Kyoto Collection
| Embryology - 28 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Congenital Anomaly Research Center, Kyoto University Graduate School of Medicine, Japan.
Prof. Kohei Shiota, Dr Mark Hill, Prof. Shigehito Yamada and Prof. Mineo Yasuda |
|
| iBook - Kyoto Embryo Collection | |
|---|---|

|
|
Sample Publications
- Developing the Digital Kyoto Collection in Education and Research[1] "The Kyoto embryo collection was begun in 1961 by Dr. Hideo Nishimura. The collection has been continuously developed and currently contains over 44,000 human normal and abnormal specimens. Beginning online in 1997, the internet provided an opportunity to make embryos from the collection widely available for research and educational purposes (http://tiny.cc/Embryo). These embryonic development resources have been continuously published and available from that time until today. Published in Japanese as an Atlas of Embryonic Development. Published online as the Kyoto Human Embryo Visualization Project (http://atlas.cac.med.kyoto-u.ac.jp) and also as the Human Embryo Atlas (http://tiny.cc/Human_Embryo_Atlas). Published now electronically as a digital eBook (http://tiny.cc/Kyoto_Collection_eBook). This new digital format allows incorporation of whole embryo and histology manipulable images, labels, and a linked glossary. New imaging modalities of magnetic resonance imaging (MRI) and episcopic fluorescence image capture (EFIC) can also be easily displayed as animations. For research, the collection specimens and histological sections have been extensively studied and published in several hundred papers, discussed here and elsewhere in this special edition. I will also describe how the Kyoto collection will now form a major partner of a new international embryology research group, the Digital Embryology Consortium (https://human-embryology.org). The digital Kyoto collection will be made available for remote researcher access, analysis, and comparison with other collections allowing new research and educational applications. This work was presented at the 40th Anniversary Commemoration Symposium of the Congenital Anomaly Research Center, Graduate School of Medicine, Kyoto University, Japan, November, 2015."
- Cartilage formation in the pelvic skeleton during the embryonic and early-fetal period.[2] "The pelvic skeleton is formed via endochondral ossification. However, it is not known how the normal cartilage is formed before ossification occurs. Furthermore, the overall timeline of cartilage formation and the morphology of the cartilage in the pelvis are unclear. In this study, cartilage formation in the pelvic skeletons of 25 human fetuses (crown-rump length [CRL] = 11.9-75.0 mm) was observed using phase-contrast computed tomography and 7T magnetic resonance imaging. The chondrification center of the ilium, ischium, and pubis first appeared simultaneously at Carnegie stage (CS) 18, was located around the acetabulum, and grew radially in the later stage. The iliac crest formed at CS20 while the iliac body's central part remained chondrified. The iliac body formed a discoid at CS22. The growth rate was greater in the ilium than in the sacrum-coccyx, pubis, and ischium. Connection and articulation formed in a limited period, while the sacroiliac joint formed at CS21. The articulation of the pubic symphysis, connection of the articular column in the sacrum, and Y-shape connection of the three parts of the hip bones to the acetabulum were observed at CS23; the connection of the ischium and pubic ramus was observed at the early-fetal stage. Furthermore, the degree of connection at the center of the sacrum varied among samples. Most of the pelvimetry data showed a high correlation with CRL. The transverse and antero-posterior lengths of the pelvic inlet of the lesser pelvis varied among samples (R2 = 0.11). The subpubic angle also varied (65-90°) and was not correlated with CRL (R2 = 0.22). Moreover, cartilaginous structure formation appeared to influence bone structure. This study provides valuable information regarding the morphogenesis of the pelvic structure."
- A detailed comparison of mouse and human cardiac development[3] "Episcopic fluorescence image capture (EFIC) was performed on 66 wild-type mouse embryos from embryonic day (E) 9.5 to birth; 2-dimensional and 3-dimensional datasets were compared with EFIC and magnetic resonance images from a study of 52 human fetuses (Carnegie stage 13-23). Time course of atrial, ventricular, and outflow septation were outlined and followed a similar sequence in both species. Bilateral venae cavae and prominent atrial appendages were seen in the mouse fetus; in human fetuses, atrial appendages were small, and a single right superior vena cava was present. In contrast to humans with separate pulmonary vein orifices, a pulmonary venous confluence with one orifice enters the left atrium in mice. The cardiac developmental sequences observed in mouse and human fetuses are comparable, with minor differences in atrial and venous morphology. These comparisons of mouse and human cardiac development strongly support that mouse morphogenesis is a good model for human development."
- Morphogenesis of the spleen during the human embryonic period[4] "Between Carnegie stages (CSs) 14 and 17, the spleen was usually recognized as a bulge in the dorsal mesogastrium (DM), and after CS 20, the spleen became apparent. Intrasplenic folds were observed later. A high-density area was first recognized in 6 of the 58 cases at CS 16 and in all cases examined after CS 18. The spleen was recognized neither as a bulge nor as a high-density area at CS 13. The mesothelium was pseudostratified until CS 16 and was replaced with high columnar cells and then with low columnar cells. The basement membrane was obvious after CS 17. The mesenchymal cells differentiated from cells in the DM, and sinus formation started at CS 20. Hematopoietic cells were detected after CS 18. The vessels were observed at CS 14 in the DM. Hilus formation was observed after CS 20. The parallel entries of the arteries and veins were observed at CS 23. The rate of increase in spleen length in relation to that of stomach length along the cranial-caudal direction was 0.51 ± 0.11, which remained constant during CSs 19 and 23, indicating that their growths were similar."
- Human embryo imaging with a super-parallel magnetic resonance (MR) microscope[5]
- Polydactyly in human embryos[6] "129 embryos with polydactyly in 36,380 human conceptuses obtained through induced abortion during the period from 1962 to 1974."
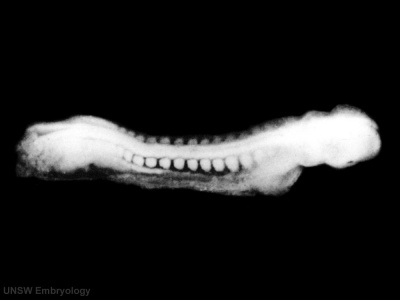
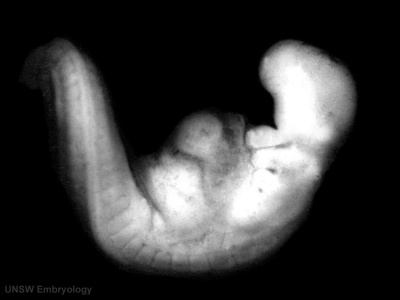
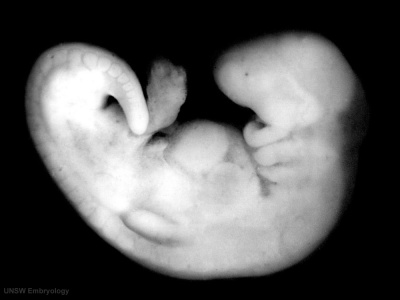
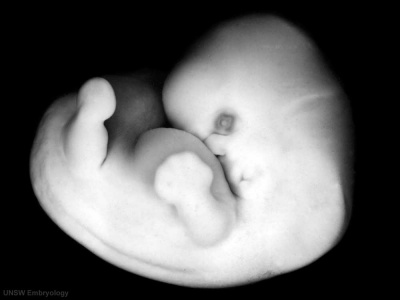
Sample Embryos
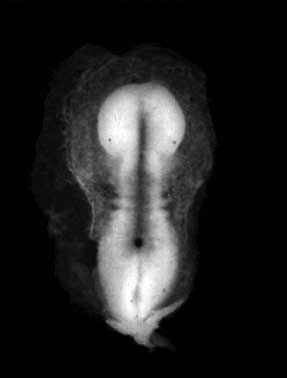
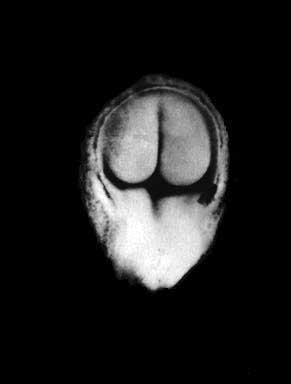
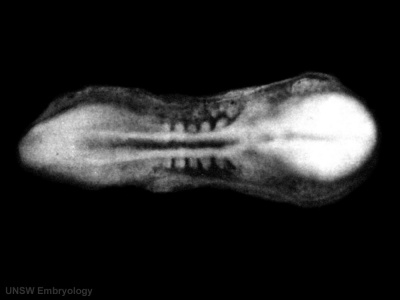
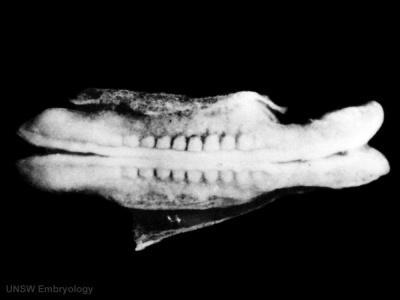
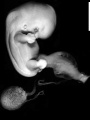
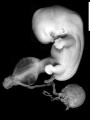
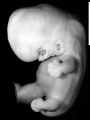
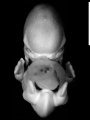
Image source: The Kyoto Collection images are reproduced with the permission of Prof. Kohei Shiota and Prof. Shigehito Yamada, Anatomy and Developmental Biology, Kyoto University Graduate School of Medicine, Kyoto, Japan for educational purposes only and cannot be reproduced electronically or in writing without permission.
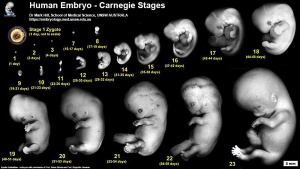
| Kyoto Specimens (Last Updated - April 7, 2014) Specimens 23,813 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Week: | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Carnegie stage: | 1 2 3 4 | 5 6 | 7 8 9 | 10 11 12 13 | 14 15 | 16 17 | 18 19 | 20 21 22 23 |
Stage 7
- Link: Carnegie stage 7
Stage 8
- Link: Carnegie stage 8
Stage 9
- Link: Carnegie stage 9
Stage 10
- Link: Carnegie stage 10
Stage 11
Stage 12
- Link: Carnegie stage 12
Stage 13
- Link: Carnegie stage 13
Stage 14
- Link: Carnegie stage 14
Stage 15
- Link: Carnegie stage 15
Stage 16
- Link: Carnegie stage 16
Stage 17
- Link: Carnegie stage 17
Stage 18
- Kyoto 25783 Links: right+yolk | left+yolk | left | ventral | right | dorsal | Kyoto Collection | Carnegie stage 18
- Link: Carnegie stage 18
Stage 19
- Link: Carnegie stage 19
Stage 20
- Link: Carnegie stage 20
Stage 21
- Link: Carnegie stage 21
Stage 22
- Link: Carnegie stage 22
Stage 23
- Link: Carnegie stage 23
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Related Images
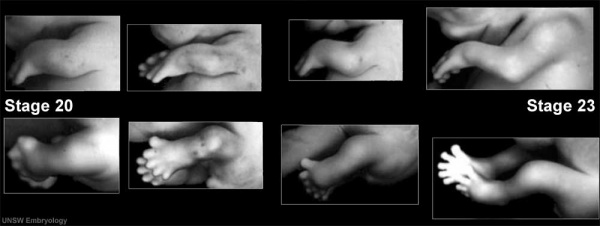
Limb Development (week 8)
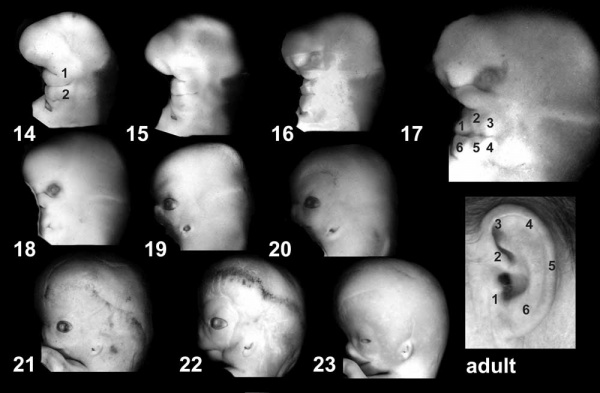
External ear stages
Recent Papers
The Kyoto Collection of Human Embryos and Fetuses: History and Recent Advancements in Modern Methods
The Kyoto Collection of Human Embryos and Fetuses[7], the largest collection of human embryos worldwide, was initiated in the 1960s, and the Congenital Anomaly Research Center of Kyoto University was established in 1975 for long-term storage of the collection and for the promotion of research into human embryonic and fetal development.
Currently, the Kyoto Collection comprises approximately 45,000 specimens of human embryonic or fetal development and is renowned for the following unique characteristics:
- the collection is considered to represent the total population of fetal specimens nationwide in Japan,
- it comprises a large number of specimens with a variety of external malformations,
- for most specimens there are clinical and epidemiological data from the mothers and the pregnancies concerned.
Therefore, the specimens have been used extensively for morphological studies and could potentially be used for epidemiological analysis. Recently, several new approaches such as DNA extraction from formalin-fixed specimens or geometric morphometrics have been adopted and it is to be expected that further technological advances will facilitate new studies on the specimens of the Kyoto Collection as well as of other human embryo collections worldwide. Permanent preservation of the Kyoto Collection is, therefore, of paramount importance so that it will continue to contribute to human embryological studies in the future.
References
- ↑ Hill MA. (2018). Developing the Digital Kyoto Collection in Education and Research. Anat Rec (Hoboken) , , . PMID: 29663673 DOI.
- ↑ Okumura M, Ishikawa A, Aoyama T, Yamada S, Uwabe C, Imai H, Matsuda T, Yoneyama A, Takeda T & Takakuwa T. (2017). Cartilage formation in the pelvic skeleton during the embryonic and early-fetal period. PLoS ONE , 12, e0173852. PMID: 28384153 DOI.
- ↑ Krishnan A, Samtani R, Dhanantwari P, Lee E, Yamada S, Shiota K, Donofrio MT, Leatherbury L & Lo CW. (2014). A detailed comparison of mouse and human cardiac development. Pediatr. Res. , 76, 500-7. PMID: 25167202 DOI.
- ↑ Endo A, Ueno S, Yamada S, Uwabe C & Takakuwa T. (2015). Morphogenesis of the spleen during the human embryonic period. Anat Rec (Hoboken) , 298, 820-6. PMID: 25403423 DOI.
- ↑ Matsuda Y, Ono S, Otake Y, Handa S, Kose K, Haishi T, Yamada S, Uwabe C & Shiota K. (2007). Imaging of a large collection of human embryo using a super-parallel MR microscope. Magn Reson Med Sci , 6, 139-46. PMID: 18037794
- ↑ Shiota K & Matsunaga E. (1978). A genetic and epidemiologic study of polydactyly in human embryos in Japan. Jinrui Idengaku Zasshi , 23, 173-92. PMID: 691840 DOI.
- ↑ Yamaguchi Y & Yamada S. (2018). The Kyoto Collection of Human Embryos and Fetuses: History and Recent Advancements in Modern Methods. Cells Tissues Organs (Print) , 205, 314-319. PMID: 30064127 DOI.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 28) Embryology Kyoto Collection. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Kyoto_Collection
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G