Developmental Signals - Wnt: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 1: | Line 1: | ||
{{Header}} | {{Header}} | ||
==Introduction== | ==Introduction== | ||
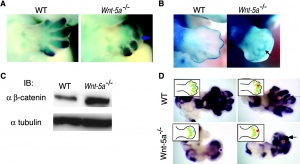
[[File:Canonical_Wnt_signaling_in_Wnt5a_KO_limb_buds.jpg|thumb|300px|Canonical Wnt signaling in normal mouse and Wnt5a KO limb buds | [[File:Canonical_Wnt_signaling_in_Wnt5a_KO_limb_buds.jpg|thumb|300px|Canonical Wnt signaling in normal mouse and Wnt5a KO limb buds{{#pmid:12952940|PMID12952940}}]] | ||
A secreted glycoprotein patterning switch with different roles in different tissues and signaling has generally been divided into the canonical and non-canonical pathways. The name was derived from two drosophila phenotypes '''wingless''' and '''int''' and the gene was first defined as a protooncogene, int1. | A secreted glycoprotein patterning switch with different roles in different tissues and signaling has generally been divided into the canonical and non-canonical pathways. The name was derived from two drosophila phenotypes '''wingless''' and '''int''' and the gene was first defined as a protooncogene, int1. | ||
| Line 17: | Line 17: | ||
== Some Recent Findings == | == Some Recent Findings == | ||
[[File:Mouse somitogenesis genes.jpg|thumb|Mouse somitogenesis genes | [[File:Mouse somitogenesis genes.jpg|thumb|Mouse somitogenesis genes{{#pmid:24304493|PMID24304493}}]] | ||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* '''WNT3 involvement in human bladder exstrophy and cloaca development in zebrafish''' | * '''WNT3 involvement in human bladder exstrophy and cloaca development in zebrafish'''{{#pmid:26105184|PMID26105184}} "Bladder exstrophy, a severe congenital urological malformation when a child is born with an open urinary bladder, is the most common form of bladder exstrophy-epispadias complex (BEEC) with an incidence of 1:30,000 children of Caucasian descent. Recent studies suggest that WNT genes may contribute to the etiology of bladder exstrophy. Here, we evaluated WNT-pathway genes in 20 bladder exstrophy patients using massively parallel sequencing. In total 13 variants were identified in WNT3, WNT6, WNT7A, WNT8B, WNT10A, WNT11, WNT16, FZD5, LRP1 and LRP10 genes and predicted as potentially disease causing, of which seven variants were novel. One variant, identified in a patient with a de novo nonsynonymous substitution in WNT3 (p.Cys91Arg), was further evaluated in zebrafish." [[Renal System - Abnormalities]] | ||
* '''Ror2 regulates branching, differentiation, and actin-cytoskeletal dynamics within the mammary epithelium''' | |||
* '''The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network''' | * '''Ror2 regulates branching, differentiation, and actin-cytoskeletal dynamics within the mammary epithelium'''{{#pmid:25624393|PMID25624393}} "Wnt signaling encompasses β-catenin-dependent and -independent networks. How receptor context provides Wnt specificity in vivo to assimilate multiple concurrent Wnt inputs throughout development remains unclear. Here, we identified a refined expression pattern of Wnt/receptor combinations associated with the Wnt/β-catenin-independent pathway in mammary epithelial subpopulations. Moreover, we elucidated the function of the alternative Wnt receptor Ror2 in mammary development and provided evidence for coordination of this pathway with Wnt/β-catenin-dependent signaling in the mammary epithelium. ... The current study presents a model of Wnt signaling coordination in vivo and assigns an important role for Ror2 in mammary development." | ||
* '''Wnt3 and Wnt3a are required for induction of the mid-diencephalic organizer in the caudal forebrain''' | |||
* '''Development of a Bioassay for Detection of Wnt-Binding Affinities for Individual Frizzled Receptors''' | * '''The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network'''{{#pmid:24304493|PMID24304493}} "In vertebrate development, the segmental pattern of the body axis is established as somites, masses of mesoderm distributed along the two sides of the neural tube, are formed sequentially in the anterior-posterior axis. This mechanism depends on waves of gene expression associated with the Notch, Fgf and Wnt pathways." | ||
* '''Wnt3 and Wnt3a are required for induction of the mid-diencephalic organizer in the caudal forebrain'''{{#pmid:22475147|PMID22475147}} "A fundamental requirement for development of diverse brain regions is the function of local organizers at morphological boundaries. These organizers are restricted groups of cells that secrete signaling molecules, which in turn regulate the fate of the adjacent neural tissue. The thalamus is located in the caudal diencephalon and is the central relay station between the sense organs and higher brain areas. ...We have identified canonical Wnt signaling as a novel pathway, that is required for proper formation of the MDO and consequently for the development of the major relay station of the brain - the thalamus. We propose that Wnt ligands are necessary to maintain the primordial tissue of the organizer during somitogenesis by suppressing Tp53-mediated apoptosis." | |||
* '''Development of a Bioassay for Detection of Wnt-Binding Affinities for Individual Frizzled Receptors'''{{#pmid:20227380|PMID20227380}} "Using a novel ELISA-based technique we were able to determine the first measurements of binding affinity for specific Wnt interactions. This study shows that purified Wnt3a, Wnt7a, and Wnt5a have different binding specificities for Frizzled receptors (Fzds) and secreted frizzled related proteins (SFRPs)." | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 59: | Line 63: | ||
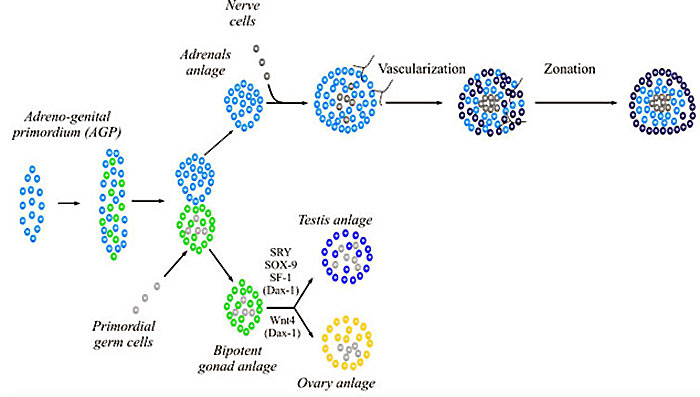
[[File:Adrenal and gonad early development.jpg]] | [[File:Adrenal and gonad early development.jpg]] | ||
WNT4 adrenal and gonad early development | WNT4 adrenal and gonad early development{{#pmid:14594453|PMID14594453}} | ||
| Line 67: | Line 71: | ||
The 4 vertebrate secreted R-spondin proteins are agonists of the canonical Wnt/β-catenin signaling pathway (see reviews{{#pmid:22439850|PMID22439850}}{{#pmid:22982762|PMID22982762}}). | The 4 vertebrate secreted R-spondin proteins are agonists of the canonical Wnt/β-catenin signaling pathway (see reviews{{#pmid:22439850|PMID22439850}}{{#pmid:22982762|PMID22982762}}). | ||
:PubMed Search: [http://www.ncbi.nlm.nih.gov/pubmed/?term=R-spondin ''R-spondin''] | |||
==Function== | ==Function== | ||
[[File:Mouse Wnt signaling 01.jpg|thumb|Mouse E10.5 Wnt signaling | [[File:Mouse Wnt signaling 01.jpg|thumb|Mouse E10.5 Wnt signaling{{#pmid:21176145|PMID21176145}}]] | ||
===Body Axis=== | ===Body Axis=== | ||
'''Wnt signaling and the polarity of the primary body axis''' | '''Wnt signaling and the polarity of the primary body axis'''{{#pmid:20005801|PMID20005801}} "Data from diverse deuterostomes (frog, fish, mouse, and amphioxus) and from planarians (protostomes) suggest that Wnt signaling through beta-catenin controls posterior identity during body plan formation in most bilaterally symmetric animals." | ||
===Sensory=== | ===Sensory=== | ||
====Hearing==== | ====Hearing==== | ||
Wnt9a Can Influence Cell Fates and Neural Connectivity across the Radial Axis of the Developing Cochlea. | Wnt9a Can Influence Cell Fates and Neural Connectivity across the Radial Axis of the Developing Cochlea.{{#pmid:28821654|PMID28821654}} | ||
:"Vertebrate hearing organs manifest cellular asymmetries across the radial axis that underlie afferent versus efferent circuits between the inner ear and the brain. Therefore, understanding the molecular control of patterning across this axis has important functional implications. Radial axis patterning begins before the cells become postmitotic and is likely linked to the onset of asymmetric expression of secreted factors adjacent to the sensory primordium. This study explores one such asymmetrically expressed gene, Wnt9a, which becomes restricted to the neural edge of the avian auditory organ, the basilar papilla, by embryonic day 5 (E5). Radial patterning is disrupted when Wnt9a is overexpressed throughout the prosensory domain beginning on E3. Sexes were pooled for analysis and sex differences were not studied. Analysis of gene expression and afferent innervation on E6 suggests that ectopic Wnt9a expands the neural-side fate, possibly by re-specifying the abneural fate." | :"Vertebrate hearing organs manifest cellular asymmetries across the radial axis that underlie afferent versus efferent circuits between the inner ear and the brain. Therefore, understanding the molecular control of patterning across this axis has important functional implications. Radial axis patterning begins before the cells become postmitotic and is likely linked to the onset of asymmetric expression of secreted factors adjacent to the sensory primordium. This study explores one such asymmetrically expressed gene, Wnt9a, which becomes restricted to the neural edge of the avian auditory organ, the basilar papilla, by embryonic day 5 (E5). Radial patterning is disrupted when Wnt9a is overexpressed throughout the prosensory domain beginning on E3. Sexes were pooled for analysis and sex differences were not studied. Analysis of gene expression and afferent innervation on E6 suggests that ectopic Wnt9a expands the neural-side fate, possibly by re-specifying the abneural fate." | ||
| Line 82: | Line 89: | ||
====Vision==== | ====Vision==== | ||
[[File:Lens-neural crest signaling 01.jpg|thumb|Lens-neural crest signaling | [[File:Lens-neural crest signaling 01.jpg|thumb|Lens-neural crest signaling{{#pmid:21468017|PMID21468017}}]] | ||
See figure from recent article, proposed embryological model summarizing how neural crest cells (NCCs) organize the eye: NCCs (blue) secrete TGF-βs, which signal to the non-lens ectoderm and dorsal optic vesicle. As a consequence, Wnt2b (red) is induced, and together they repress lens formation in the non-lens ectoderm. This leads to the alignment of Pax6 expression in the future lens and neural retina (grey). | See figure from recent article, proposed embryological model summarizing how neural crest cells (NCCs) organize the eye: NCCs (blue) secrete TGF-βs, which signal to the non-lens ectoderm and dorsal optic vesicle. As a consequence, Wnt2b (red) is induced, and together they repress lens formation in the non-lens ectoderm. This leads to the alignment of Pax6 expression in the future lens and neural retina (grey).{{#pmid:21468017|PMID21468017}} | ||
:'''Links:''' [[Vision_-_Lens_Development|Lens]] | :'''Links:''' [[Vision_-_Lens_Development|Lens]] | ||
===Neural=== | ===Neural=== | ||
[[File:Chicken-_neural_Wnt_expression.jpg|thumb|Chicken embryo neural Wnt 1 and Wnt 6 expression | [[File:Chicken-_neural_Wnt_expression.jpg|thumb|Chicken embryo neural Wnt 1 and Wnt 6 expression{{#pmid:19732418|PMID19732418}}]] | ||
"Wnts are antagonised by Shh signalling. By demonstrating that Wnt4 expression in the thalamus is repressed by Shh from the ZLI we reveal an additional level of interaction between these two pathways and provide an example for the cross-regulation between patterning centres during forebrain regionalisation." | "Wnts are antagonised by Shh signalling. By demonstrating that Wnt4 expression in the thalamus is repressed by Shh from the ZLI we reveal an additional level of interaction between these two pathways and provide an example for the cross-regulation between patterning centres during forebrain regionalisation."{{#pmid:19732418|PMID19732418}} | ||
'''Wnt receptor Frizzled-1 in presynaptic differentiation and function''' | '''Wnt receptor Frizzled-1 in presynaptic differentiation and function'''{{#pmid:19883499|PMID19883499}} "We examined the distribution of the Wnt receptor Frizzled-1 in cultured hippocampal neurons and determined that this receptor is located at synaptic contacts co-localizing with presynaptic proteins. Frizzled-1 was found in functional synapses detected with FM1-43 staining and in synaptic terminals from adult rat brain. Interestingly, overexpression of Frizzled-1 increased the number of clusters of Bassoon, a component of the active zone, while treatment with the extracellular cysteine-rich domain (CRD) of Frizzled-1 decreased Bassoon clustering, suggesting a role for this receptor in presynaptic differentiation. Consistent with this, treatment with the Frizzled-1 ligand Wnt-3a induced presynaptic protein clustering and increased functional presynaptic recycling sites, and these effects were prevented by co-treatment with the CRD of Frizzled-1." | ||
| Line 96: | Line 103: | ||
===Muscle=== | ===Muscle=== | ||
'''Wnt signaling promotes AChR aggregation at the neuromuscular synapse''' | '''Wnt signaling promotes AChR aggregation at the neuromuscular synapse'''{{#pmid:19020093|PMID19020093}} | ||
:"Wnt proteins regulate the formation of central synapses by stimulating synaptic assembly, but their role at the vertebrate neuromuscular junction (NMJ) is unclear. Wnt3 is expressed by lateral motoneurons of the spinal cord during the period of motoneuron-muscle innervation. ...Wnt3 does not signal through the canonical Wnt pathway to induce cluster formation. Instead, Wnt3 induces the rapid formation of unstable AChR micro-clusters through activation of Rac1, which aggregate into large clusters only in the presence of agrin." | :"Wnt proteins regulate the formation of central synapses by stimulating synaptic assembly, but their role at the vertebrate neuromuscular junction (NMJ) is unclear. Wnt3 is expressed by lateral motoneurons of the spinal cord during the period of motoneuron-muscle innervation. ...Wnt3 does not signal through the canonical Wnt pathway to induce cluster formation. Instead, Wnt3 induces the rapid formation of unstable AChR micro-clusters through activation of Rac1, which aggregate into large clusters only in the presence of agrin." | ||
| Line 102: | Line 109: | ||
===Vascular=== | ===Vascular=== | ||
'''Canonical Wnt signaling regulates organ-specific assembly and differentiation of CNS vasculature''' | '''Canonical Wnt signaling regulates organ-specific assembly and differentiation of CNS vasculature'''{{#pmid:19023080|PMID19023080}} "The central nervous system (CNS) vasculature consists of a tightly sealed endothelium that forms the blood-brain barrier, whereas blood vessels of other organs are more porous. Wnt7a and Wnt7b encode two Wnt ligands produced by the neuroepithelium of the developing CNS coincident with vascular invasion. Using genetic mouse models, we found that these ligands directly target the vascular endothelium and that the CNS uses the canonical Wnt signaling pathway to promote formation and CNS-specific differentiation of the organ's vasculature" | ||
===Respiratory=== | ===Respiratory=== | ||
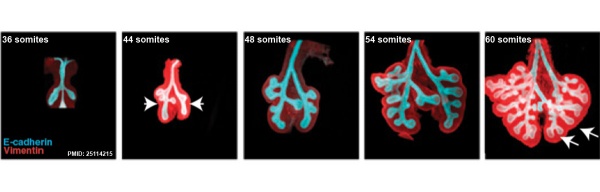
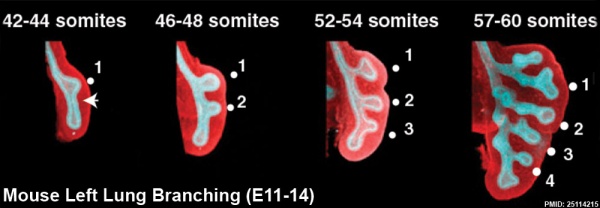
This study used knockout mice to show the role of Wnt signalling in bronchial branching morphogenesis between E10 to E14. | This study used knockout mice to show the role of Wnt signalling in bronchial branching morphogenesis between E10 to E14.{{#pmid:25114215|PMID25114215}} The images below are from a normal branching sequence with a mesenchyme (red) and epithelium (blue) marker. Knocking out Frizzled 2 receptor impacted on both tube shape and branch-point formation in the developing lung. | ||
[[File:Mouse respiratory 36 to 60 somites.jpg|600px]] | [[File:Mouse respiratory 36 to 60 somites.jpg|600px]] | ||
| Line 115: | Line 122: | ||
==Signaling Pathway== | ==Signaling Pathway== | ||
[[File:Wnt signal pathway.jpg|thumb|400px|The Wnt signal pathways | [[File:Wnt signal pathway.jpg|thumb|400px|The Wnt signal pathways{{#pmid:12952939|PMID12952939}}]] | ||
At least 3 separate signaling pathways have been identified in relation to Wnt singnaling. | At least 3 separate signaling pathways have been identified in relation to Wnt singnaling. | ||
| Line 171: | Line 178: | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:20010950}} | |||
{{#pmid:20005801}} | |||
{{#pmid:19864126}} | |||
{{#pmid:19736321}} | |||
'''Search PubMed:''' [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=pubmed&cmd=search&term=Wnt Wnt] | '''Search PubMed:''' [http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=pubmed&cmd=search&term=Wnt Wnt] | ||
| Line 192: | Line 202: | ||
{{Glossary}} | {{Glossary}} | ||
{{Footer}} | {{Footer}} | ||
[[Category:Developmental Signal]] [[Category:Wnt]] [[Category:Molecular]] | [[Category:Developmental Signal]] [[Category:Wnt]] [[Category:Molecular]] | ||
Revision as of 07:53, 21 February 2018
| Embryology - 24 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
A secreted glycoprotein patterning switch with different roles in different tissues and signaling has generally been divided into the canonical and non-canonical pathways. The name was derived from two drosophila phenotypes wingless and int and the gene was first defined as a protooncogene, int1.
Humans have 19 identified members, with the major subgroup of Wnts (WNT1, WNT3A, WNT8) signaling through activation of beta-catenin dependent transcription from at least 4 WNT genes encoding secreted glycoproteins. At least one wnt receptor called frizzled (FZD). Wnt7a is secreted protein and binds to extracellular matrix. The mechanism of Wnt distribution (free diffusion, restricted diffusion and active transport) and all its possible cell receptors are still being determined.
In the gastrointestinal system, Wnt maintains the pool of undifferentiated intestinal progenitor cells and control maturation and correct positioning of the Paneth cell (a differentiated intestinal cell type).
The Wnt/beta-catenin signal pathway activation has also been identified in several cancers and appears to drive cell-cycle progression.
If you are interested in this family of proteins, look also at Roel Nusse -The Wnt Homepage.
| Factor Links: AMH | hCG | BMP | sonic hedgehog | bHLH | HOX | FGF | FOX | Hippo | LIM | Nanog | NGF | Nodal | Notch | PAX | retinoic acid | SIX | Slit2/Robo1 | SOX | TBX | TGF-beta | VEGF | WNT | Category:Molecular |
Some Recent Findings
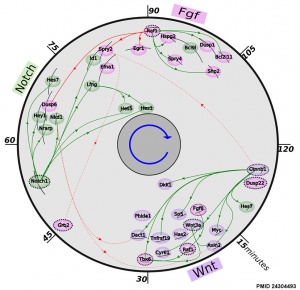
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References <pubmed limit=5>Wnt</pubmed> |
Human Wnt Family
| Table - Human Wnt Family | |||||
| Approved Symbol |
Approved Name | Previous Symbols |
Synonyms | Chromosome | |
|---|---|---|---|---|---|
| WNT1 | Wnt family member 1 | INT1 | 12q13.12 | ||
| WNT2 | Wnt family member 2 | INT1L1 | IRP | 7q31.2 | |
| WNT2B | Wnt family member 2B | WNT13 | XWNT2 | 1p13.2 | |
| WNT3 | Wnt family member 3 | INT4 | "MGC131950, MGC138321, MGC138323" | 17q21.31-q21.32 | |
| WNT3A | Wnt family member 3A | 1q42.13 | |||
| WNT4 | Wnt family member 4 | WNT-4 | 1p36.12 | ||
| WNT5A | Wnt family member 5A | hWNT5A | 3p14.3 | ||
| WNT5B | Wnt family member 5B | 12p13.33 | |||
| WNT6 | Wnt family member 6 | 2q35 | |||
| WNT7A | Wnt family member 7A | 3p25.1 | |||
| WNT7B | Wnt family member 7B | 22q13.31 | |||
| WNT8A | Wnt family member 8A | WNT8D | 5q31.2 | ||
| WNT8B | Wnt family member 8B | 10q24.31 | |||
| WNT9A | Wnt family member 9A | WNT14 | 1q42.13 | ||
| WNT9B | Wnt family member 9B | WNT15 | WNT14B | 17q21.32 | |
| WNT10A | Wnt family member 10A | 2q35 | |||
| WNT10B | Wnt family member 10B | "WNT-12, SHFM6" | 12q13.12 | ||
| WNT11 | Wnt family member 11 | 11q13.5 | |||
| WNT16 | Wnt family member 16 | 7q31.31 | |||
| Links: Developmental Signals - Wnt | OMIM Wnt1 | HGNC | Bmp Family | Fgf Family | Pax Family | R-spondin Family | Sox Family | Tbx Family | Wnt Family | |||||
| |||||
| Human WNT Family | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
WNT4
| Table - Human Wnt Family | ||||
| Approved Symbol |
Approved Name | Previous Symbols |
Synonyms | Chromosome |
|---|---|---|---|---|
| WNT4 | Wnt family member 4 | WNT-4 | 1p36.12 | |
WNT4 adrenal and gonad early development[7]
- PubMed Search: WNT4
R-spondin
The 4 vertebrate secreted R-spondin proteins are agonists of the canonical Wnt/β-catenin signaling pathway (see reviews[8][9]).
- PubMed Search: R-spondin
Function
Body Axis
Wnt signaling and the polarity of the primary body axis[11] "Data from diverse deuterostomes (frog, fish, mouse, and amphioxus) and from planarians (protostomes) suggest that Wnt signaling through beta-catenin controls posterior identity during body plan formation in most bilaterally symmetric animals."
Sensory
Hearing
Wnt9a Can Influence Cell Fates and Neural Connectivity across the Radial Axis of the Developing Cochlea.[12]
- "Vertebrate hearing organs manifest cellular asymmetries across the radial axis that underlie afferent versus efferent circuits between the inner ear and the brain. Therefore, understanding the molecular control of patterning across this axis has important functional implications. Radial axis patterning begins before the cells become postmitotic and is likely linked to the onset of asymmetric expression of secreted factors adjacent to the sensory primordium. This study explores one such asymmetrically expressed gene, Wnt9a, which becomes restricted to the neural edge of the avian auditory organ, the basilar papilla, by embryonic day 5 (E5). Radial patterning is disrupted when Wnt9a is overexpressed throughout the prosensory domain beginning on E3. Sexes were pooled for analysis and sex differences were not studied. Analysis of gene expression and afferent innervation on E6 suggests that ectopic Wnt9a expands the neural-side fate, possibly by re-specifying the abneural fate."
- Links: Inner Ear | Chicken Development
Vision
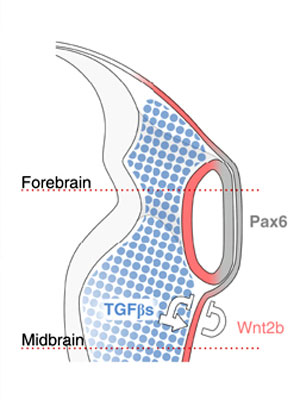
See figure from recent article, proposed embryological model summarizing how neural crest cells (NCCs) organize the eye: NCCs (blue) secrete TGF-βs, which signal to the non-lens ectoderm and dorsal optic vesicle. As a consequence, Wnt2b (red) is induced, and together they repress lens formation in the non-lens ectoderm. This leads to the alignment of Pax6 expression in the future lens and neural retina (grey).[13]
- Links: Lens
Neural
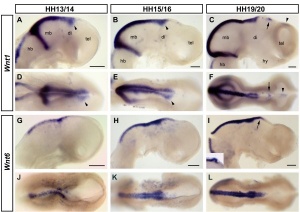
"Wnts are antagonised by Shh signalling. By demonstrating that Wnt4 expression in the thalamus is repressed by Shh from the ZLI we reveal an additional level of interaction between these two pathways and provide an example for the cross-regulation between patterning centres during forebrain regionalisation."[14]
Wnt receptor Frizzled-1 in presynaptic differentiation and function[15] "We examined the distribution of the Wnt receptor Frizzled-1 in cultured hippocampal neurons and determined that this receptor is located at synaptic contacts co-localizing with presynaptic proteins. Frizzled-1 was found in functional synapses detected with FM1-43 staining and in synaptic terminals from adult rat brain. Interestingly, overexpression of Frizzled-1 increased the number of clusters of Bassoon, a component of the active zone, while treatment with the extracellular cysteine-rich domain (CRD) of Frizzled-1 decreased Bassoon clustering, suggesting a role for this receptor in presynaptic differentiation. Consistent with this, treatment with the Frizzled-1 ligand Wnt-3a induced presynaptic protein clustering and increased functional presynaptic recycling sites, and these effects were prevented by co-treatment with the CRD of Frizzled-1."
- Links: Neural System Development)
Muscle
Wnt signaling promotes AChR aggregation at the neuromuscular synapse[16]
- "Wnt proteins regulate the formation of central synapses by stimulating synaptic assembly, but their role at the vertebrate neuromuscular junction (NMJ) is unclear. Wnt3 is expressed by lateral motoneurons of the spinal cord during the period of motoneuron-muscle innervation. ...Wnt3 does not signal through the canonical Wnt pathway to induce cluster formation. Instead, Wnt3 induces the rapid formation of unstable AChR micro-clusters through activation of Rac1, which aggregate into large clusters only in the presence of agrin."
Vascular
Canonical Wnt signaling regulates organ-specific assembly and differentiation of CNS vasculature[17] "The central nervous system (CNS) vasculature consists of a tightly sealed endothelium that forms the blood-brain barrier, whereas blood vessels of other organs are more porous. Wnt7a and Wnt7b encode two Wnt ligands produced by the neuroepithelium of the developing CNS coincident with vascular invasion. Using genetic mouse models, we found that these ligands directly target the vascular endothelium and that the CNS uses the canonical Wnt signaling pathway to promote formation and CNS-specific differentiation of the organ's vasculature"
Respiratory
This study used knockout mice to show the role of Wnt signalling in bronchial branching morphogenesis between E10 to E14.[18] The images below are from a normal branching sequence with a mesenchyme (red) and epithelium (blue) marker. Knocking out Frizzled 2 receptor impacted on both tube shape and branch-point formation in the developing lung.
- Links: Respiratory | Mouse Development
Signaling Pathway
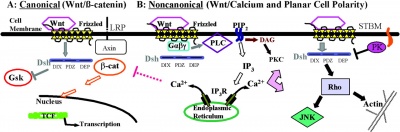
At least 3 separate signaling pathways have been identified in relation to Wnt singnaling.
- Canonical pathway
- Non-canonical or planar cell polarity pathway
- Wnt-Calcium ion pathway
Canonical Pathway
- Wnt binds the surface receptor Frizzled (Fz) and LRP5/6 receptor complex
- Induces the stabilization of beta-catenin (through the DIX and PDZ domains of Dishevelled and other factors including Axin, glycogen synthase kinase 3 and casein kinase 1)
- Beta-catenin translocates into the nucleus
- Beta-catenin complexes with members of the LEF/TCF family of transcription factors.
- Transcriptional induction of target genes.
- Beta-catenin is then exported from the nucleus and degraded via the proteosomal machinery.
Example - early development of the sinoatrial node cells in the heart.
Non-Canonical Pathway
(Planar Cell Polarity Pathway)
- Wnt binds the surface receptor Frizzled (Fz) independent of the LRP5/6 receptor complex.
- Acts through Dishevelled (PDZ and DEP domains)
- Mediates cytoskeletal (microfilament) changes through activation of the small GTPases Rho and Rac.
Wnt-Calcium Ion Pathway
- Wnt binds the surface receptor Frizzled (Fz) and mediates activation of heterotrimeric G-proteins.
- Acts internally through Dsh, phospholipase C, calcium-calmodulin kinase 2 (CamK2) and protein kinase C (PKC).
- has various cellular functions including cell adhesion and motility.
Protein Data
- FUNCTION: PROBABLE DEVELOPMENTAL PROTEIN. SIGNALING BY WNT-7A ALLOWS SEXUALLY DIMORPHIC DEVELOPMENT OF THE MULLERIAN DUCTS (BY SIMILARITY).
- SUBCELLULAR LOCATION: POSSIBLY SECRETED AND ASSOCIATES WITH THE EXTRACELLULAR MATRIX.
- TISSUE SPECIFICITY: EXPRESSION IS RESTRICTED TO PLACENTA, KIDNEY, TESTIS, UTERUS, FETAL LUNG, AND FETAL AND ADULT BRAIN.
- SIMILARITY: BELONGS TO THE WNT FAMILY.
- FUNCTION: PROBABLE DEVELOPMENTAL PROTEIN. SIGNALING BY WNT-7A ALLOWS SEXUALLY DIMORPHIC DEVELOPMENT OF THE MULLERIAN DUCTS.
- SUBCELLULAR LOCATION: POSSIBLY SECRETED AND ASSOCIATES WITH THE EXTRACELLULAR MATRIX.
- DISEASE: MALE MICE LACKING WNT-7A FAIL TO UNDERGO REGRESSION OF THE MULLERIAN DUCT AS A RESULT OF THE ABSENCE OF THE RECEPTOR FOR MULLERIAN-INHIBITING SUBSTANCE. WNT7A-DEFICIENT FEMALES ARE INFERTILE BECAUSE OF ABNORMAL DEVELOPMENT OF THE OVIDUCT AND UTERUS, BOTH OF WHICH ARE MULLERIAN DUCT DERIVATIVES.
- SIMILARITY: BELONGS TO THE WNT FAMILY.
(data from Expasy)
OMIM
About OMIM "Online Mendelian Inheritance in Man OMIM is a comprehensive, authoritative, and timely compendium of human genes and genetic phenotypes. The full-text, referenced overviews in OMIM contain information on all known mendelian disorders and over 12,000 genes. OMIM focuses on the relationship between phenotype and genotype. It is updated daily, and the entries contain copious links to other genetics resources." OMIM
References
- ↑ Topol L, Jiang X, Choi H, Garrett-Beal L, Carolan PJ & Yang Y. (2003). Wnt-5a inhibits the canonical Wnt pathway by promoting GSK-3-independent beta-catenin degradation. J. Cell Biol. , 162, 899-908. PMID: 12952940 DOI.
- ↑ 2.0 2.1 Fongang B & Kudlicki A. (2013). The precise timeline of transcriptional regulation reveals causation in mouse somitogenesis network. BMC Dev. Biol. , 13, 42. PMID: 24304493 DOI.
- ↑ Baranowska Körberg I, Hofmeister W, Markljung E, Cao J, Nilsson D, Ludwig M, Draaken M, Holmdahl G, Barker G, Reutter H, Vukojević V, Clementson Kockum C, Lundin J, Lindstrand A & Nordenskjöld A. (2015). WNT3 involvement in human bladder exstrophy and cloaca development in zebrafish. Hum. Mol. Genet. , 24, 5069-78. PMID: 26105184 DOI.
- ↑ Roarty K, Shore AN, Creighton CJ & Rosen JM. (2015). Ror2 regulates branching, differentiation, and actin-cytoskeletal dynamics within the mammary epithelium. J. Cell Biol. , 208, 351-66. PMID: 25624393 DOI.
- ↑ Mattes B, Weber S, Peres J, Chen Q, Davidson G, Houart C & Scholpp S. (2012). Wnt3 and Wnt3a are required for induction of the mid-diencephalic organizer in the caudal forebrain. Neural Dev , 7, 12. PMID: 22475147 DOI.
- ↑ Carmon KS & Loose DS. (2010). Development of a bioassay for detection of Wnt-binding affinities for individual frizzled receptors. Anal. Biochem. , 401, 288-94. PMID: 20227380 DOI.
- ↑ Val P, Lefrançois-Martinez AM, Veyssière G & Martinez A. (2003). SF-1 a key player in the development and differentiation of steroidogenic tissues. Nucl. Recept. , 1, 8. PMID: 14594453 DOI.
- ↑ de Lau WB, Snel B & Clevers HC. (2012). The R-spondin protein family. Genome Biol. , 13, 242. PMID: 22439850 DOI.
- ↑ Jin YR & Yoon JK. (2012). The R-spondin family of proteins: emerging regulators of WNT signaling. Int. J. Biochem. Cell Biol. , 44, 2278-87. PMID: 22982762 DOI.
- ↑ Ferrer-Vaquer A, Piliszek A, Tian G, Aho RJ, Dufort D & Hadjantonakis AK. (2010). A sensitive and bright single-cell resolution live imaging reporter of Wnt/ß-catenin signaling in the mouse. BMC Dev. Biol. , 10, 121. PMID: 21176145 DOI.
- ↑ Petersen CP & Reddien PW. (2009). Wnt signaling and the polarity of the primary body axis. Cell , 139, 1056-68. PMID: 20005801 DOI.
- ↑ Munnamalai V, Sienknecht UJ, Duncan RK, Scott MK, Thawani A, Fantetti KN, Atallah NM, Biesemeier DJ, Song KH, Luethy K, Traub E & Fekete DM. (2017). Wnt9a Can Influence Cell Fates and Neural Connectivity across the Radial Axis of the Developing Cochlea. J. Neurosci. , 37, 8975-8988. PMID: 28821654 DOI.
- ↑ 13.0 13.1 Grocott T, Johnson S, Bailey AP & Streit A. (2011). Neural crest cells organize the eye via TGF-β and canonical Wnt signalling. Nat Commun , 2, 265. PMID: 21468017 DOI.
- ↑ 14.0 14.1 Quinlan R, Graf M, Mason I, Lumsden A & Kiecker C. (2009). Complex and dynamic patterns of Wnt pathway gene expression in the developing chick forebrain. Neural Dev , 4, 35. PMID: 19732418 DOI.
- ↑ Varela-Nallar L, Grabowski CP, Alfaro IE, Alvarez AR & Inestrosa NC. (2009). Role of the Wnt receptor Frizzled-1 in presynaptic differentiation and function. Neural Dev , 4, 41. PMID: 19883499 DOI.
- ↑ Henriquez JP, Webb A, Bence M, Bildsoe H, Sahores M, Hughes SM & Salinas PC. (2008). Wnt signaling promotes AChR aggregation at the neuromuscular synapse in collaboration with agrin. Proc. Natl. Acad. Sci. U.S.A. , 105, 18812-7. PMID: 19020093 DOI.
- ↑ Stenman JM, Rajagopal J, Carroll TJ, Ishibashi M, McMahon J & McMahon AP. (2008). Canonical Wnt signaling regulates organ-specific assembly and differentiation of CNS vasculature. Science , 322, 1247-50. PMID: 19023080 DOI.
- ↑ Kadzik RS, Cohen ED, Morley MP, Stewart KM, Lu MM & Morrisey EE. (2014). Wnt ligand/Frizzled 2 receptor signaling regulates tube shape and branch-point formation in the lung through control of epithelial cell shape. Proc. Natl. Acad. Sci. U.S.A. , 111, 12444-9. PMID: 25114215 DOI.
- ↑ Westfall TA, Brimeyer R, Twedt J, Gladon J, Olberding A, Furutani-Seiki M & Slusarski DC. (2003). Wnt-5/pipetail functions in vertebrate axis formation as a negative regulator of Wnt/beta-catenin activity. J. Cell Biol. , 162, 889-98. PMID: 12952939 DOI.
Reviews
Inestrosa NC & Arenas E. (2010). Emerging roles of Wnts in the adult nervous system. Nat. Rev. Neurosci. , 11, 77-86. PMID: 20010950 DOI.
Petersen CP & Reddien PW. (2009). Wnt signaling and the polarity of the primary body axis. Cell , 139, 1056-68. PMID: 20005801 DOI.
Franco CA, Liebner S & Gerhardt H. (2009). Vascular morphogenesis: a Wnt for every vessel?. Curr. Opin. Genet. Dev. , 19, 476-83. PMID: 19864126 DOI.
van Amerongen R & Nusse R. (2009). Towards an integrated view of Wnt signaling in development. Development , 136, 3205-14. PMID: 19736321 DOI.
Search PubMed: Wnt
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- The Wnt gene Homepage by Roel Nusse
- Atlas of Genetics and Cytogenetics in Oncology and Haematology - The WNT Signaling Pathway and Its Role in Human Solid Tumors
- Mouse Wnt genes
- Comparative table of all vertebrate Wnt genes
- alignments of all mammalian Wnt genes
- Other Wnt pages
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 24) Embryology Developmental Signals - Wnt. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Signals_-_Wnt
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G