Detailed Cardiac - Systemic Venous Sinus
| Embryology - 28 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Detailed Cardiac: Systemic Venous Sinus | Pulmonary Vein | Superior Interatrial Fold | Atrioventricular Cushions | Atrioventricular Canal | Interventricular Communication | Subpulmonary Infundibulum | Arterial Roots | Intrapericardial Arterial Trunks | Extrapericardial Arterial Channels | Sinus Node | Atrioventricular Conduction Axis |
Formation and Subsequent Remodelling of the Tributaries of the Systemic Venous Sinus
The systemic venous sinus, also called the "sinus venosus", is a thin walled cavity forming the input to developing heart which has 3 venous inputs (vitelline vein, umbilical vein, common cardinal vein). Later in heart development this structure gets incorporated into the wall of the future right atrium. The sinuatrial orifice, is the opening between the sinus venosus and right atrium which has two valve leaflets to prevent backflow of blood. The sinus venarum, is the smooth-walled portion of the adult right atrium; originally the left horn of the sinus venous.
In the chicken model, this region develops at the junction between the splanchnic and somatic mesoderm.[1]
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Systemic Venous Sinus Development <pubmed limit=5>Systemic Venous Sinus Development</pubmed>
<pubmed limit=5>Coronary Sinus Development</pubmed> |
Remodelling of the Venous Tributaries
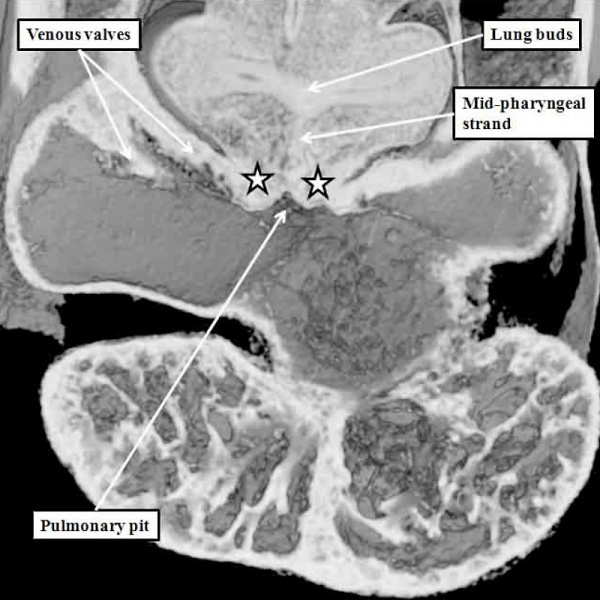
Further development of the atrial chambers, which occurs concomitant with the process of ballooning, requires remodeling of the venous channels which bring the blood back to the heart. There are three such sets of venous tributaries.They bring blood from the embryo, the yolk sac, and the placenta, respectively. The three sets unify to form the so-called sinus horns, which then drain to the initial atrial part of the heart tube. At the initial stages, when the blood enters the atrial component of the heart on each side through the so-called sinus horns, there is minimal formation of the lungs, so there are no pulmonary veins. It is the systemic venous return, therefore, which enters both sides of the atrial part of the tube. This part of the tube, moreover, remains connected to the body of the embryo through a stalk, known as the dorsal mesocardium. The dorsal mesocardium is bordered by two prominences known as the pulmonary ridges. The connection with the pharyngeal mesenchyme through the heart stalk will eventually, subsequent to the development of vascular channels within the lung buds, function as the portal of entry for the pulmonary vein (Figure 4).
Fig. 4. The section is from another mouse embryo early at E10.5. It shows the continuity of the atrial component of the heart tube with the pharyngeal mesenchyme through the connection known as the dorsal mesocardium, or heart stalk. This connects the heart with the body of the embryo, producing raised flanges known as the pulmonary ridges. These flank the pulmonary pit (white stars with black borders). Note the presence of a midline strand within the pharyngeal mesenchyme. This will eventually canalize to form the pulmonary vein.
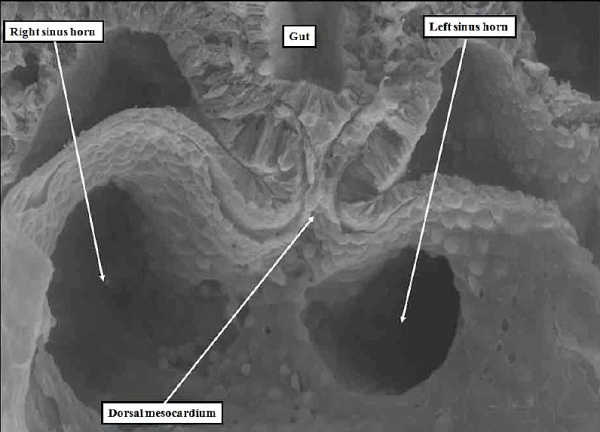
Prior to canalization of the pulmonary vein, however, there is a shift of the blood entering the heart through the systemic venous tributaries. As already emphasized, the channels bringing the blood from the embryo back to the heart initially enter the atrial component of the heart tube in symmetrical fashion (Figure 5).
Fig. 5. The image is a scanning electron micrograph through the dorsal mesocardium prepared from a mouse embryo at E8.5, earlier than the specimen shown in Figure 4. The lungs have yet to develop at this early stage. The venous tributaries open in relatively symmetrical fashion to the atrial component of the heart tube through the channels known as the horn of the systemic venous sinus.
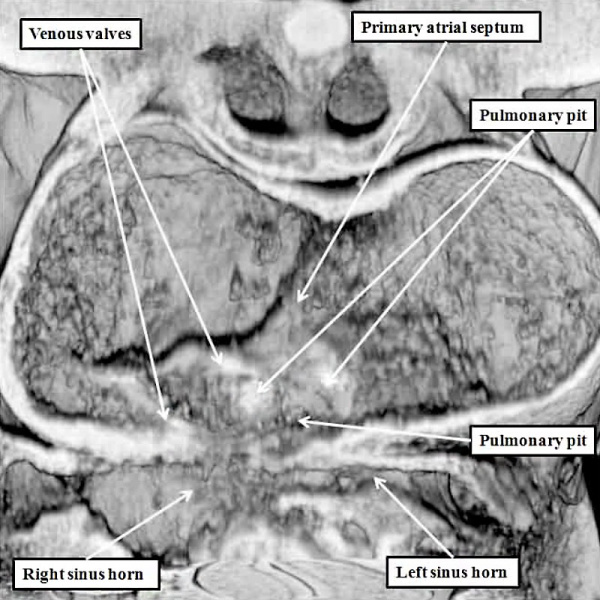
At the earlier stage, as shown in Figure 5, there are no obvious boundaries between the atrial chamber and the systemic venous channels, with the lungs not yet having been formed. With ongoing development, the venous channels move rightwards so that, eventually, they open only to the right side of the initially common atrial chamber. As the channels move rightwards, so flaps of myocardium are formed at their junction with the atrial chamber, producing the structures known as the venous valves. It is at this stage, furthermore, during the early part of E10.5 in the mouse, that the first sign is seen of atrial septation (Figure 6).
Fig. 6. The image is prepared from the same dataset as Figure 4. It shows a cut through the junction between the venous tributaries and the atrial component of the heart tube, with the boundary between the two now marked by the venous valves. As can be seen, the junction is to the right side of the common atrial chamber. Note the pulmonary pit between the pulmonary ridges, which mark the site of the dorsal mesicardium. Note also the appearance of the atrial septum in the roof of the common atrial chamber.
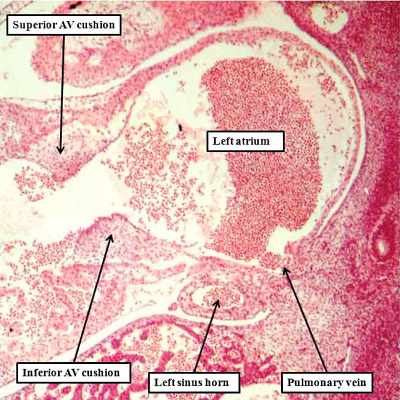
It is subsequent to the rightward shift of the systemic venous tributaries, as shown in Figure 6, that the lung buds grow and develop their own vasculature. The intraparenchymal pulmonary veins initially form a plexus that has connections with the adjacent systemic venous channels developing within the body of the embryo itself. With normal development, however, the pulmonary veins unite in the midline, with formation of a common pulmonary vein. This canalizes from the mid-pharyngeal strand, which was recognizable even before the establishment of the intraparenchymal plexuses (Figure 4). The common pulmonary vein then opens to the heart through the pulmonary pit, itself adjacent to the left sinus horn in the left atrioventricular junction (Figure 7).
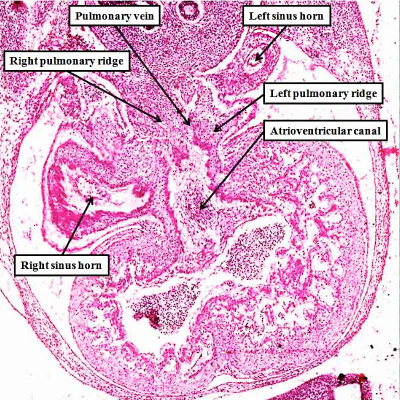
Fig. 7. The images are from different human embryos at Carnegie stage 14, at the end of the fourth week of development. The left hand panel, which is a sagittal section, shows the entrance of the pulmonary vein adjacent to the left sinus horn and the atrioventricular junction. The right hand panel, sectioned in frontal fashion, shows how the vein enters the atrial component of the heart tube between the pulmonary ridges, confirming its adjacency to the atrioventricular canal.
Should the intrapulmonary plexuses fail to unite so as to drain to the heart through the common pulmonary vein, however, they are able to retain and expand their connections to the systemic venous plexuses developing within the embryo itself. It is retention of these initial connections, with failure of junction with the pulmonary vein, which explains the lesion known as anomalous pulmonary venous connection. Depending on how much of the developing lungs drain anomalously, the lesion can be partial or complete. The pulmonary venous blood is able to return to the heart through any of the systemic venous channels. This means that, when the connection is totally anomalous, the blood can return to the heart through a superior caval venous channel, through the left sinus horn directly to the heart, or through the inferior caval vein, usually via the hepatic portal venous system. The situation in which there is only partial anomalous return can then, as we will see, influence the overall development of the atrial chambers, with one variant of partially anomalous pulmonary venous connection underscoring the appearance of the interatrial communication known as the sinus venosus defect.
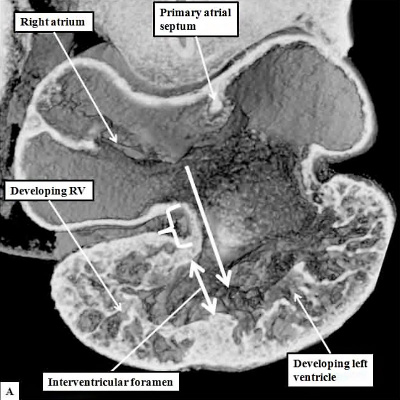
If we return to consider the stage at which the initial atrial septum, known as the primary septum, or “septum primum”, was first seen growing as a protuberance from the atrial roof (Figure 6), the atrial chamber itself is in connection only with the inlet component of the ventricular loop, this being the part that is ballooning to give rise to the developing left ventricle. This stage of development in the mouse, at E10.5, when the atrioventricular canal remains exclusively supported by the developing left ventricle, and the outflow tract is supported exclusively by the right ventricle, is equivalent to Carnegie stages 12 through 14 in the human embryo. The human embryo itself, at this early stage, is no more than four to five weeks old. At this early stage, furthermore, all the blood entering the atrial chambers is able to gain access only to the developing left ventricle (Figure 8, left hand panel), while the developing outflow tract is supported exclusively by the developing right ventricle. This can be likened to the situations, seen in malformed hearts, of double inlet left ventricle, and double outlet right ventricle (Figure 8, right hand panel).
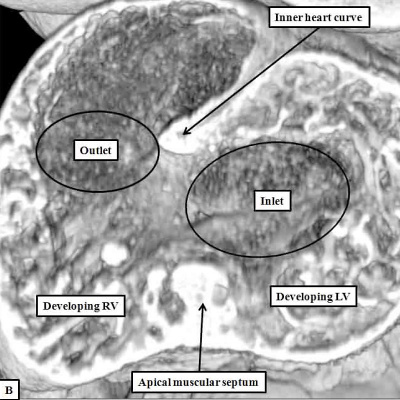
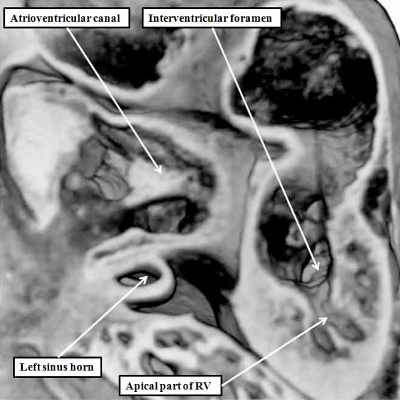
Fig. 8. The cut shown in Panel A, prepared from a mouse at E10.5, is taken through the atrioventricular canal, which opens exclusively to the apical part of the left ventricle, which is ballooning from the inlet part of the ventricular loop (long white arrow). The venous tributaries now open to the right side of the initially common atrium. The primary atrial septum can now be seen growing downwards from the atrial roof. The bracket shows the right side of the atrioventricular junction. There is no direct access from the right atrium to the developing right ventricle. Blood entering the right atrium must pass through the atrioventricular canal, and the interventricular foramen (double headed white arrow) so as to enter the cavity of the developing right ventricle. Panel B is taken from another mouse embryo at E10.5. As had been shown in panel A, the atrioventricular canal (inlet) opens exclusively to the developing left ventricle (LV). The cut in Panel B also shows that the outflow tract (outlet) is supported exclusively by the developing right ventricle (RV). Note that the muscular ventricular septum is developing between the ballooning apical components of the two ventricles. So as to reach the outflow tract, all the atrial blood must pass through the embryonic interventricular foramen, which is bounded cranially by the inner heart curvature, and caudally by the muscular ventricular septum.
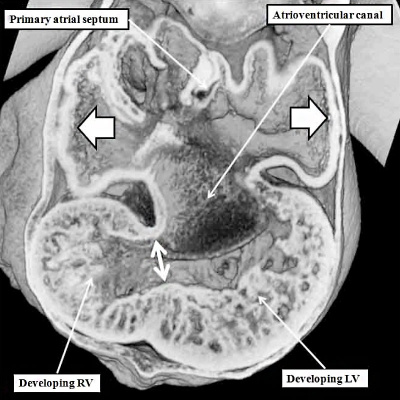
The arrangement as found in the mouse heart, at E10.5, when the atrioventricular canal is supported exclusively by the developing left ventricle, with the outflow tract arising exclusively above the cavity of the developing right ventricle, is replicated in the developing human heart at Carnegie stage 14 (Figure 9).
Fig. 9. The images are prepared from a human heart at Carnegie stage 14 using high resolution episcopic microscopy. The left hand panel shows a section through the atrioventricular (AV) canal, which is supported exclusively above the cavity of the developing left ventricle. Note again the parallel expansion of the atrial appendages from the atrial component of the tube (white arrows with black borders), while the apical components of the ventricles are developing in series. As is shown in the right hand panel, from the same heart but sectioned so as to show the apical part of the developing right ventricle (RV), the outflow tract is supported by the developing right ventricular chamber.
Relationship to the Functionally Univentricular Heart
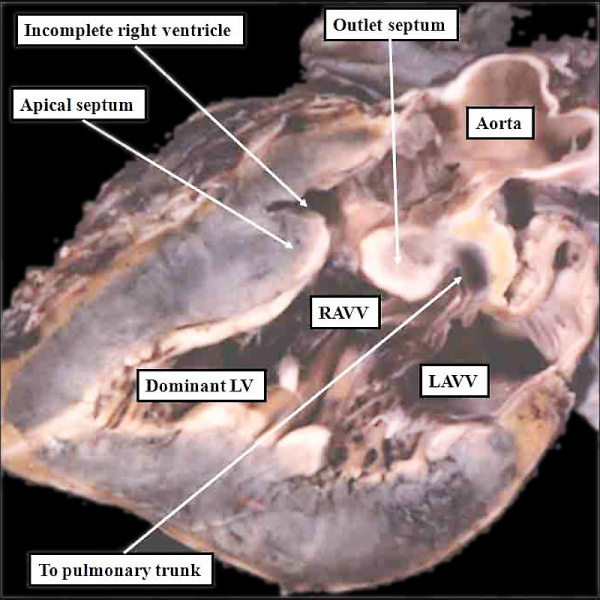
As I will show, the next key stage in cardiac development is rightward expansion of the atrioventricular canal, such that the cavity of the right atrium is able directly to drain to the developing right ventricle. As can be seen in Figures 8 and 9, however, the parietal wall of the right atrium at the rightward margin of the atrioventricular canal is already in continuity with the roof of the right ventricle, even though the circumference of the atrioventricular canal itself is supported above the cavity of the developing left ventricle (right hand panel, Figure 8). If the atrioventricular canal failed to expand, yet the remainder of the heart continued its development, the end result would be that the atrial chambers would retain their connection with the left ventricle. This is the derangement that produces the abnormal situation known as double inlet left ventricle (Figure 10). In this arrangement, the left ventricle remains as the dominant chamber in the ventricular mass, with the right ventricle being incomplete, since it self-evidently lacks its inlet component. In most instances, the right and left atriums, connecting to the dominant left ventricle, possess their own atrioventricular valves, but in a minority of cases, the atrioventricular junctions can be guarded by a common atrioventricular valve, replicating more closely the situation seen in the developing heart (right hand panel, Figure 7). The outflow tract, however, is usually divided in the malformed heart, showing that development of the outflow tract has continued, albeit not always in normal fashion, since in most instances it is the aorta that remains supported by the incomplete right ventricle, with the pulmonary trunk arising from the dominant left ventricle, as in the heart shown in Figure 10.
Fig. 10. The image shows a congenitally malformed heart in which both the right and left atrioventricular valves (RAVV. LAVV) are connected with the dominant left ventricle. The right ventricle is incomplete, and is supplied through a ventricular septal defect, comparable to the embryonic interventricular communication shown in Figure 8, right hand panel. Note that, in this heart, the aorta arises from the incomplete right ventricle, and the pulmonary trunk from the dominant left ventricle. This is the arrangement usually described as “transposition”, but better accounted for in terms of discordant ventriculo-arterial connections.
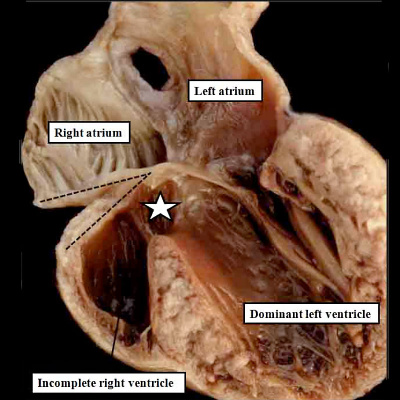
The heart with double inlet left ventricle has, for many years, been described as having a “single ventricle”. Because the atrial chambers connect with only the dominant left ventricle, and the left ventricle then pumps the entirety of the blood volume, the haemodynamic arrangement can appropriately be considered functionally univentricular. In anatomic terms, however, there is a small and incomplete right ventricle accompanying the dominant left ventricle, so the arrangement cannot logically be described in terms of a “single ventricle”. This is just one of the many terminological problems that confront those seeking to understand the anatomy of the congenitally malformed heart. When we consider the developmental implications, nonetheless, the lesion must have required an insult at around the time of 4.5 weeks during human development, or embryonic day 10.5 in the mouse. This is because, as I will describe in the next section, by 5 weeks of development in the human heart, the atrioventricular canal has expanded, such that the cavity of the right atrium is placed in direct communication with the cavity of the right ventricle. In the mouse, this process occurs during the early part of E11.5. A malformation in humans with close similarities to double inlet left ventricle, also involving the atrial chambers connecting only with the dominant left ventricle, is seen in so-called tricuspid atresia. The essence of this lesion is again failure of rightward expansion of the atrioventricular canal, but with growth of the atrial septum so that the right atrium is cut off from the atrioventricular canal. In the postnatal heart, this is seen as absence of the right atrioventricular connection (Figure 11).
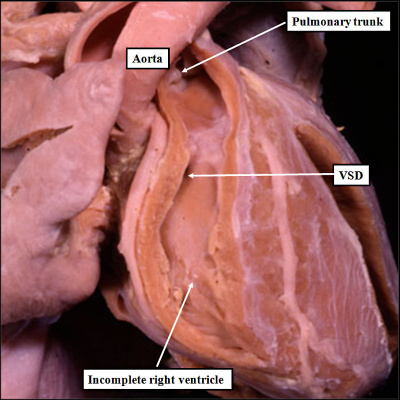
Fig. 11. The images show two different hearts with the lesion known as classical tricuspid atresia. As is seen in the left hand panel, there has been failure of expansion of the atrioventricular junctions, so that the floor of the right is separated from the roof of the right ventricle by the right atrioventricular groove (dashed black lines). Only the dominant left ventricle has an inlet, with the blood entering the incomplete right ventricle through the ventricular septal defect (star). The right hand panel, showing a different heart, is dissected to reveal the structure of the incomplete right ventricle. In this heart, as in most example of tricuspid atresia, it gives rise to the pulmonary trunk. The ventricular septal defect (VSD) is restrictive in this heart.
The heart with tricuspid atresia, like double inlet left ventricle, is also functionally univentricular. Other variants of hearts with double inlet and absent atrioventricular connection can have the right ventricle as the dominant ventricular chamber, implying excessive expansion of the atrioventricular canal, and hence later timing of the process disturbing normal morphological development. The lesions illustrated with the atrioventricular junctions, representing the remnants of the embryonic atrioventricular canal, connecting only with the morphologically left ventricle, nonetheless, serve to show the importance of appreciating the timing of the various events occurring during cardiac development. In the settings with univentricular connection to the dominant left ventricle, the insult must have occurred within the initial five weeks of human development. On very rare occasions, it is also possible to find double inlet to a truly solitary ventricle. This implies failure of ballooning of the apical ventricular components, with the insult presumably occurring even earlier than the one producing double inlet left ventricle.
References
Terms
- angioblastic cords - Groups or ‘columns’ of embryonic precursor cells which will form the walls of both arteries and veins.
- apex - Anatomical term referring to the most inferior, left, downwards pointing part of the heart.
- aorta - The largest artery in the human body originating in the left ventricle. The aorta ascends, arches over the heart and then descends through the abdomen.
- aortic arch arteries - (pharyngeal arch arteries) Each early developing pharyngeal arch contains a lateral pair of arteries arising from the aortic sac, above the heart, and running into the dorsal aorta. Later in development these arch arteries are extensively remodelled to form specific components of the vascular system.
- aortic valve - Three-leaflet valve located at the junction between the left ventricle and aortic entrance.
- aortic vestibule - Smooth-walled portion of the left ventricle directly below the aortic valve.
- aorticopulmonary septum - Division between the aorta and the pulmonary trunk formed from the bulbar ridges.
- atresia - Abnormal closure or absence of a body vessel or orifice.
- atrioventricular canal - Junction between the primitive atrium and primitive ventricle in the embryo. This canal splits to later form two atrioventricular canals which consequently form the valves of the adult heart.
- bulbar ridges - Endocardial cushion tissue located in the bulbus cordis extending into the truncus arteriosus thus forming ridges. These fuse together to form the aorticopulmonary septum.
- bulbus cordis - A region of the early developing heart tube forming the common outflow tract, will differentiate to form three regions of the heart.
- cardiac jelly - Term used in early heart development to describe the initial gelatinous or sponge-like connective tissue separating the myocardium and heart tube endothelium.
- cardiogenic region - The area in the embryo where the precursor cells for heart development lie.
- chordae tendineae - Cord-like tendons connecting the papillary muscles to the leaflets of the mitral and tricuspid valves.
- congenital heart disease - Abnormal structure or function of the heart due to a developmental defect arising prior to birth.
- connective tissue - Fibrous tissue that acts to support body structures or bind other forms of tissue.
- conus arteriosus - An embryological heart outflow structure, that forms in early cardiac development and will later divides into the pulmonary artery and aorta. Term is also used clinically to describe the malformation of the cardiac outflow pattern, where only one artery arises from the heart and forms the aorta and pulmonary artery.
- coronary sinus - a venous sinus emptying into the right atrium that collects blood from the myocardium of the heart. The coronary sinus is the largest cardiac vein.
- cyanosis - Blue colouration of the skin and mucous membrane due to poor oxygenation of the blood.
- dorsal aortae - Two largest arteries either side of the midline which later fuse to form the descending portion of the aorta.
- dorsal mesocardium - The mesentery attaching the heart to the dorsal wall of the pericardial coelom. This breaks down to form a space known as the transverse pericardial sinus.
- dyspnoea - Shortness of breath.
- endocardial cushions - Swellings of migrated cells on the inner lining of the heart located in the atrioventricular canal.
- endocardial heart tubes - Two tubes formed from the cardiogenic plate in the developing embryo. These form the primordium of the truncus arteriosus, the atrium and the ventricles; later invested with myocardium.
- endocardium - The epithelial membrane lining the inside surface of heart, which along with the endothelial layer forms a continuous lining of the entire cardiovascular system. The endocardium, like the majority of the heart is mesoderm in origin.
- endothelium - A simple squamous epithelium lining blood vessels.
- epicardium - The outer layer of heart tissue.
- fibrous trigone - (trigonum fibrosum) term describing the dense connective tissue between the aortic ring and the atrioventricular ring and has a left and right component. The right fibrous trigone (trigonum fibrosum dextrum) lies between the aortic ring and the right atrioventricular ring. The left fibrous trigone (trigonum fibrosum sinistrum) lies between the aortic ring and the left atrioventricular ring.
- foramen ovale - Shunt allowing blood to enter the left atrium from the right atrium. It is located in the septum secundum.
- foramen primum - Original space between the septum primum and the fused endocardial cushions as the septum primum grows towards the cushions.
- foramen secundum - Refers to the coalesced perforations in the septum primum after it has fused with the endocardial cushions.
- fossa ovalis - (oval fossa, annulus ovalis) Region in the postnatal atrial septum where the foramen ovale was located during embryonic development. Appears as an indentation in the right atrium septum.
- heart murmur - Extra heart sounds appearing upon auscultation due to turbulent blood flow.
- hypertrophy - Increase in size of an organ or tissue due to enlargement of component cells.
- inferior vena cava - (IVC) Large vein which carries deoxygenated blood from the lower half of the body to the right atrium.
- inflow tract - Entrance of blood into the heart tube; the sinus venosus portion of the tube.
- infundibulum - Smooth-walled portion of the right ventricle directly below the pulmonary valve.
- interventricular septum - Wall of muscular tissue growing from the base of the heart dividing the primitive ventricle into the left and right ventricles.
- interventricular foramen - Space between the interventricular septum and the fused endocardial cushions. The foramen closes when the septum fuses with the endocardial cushions and bulbar ridges.
- intraembryonic coelom - Initial embryonic space in lateral plate mesoderm that will be separated to form the three major body cavities: pericardial, pleural and peritoneal cavity. The lateral plate mesoderm is divided by this space into splanchnic and somatic mesoderm.
- left horn of sinus venosus - The left side of the sinus venosus (initially symmetrical with the right) collecting blood from half of the paired veins: common cardinal veins, umbilical veins and vitelline veins. Later the left horn diminishes and becomes the small coronary sinus.
- mitral valve - (Bicuspid valve) two leaflet valve located on the left side of the heart, that is between the left atrium and ventricle.
- myocardium - The middle layer of the heart wall composed of cardiac muscle.
- neural crest mesenchyme - Connective tissue arising from critical cells in the cranial region of the embryo. These paired dorsal lateral streaks of cells migrate throughout the embryo and can differentiate into many different cell types (= pluripotential). Those that remain on the dorsal neural tube form the sensory spinal ganglia (DRG), those that migrate ventrally form the sympatheitic ganglia. Neural crest cells also migrate into the somites and regions through the entire embryo.
- outflow tract - Exit of blood from the heart tube formed by the truncus arterioles.
- oval fossa - (fossa ovalis, annulus ovalis) Region in the postnatal atrial septum where the foramen ovale was located during development. Appears as an indentation in the right atrium septum.
- papillary muscles - Small muscles found on the inner myocardium of the left and right ventricles. They are attached to the mitral and tricuspid valves via the chordae tendineae and serve to limit the movements of the valves.
- patent foramen ovale - Abnormality in atrial septum due to failure of the atrial septum to close at the foramen ovale. (More? Atrial Septal Defects)
- pericardial coelom - (pericardial cavity) The anatomical body cavity in which the heart lies. The pericardial cavity forms in the lateral plate mesoderm above the buccopharyngeal membrane, as part of the early intraembryonic coelom. This cavity is initially continuous with the two early pleural cavities. Note the single intraembryonic coelom forms all three major body cavities: pericardial cavity, pleural cavity, peritoneal cavity.
- primordial atrium - Common cavity in the upper portion of the developing heart. Later divides to form the left and right atria.
- primordial ventricle - Common cavity in the lower portion of the developing heart. Later divides to form the left and right ventricles.
- pulmonary circulation - Carries blood between the heart and lungs.
- pulmonary trunk - A vessel that arises from the right ventricle of the heart, extends upward, and divides into the right and left pulmonary arteries that transport deoxygenated blood to the lungs.
- pulmonary valve - Three-leaflet valve located at the junction between the right ventricle and the pulmonary trunk.
- pulmonary veins - Four veins that allow oxygenated blood from the lungs to empty into the left atrium.
- right horn of sinus venosus - The right side of the sinus venosus (initially symmetrical with the left) collecting blood from half of the paired veins: common cardinal veins, umbilical veins and vitelline veins. Later the right horn dilates, receiving all the veins, and becomes the sinus venarum of the right atrium.
- second heart field - (SHF) splanchnic mesoderm that forms progenitors important for heart formation.
- semilunar valves - Flaps of endocardium and connective tissue reinforced by fibres which prevent the valves from turning inside out. They are shaped like a half moon, hence the name semilunar. The semilunar valves are located between the aorta and the left ventricle and between the pulmonary artery and the right ventricle.
- septum primum - Original structure growing from the roof of the heart towards the endocardial cushions dividing the primitive atrium.
- septum secundum - Second structure growing to the right of the septum primum dividing the primitive atrium.
- sinoatrial node - Specialised cardiomyocyte pacemaking region of the heart located at the entry of the right superior caval vein (SVC) into the right atrium. (More? Detailed Cardiac - Sinus Node)
- sinus venosus - (systemic venous sinus) An early developmental cardiovascular structure, thin walled cavity, forming the input to developing heart which has 3 venous inputs (vitelline vein, umbilical vein, common cardinal vein). Later in heart development this structure gets incorporated into the wall of the future right atrium. (More? Systemic Venous Sinus)
- sinuatrial orifice - The opening between the sinus venosus and right atrium which has two valve leaflets to prevent backflow of blood.
- sinus venarum - Smooth-walled portion of the adult right atrium; originally the left horn of the sinus venous.
- splanchnic mesoderm - Gastrointestinal tract (endoderm) associated mesoderm formed by the separation of the lateral plate mesoderm into two separate components by a cavity, the intraembryonic coelom. Splanchnic mesoderm is the embryonic origin of the second heart field, gastrointestinal tract connective tissue, smooth muscle, blood vessels and contribute to organ development (heart, pancreas, spleen, liver).
- stenosis - Abnormal narrowing of a blood vessel or orifice.
- superior vena cava - (SVC) Short vein which carries deoxygenated blood from the upper half of the body to the right atrium.
- systemic circulation - Carries oxygenated blood away from the heart to the other body organs and returns to the heart deoxygenated.
- tachypnoea - Abnormally rapid breathing rate.
- trabeculae - (trabeculations)Muscular beams located within the ventricles and parts of the atria of the heart.
- transverse pericardial sinus - Dorsal area within the pericardial coelom, initially occupied by the dorsal mesocardium.
- tricuspid valve - Three leaflet valve located in the right atrioventricular canal i.e. between the right atrium and ventricle.
- tricuspid atresia - Abnormality of the tricuspid valve between the right atrium and right ventricle.
- truncus arteriosus - An embryological heart outflow structure, that forms in early cardiac development and will later divides into the pulmonary artery and aorta. Term is also used clinically to describe the malformation of the cardiac outflow pattern, where only one artery arises from the heart and forms the aorta and pulmonary artery.
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Cite this page: Hill, M.A. (2026, Mayıs 28) Embryology Detailed Cardiac - Systemic Venous Sinus. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Detailed_Cardiac_-_Systemic_Venous_Sinus
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G