Trophoblast: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 17: | Line 17: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''The trophoblast plug during early pregnancy: a deeper insight'''<ref name=PMID27510415><pubmed>PMID27510415</pubmed></ref> | |||
"During the first trimester of pregnancy, foetal endovascular trophoblasts invade into maternal spiral arteries, accumulate and form plugs in the lumen of the vessels. These plugs only allow blood plasma to seep through. Hence, during the first trimester of pregnancy, a first flow of fluids through the placental intervillous space is established, resulting in a physiological oxygen gradient between mother and foetus. The trophoblast plugs block spiral arteries until the beginning of the second trimester (11-14 weeks)." | |||
* '''MSX2 Induces Trophoblast Invasion in Human Placenta'''<ref name=PMID27088357><pubmed>27088357</pubmed></ref> "Normal implantation depends on appropriate trophoblast growth and invasion. Meanwhile, overexpression of MSX2 and treatment with the MSX2-specific siRNA resulted in decreased and increased E-cadherin expression, respectively, in JEG-3 cells. Lastly, the protein expression levels of MSX2 were significantly lower in human pre-eclamptic placental villi than in the matched control placentas. Collectively, our results suggest that MSX2 may induce human trophoblast cell invasion, and dysregulation of MSX2 expression may be associated with pre-eclampsia." | * '''MSX2 Induces Trophoblast Invasion in Human Placenta'''<ref name=PMID27088357><pubmed>27088357</pubmed></ref> "Normal implantation depends on appropriate trophoblast growth and invasion. Meanwhile, overexpression of MSX2 and treatment with the MSX2-specific siRNA resulted in decreased and increased E-cadherin expression, respectively, in JEG-3 cells. Lastly, the protein expression levels of MSX2 were significantly lower in human pre-eclamptic placental villi than in the matched control placentas. Collectively, our results suggest that MSX2 may induce human trophoblast cell invasion, and dysregulation of MSX2 expression may be associated with pre-eclampsia." | ||
* '''OVO-like 1 regulates progenitor cell fate in human trophoblast development'''<ref>Renaud, SJ. entail., OVO-like 1 regulates progenitor cell fate in human trophoblast development [http://www.pnas.org/content/112/45/E6175.abstract.html?etoc PNAS]</ref> "Epithelial barrier integrity is dependent on progenitor cells that either divide to replenish themselves or differentiate into a specialized epithelium. This paradigm exists in human placenta, where cytotrophoblast cells either propagate or undergo a unique differentiation program: fusion into an overlying syncytiotrophoblast. Syncytiotrophoblast is the primary barrier regulating the exchange of nutrients and gases between maternal and fetal blood and is the principal site for synthesizing hormones vital for human pregnancy. How trophoblast cells regulate their differentiation into a syncytium is not well understood. In this study, we show that the transcription factor OVO-like 1 (OVOL1), a homolog of Drosophila ovo, regulates the transition from progenitor to differentiated trophoblast cells. OVOL1 is expressed in human placenta and was robustly induced following stimulation of trophoblast differentiation. Disruption of OVOL1 abrogated cytotrophoblast fusion and inhibited the expression of a broad set of genes required for trophoblast cell fusion and hormonogenesis. OVOL1 was required to suppress genes that maintain cytotrophoblast cells in a progenitor state, including MYC, ID1, TP63, and ASCL2, and bound specifically to regions upstream of each of these genes." [http://www.omim.org/entry/602313 OMIM - OVOL1] | * '''OVO-like 1 regulates progenitor cell fate in human trophoblast development'''<ref>Renaud, SJ. entail., OVO-like 1 regulates progenitor cell fate in human trophoblast development [http://www.pnas.org/content/112/45/E6175.abstract.html?etoc PNAS]</ref> "Epithelial barrier integrity is dependent on progenitor cells that either divide to replenish themselves or differentiate into a specialized epithelium. This paradigm exists in human placenta, where cytotrophoblast cells either propagate or undergo a unique differentiation program: fusion into an overlying syncytiotrophoblast. Syncytiotrophoblast is the primary barrier regulating the exchange of nutrients and gases between maternal and fetal blood and is the principal site for synthesizing hormones vital for human pregnancy. How trophoblast cells regulate their differentiation into a syncytium is not well understood. In this study, we show that the transcription factor OVO-like 1 (OVOL1), a homolog of Drosophila ovo, regulates the transition from progenitor to differentiated trophoblast cells. OVOL1 is expressed in human placenta and was robustly induced following stimulation of trophoblast differentiation. Disruption of OVOL1 abrogated cytotrophoblast fusion and inhibited the expression of a broad set of genes required for trophoblast cell fusion and hormonogenesis. OVOL1 was required to suppress genes that maintain cytotrophoblast cells in a progenitor state, including MYC, ID1, TP63, and ASCL2, and bound specifically to regions upstream of each of these genes." [http://www.omim.org/entry/602313 OMIM - OVOL1] | ||
Revision as of 14:22, 23 August 2016
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction

(Greek, trophe = "nutrition" and -blast, a primordial cell) During early development the trophoblast cells have an important contribution to extraembryonic tissues (fetal placenta and membranes) and processes of early development (adplantation, implantation and endocrine support of pregnancy).
In humans, week 1 blastocyst formation the outer layer of cells (adjacent to the zona pellucida) form a flat squamous epithelial layer of cells, the trophoblast layer. Week 2 following blastocyst hatching the trophoblast layer is involved with initial adhesion to the uterine wall and subsequent implantation within the wall. During this period the trophoblast layer differentiates into two distinct layers (syncitiotrophoblast and cytotrophoblast).
- Links: Blastocyst | Implantation | Week 2 | Week 3 | Placenta Development | Trophoblast - Protein Expression | Ectopic Implantation Research
Some Recent Findings
"During the first trimester of pregnancy, foetal endovascular trophoblasts invade into maternal spiral arteries, accumulate and form plugs in the lumen of the vessels. These plugs only allow blood plasma to seep through. Hence, during the first trimester of pregnancy, a first flow of fluids through the placental intervillous space is established, resulting in a physiological oxygen gradient between mother and foetus. The trophoblast plugs block spiral arteries until the beginning of the second trimester (11-14 weeks)."
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Trophoblast <pubmed limit=5>Trophoblast</pubmed> |
| Older papers |
|---|
|
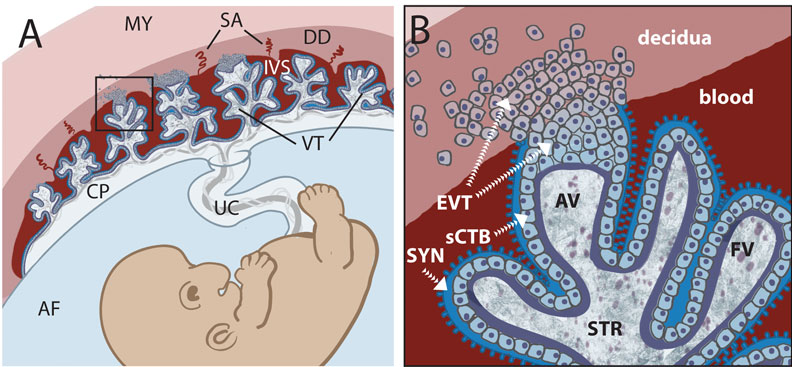
Trophoblast and Placental Villi
Early placental development cartoon showing trophoblast contribution to placental villi.[11]
Legend
- SYN - syncytiotrophoblasts
- sCTB - subsyncytial cytotrophoblasts (this layer grows increasingly discontinuous in later trimesters)
- EVT - extravillous cytotrophoblasts (anchor the villous tree in the decidua)
Cytotrophoblasts
Hyperglycosylated human Chorionic Gonadotropin (hCG) promotes the growth of cytotrophoblast cells and the endometrial invasion by these cells during implantation.[12]
Extravillous Cytotrophoblasts
Maintain open maternal spiral arteries emptying into the maternal lacunae[13]
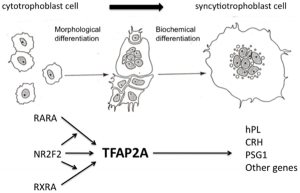
Syncytiotrophoblasts
- form by fusion of rapidly dividing cytotrophoblast cells
- secrete proteolytic enzymes, enzymes break down extracellular matrix around cells
- Allow passage of blastocyst into endometrial wall, totally surround the blastocyst
- generate spaces, lacunae, that fill with maternal blood
- secrete Human Chorionic Gonadotropin (hCG), hormone, maintains decidua and Corpus Luteum, basis of pregnancy diagnostic test, present in urine is diagnostic of pregnancy
- Later in development placenta will secrete hCG
Cell–cell Fusion Activity
Two pairs of envelope genes of retroviral origin, syncytins, have fusogenic properties. Human endogenous retroviruses (HERVs) make up about 8% of the human genome. Recent study has shown that placentae from intrauterine growth restriction have impaired cell fusion and differentiation that correlates with reduced levels of HERV envelope genes.[14]
Human - syncytin 1 and syncytin 2
- Syncytin 2 is an envelope gene from the human endogenous retrovirus FRD (HERV-FRD)[15]
- Syncytin 2 receptor is Major Facilitator Superfamily Domain Containing 2 (MFSD2) (at chromosomal position 1p34.2)
- belongs to family of presumptive carbohydrate transporters with 10-12 membrane-spanning domains
- Syncytin 1 (-FRD) receptor is ASCT-1/-2
Mouse - syncytin A and syncytin B
- syncytin A is essential for trophoblast cell differentiation and syncytiotrophoblast morphogenesis[16]
- Links: OMIM Syncytin-2
Human Chorionic Gonadotropin
hCG sources - produced by villous syncytiotrophoblast cells, hyperglycosylated hCG produced by cytotrophoblast cells, free beta-subunit made by multiple primary non-trophoblastic malignancies, and pituitary hCG made by the gonadotrope cells of the anterior pituitary.
- levels peak at 8 to 10 weeks of pregnancy, then decline and are lower for rest of pregnancy
- 0-1 week: 0-50 mIU/ml
- 1-2 weeks: 40-300 mIU/ml
- 3-4 weeks: 500-6,000 mIU/ml
- 1-2 months: 5,000-200,000 mIU/ml
- 2-3 months: 10,000-100,000 mIU/ml
- 2nd trimester: 3,000-50,000 mIU/ml
- 3rd trimester: 1,000-50,000 mIU/ml
Non-pregnant females: <5.0 mIU/ml Postmenopausal females: <9.5 mIU/ml
HLA-G
An acronym for histocompatibility antigen, class I, G (also called: Human Leukocyte Antigen G, (HLA-6.0; HLA60, T-CELL A LOCUS, TCA) and is expressed on placental cytotrophoblast cells and other adult tissues. This distinct tissue distribution differs from the other HLA antigens (HLA-A, HLA-B, HLA-C) leading to the description as a non-classical class I antigen. May have a role in protecting the fetus from the maternal immune response.
Human gene is located at 6p22.1 and there exist several protein isoforms from alternative splicing of messenger RNAs, membrane-bound isoforms (HLA-G 1-4) and soluble soluble (HLA-G 5-6)[17]. The molecule is a heterodimer consisting of both a heavy chain and a light chain (beta-2 microglobulin). The membrane-bound isoform heavy chain is anchored in the membrane and increased expression of the soluble form is related to higher implantation rates. Changes in HLA-G expression have been associated with increased miscarriage rates.[18] Killer cell immunoglobulin-like receptor (KIR) 2DL4 (KIR2DL4) has been shown to act as a receptor for the soluble HLA-G, leading to a stimulation of resting natural killer (NK) cells.[19]
- Links: HLA-G | OMIM142871
Trophoblast Cell Lines
A useful component of current placentation and trophoblast research are a number of trophoblast cell lines that have been derived from a number of different sources including term placenta, choriocarcinomas and transformation of primary isolated cells. BeWo cell line is the earliest human choriocarcinoma, established in 1968[20]. Most appear to be examples of extravillous trophoblast cells, some examples of various human "trophoblast" cell lines are listed below.
- BeWo - human choriocarcinoma[20], hypotetraploid (modal number = 86; range = 71 to 178) (ATCC CCL-98)
- JEG-3 - choriocarcinoma HTR8/SVneo a transformed extravillous trophoblast line
- SGHPL-4 - [21][22]
- TEV-1 - (HPV16) E6/E7 infection first-trimester extravillous trophoblast cell line[23]
- ACH-3P - fusion of primary human first trimester trophoblasts (GA week 12) with a human choriocarcinoma cell line (AC1-1).[24]
- HChEpC1b - retroviral infection by E6/E7/hTERT[25]
History
The name "Trophoblast" was used for the first time by Ambrosius Arnold Willem Hubrecht (1853 – 1915) at the meeting of the Anatomical Congress at Wiirzburg in 1888, and its earliest definition is found in the report of that meeting in Nos. 17 and 18 of the Anatomischer Anzeiger, Bd. III. "We there read, concerning a very early stage of the hedgehog (p. 510) : Die aussere Wand der Keimblase ist verdickt (drei bis vierschichtig) und besitzt wabige Lacunen. Fur diese aussere (epiblastische) Schicht sei der. Name Trophoblast gewahlt."
- Links: The Trophoblast - A Rejoinder (1904) | Ambrosius Arnold Willem Hubrecht | Hubrecht Collection | Embryologists | Embryology History
Abnormalities
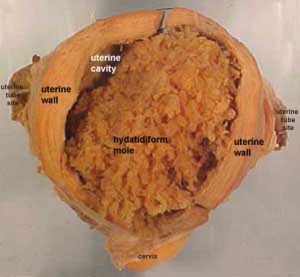
Hydatidiform Mole
A type of placental abnormality when only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole", which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus. The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo.
There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
This topic is also covered in Placenta - Abnormalities
Trophoblast Infections
- Malaria (More? Abnormal Development - Malaria)
- Chlamydia trachomatis[26]
- Herpesvirus 8 (HHV-8)[27]
- Group B streptococcus (GBS)[28]
References
- ↑ <pubmed>19924284</pubmed>| PMC2773928 | PLoS One
- ↑ <pubmed>16681851</pubmed>| PMC1479373 | J Transl Med.
- ↑ <pubmed>PMID27510415</pubmed>
- ↑ <pubmed>27088357</pubmed>
- ↑ Renaud, SJ. entail., OVO-like 1 regulates progenitor cell fate in human trophoblast development PNAS
- ↑ <pubmed>25203285</pubmed>
- ↑ <pubmed>24186980</pubmed>| Genes Dev.
- ↑ <pubmed>22423048</pubmed>
- ↑ <pubmed>20195529</pubmed>| PLoS One
- ↑ <pubmed>20338637</pubmed>
- ↑ <pubmed>20107601</pubmed>| PLoS
- ↑ <pubmed>20735820</pubmed>
- ↑ <pubmed>20359743</pubmed>
- ↑ <pubmed>20664994</pubmed>
- ↑ <pubmed>18988732</pubmed>| PNAS
- ↑ <pubmed>19564597</pubmed>
- ↑ <pubmed>16339579</pubmed>
- ↑ <pubmed>24557735</pubmed>
- ↑ <pubmed>16366734</pubmed>
- ↑ 20.0 20.1 <pubmed>5753554</pubmed>
- ↑ <pubmed>9804259</pubmed>
- ↑ <pubmed>16226120</pubmed>
- ↑ <pubmed>15866109</pubmed>
- ↑ <pubmed>18093301</pubmed>
- ↑ <pubmed>19148547</pubmed>
- ↑ <pubmed>19265152</pubmed>
- ↑ <pubmed>19115001</pubmed>
- ↑ <pubmed>18599257</pubmed>
Reviews
<pubmed>19876840</pubmed>
Articles
<pubmed>20081188</pubmed> <pubmed>20033790</pubmed> <pubmed>19617545</pubmed> <pubmed>19564597</pubmed> <pubmed>19582159</pubmed>
Search PubMed
Search October 2010
- Syncytiotrophoblast - All (10230) Review (1104) Free Full Text (2508)
- Cytotrophoblast - All (9828) Review (1062) Free Full Text (2401)
Search Pubmed: Trophoblast | syncytiotrophoblast | cytotrophoblast |
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Trophoblast. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Trophoblast
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G