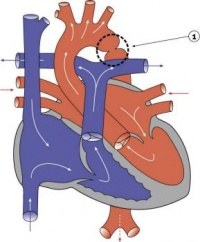
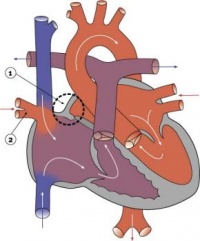
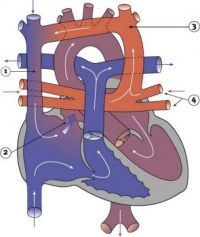
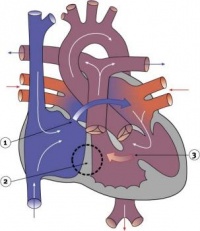
Fetal ECHO Meeting 2012
Introduction
Fetal ECHO Meeting 5th-8th October 2012 ROYAL PRINCE ALFRED HOSPITAL, SYDNEY, AUSTRALIA
This presentation will introduce development of the heart from its earliest origins through to the end of the embryonic period. The main morphological changes in the human heart will be shown in the context of the normal human timeline of development.
Developmental Timings
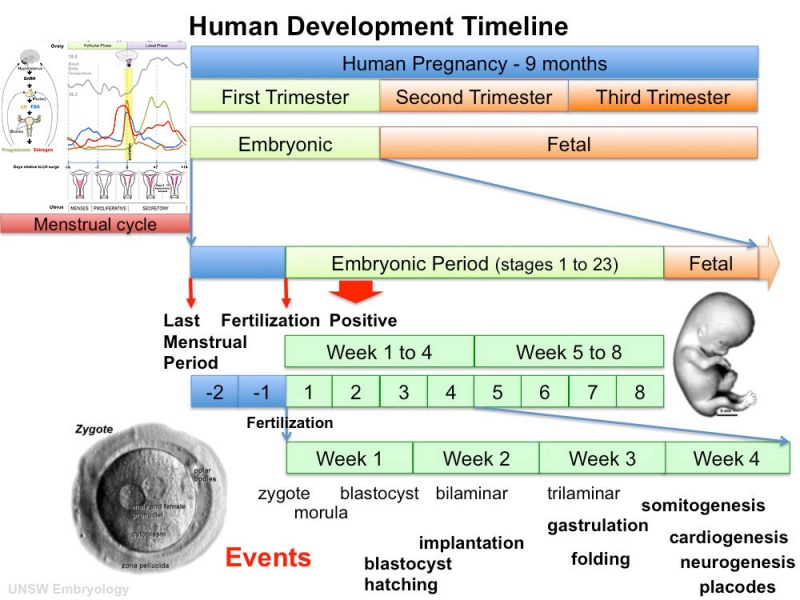
- Clinical timings refer to Gestational Age (GA) from the last day of the Last Menstrual Period (LMP).
- Embryonic timings refer to Ovulation age (OA) from day 14 of the cycle when fertilization occurs.
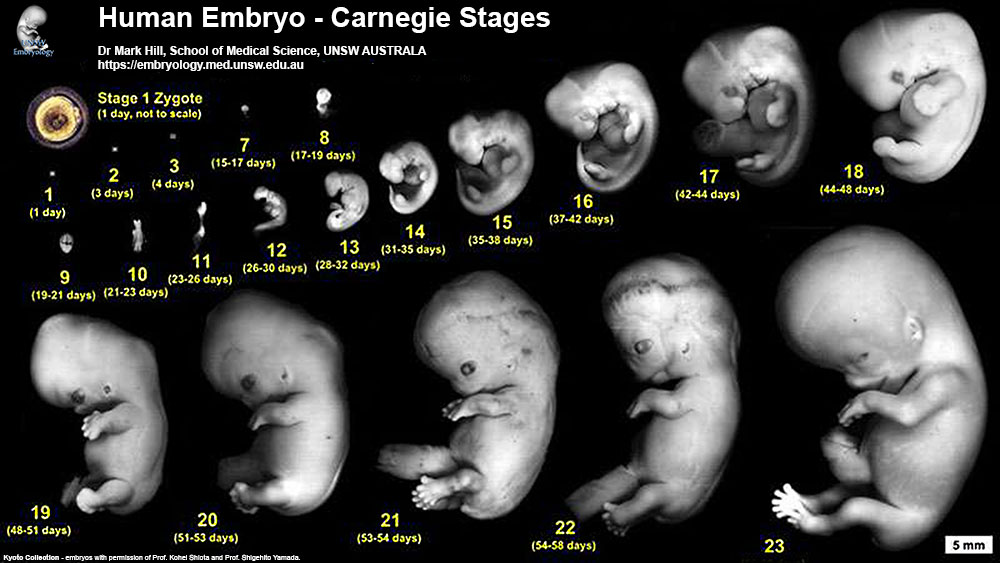
Human Embryo Development
Weeks shown are ovulation age (OA) for gestational age (GA) add 2 weeks.
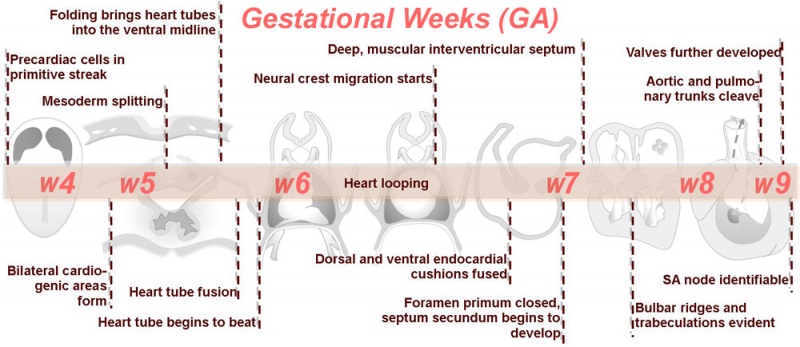
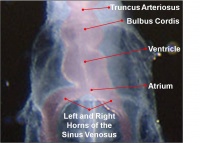
Heart Embryonic Timeline
Weeks shown above are for clinical Gestational Age (GA). Ages shown below are Ovulation Age (OA), subtract 2 weeks from GA.
| Begin Advanced | Heart Fields | Heart Tubes | Cardiac Looping | Cardiac Septation | Outflow Tract | Valve Development | Cardiac Conduction | Cardiac Abnormalities | Molecular Development |
Heart Field
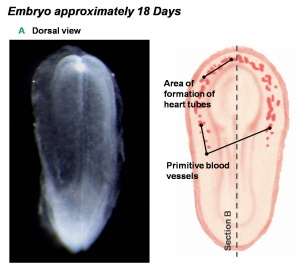
Dorsal view of 18 day embryo |
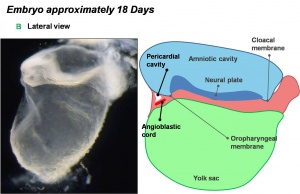
Lateral view of 18 day embryo | ||||||
|
|
- The heart primordium arises predominantly from the primary heart field in splanchnic mesoderm forming in the cardiogenic region of the trilaminar embryo.
- The cardiogenic region can be thought of as bilateral fields that merge cranially to form a horseshoe-shaped field.
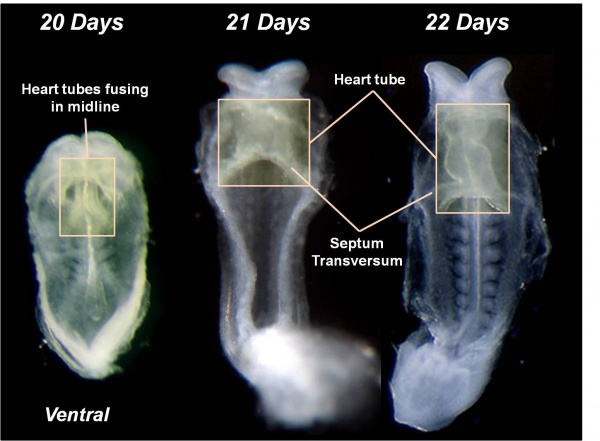
- During the third week (PO) of development (approximately day 18) angioblastic cords develop in this cardiogenic mesoderm and canalise to form bilateral endocardial heart tubes.
- The secondary heart field has been described as pharyngeal mesenchyme that contributes myocardium and smooth muscle to the arterial pole.
Heart Tube
- The endothelium of the heart tube forms the internal endocardium.
- The epicardium develops from mesothelial cells arising from the sinus venosus, that spread cranially over the myocardium.
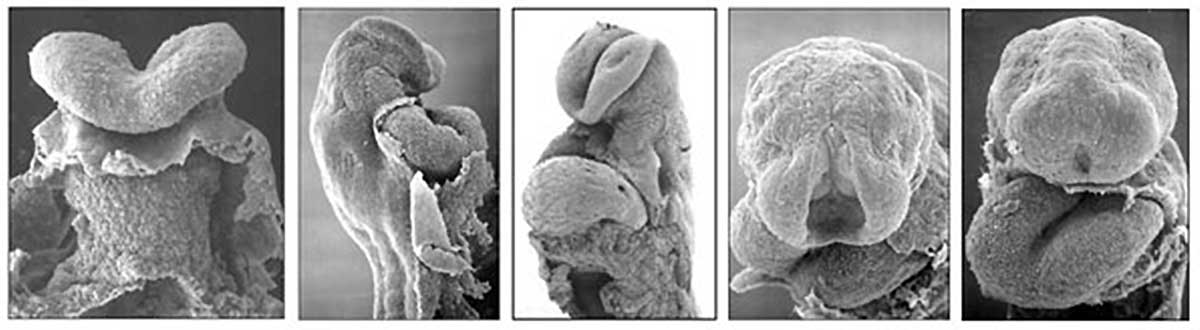
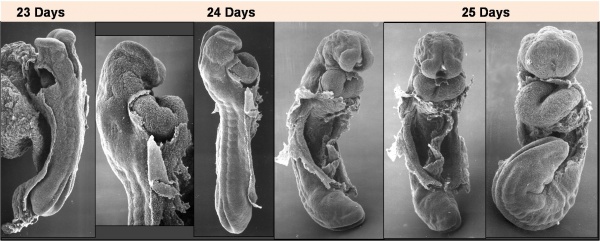
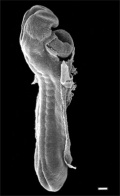
Cardiac Looping
Image day 21 to 25 (GA Week 5)
- First stage occurs from late in the sixth week (GA) to early in the seventh week (GA)
- studied in chick embryos whose initial, straight heart tube is representative of that developing in humans.
- Second stage (creating S-shape) occurs as the ventricular bend moves caudally and the distance between the outflow and inflow tracts diminishes.
- dorsal mesocardium degenerates forming the transverse pericardial sinus (a point of communication across the pericardial coelom).
- atrial and outflow poles converge and myocardial cells are added, forming the truncus arteriosus.
- Final stage is the wedging of the aorta between the atrioventricular (AV) valves.
- occurs during septation and is dependent on the retraction and rotation of the myocardium by 45°.
Left-Right Asymmetry
- Heart is the first organ in the body to express left-right asymmetry in the form of looping.
- Axis of the heart is established during gastrulation.
- Left - Shh is expressed in the left side of Henson’s node in the primitive streak. Leads to expression of Nodal and Pitx2 in left sided mesoderm. Pitx2 expressed in the left side of the heart tube is then responsible for asymmetric organogenesis.
- Right - Shh inhibited by the activation of Activin IIa receptors.
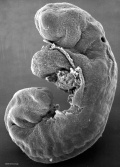
|
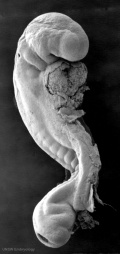
|
|
| Stage 11 | Stage 12 | Stage 13 |
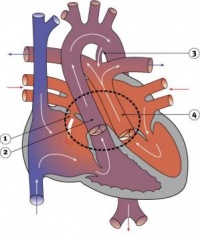
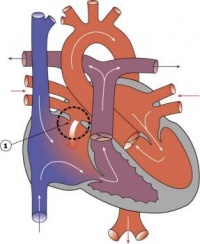
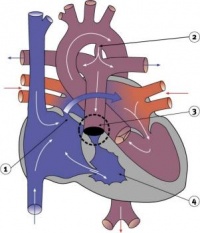
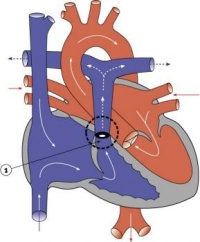
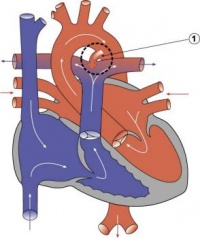
Septation
Atrioventricular Canal
- Partitioning of the primitive heart occurs between the middle of the week 6 (GA) and the end of week 7 (GA).
- Division of the atrioventricular canal is described below while
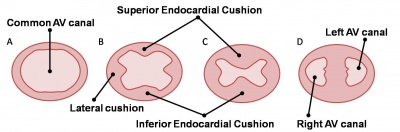
Two endocardial cushions form on the dorsal and ventral surfaces of the AV canal. Following expansion of the cardiac jelly, epithelial to mesenchymal transformation (EMT) of the endocardial cells in the canal occurs forming the cushions. Synergistic signalling between BMP and TGFβ facilitates EMT. The cushions grow as they are invaded by mesenchymal cells from the endocardium during the fifth week, eventually fusing to create the right and left AV canals, hence partially separating the primitive atrium and ventricle.
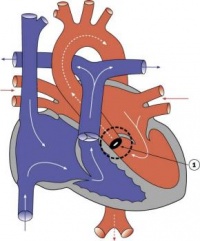
Atrial and Ventricular Septation
| Development of the Atria |
|---|
| Membranous tissue forming the septum primum grows from the roof of the atrium, dividing it into left and right halves. The septum primum originates from myocardium that differentiates from splanchnic mesoderm near the venous pole and approaches the endocardial cushions. The foramen primum refers to the decreasing communication between the septum primum and endocardial cushions. The junction of the septum primum and endocardial cushions becomes myocardialised by ingrowth of myocardial cells, although the centre is maintained as dense connective tissue and is referred to as Todaro’s tendon. Apoptosis-induced perforations appear in the centre of the septum primum to produce the foramen secundum. At this time, the strong, muscular septum secundum grows immediately to the right of the septum primum and gradually overlaps the foramen secundum during the fifth and sixth weeks of development. Part of the atrial septum and dorsal right atrium, as well as the septum secundum develop from left-sided mesenchyme. The incomplete partition of the atrium by the septum secundum forms the foramen ovale. Blood flows through the foramen ovale and foramen secundum to the left atrium. The remaining portion of the septum primum acts as the valve of the foramen ovale. Blood cannot flow in the opposite direction as the muscular strength of the septum secundum prevents prolapse of the septum primum.
Remodelling of the venous pole, including the further induction of myocardial cells, contributes to the development of the atria. In the mouse, myocardial differentiation occurs in the dorsal mesocardium and cells are then recruited to the venous pole. The development of two left to right shunts in the venous system leads to an increase in the right horn of the sinus venosus and consequentially a decrease in left horn by the end of the fourth week. The sinuatrial orifice correspondingly shifts to the right thus becomes located in the right atrium. Hence the right atrium receives the superior vena cava (SVC) and inferior vena cava (IVC) in the adult. In mice and chicks the left sinus horn develops as the left SVC, however this regresses to form the coronary sinus in humans. Thus the sinus venosus gradually becomes incorporated into the right atrium. It contributes to the smooth walled part of the adult right atrium, referred to as the sinus venarum. The trabeculated right atrium corresponds to the primordial atrium; the division between these structures is indicated by the inner crista terminalis and outer sulcus terminalis. The primordial pulmonary vein develops in the dorsal wall of the left atrium. As the atrium increases in size it incorporates more of the branches of the pulmonary vein, culminating in its receiving the four pulmonary veins. The smooth wall of the adult left atrium originated from the primordial pulmonary vein, while the trabeculated wall represents the primordial atrium. |
| Development of the Ventricles |
|---|
|
Minor trabeculations appear during early development of the primordial ventricle. Following growth of the ventricles further trabeculations appear and grow as larger, muscular structures. Some authors tout the idea that as trabeculations grow they coalesce resulting in the formation of the ventricular septum. However, the more commonly described theory of septation begins with the appearance of a primordial muscular interventricular (IV) ridge developing in the floor of the ventricle near the apex. As either side of the ventricle grows and dilates, their medial walls fuse forming the prominent IV septum. The foramen located between the cranial portion of the IV septum and the endocardial cushions, the IV foramen, closes by the end of the seventh week as the bulbar ridges fuse with the endocardial cushions. |
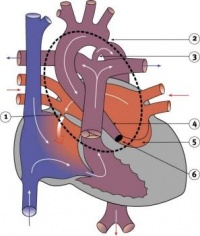
Outflow Tract Septation
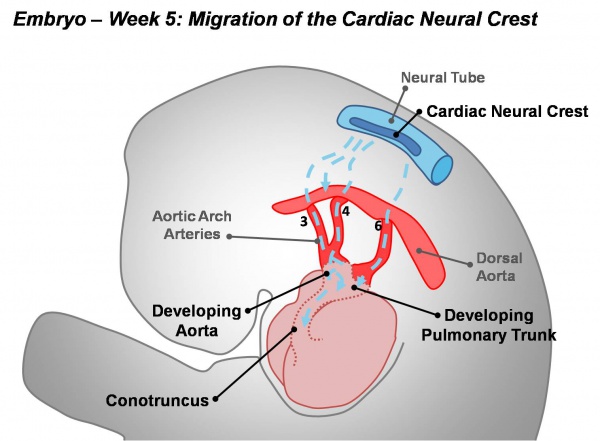
- Cardiac neural crest - levels associated with somites 1 and 3 of the neural tube migrate through the pharyngeal arches to contribute to the conotruncal septum.
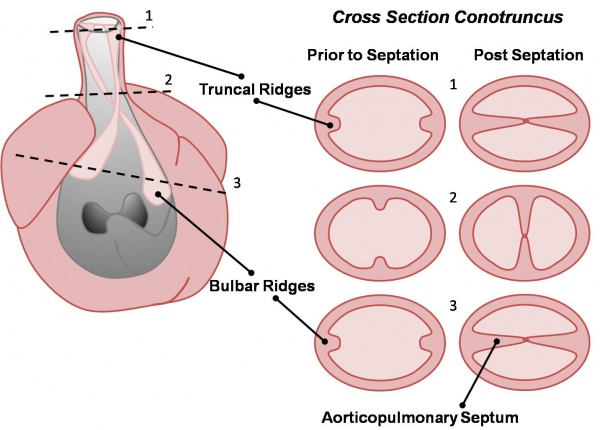
- Week 7 (GA) - proliferation of pharyngeal mesenchyme in the bulbus cordis forms bulbar ridges, continuous in the truncus arteriosus. Cardiac neural crest migrates into these ridges, condensing as cellular columns to support the outflow tract septum. The ridges form a 180° spiral to create the helical aorticopulmonary septum. Myocardialisation of the ridges gives a zippering effect resulting in fusion.
- Week 8 (GA) - fusion occurs distal to proximal direction, allowing for cleavage of the aorta and pulmonary trunk. The spiralling nature of the ridges causes the pulmonary trunk to twist around the aorta. The bulbus cordis accounts for the smooth conus arteriosus (or infundibulum) in the right ventricle and the aortic vestibule in the left ventricle.
- Links: Movie - Outflow tract
Heart Week 7 and 10 (GA)
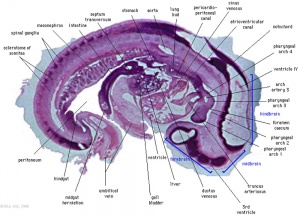
|
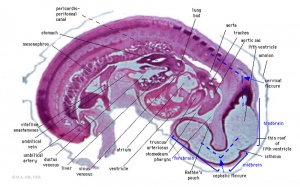
|
| G6L | G7L |
| Stage 13 Embryo (GA Week 7) | ||||||
|---|---|---|---|---|---|---|
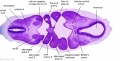
|
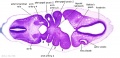
|
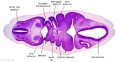
|
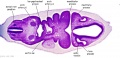
|
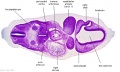
|
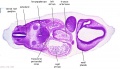
|
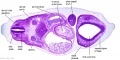
|
| B1L | B2L | B3L | B4L | B5L | B6L | B7L |
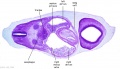
|
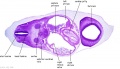
|
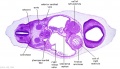
|
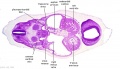
|
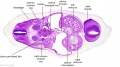
|
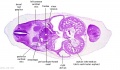
|
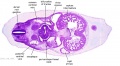
|
| C1L | C2L | C3L | C4L | C5L | C6L | C7L |
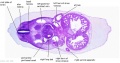
|
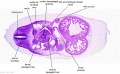
|
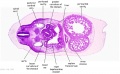
|
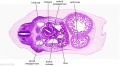
|
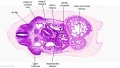
|
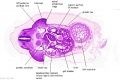
|
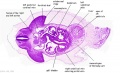
|
| D1L | D2L | D3L | D4L | D5L | D6L | D7L |
| Stage 22 Embryo (GA Week 10) | ||
|---|---|---|
| Section | Name | Description |
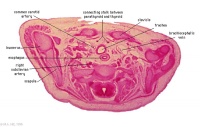
|
C6L | L and R common carotid arteries. L and R internal jugular veins. |
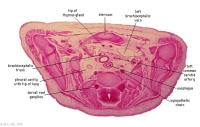
|
C7L | L brachiocephalic vein (transverse anastomosis of L, R jugular veins). L common carotid artery. Brachiocephalic trunk. |
|
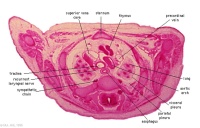
D1L | Aortic arch. Thymus (retrosternal). Trachea. Oesophagus. Large L and R jugular veins. |
|
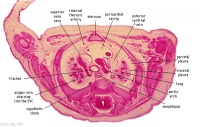
D2L | Aortic arch. L jugular lymph sac lying laterally with hemiazygos joining it. Trachea. Oesophagus. Draining of azygos vein into R jugular vein. |
|
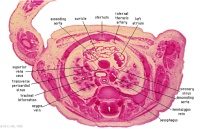
D3L | Ascending aorta attached cranial to transverse pericardial sinus. Superior vena cava expanding into R jugular vein. Trachea (bifurcation). Oesophagus. Azygos (R) and hemiazygos (L) veins. |
|
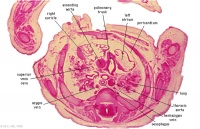
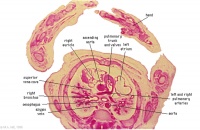
D4L | Ascending aorta. Pulmonary trunk and arteries. Thoracic aorta. Oesophagus. Bronchi. Note azygos vein on right, and precursor of hemiazygos vein on left side.
(Image excerpt different scale from previous images) |
|
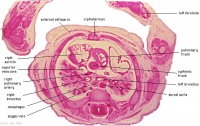
D5L | Superior vena cava. Ascending arch of aorta. Pulmonary trunk with the other two semilunar valves.
Sinus above one valve and branching of pulmonary trunk into L and R pulmonary arteries. |
|
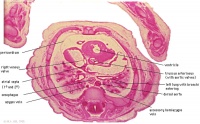
D6L | Superior vena cava. Ascending aorta with transverse pericardial sinus behind. Semilunar valve at origin of pulmonary trunk. L. atrium and auricle. Thoracic aorta. |
|
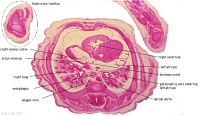
D7L | R atrium with R venous valve. R ventricle close to origin of pulmonary trunk. L.ventricular wall and three semilunar valves of aortic ostium. |
|
E1L | Section through all four chambers of heart.
left ventricle- outflow tract close to origin of ascending aorta. Deep coronary sulcus and transverse pericardial sinus. right atrium- R venous valve of the inferior vena cava, the small septum secundum, the aperture in the dorsal part of septum primum (ostium secundum). (The foramen ovale arises later with the elongation of the septum secundum). left atrium- entrance of the L and R pulmonary veins and the auricle. |
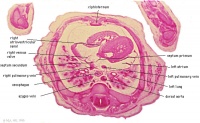
|
E2L | Section through all four chambers of heart.
Ventricles. Right venous valve. Between the tiny ridge of the septum secundum of the R atrium and the R venous valve. right atrium- drainage of the inferior vena cava (cf. E3). R atrioventricular canal. Septum primum. left atrium- drainage sites of R and L pulmonary veins. |
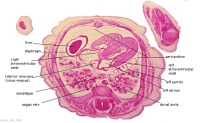
|
E3L | Section through all four chambers of heart.
Ventricles. Diaphragm and liver. Pericardium and cavity. R atrium and two cusps of the atrioventricular valve. L atrium and its auricle. Thoracic aorta. Inferior vena cava. Coronary sinus. |
Magnetic Resonance Imaging
The following movies show aspects of heart development from mid-embryonic to the end of embryonic development.[1] Original image resolution of the scans ranges from 30-150 microns/pixel.
Gestational Age
- Week 6 to 7 - Stage 13 Heart (atrioventricular junction)
- Week 8 - Stage 16 Heart (outflow tract)
- Week 9 - Stage 18 Heart AV valves
- Week 10 - Stage 23 Heart septation (ventricular+inlet)
Image source: The Kyoto Collection embryo MRIs are reproduced with the permission of Prof. Shige Yamada, Cecilia Lo and Kohei Shiota. This material is provided for educational purposes only and cannot be reproduced electronically or in writing without permission. MRI Atlas of Human Embryo Ref:<pubmed>20503356</pubmed>| PMC3401072
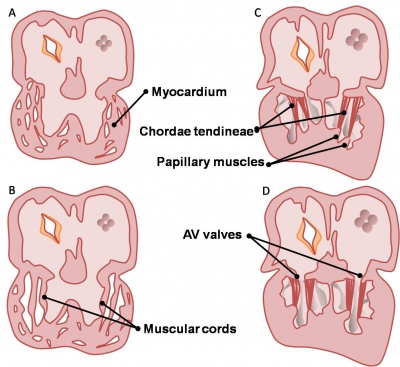
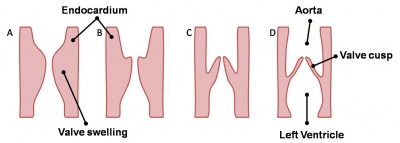
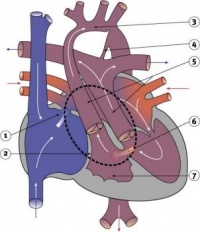
Valve Development
|
Development of the mitral and tricuspid valves
|
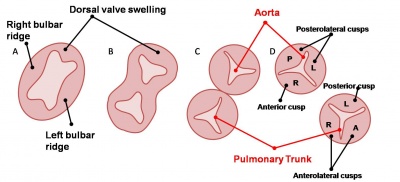
Development of the semilunar valves |
Development of the semilunar cusps |
Aortic and pulmonary valves (semilunar valves)
- formed from the bulbar ridges and subendocardial valve tissue.
- primordial semilunar valve consists of a mesenchymal core covered by endocardium.
- mechanisms of valve remodelling may involve apoptotic pathways.
Cardiac Conduction
|
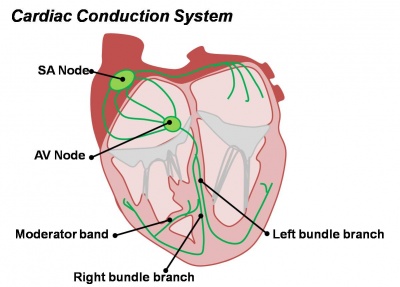
Diagram of the adult cardiac conduction system |
Cardiovascular Movies
|
|
Embryonic Heart Rate
In a 1996 study normal successful human gestations were defined by EHR criteria at different early embryonic (34-56 days GA, from last menstrual period) developmental stages (at the earliest stages when embryo length is difficult to measure gestational sac diameters are included). [2]
- Stage 9-10 2 mm embryo (gestational sac diameter of 20 mm) EHR at least 75 beats / minute
- Stage 11-12 5 mm embryo (gestational sac diameter of 30 mm) EHR at least 100 beats / minute
- Stage 16 10 mm embryo EHR at least 120 beats / minute
- Stage 18 15 mm embryo EHR at least 130 beats / minute
Molecular
ile:Molecular_&_Genetic_Cardiac_Development_Factors.jpg
Fetal Heart
Beyond the scope of this current talk.
Study using 3 and 4-dimensional fetal echocardiography between 12 and 41 weeks gestational age measured outflow tract angles.[3]
- ductal arch and thoracic aorta angle decreased with age.
- between the ductal arch and the aortic arch angle increased with age.
- between the left outflow tract (LOT) and right outflow tract (ROT) angle increased with age.
Abnormalities
| Abnormalities | |||
|---|---|---|---|
| Diagram | Abnormality | Epidemiology | Description |
| Ventricular Septal Defect | 25% of CHD; more frequent in males | Result from growth failure of the membranous IV septum or endocardial cushions leading to a lack of closure of the IV foramen. Many VSDs are actually from defects in the outflow tract septum. 30-50% close spontaneously; large VSDs result in dyspnoea and cardiac failure in infancy. | |
| Tetralogy of Fallot | 9-14% of CHD | Comprises a classic group of four defects: pulmonary stenosis, VSD, dextroposition of the aorta and right ventricle hypertrophy. An additional ASD creates a ‘pentalogy’ of Fallot. Results in cyanosis. | |
| Transposition of the Great Vessels | 10-11% of CHD | The most common form of transposition is where the aorta arises from the right ventricle while the pulmonary trunk arises from the left. This occurs either via abnormal rotation of the arterial pole or abnormal development of the outflow septum. In order for a newborn to survive prior to surgery there must be mixing of systemic and pulmonary circulations through a VSD, ASD or patent ductus arteriosus. It is the most common cause of cyanotic heart disease in newborns and is surgically corrected. | |
| Atrial Septal Defect | 6-10% of CHD; more common in females | The atrial septal myocardium is derived from multiple sources making various places susceptible to defects. The most common is a patent foramen ovale. Others include an ostium secundum defect and ostium primum defect (where there is usually some defect with the AV cushions as well). Defects in the sinus venosus act as ASDs yet are really defects in the wall separating the right pulmonary veins and SVC. A common atrium results from a complete lack of atrial septation. ASDs results in cyanosis due to a right-to-left shunt. | |
| Pulmonary Atresia & Pulmonary Stenosis | 10% of CHD | Unequal division of trunks causes cusps to fuse to form a dome with a narrow/non-existent lumen and hence obstruction to blood flow from the right ventricle to the pulmonary artery. The most common side of stenosis/obstruction is at the pulmonary valve itself (rather than distal or proximal to the valve). Heart-lung transplantation may be the only therapy. | |
| Patent Ductus Arteriosus | 6-8% of CHD; 2-3 times more common in females; common in preterm newborns | Failure of contraction of the muscular wall of the DA. This imposes greater pressure on the pulmonary circulation. Many will close spontaneously, however if this does not occur, surgical closure is advised to prevent the development of congestive heart failure. | |
| Aortic Stenosis | 7% of CHD | Stenosis is caused either by a muscular obstruction below the aortic valve, obstruction at the valve itself or aortic narrowing above the valve. The most common site is at the valve itself which results from a persistence of endocardial tissue that normally degenerates. Results in LV hypertrophy and heart murmurs. | |
| Hypoplastic Left Heart Syndrome | 4-8% of CHD | The left ventricle is incapable of supporting the systemic circulation, hence the right ventricle maintains both pulmonary and systemic circulations aided by an ASD. Other defects such as stenosis or atresia of the mitral and aortic valves, anomalous pulmonary venous connections and hypoplasia of the aortic arch can be responsible for hypoplastic left heart syndrome. Infants usually die within weeks. | |
| Coarctation of the Aorta | 5-7% of CHD | Aortic constriction in the region of the ductus arteriosus. It is associated with patent ductus arteriosus, VSD and aortic stenosis. Closure of the ductus arteriosus in neonates with a coarctation imposes a greater afterload on the left ventricle which can lead to congestive heart failure. Treatment aims at maintaining the ductus arteriosus via prostaglandins. | |
| Partial/Total Anomalous Pulmonary Venous Connection | <4% of CHD; more common in females | In partial anomalous pulmonary venous drainage (PAPVD) a portion of the pulmonary venous blood flow returns to the right atrium. When over 50% of the veins return to the right atrium the condition is clinically significant. All types of total anomalous pulmonary venous connection (TAPVC) (those compatible with survival) are accompanied by an atrial septal defect. Pulmonary veins in TAPVC tend to open into one of the systemic veins before returning to the right atrium. The overloaded pulmonary circuit leads to cyanosis, tachypnoea and dyspnoea. Treatment is via surgical redirection. | |
| Tricuspid Atresia | 1-3% of CHD | Complete lack of formation of the tricuspid valve which results in an hypoplastic right ventricle. The pulmonary circulation can be maintained via a VSD, and an ASD is necessary for survival. Results in cyanosis and tachypnoea. Treatment is initially via administration of prostaglandins followed by surgery to place a shunt to maintain the pulmonary circulation. | |
| Double Outlet Right Ventricle | 1-1.5% of CHD | Both large arteries arise wholly or mainly from the right ventricle. This defect is considered to be present if the aorta obtains 50% of its blood from the right ventricle. The defect arises due to absence of the secondary heart field such that myocardium is not added to the outflow tract during looping. Arrangement of the atrioventricular valves, coronary arteries and the ventriculoarterial connections as well as clinical manifestations are variable. | |
| Interrupted Aortic Arch | Very rare | An extreme form of coarctation of the aorta involving a gap in the ascending or descending thoracic aorta that results from abnormal proliferation/migration/function of neural crest cells. Different types of interrupted aortic arch are defined based on their relation to the arterial branches of the aortic arch. It is greatly associated with other defects such as a patent ductus arteriosus or VSD. It is treated with prostaglandin to maintain ductus arteriosus followed by surgery. |
References
- ↑ <pubmed>20503356</pubmed>| PMC3401072 | MRI Atlas of Human Embryo
- ↑ <pubmed>8921130</pubmed>
- ↑ <pubmed>17384040</pubmed>| MC2190734 J Ultrasound Med.
Online Textbooks
- Developmental Biology (6th ed.) Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000. Figure 15.6 Cascade of heart development
Search Bookshelf heart development
Reviews
<pubmed>21940548</pubmed> <pubmed>17224285</pubmed>| PMC1858673 <pubmed></pubmed>
Articles
<pubmed></pubmed> <pubmed></pubmed> <pubmed></pubmed> <pubmed>9475206</pubmed>
Search Pubmed
Search Pubmed heart development
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Cardiovascular Ultrasound is an open access, peer-reviewed, online journal covering clinical, technological, experimental, biological, and molecular aspects of ultrasound applications in cardiovascular physiology and disease. Search - Fetal
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 18) Embryology Fetal ECHO Meeting 2012. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Fetal_ECHO_Meeting_2012
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G