the mesoderm is thickened; the cells of which it is composed resemble those of a sclerotome, having clear-cut rounded nuclei rather than the irregular and more indefinite nuclei of the mesenchyme cells, and being deeply stained; the cels are packed more closely than those in the loose mesenchyme; there is a definite thickening of the ectoderm over the limb bud swelling, as described by Gage (1905) in a specimen which showed the limb buds in a more advanced stage; there is an abrupt line of demarcation between the cels of the limb bud and those of the through an embryo of 2-7mm. in length,which is very like a section through the arm bud in my embryo. somite, even though, at the widest part of the somite, the two structures are in close apposition.
With regard to a contribution from cels of the myotome towards the limb bud, there is a close resemblance between this specimen and embryo XII, 241 mm. in length, belonging to Dr Mall's collection; of this embryo W. H. Lewis (1902) states (p. 149): "The origin of the cells which cause this swelling I am notable to determine, though there are suspicious looking processes from the myotome". Zechel (1924), referring to Grosser's embryo Kl32, of 3-4 mm. length, writes of an out wandering of cells from the lateral plate of the myotome into the anlage of the upper limb.
Lewis, in spite of the " suspicious-looking processes ", shows, in his Fig. 2, a quite definite separation between "arm bud" and "8th cervical muscle plate"; this is just what I find in the present specimen, and I have the impression that there is, in this specimen, no contribution from the myotome towards the limb bud.
The posterior limb bud is even more difficult to identify than the anterior; there is a symmetrical swelling on each side of the body at the level of the last two somites (lumbar 1 and 2) and it extends over the succeeding unsegmented mesoderm. This swelling is recognizable in the photographs (PI. I, figs. 1 and 2) and is, presumably, the Wolffian ridge; there is nothing particularly characteristic about any part to fit by which it may be identified as a limb bud, but on the other hand it does appear to have a definite relationship to the Wolffian duct, for at this point the duct comes close to the surface and its course is exactly marked out by the groove which forms the ventral limit of the swelling; Bremer (1906) shows a rather similar swelling in his 4 mm. embryo, but states (p. 459), "There is no trace of posterior limb", in his figures the Wolffian duct shows a similar relationship to the swelling to what it does in my specimen.
Alimentary Tract
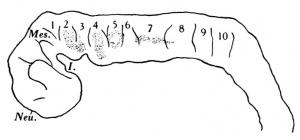
The pharynx is open anteriorly, for the pharyngeal membrane has ruptured and its remains can be seen (Text-fig. 1) adhering to the ventral and dorsal walls.There are three pharyngeal pouches on each side , all of which reach the ectoderm, and there are three bronchial grooves on the exterior.
The first two pouches on each side have well developed dorsal diverticula and much smaller ventral diverticula.
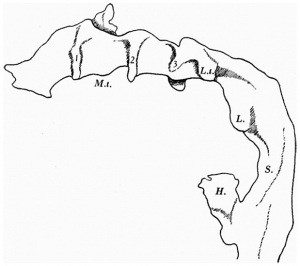
The pharynx, as a whole (Text-fig. 2), is concave dorsally from side to side on its dorsal surface and concave ventrally on its ventral surface.
The floor is marked in its most anterior part by a deep median groove, and a much shallower groove likewise marks the dorsal wall, or roof. At the point of attachment of the pharyngeal membrane the groove on the roof of the pharynx is continued on to the roof of the mouth, and there it divides into two limbs with a rounded ridge between them; this ridge is due to the down growth of the forebrain and emphasises the close relationship between brain and pharynx at this point; just to the cranial side of the pharyngeal membrane the pouch of Rathke shows as a small diverticulum of the mouth cavity; Seessel's pouch cannot be identified.
The deep median groove on the floor of the pharynx also is prolonged forwards to the mouth; it is from this groove that one would expect to find the outgrowth of the median thyroid rudiment, and there is to be seen, in four sections, just at the junction of the first and second bronchial arches, a small outgrowth from the pharyngeal floor towards the ventral aorta; I believe that this is the median thyroid rudiment, though it is smaller than in other embryos of about the same age.
In the section just before the deepest part of the outgrowth of the thyroid there is the beginning of a very curious structure which consists of a hollow tubular evagination of the pharyngeal floor projecting freely into the lumen of the pharynx in a cranial direction and covering a distance of seven sections.
This structure begins on the pharyngeal floor as a slight ridge which then becomes pinched off from the floor so that it becomes a tube lying free in the lumen of the pharynx.
I thought at first that it might be an internal gil filament. Since Grosser (1912) first described them, such structures have been seen in embryos of about this age, and younger, by Heuser (1930), Atwell (1930), Veit & Esch (1922), Politzer (1928a), Davis (1923), and Girgis (1926). In all these cases, however, there are merely proliferations or ridges of the entoderm and not long, freely projecting structures such as I have described; furthermore, the correctness of Grosser's interpretation has been questioned by Boyden (1918), who considered that the structures described by the above observers are more comparable with the outpocketings which occur in the pre-auditory region of the chick, and that they probably are not rudimentary internal gils (a comment with which He user appears to agree) for the following reasons: gils are essentially ectodermal derivatives; gils are least frequently developed in association with the first pouches in water breathing vertebrates; the embryonic stages at which these structures have been observed are too early, for al the pouches have not yet reached the ectoderm.The truth of thes statement that gilsare essentially ectodermal derivatives is,I believe, stilopen to question. These rudimentary structures (they are not present in my embryo) seem always to grow from the ventral wall, and always occur near the closing membrane of the first pouch; the structure that I am describing in the present embryo grows from the ventral wall also, but is attached close to the middle line and as far away as it can be from the first closing membrane; it thus seems very unlikely that it is a rudimentary internal gil, though it does appear as if, during life, it had been waving about freely in the pharyngeal lumen an ideal position for a gil filament-but there is no evidence oxblood cels in its lumen but only a few mesoderm cels. Its close relationship to the thyroid led me to consider the possibility of its association with an endostyle, but this, I believe, is out of the question; in short, I am unable to offer any interpretation of this structure.
Attention has been called to the small size of the median thyroid rudiment; the lateral rudiments, on the other hand, show very well, and the condition is very like that described and figured by Weller (1933), in an embryo 4 mm. long. It was not until I had read Weller's paper that I realised the significance of what I at first took to be 4th pouches, though I had been surprised at their size compared with the 3rd.
The lateral thyroid rudiment shows (Text-fig.2) as a rather large outgrowth from the pharynx just to the caudal side of the small 3rd pouch; the cavity of the pharynx is continued into the outgrowth and round its margin the epithelium is thickened. It may be possible that the small size of the median rudiment is correlated with the greater development of the lateral rudiment.
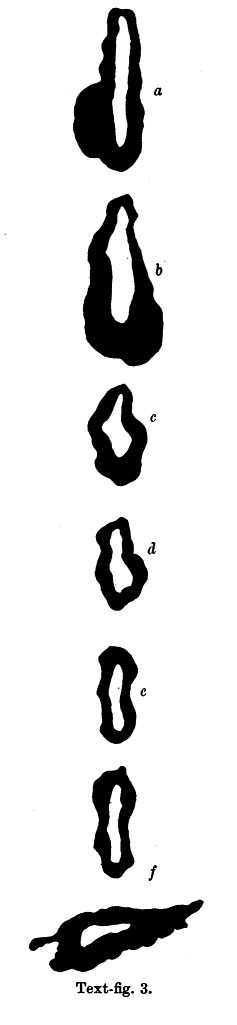
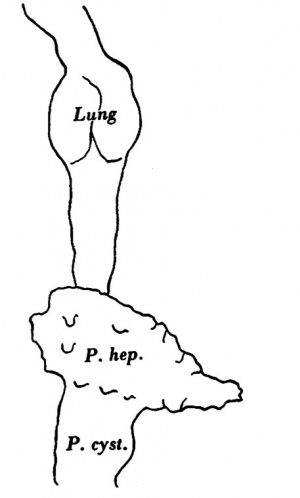
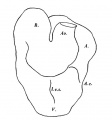
The part of the alimentary tract that succeeds the pharynx has been a source of difficulty in interpretation to several observers; in the present specimen it shows, at first, a slit-like lumen and walls of uniform thickness, but, after eight sections, there appears a rather spherical outgrowth on the right side (Text-fig. 3a) and, after a few more sections, there is a corresponding dilatation on the left; the difference in level of the two outgrowths is due to a slight obliquity of the sections through this part of the gut. These two out-growths, which I will call the cranial swellings, arise from the ventral part of the gut,whereas the dorsal part of the gut has a slit-like lumen and is separated from the ventral expanded part by a slight constriction; beyond this cranial swelling the gut shows a lumen which is suddenly much reduced in ventro- dorsal diameter, being the continuation merely of the slit-like dorsal part of the more cranial portion of the gut; farther on the gut has a pear-shaped lumen, wide ventrally where it forms what I will call the caudal swelling (Text-fig.3d), and slit-like dorsally, and it becomes continuous, in the septum transversum with the part of the gut from which the liver arises (Text-fig. 3f). The cranial swelling shows a slight indication of division into two; its walls are much thickened; it is to the cranial side of the septum transversum; it lies dorsal to the sinus venosus and atrium, just where these two cavities communicate; the swellings is greater towards the left side than to the right, and on the left side it grows towards the pericardial cavity and on the right towards the pleural passage. The caudal swelling ismuch smaller; itshows no evidence of duplicity; it is close to the hepatic diverticulum, and it shows no thickening of the ventral wall, which is, indeed, rather thinner than the dorsal wall. I show (Text-fig.3) a series of outlines of sections of the gut at different levels, which may be compared with similar figures by van den Broek, and by Girgis and others, and from which it will be seen that I am in disagreement with van den Broek and Girgis, both of whom show what they describe as lung, in the same section as the liver.
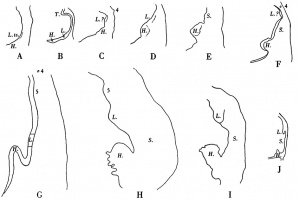
In Text-fig.4 I show a drawing of this region in the present specimen, together with tracings of the same area in the embryos described by Corner (1929), Heuser (1930), Low (1908), Davis (1923), Thompson (1907, 1908), Girgis (1926), Johnson (1917), and Waterston (1914). In the earlier stages (Corner and Heuser) lung and liver are very close to each other; in Low's specimen I have marked a doubtful swelling which I believe represents the lung bud,but from his statements I do not think that this is the groove which Low considers (p. 245) "may indicate the lung groove", but that he would place it some distance further cranialwards. In Davis' specimen liver and lung stilare in close apposition, but in the embryo described by Thompson there is a sudden increase in the distance between the two rudiments, but what Thompson interprets as stomach Grosser (1912) would call lung bud, and Davis after a study of his own specimen naturally takes the same view as Grosser; Girgis, in a 22-somite embryo, and van den Broek, in one of 28 somites, show the lung and liver rudiments so close to each other that parts of both are cut in the same section, a fact which is in keeping with Grosser's interpretation of Thompson's specimen; in Johnson's specimen, of 24 somites and in the present one, of 25 somites, there is a much larger interval between lung and liver, and I agree with the interpretation of Johnson, as far as these two rudiments are concerned, though I am not so certain about his ?5th pharyngeal pouch; in Waterston's embryo, of 27 somites, there is again a wide interval between lung and liver, and he shows a lung bud which can surely be interpreted as nothing but lung. In the earlier stages to which I have referred the lung bud is shown as being bifid at its caudal end, but such a bifid condition is not present in the more caudal swelling of Thompson's embryo, whereas it is present in the cranial swelling which Thompson calls lung bud. Furthermore, if Grosser is correct in his interpretation of the more caudal swelling as lung bud, itwould mean that this structure would come to an end right up against the dorsal surface of the liver bud, behind which it is hidden. The resemblance between Thompson's specimen and my own is so close that I find it difficult to believe that Grosser's interpretation is correct.
In some of the figures referred to above (C, F, G, H) the positions of the 4th pharyngeal pouches and of doubtful 5th pouches are indicated; in Fig. F, Grosser would interpret what I have labelled L? as 5th pouch?. In view of Weller's (1933) work, I am a little doubtful as to the correctness even of the designation of certain of the structures interpreted as 4th pouches, and more so stilwith regard to the doubtful 5th pouches; for examples I refer to the figures of Low, Thompson, Girgis, Johnson and Davis, though, owing to the absence of figures of the whole pharynx, it is not possible to be certain ast o the correctness of their interpretation.
In the present specimen the 3rd pouch is quite small and I do not believe that what I have called lung bud could be interpreted as 5th pouch, especially as I have not been able to recognize a 4th pouch.
It would thus appear that after the 20-somite stage, as exemplified by Davis'specimen,there is a rapid growth of the foregut, and liver and lung thus come to be widely separated from each other; such growth may have an influence in obliterating the dorsal concavity of the body of the embryo by allowing more room for the developing heart, and it has been noticed that this concavity does become less in later stages.
Liver
The part of the gut which succeeds the stomach shows an increase in its ventro-dorsal diameter and from it the liver arises; the liver diverticulum grows from the gut at first ventralwards and then towards the left, so that the lumen of gut and of liver diverticulum together form an L-shaped cavity; the liver then grows cranialwards and to the right,caudal wards and to the left and thus, as shown also by Thompson (1908), the liver as a whole lies obliquely across the ventral surface of the gut (Text-fig. 5).
Two very obviously different parts of the liver outgrowth can be recognised well in a model (Text-figs. 1 and 5); a large cranial portion with uneven surface extending on each side of the gut, and a small smooth caudal portion confined to the ventral surface of the gut and not extending in a lateral direction; the uneven surface of the cranial portion is due to the outgrowth of liver cords from it, and it is the "pars hepatica", of Maurer, of the diverticulum; the smoothness of the smaller caudal portion is due to the absence of liver cords, and it is the "pars cystica" of the liver outgrowth.
Liver cords grow out from the pars hepatica in a cranial and in a ventral direction, to the right and to the left, forming in all about twenty-five main stems, of which most grow ventralwards, about an equal number to the right and to the left,and least of a lina cranial direction.
In the septum transversum. the distinction between the cells of the liver cords and of the mesenchyme is quite clear, the liver cords being recognized by the large size of their cels, and, as van den Broek (1911) pointed out,by their being more darkly stained, and by their being more closely packed together than the mesenchyme cels. All the liver cords are solid outgrowths from the walls of the diverticulum and Lung there is not any extension into them from the adjacent cavity of the gut.
Bremer (1906), in his description of an embryo of 4mm., called attention to the relationship between the developing liver and the vessels in the septum transversum, and expressed the opinion that the branching of the vitelline veins was independent of the liver cords, and that the ultimate close relationship of the two structures was the inevitable result of their mutual growth. The same conditions are found in the present specimen, in which it is only at the cranial part of the liver diverticulum that vitelline and umbilical veins join, so that it is to the vitelline veins that the liver is most closely related. Without giving a tedious description of the branching of the vitelline veins,it may be said that there is one main vessel on each side which passes through the septum transversum, and that though bits of the vessel may get pinched of, such pinching off appears to be due to mesenchymal rather than to hepatic invasion, and the end result is that there is formed a complicated network of vessels, many of which come to be blood spaces completely isolated from each other and surrounded with liver cords; yet it would be untrue, I believe, to describe the liver cords as breaking up the veins, and I would rather say that though by their mutual growth liver cords and vessels do come into close contact, the cords are not the principal factor in the branching of the veins, though they may by their pressure encourage such branching.
Yolk Sac and Hindgut
Caudal to the liver bud the ventrodorsal diameter of the gut becomes reduced and, as the opening into the yolk sac is approached, it is restricted also from side to side; the wall of the gut remains thick in its dorsal part but ventralwards becomes much thinner, and it is this thin portion which is continued into the yolk sac, so that there is a very striking difference between the thick dorsal part of the gut on the one hand and the thin ventral part and yolk sac wall on the other. This is similar to the condition described by Johnson (1917), but different from that illustrated by Davis (1923) and Politzer (1928a).
The yolk stalk can be seen very well in the photographs, leading out from the widely open ventral body wall; the yolk sac has collapsed on three sides, and the blood islands in its wall make knob-like projections on its surface. Caudal to the opening of the yolk sac the cavity of the gut becomes gradually reduced but retains a pear-shaped outline with the wider end directed entral-wards, until, after about 75 sections, it opens into the cloaca.
Cloaca and Allantois
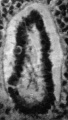
The cloaca, with the allantois arising from its cranial and ventral part, is shown in Fig. 1. The allantois follows the course of the body stalk, passing at first cranialwards and then turning caudalwards; it thus presents a curve convex in a cranial direction, with ventral and dorsal limbs, and its total length is 1-45 mm., which is rather more than is found in other embryos of a similar stage of development.
The calibre of the allantois is nearly uniform throughout, but the ventral, or caudal limb, is constricted here and there, and at its termination is considerably widened and bent upon itself in an S-shaped manner.
A lumenispresentthroughout,wideatthebeginningoftheduct,then gradually decreasing towards the end, where it becomes dilated again, the S-shaped portion having the greatest diameter of the whole duct.
The concavity of the allantoic loop is occupied by the great blood lake formed by the fusion of the umbilical veins in the body stalk; only one anasto- mosis between the two umbilical arteries is present, it occurs only a short distance before the end of the allantois and it does not appear to have any con- stricting effect on the duct, as Atwell (1930) suggested might be the case.
In the body stalk, considerably ventral and slightly to the right of the allantois, there is a curious structure (PI. I, fig. 3) resembling the "Epithel- blaschen" described by Veit & Esch (1922) in an embryo of the 4th week, with 8 pairs of somites; it is a closed cylindrical sac, like a portion of the allantois in appearance and, like the allantois, is surrounded by a clear space which serves to distinguish it at once from a blood vessel. This vesicle does not appear to have been constricted off from the main part of the allantois, as Davis (1923) suggested might have been the case in the specimen described by Veit & Esch, but it is a completely isolated structure; its wall is one cell thick, its cavity is empty, except for a little epithelial debris, and it tapers rather to a point at each end, which is the one feature in favour of its having been constricted off from the allantois.
The cloaca is expanded ventrodorsally and flattened from side to side;itis continuous caudally with the tail gut, which itself extends to within three sections of the tip of the definitive tail. From the caudal margin of the body stalk to the beginning of the tail the ventral body wall of the embryo is marked by a median groove which gradually deepens as it is traced in a caudal direction; throughout the length of this groove ectoderm and entoderm are in contact, at least there is no mesoderm intervening, though the entoderm is drawn ventralwards in a thread-like manner so that, towards the caudal end, the connexion between entoderm and ectoderm is very slender; this area of contact is the cloacal membrane (Text-fig. 1).
The sides of the cloaca are marked, nearer the dorsal than the ventral border, by a groove which runs parallel with the dorsal edge of the gut and which is continued round the curve of the cloaca towards the tail; this groove indicates the future subdivision of the cloaca into rectum and urogenital sinus. Of these two parts of the cloaca the ventral urogenital sinus is by far the larger, and is expanded from side to side in contrast with the laterally compressed rectum. Watt (1915), in his description of twin embryos of 17-19 somites, referred to the impending subdivision of the cloaca and to a groove similar to that to which I have called attention; he showed, in his Fig. 7c, of embryo V, in contrast with the present specimen, a wide dorsal "rectal bay" and a laterally compressed ventral "bladder bay", whilst his embryo VI, shown in Fig. 7b, is much more like the present specimen.
From a study of this embryo I have the impression that the dorsal rectal portion of the cloaca is carried caudalwards, caudal to the bladAdr portion and is then continued ventral to the urogenital sinus and so into the tail, so that a section through the cloaca near the tip of the tail shows a dorsal laterally compressed portion, which is the rectum, an intermediate expanded portion, which is the urogenital sinus, and a ventral compressed portion, which is the caudal part of the rectum, or the tail gut; with the straightening out of the tail and the atrophy of the tail gut, the rectum will be left opening dorsally and the urogenital sinus ventrally, after the rupture of the cloacal membrane.
Urogenital System
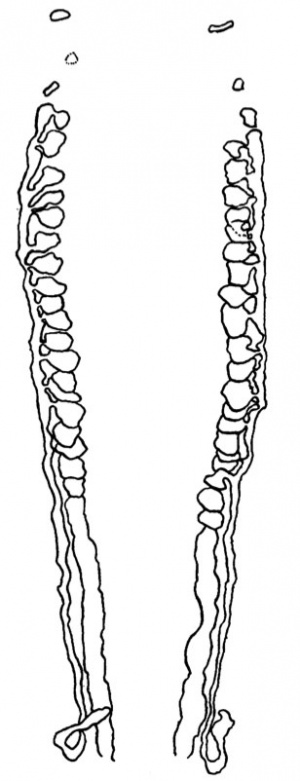
This is composed of a few isolated pronephric rudiments and of a series of mesonephric tubules, or vesicles, which are connected with each other through a longitudinal mesonephric duct that runs caudalwards and just fails to open into the cloaca (Text-fig.6).
The pronephric rudiments are three on each side.
On the left side the first indication of a pronephric tubule occurs as a thickening and invagination of the coelomic epithelium on a level with the middle of the hepatic diverticulum and with about the middle of the 8th somite; the invagination is directed at first dorsalwards and then makes connexion with a small, blind and almost solid, though still hollow, vesicle. The second rudiment is much smaller; it is placed opposite the caudal half of the 9th somite, caudal to the hepatic diverticulum, and it arises in the same manner as the first, but the invagination is directed lateralwards rather than dorsalwards and endsina small, solid clump of cels, the whole structure being much smaller than the first and not extending so far in a dorsal direction. The third rudiment is larger than either the first or second; it is related to about the middle of the 10th somite, and is essentially similar to the first and second tubules and ends in a definitely hollow vesicle.
The next tubule, which begins towards the caudal part of the 10th somite, is the first which joins the Wolffian duct; it arises,like the previous three tubules, by a connexion with the coelomic epithelium and then turns in a caudal and dorsal direction to join the Wolffian duct, which has a blind cranial end beginning at almost the same level as the tubule.
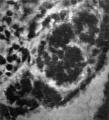
There next occurs a series of 20 vesicles, or tubules, which appear to arise in situ, though in nearly every case there can be recognised the remains of the nephrogenic cord through which they were connected originally with the coelomic epithelium; al of these vesicles are quite separate from each other, all possess a lumen, and al, except the last two and Nos. 15 and 17, make connexion with the Wolffian duct. A good guide to the point of fusion of vesicle and duct is found in the presence of a clear space, which surrounds vesicle and duct individually where these two structures are separate (PI. I, fig. 5), but which becomes common to both duct and vesicle where these are united; such spaces are of common occurrence in human embryos, and it has been suggested by Prof. Evatt, in verbal communications to the Anatomical Society, that they are early lymphatic spaces. Caudal to the 20th vesicle there is a nodular cord of mesoderm which fades away towards the tail and shows a series of nine or ten dilatations, the beginnings of new vesicles.
On the right side the first pronephric rudiment arises in a manner similarto that on the left side, there being a thickening of the coelomic epithelium, an invagination, a dorsolateral and caudal extension terminating in a blind and hollow vesicle.
The first pronephric tubule lies at the level of the cranial part of the 8th somite. At the extreme caudal end of this somite there is a definite thickening of the coelomic epithelium, and in the mesenchyme a few cels which may represent a degenerated tubule. The third rudiment is similar to the first and is related to the cranial part of the 9th somite; it is succeeded, at the caudal end of this somite, by a tubule which, as on the left side, turns caudalwards to join a well-defined duct. Beyond this point there is a series of 19 tubules, or vesicles, all of which show a lumen, and all , with the exception of the last four and No.13, are connected with the Wolffian duct. As on the left side,thelast tubule isfollowed by a nodular cord which has about ten dilatations.
The Wolfian duct begins in the manner already described, opposite the 10th somite on the left side and opposite the 9th somite on the right. A lumencan be traced throughout its entire length, being especially large towards the cranial end of the duct and frequently showing enlargements after the union with it of a mesonephric vesicle. At its caudal end the duct is bent, inconformity with the curvature of the tail region of the embryo, and ends blindly close to the cloaca; the duct has a blunt and rather small and tapering end, but just before its termination it is much larger and it is at this point that it attains its greatest diameter; where the duct turns round, following the curvature of the tail, it is compressed from side to side and is much smaller than it is just before its termination.
The Wolffian duct comes to an end before reaching the cloaca, and close under the ectoderm; yet the relationship between ectoderm and duct is not so close at the very end of the duct as it is some distance further cranialwards; I show (Text-fig. 7) camera lucida drawings of the last 11 sections of the duct and adjacent ectoderm; at about the bend of the duct or slightly to the caudal side of this, the duct is so close to the ectoderm that I was at first under the impression that an actual opening of the duct on to the surface was present at this point, especially as the ectoderm here is rather irregular; such an opening, however, does not exist, and it is only that there is very close relationship and, indeed, actual contact between the duct and the ectoderm, as may be seen in the figure.
It thus appears that in this embryo there is no contribution of the ectoderm towards the formation of the duct.
The course of the more caudal part of the Wolffian duct can be marked out almost exactly on the surface of the embryo, for the caudal part of the coelom, or inferior peritoneal canals, underlies a prominence - the Wolffian ridge - on each side of the body; this prominence is separated from the more ventral part of the body of the embryo by a groove which turns forwards towards the tail, and at the bottom of this groove, which can be seen in the photograph of the intact embryo, there lies the caudal part of the duct; this is just like the condition described by Bremer (1906) in an embryo of 4mm. length, in which the Wolffian duct failed to reach the cloaca on one side.
The pronephric tubules have been described above as arising from an invagination of the coelomic epithelium, whereas the mesonephric tubules appear to arise in situ; there is, however, on the left side at about the level of the middle of the third mesonephric vesicle, a very well-marked nephrostome (P1.I,fig.5); itisadeepand wide invagination of the coelomic epithelium and is directed medialwards and dorsalwards; it has no connexion with the vesicle but lies ventral to it. In association with the first mesonephric vesicle on the right side there isa less well marked, but stildeep, invagination of the epthelium,which I believe tone a nephrostome; these are the only nephrostomes present, though nearly al the vesicles have a connexion with the coelomic epithelium through the remains of the nephrogenic cord. When, in a reconstruction, the dorsal wall of the body cavity is viewed from the front the line of connexion, through the nephrogenic cord, between nephric rudiments and the coelomic epithelium is found not to be straight but to approach the middle line as it is traced in a caudal direction, and it shows an abrupt medial bend at the point of transition from pronephric vesicles to mesonephric vesicles. Watt (1915) has referred to the thickening of the coelomic epithelium along the mesentery and to the medial side of the urogenital fold; a urogenital fold is not well marked in this specimen, though there is a clear indication of it in the pronephric region, where it is limited medially by the nephrostomes of the pronephric tubules; in the mesonephric region the vesicles by their size cause an elevation of the dorsal wall of the coelom, and this may be regarded as the urogenital fold; it is the epithelium of the dorsal wall of the coelom that is so characteristic in the mesonephric area, and the arrangement of the cells is so definite that it may almost be employed as a means of differentiating the mesonephric region from the pronephric; over the mesonephric rudiments the epithelium forms only a thin layer of cels arranged with their long axes parallel to the surface,but just where theremains of the nephrogenic cord are connected with the epithelium there is a sudden and very striking change, the cels here being arranged with their long axes atrightanglestothesurfaceand thus seeming to form a much thicker layer than over the vesicles; this arrangemeant of the cels at right angles to the surface is continued from the nephrogenic cord along the sides of the mesentery; in the pronephric region al the cells of the dorsal wall of the coelom appear to be arranged at right angles to the surface, and seem merely to be smaller over the pronephric rudiments than in the adjacent epithelium. There are no glomeruli present.
The arrangement of the nephric system in this embryo corresponds very much with that described for other specimens of about the same age, for example, MacCallum's (1902) of 19 somites, Davis' (1923) of 20 somites, Girgis' (1926) of 22 somites, Thompson's (1907) of 23 somites, and Johnson's (1917) of 24 somites; but Davis and Girgis consider that the pronephros extends as far as the 12th (Davis) and the 13th (Girgis) somites; in the present specimen I believe that the pronephros does not extend beyond the level of 10th somite, because on each side the first three tubules, which I have called pronephric, form an isolated group, they are al rudimentary, and they all appear to arise in a similar manner by an invagination of the coelomic epithelium, whilst those vesicles, which I have called mesonephric, form a continuous series, they are alwell developed, they alseem to have arisen in situ, and they all are alike in appearance.
Nervous System
The brain and spinal cord form an almost completely closed tube, the anterior neuropore being closed and the posterior being just in the process of closing.
There are three brain vesicles, and in the following account they are described as being in the position shown in the figures.
Forebrain
This is a globular mass which overhangs the pharynx and mouth region; at the point indicated in Text- fig. 8 there can be seen the remains of the anterior neuropore, the closure of which has been just completed, and the rolled-in edges of the brain wall show very well in the sections and it can be seen that the head ectoderm also istucked a litleinto the groove at the point of closure; the condition is very like that figured and described by Sternberg (1927) in an embryo of 25 somites, and 2-7mm. in length. Just to the cranial side of the pharyngeal membrane there is a slight downgrowth of the floor of the brain which marks the infundibular region (Text-figs. 1 and 8), and at this point the brain wall and mouth cavity are in contact and it is here that the notochord comes to an end.
From the sides of the forebrain there arise the optic vesicles,but there is no evidence of a lens thickening in the overlying ectoderm.
With regard to the markings on the surface of the brain interpretation is not easy, for there has been an undoubted shrinkage of the brain, a shrinkage which appears to involve chiefly the ependymal and mantle zones, whilst the marginal zone does not seem to be much altered. Bearing this fact in mind, it maybe stated that the forebrain, between the main mass of the optic vesicles, is constricted from side to side, whilst on a level with the tip of the vesicles it becomes expanded. At this level a groove is seen on the roof, when the brain is viewed in profile, and it isa similar groove that Wen (1928) showed in his Fig.c,of a 22-somite embryo, and which he considered as marking the separation between forebrain and midbrain. The side of the brain just behind this point is marked by two grooves which together form a V with apex directed towards the infundibular region; Wen showed similar grooves in his 22-somite specimen, and he regarded the more posterior as dividing the midbrain into anterior and posterior parts, and the more anterior as marking the separation on the side of the head between forebrain and midbrain. Johnson (1917), in his specimen of 24 somites, shows a groove corresponding with the posterior limb of the V-shaped groove and he considers that it is this groove which separates forebrain and midbrain, whereas Wen regards this groove as dividing the midbrain into two parts; in this I am in agreement with Johnson,as far as my embryo is concerned, that is to say I believe that the posterior limb of the V-shaped groove marks the separation between forebrain and midbrain; the anterior limb of this groove I believe to be due to the overhanging growth, in front of it, of a developing cerebral hemisphere, such as Watt (1915) shows in hisembryoVI. An oblique groove marks clearly the separation between midbrain and hindbrain, the latter being recognized by its neuromeres.
The midbrain is the smallest of the three segments, and here the forebrain and hindbrain are bent so as to form a right angle with each other.
The hindbrain is the largest segment. The roof already shows the thinning of the future roof of the fourth ventricle, and when the embryo was in alcohol it was possible to see, through this thin roof, a series of alternating expansions and constrictions- the neuromeres.
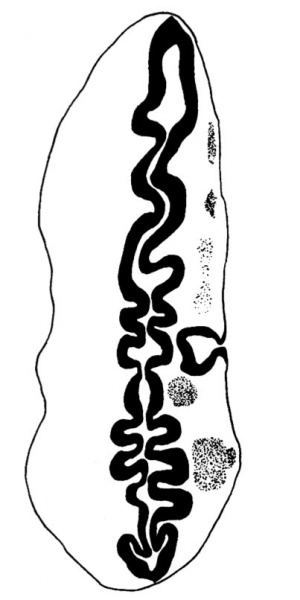
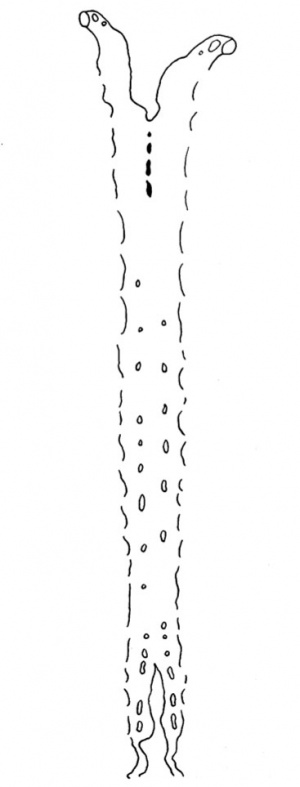
The neuromeres resemble closely those described by other observers, but they show more clearly than in other specimens (Text-fig. 9); there are ten of these segments which involve the lateral wall of the brain. The 2nd, 4th, 6th and 7th involve also the flor, to whichtheygivean irregular outline;itisthe grooves between the projections of the floor caused by these neuromeres, and especially the 4th, which are referred to by Thompson (1907), Bremer (1906), and van den Broek (1911),as having given rise to a difficulty in their interpretation, and van den Broek suggested that they might have some association with the Vth and VIlIth ganglia, which is indirectly true in view of the relation of these ganglia to the neuromeres which are responsible for the production of these grooves. The ganglion of the trigeminal nerve lies lateral to segments 2 and 3; the acoustico-facial ganglion is lateral to segment 4; the otocyst is lateral to segment 5; the glossopharyngeal ganglion is lateral to segment 7, and the vagus ganglion is lateral to segments 7 and 8 (Text-figs. 8 and 9), but the glossopharyngeal and vagus ganglia are dificultto define and appear only as rather diffuse clumps of cels.The first somite is opposite the 8th neuromere, the 2nd somite is opposite the 9th neuromere, and the 3rd is opposite the 9th and 1oth neuromeres and the intervening groove.
Spinal cord
The more cranial part of the spinal cord shows three faintly marked segments, succeeding those of the hindbrain and, like them,having no segmental relationship to the somites. In its cranial two-thirds the lateral surface of the spinal cord is marked by a longitudinal groove corresponding in position with that of the future sulcus limitans on the inside; the diameter of the spinal cord is reduced in its middle part just beyond the dorsal concavity of the body, and it then increases, especially in a ventrodorsal direction, towards the tail; intends, 30 sections before reaching the tip of the tail, in close apposition to the primitive streak; it is here that the posterior neuropore can be seen just in the act of closing, the condition being essentially similar to what is found at the anterior end of the brain only showing an earlier stage in the process.
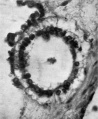
Otocyst
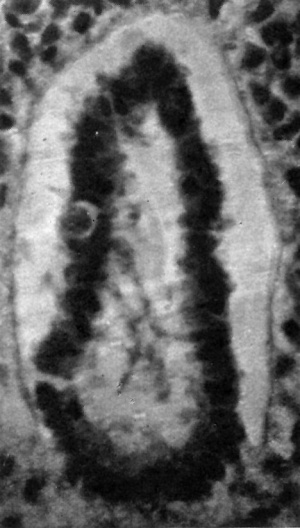
This is a hollow, oval vesicle, measuring 0-18 mm. in a craniocaudal direction, and 0.14 mm. from side to side; itisthus of much the same size as the otocyst of embryos described by Davis (1923), Watt F (1915), and van den Broek (1911). The sac is open on to the exterior (Text-fig. 9) through an orifice that measures 0*02mm. in diameter, and which is, as in van den Broek's specimen, nearer the cranial pole than thecaudalpoleofthevesicle. From the ventromedial surface of the sac there projects forwards a hollow diverticulum for a distance of 0 04 mm. Waterston (1914) shows a rather similar diverticulum, but in his specimen the-projection is in a dorsomedial direction.The picture presented by my specimen is very like Arey's (1930) Fig. 488, of a section through a 6-mm. pig embryo, and he refers (p. 492) to the ventromedial diverticulum as "a median out-pocketing where the endolymph duct attaches"; yet though the vesicle in this pig embryo antinomy specimen are so alike,mine must be much the younger,and it is stil open to the outside, and it is not til after the closure of the opening that there arises, from the point of closure, the endolymph duct which then shifts in a ventromedial direction,so that however tempting it maybe to interpret it otherwise,it is probable that this diverticulum is a folding due to shrinkage. The vesicle as a whole overlies the 5th neuromere and corresponds almost exactly with it in size; the diverticulum referred to projects towards the groove between the 4th and 5th neuromeres and towards the acoustico-facial ganglion; at the more cranial part of the vesicle, at the level of its opening to the exterior, the relationship between the vesicle and the acoustico-facial ganglion is a very close one, the ganglion appearing to be moulded round the ventral surface of the vesicle. The structure of the vesicle is essentially similar to that of the adjacent wall of the hindbrain and it shows the same arrangement of mantle and marginal zones.
Neural Crest
At the level of the 5th somite a neural crest can be recognised with certainty; from the 5th to the 2nd somite there is a collection of darkly stained nerve cells occupying the position of a neural crest, but they cannot be differentiated clearly from the surrounding mesenchyme, nor do they seem to have any connexion with the cels of the vagus ganglion. A definite neural crest can be traced from the 5th to the 21st somite; it shows no point of maximum development, suchasWen (1928) suggested occurring at the level of the 5th somite in his 17-somite embryo, but from about the 9th or 10th somite there is a gradual reduction in the development of the crest when it is traced towards either the head or the tail.
With regard to the cranial nerve ganglia, the trigeminal and acoustico- facial ganglia are large and well defined, but the glossopharyngeal and vagus ganglia are not seen so clearly. The ganglia, especially the trigeminal and acoustico-facial, in the more cranial sections, i.e. nearer the roof of the brain, appears circular masses of cells lying close to the marginal zone of the neural tube,but when traced in a caudal direction there is a migration of the cells in a lateral direction,whereby thecelscome to lie close up against the ectoderm of thehead,whichismuch thickened at the site of contact of the ganglionic cels. Such ectodermal thickenings are developed in association with each of the four ganglia, trigeminal, acoustico-facial, glossopharyngeal and vagus, and they involve the whole of the 1st, 2nd and 3rd branchial arches with which the ganglia are associated, and the thickening is prolonged in a caudal direction beyond the 3rd arch on to the cardiac area. In connexion with these ganglia, reference may be made to the arrangement of the cels in the walls of the neuromeres; showing particularly well in the case of the 4th neuromere, there is a great thickening of the brain wall at each of the neuromeres, whilst between the neuromeres the wall is much thinner; such thickenings of the brain wall in the region of a neuromere are suggestive of there being an association between them and afferent or efferent nerve fibres; though no nerve fibres can be identified in this embryo, there is an undoubted close connexion between the 4th neuromere and the acoustico-facial ganglion; connexion between the other cranial ganglia and the neuromeres is not so clear, there being one ganglionic mass related to more than one neuromere.
Vascular System
Heart
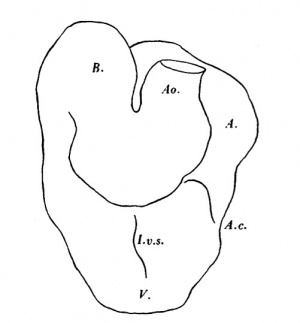
As can be seen from the drawing of a model (Text-fig. 10), the heart consists of (1) a sinus venosus, in the upper left part of which is the opening into (2) the atrium which is, for the most part, a large single chamber, but which in its caudal part shows by an external groove and an internal ridge a commencing division into right and left chambers. The atrium leads by a constricted canal into (3) the ventricle, which forms the most caudal part of the heart and also shows the beginning of an interventricular septum and which then turns obliquely cranialwards and to the right; at the summit of its arch the ventricle joins (4) the bulbus, which passes caudalwards and to the left, extending from the most cranial limit of the heart to about half-way down it, where it turns abruptly cranialwards into (5) the aorta. The sharpness of the various curvatures is emphasized more in the endothelial heart tube than in the myocardial. The caudal part of the sinus venosus lies in the septum transversum and is slung for some distance to the cranial side of its union with the atrium to the middorsal line by a thick mesentery which is common to the heart and the gut, the heart lying ventral to the lung bud region, and the two organs being separated from each other only by the thickness of the irrespective walls. A mesentery slings the arterial end of the heart to the ventral body wall for a short distance until the aorta becomes embedded in the body wall. Elsewhere the heart tube is free.
With regard to structure; the sinus venosus is different from the rest of the heart and has not the same thick myocardial wall, but is more of the nature of a largely dilated vessel, it is indeed a true venous sinus.
In those parts of the heart where the walls are thin there is a considerable interval between the endocardial and myocardial heart tubes, which is filed with the loose trabecular tissue which has been so often described. Inthe thickest part of the heart, e.g. the ventricle, such a submyocardial space scarcely exists owing to the thickness of the muscular wall of the heart, and here the endocardial tube is formed of extremely thin, elongated cells placed endtoend;where there is a large sub myocardial space it can be seen that the trabecular tissue forms, on the deep surface of the space, a definite limiting membrane which abuts against the endocardial tube.
There is a difference in the appearance of the myocardium in the various parts of the heart; the sinus is by far the thinnest, and really has no myocardial coattail; a myocardial coat can be recognised in the atrium, where it forms a fairly thick and compact layer of cels,quite different from what is found in the wall of the ventricle; in the ventricle the myocardium is thicker but the cells are more loosely arranged, they are large and form columns or chains enclosing clear spaces between them; the myocardium of the bulbus forms a layer intermediate in thickness between that of the sinus and the atrium.
Blood Vessels
The general plan of the aorta can be seen in the figure (11). It passes cranial wards from the bulbus and then bifurcates, and the two divisions turn round in a caudal direction to form the first pair of aortic arches, and from the summit of each there arises the internal carotid artery. There is a second pair of aortic arches caudal to the first,and a third pair is almost completed by a considerable outgrowth from the ventral aorta and a much smaller ventral sprout from the dorsal aorta. The dorsal aortae then pass on along each side of the middle line as far as the caudal border of the 5th somite, or the junction of brain and spinal cord, where they join here and there; but true union does not occur til the level of the middle of the 7th somite,or at the level of the cranial end of the liver;from this point a single dorsal aorta passes towards the tail as far as the origin of the allantois, at the level of the 20th somite, where a division into two caudal aortae takes place, and here too the large umbilical plexus of vessels arises. The condition is thus rather in advance of that found in Girgis' (1926) embryo of 22 somites,and is very like that in Johnson's (1917) specimen of 24 somites.
Ventral branches, not strictly segmentally arranged, arise from the ventral surface of the aorta; there are 12 on the left side and 14 on the right. Dorsal and lateral branches are present also, but are not so definite as the ventral series. The picture formed by these various branches of the aorta and by the post-cardinal and sub-cardinal veins is almost identical with H. M. Evans' (1912) Fig. 436, p. 632, of the vessels in an embryo of 23 somites.
The more caudal of the ventral branches of the aorta, which rise in between the 20th and 23rd somites, i.e. between the 9th and 12th thoracic segments, are those which give origin to the umbilical arteries; these two vessels run, one on each side of the allantois as it leaves the gut, but ventral and dorsal, the left being ventral, to the allantois as itliesinthebodystalk; a branch connects the two arteries round the left side of the allantois just before its expanded end, and that is the only communication between the two vessels; the umbilical arteries then break up into branches in the chorion.
In the chorion there arise two veins which unite in the body stalk, like the limbs of a Y, to form a single large vessel, or blood lake, which occupies the concavity of the allantoic loop; this lake is then split into two, a right and a left umbilical vein, by the umbilical arteries which course through the blood lake on their way from aorta to chorion. The two umbilical veins then run towards the body of the embryo on each side of the allantois, which, together with the two arteries, separates the veins from each other. Beyond the body stalk the umbilical veins come to occupy the ventrolateral body wall, where they raise up large ridges which can be recognized in the intact embryo, the right vessel being the larger of the two;these ridges form the boundaries of the opening from the body cavity into the extra-embryonic coelom and have attached to them the line of amniotic reflection. The two veins then turn medialwards in the septum transversum, and are there joined by the vitelline veins, the combined vitello-umbilical trunk opening into the sinus venous.
The anterior and posterior cardinal veins are not easy tof ollow; the anterior cardinal vein is formed by tributaries from the region of the optic vesicle and receives tributaries also from the dorsal region of the head; the vein then passes on the lateral side of the otocyst to the level of the 5th somite, where it turns ventralwards and is joined by the posterior cardinal vein to form the duct of Cuvier.
The posterior cardinal vein can be traced in a caudal direction as far as the level of the 17th somite, beyond which point it cannot be identified; it is placed dorsally between the Wolffian duct and the mesonephric vesicles.
The right duct of Cuvier opens independently into the sinus venous on the cranial side of the vitello-umbilical trunk, whilst on the left side it joins the vitello-umbilical trunk before that vessel opens into the sinus. The vitelline veins enter the body of the embryo along the cranial wall of the yolk stalk and pass into the septum transversum. On careful examination of the septum,I was surprised to find how few are the vessels in it, there being scarcely any in the ventral part of the septum,but in its dorsal part there are two main vessels on the left side, whilst on the right side the arrangement is rather more complicated, the vitelline vein being broken pinto several branches with which the liver cords are in contact, so that here and there a vessel is found to be surrounded completely with liver cords. But,as I have stated above,I do not believe that the liver cords have been the principal factors in the branching of the vitelline veins.The various branches of the vitelline veins ultimately unite into a single vessel on each side which joins the umbilical vein to form a vitello-umbilical trunk, and this, on the left side, is joined by the duct of Cuvier.
Coelom
The coelom consists of pericardial cavity and peritoneal cavity which communicate with each other through large pleuro-peritoneal canals and which are indirectly open to the extra-embryonic coelom round the attachment of the yolk stalk. The separation of the pericardial cavity from the pleuro-peritoneal canals is caused by a ridge on the lateral coelomic wall which overlies the duct of Cuvier, though it does not appear to be due to this vessel.
The two pleuro-peritoneal canals are four-sided tubes, open at the cranial end into the pericardial cavity through the dissolution of their ventral walls, and at the caudal end they open into the peritoneal cavity and the extra-embryonic coelom round the yolk stalk. Each canal has four walls, medial, lateral, ventral and dorsal; the medial wall is everywhere thicker than the others and is composed of about five layers of cels; the lateral wall is the thinnest; the ventral and the dorsal walls are rather thick and in the region of the septum transversum the ventral walls are raised into prominent ridges by the vitelline veins.
Cranially and caudally the ventral walls of the canals open out into the pericardial cavity and into the extra-embryonic coelom respectively, whilst more caudally the peritoneal cavity becomes closed offfrom the extra-embryonic coelom by the formation of the body stalk.The umbilical arteries, as they pass ventralwards from the aorta, give rise to ridges on the medial wall of the coelom and, as they run out into the body stalk, they cause similar ridges on the ventral wall. The caudal limit of the coelom is at the Wolffian ridges, to which reference has been made in connexion with the hind limb buds.
The septum transversum is an obliquely placed partition between the pericardial and peritoneal cavities, and it slopes from in front cranialwards and dorsalwards; growing into it from the dorsal side there is the liver; run- ning along its caudal edge in a cranial and dorsal direction there are the vitelline veins, which form a complicated plexus in its substance; entering its lateral sides there are the umbilical veins caudally and the ducts of Cuvier cranially; the sinus venosus lies just above its cranial edge.
Notochord
The notochord begins, as an extension cranialwards of the primitive streak, as a thick, solid column of cels.
Its appearance in section varies at different levels; at the origin of the umbilical arteries it is an oval mass of cels,compressedfrom sidetosideand with an indication of a lumen in its interior; at the middle of the body stalk it has a circular outline and the lumen is quite definite; at the cranial edge of the body stalk the section is oval but compressed ventrodorsally, and at this level a distinct open space bounded by a limiting membrane separates the noto- chord from the marginal zone of the spinal cord, whilst between the notochord and the aorta there are a few scattered mesenchyme cels;at the middle of the yolk stalk the notochord is flattened ventrodorsally, still has a lumen and is surrounded by the same space and limiting membrane, which are here in direct contact with the spinal cord dorsally and with the aorta ventrally; at the caudal part of the heart the notochord is rather smaller and more solid, with a less definite lumen, and mesenchyme celsagain separate itfrom the aorta; in the lung region it is more circular in outline and is still further separated from the aorta; where it turns forward into the head region, and is cut along its long axis,itappearsastwo slender rows of cels with a cleft-like space between them; where the notochord is related to the brain it is circular in section til just before its end, where it is flattened from before backwards. It ends at the level of the pharyngeal membrane, being, as it were, squeezed out of existence by the brain wall coming into contact with the epithelium of the mouth in the infundibular region. The notochord comes into contact with the roof of the pharynx at two points; one, midway between the first and second pharyngeal pouches, and the other, midway between the second and third pouches; there is not any appreciable difference between the diameter of the notochord at these two points and elsewhere, nor does there appear to be any thickening of the underlying pharyngeal mucous membrane, as described by Davis (1923), and by Huber (1912) in a rather later stage.
Germ Cells
An account of the appearance and arrangement of the germ cels in, among other specimens, an embryo of 4 mm., with 26-27 somites, has been given, and illustrated with beautiful photographs, by Politzer (1928b,1933). He describes the germ cels as being situated both in the wall of the gut and in the adjacent mesoderm, from the caudal end of the intestinal portal to the origin of the allantois. Special cels in corresponding positions can be recognised in the present specimen, but they are not nearly so numerous, or, as is more probable, I have failed to recognise them, for there appear to be only about 40, whereas Politzer recognized 586, of which 330 where in the mesoderm and 256 in the entoderm. In the present embryo the majority of these cels are situated in the wall of the gut and these are recognizable more easily than those in the mesoderm; of those situated in the mesoderm, some occur alongside the gut in the splanchnic mesoderm, and others are occasionally found in the mesoderm just to the medial side of the mesonephric vesicles. They al are large cels with clear cytoplasm whereby, as Politzer describes, they may be differentiated from blood cels; the nucleus is large and round and has a granular appearance. A single germ cel is shown in PI.I,fig.6.
Summary
An apparently normal embryo, nearly 3 mm. long, and with 25 somites, is described. It was obtained from a uterus removed at a post-mortem examination. It is hoped that the specimen may form a useful addition to a stage in human development in which there are not very many specimens available.
The following points of interest arise from a study of the specimen: A convenient method of orienting the sections is described.
Photographs of the embryo, before and after the removal of the amnion, are shown.
The ovulation age has been estimated-by Streeter's method of the comparison of human developmental stages with those ofMacacus rhesus-and the age of this specimen has been compared with that of other embryos of about the same stage of development. The question of the interpretation of the morphological division of the somites is discussed.
The limb buds are in a very early stage of development, the forelimb bud being just recognizable, and it is suggested that there is no contribution from the myotome towards its structure.
The thyroid gland is smaller than usual, as far as its median component is concerned, whilst the lateral thyroid rudiment is well developed.
A very peculiar structure projects as a hollow tube into the interior of the pharynx; the author is unable to interpret it and has been unable to discover in the literature anything resembling it.
The lung buds are in an interesting stage and seem to throw some light on this region in other embryos of about this stage.
The allantois is exceptionally well developed, and there is a curious vesicle near its termination, which appears to have no connexion with the allantois. The cloaca shows an early stage of division into urogenital sinus and rectum, by a pinching in of its lateral walls.
The urogenital system shows pronephric rudiments and mesonephric vesicles; the Wolffian duct is at a very interesting stage, not yet having reached the cloaca; it is suggested that the duct receives no contribution from the ectoderm.
The brain shows the neuromeres exceptionally well; ten of them are recognizable.
The heart forms an S-shaped loop; it is slung at its venous end by a dorsal mesentery.
The relation of the liver cords to the vitelline veins is discussed; it is sug- gested that the cords are not the main factor in the breaking up of the veins
Primitive germ cells are described and illustrated.
References
AREY, L. B. (1930). Developmental Anatomy, 2nd ed. Philadelphia and London: W. B. Saunders Co.
ATWELL, W. J. (1930). Contr. Embryol. Carneg. Instn, vol. XXI, No. 118, pp. 1-24.
BARDEEN, C. R. & LEWIS, W. H. (1901). Amer. J. Anat. vol. i,pp. 1-36.
BARTELMEZ, G. W. & EVANS, H. M. (1926). Contr. Embryol. Carneg. Instn, vol. XVII, No. 85, pp. 1-67
BOYDEN, E. A. (1918). Amer. J. Anat. Vol. xxil, pp. 205-36. BREMER,J.L.(1906). Amer.J.Anat.vol.v,pp.459-80.
VAN DEN BROEK, A. J. P. (1911). Anat. Hefte, Bd. xLIV, Heft 133, S. 273-304.
BROMAN,I.(1896). Morph.Arb.Bd.v,S.169-205.
BUTCHER, E. 0. (1929). Amer. J. Anat. vol. xLIv, pp. 381-440.
CORNER, G. W. (1929). Contr. Embryol. Carneg. Inetn, vol. xx, No. 112, pp. 81-102. COVENTRY,A.F.(1923). Anat.Rec.vol.xxv,pp.237-56.
DANDY, W. E. (1910). Amer. J. Anat. vol. x, pp. 85-108.
DAVIS,C.L.(1923). Contr.Embryol.Carneg.Inmtn,vol.xv,No.72,pp.1-51.
Evans HM. The development of the vascular system. In Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia. pp570-708.
EVANS,H.M.(1912). In Keibel & Mall, Manual of Human Embryology, vol.1, pp.570-709.
Gage SP. A three weeks' human embryo, with especial reference to the brain and nephric system. (1905) Amer. J Anat. 4: 409-443.
GAGE, S. P. (1905). Amer. J. Anat. vol. IV, pp. 409-44.
Gnmcis, A.(1926). J.Anat.,Lond.,vol.LX,pp.382-410.
Grosser O. VII. The development of the egg membranes and the placenta; menstruation in Keibel F. and Mall FP. Manual of Human Embryology I. (1910) J. B. Lippincott Company, Philadelphia.
GROSSER,0.(1910). In Keibel & Mall, Manual of Human Embryology, vol.1, pp. 91-179.
(1912). In Keibel & Mall, Manual of Human Embryology, vol.1, pp.446-97.
HAIN, A. M. (1934). Brit. med. J. pp. 454-5.
HEUSER, C. H. (1930). Contr. Embryol. Carneg. Instn, vol. XXII, No. 131, pp. 135-54. HUBER,G.C.(1912). Anat.Bee.vol.VI,pp.373-404.
Ingalls NW. A human embryo at the beginning of segmentation, with special reference to the vascular system. (1920) Contrib. Embryol., Carnegie Inst. Wash. Publ. 274, 11: 61-90.
JOHNSON, F. P. (1917). Contr. Embryol. Carneg. Instn, vol. VI, No. 19, pp. 125-68.
Keibel, F. & Elze, C. (1908). Normentafeln zur Entwicklungsgeechichte der Wirbeltiere, Heft 8. Jena.
Lewis WH. The development of the arm in man. (1902) Amer. J Anat. 1(2): 145-184.
Low A. Description of a human embryo of 13-14 mesodermic somites. (1908) J Anat Physiol. 42(3): 237-51. PMID 17232769 | PMC1289161
Low,A.(1908). J.Anat.andPhyeiol.vol.xmii(3rdser.vol.I),pp.237-51.
MACCALLUM,J.B.(1902). Amer.J.Anat.vol.I,pp.245-60.
POLITZER, G. (1928a). Z.ge.Anat.1.Z.Anat.Entw(e3ch.Bd.LXXXVII,S.674-727.
(1928b). Z.ge.Anat.1.Z.Anat.EntwGeech.Bd.LXXXVII,S.766-80.
(1933). Z.ge.Anat.1.Z.Anat.EntwGeech.Bd.c,S.331-61.
POLITZER,G.& STERNBERG,H.(1930). Z.ge.Anat.1.Z.Anat.EntwGeech.Bd.XCIa,S.279-379.
STERNBERG, H. (1927). Z. ges. Anat. 1. Z. Anat. EntwGeech. Bd. LXXXII, S. 747-80.
STREETER, G. L. (1932). Anat. Rec. vol. LIV, supp. p. 71.
(1933a). Anat. Rec. vol. LV, supp. p.39.
(1933b). Ann. Rep. of Director of Dept. Embryol. Carneg. Instn, Washington,for1932-3,p.4.
TEACHER, J. H. (1924). J. Obetet. Gynaec. vol. xXXI, pp. 166-217.
Thompson P. Description of a human embryo of twenty-three paired somites. (1907) J Anat Physiol, 41(3):159-71. PMID 17232726
THOMPSON, P. (1907). J. Anat. and Physiol. vol.XLI (3rdser.vol.I), pp.159-71.
(1908). J. Anat. and Physiol. vol. XLII (3rdser.vol.I), pp.170-5.
VErr,0.& ESCH,P.(1922). Z. ge. Anat. 1.Z.Anat. Entw Geech. Bd.LXm,S.343-414.
Waterston D. A very young human embryo found embedded in a "decidual cast" of the uterus. (1914) Proc R Soc Med., 7: 353-7. PMID 19978039
WATERSTON, D. (1914). J. Anat. and Physiol. vol. xLix (3rd ser. vol. x), pp. 90-118.
WATT,J.C.(1915). Contr. Embryol. Carneg. Instn, vol.I, No.2,pp.5-44.
WELLER,G.L.Jr.(1933). Contr. Embryol. Carnegie. Insta, vol. xxiv,No.141,pp.93-139.
WEN, I. C. (1928). J. comp. Neurol. vol. XLV, pp. 301-76.
WILSON, J.T. (1914). J.Anat. and Physiol. vol. XLVIII (3rd ser .vol. IX),pp.315-51.
ZECHEL, G. (1924). Z.ge.Anat.1.Z. Anat. EntwGeech. Bd.LXXIV,S.593-607
Explanation of Plate I
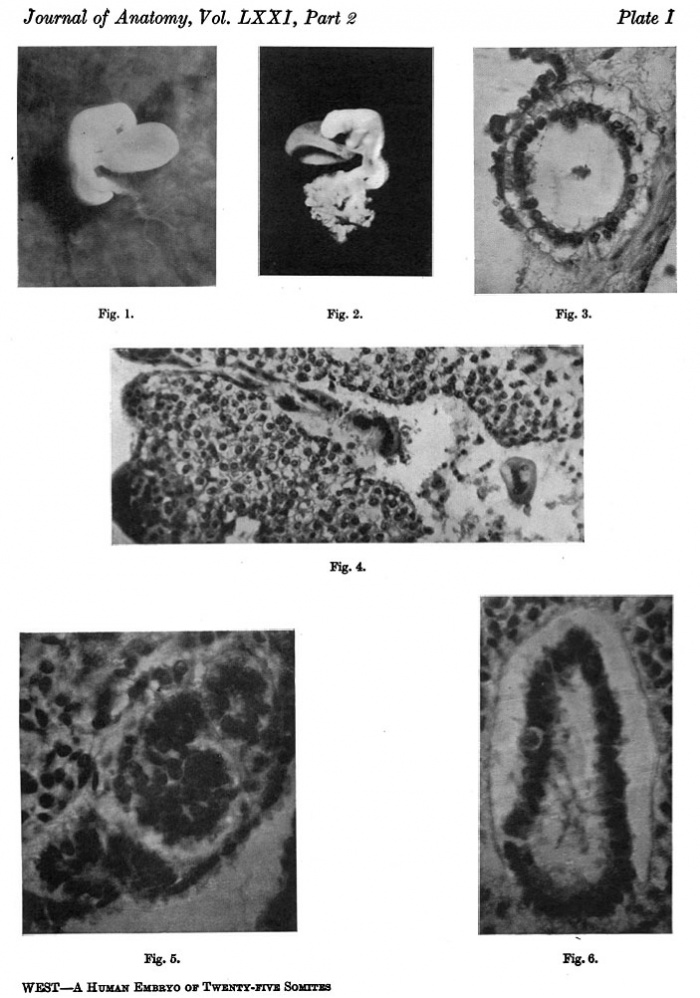
|
Fig.1. Photograph of embryo,enclosed in amnion, in chorionic sac. Fig.2. Photograph of embryo, after removal of amnion. Fig.3. Photograph of a section through the middle of the vesicle, or "Epithelblischen", near the end of the allantois in the body stalk. x345. Fig.4. Photograph of part of section through a "cell island"of the chorion, with the syncytiotrophoblast intruding into the intervillous space; towards the right is a "giant cell". x 230. Fig.5. Photograph of part of section 152, to show an ephrostome; the section passes through the 3rd mesonephric tubule turning dorsalwards and lateralwards to join the Wolffian duct. x580. Fig.6. Photograph of part of section 206, to show a germ cell in the wall of the gut on the left side. x580. |
Images
- Links: Fig 1 | Fig 2 | Fig 3 | Fig 4 | Fig 5 | Fig 6 | Fig 7 | Fig 8 | Fig 9 | Fig 10 | Fig 11 | Plate 1 | Plate 1 Fig 1 | Plate 1 Fig 2 | Plate 1 Fig 3 | Plate 1 Fig 4 | Plate 1 Fig 5 | Plate 1 Fig 6
Reference
West CM. A human embryo of twenty-five somites. (1937) J. Anat., 71(2): 169-200.1. PMID 17104635
Cite this page: Hill, M.A. (2026, April 17) Embryology Paper - A Human Embryo of Twenty-five Somites. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_A_Human_Embryo_of_Twenty-five_Somites
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
The information below was not part of the above original historic article.
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Carnegie Stage Table
Weeks shown in the table below are embryonic post ovulation age, for clinical Gestational Age (GA) measured from last menstrual period, add 2 weeks.
(not to scale) |
||||
 |
fertilized oocyte, zygote, pronuclei | |||
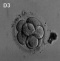 |
morula cell division with reduction in cytoplasmic volume, blastocyst formation of inner and outer cell mass | |||
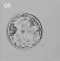 |
loss of zona pellucida, free blastocyst | |||
| attaching blastocyst | ||||
(week 2) |
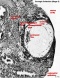 |
implantation | ||
 |
extraembryonic mesoderm, primitive streak, gastrulation | |||
| gastrulation, notochordal process | ||||
| primitive pit, notochordal canal | ||||
 |
Somitogenesis Somite Number 1 - 3 neural folds, cardiac primordium, head fold | |||
| Somite Number 4 - 12 neural fold fuses | ||||
| Somite Number 13 - 20 rostral neuropore closes | ||||
| Somite Number 21 - 29 caudal neuropore closes | ||||
| Somite Number 30 leg buds, lens placode, pharyngeal arches | ||||
| lens pit, optic cup | ||||
| lens vesicle, nasal pit, hand plate | ||||
| nasal pits moved ventrally, auricular hillocks, foot plate | ||||
| finger rays | ||||
| ossification commences | ||||
| straightening of trunk | ||||
| upper limbs longer and bent at elbow | ||||
| hands and feet turned inward | ||||
| eyelids, external ears | ||||
| rounded head, body and limbs | ||||
The embryos shown in the table are from the Kyoto and Carnegie collection and other sources.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 17) Embryology Paper - A Human Embryo of Twenty-five Somites. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_A_Human_Embryo_of_Twenty-five_Somites
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
- ↑ <pubmed>17233002</pubmed>