Neural - Prosomere
| Embryology - 25 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
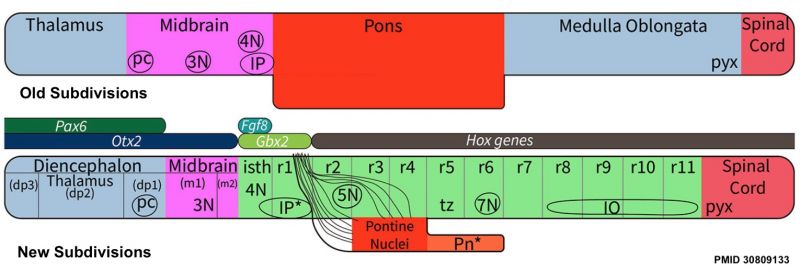
The prosemeric (neuromeric) model describes brain development based upon a series of neural tube transverse subunits (segmental) and integrates with the longitudinal (columnar) concepts. Historically described as primary and secondary vesicles, the modern interpretation of segmentation is based upon regional gene expression. Best example that corresponds to morphological features is found in the hindbrain rhombomeric HOX expression pattern.
Review - Time for Radical Changes in Brain Stem Nomenclature-Applying the Lessons From Developmental Gene Patterns[1] "The traditional subdivision of the brain stem into midbrain, pons, and medulla oblongata is based purely on the external appearance of the human brain stem. There is an urgent need to update the names of brain stem structures to be consistent with the discovery of rhomobomeric segmentation based on gene expression. The most important mistakes are the belief that the pons occupies the upper half of the hindbrain, the failure to recognize the isthmus as the first segment of the hindbrain, and the mistaken inclusion of diencephalic structures in the midbrain. The new nomenclature will apply to all mammals. This essay recommends a new brain stem nomenclature based on developmental gene expression, progeny analysis, and fate mapping."
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Prosomere | Prosomeres | prosemeric model | neuromeric model |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page. |
References
- ↑ Watson C, Bartholomaeus C & Puelles L. (2019). Time for Radical Changes in Brain Stem Nomenclature-Applying the Lessons From Developmental Gene Patterns. Front Neuroanat , 13, 10. PMID: 30809133 DOI.
- ↑ Puelles L. (2018). Developmental studies of avian brain organization. Int. J. Dev. Biol. , 62, 207-224. PMID: 29616730 DOI.
- ↑ Sousa AMM, Meyer KA, Santpere G, Gulden FO & Sestan N. (2017). Evolution of the Human Nervous System Function, Structure, and Development. Cell , 170, 226-247. PMID: 28708995 DOI.
Whalley K. (2017). Gene expression: Evolving expression patterns. Nat. Rev. Neurosci. , 19, 7. PMID: 29238089 DOI.
Alvarez-Bolado G, Grinevich V & Puelles L. (2015). Editorial: Development of the hypothalamus. Front Neuroanat , 9, 83. PMID: 26157363 DOI.
Ferran JL, Puelles L & Rubenstein JL. (2015). Molecular codes defining rostrocaudal domains in the embryonic mouse hypothalamus. Front Neuroanat , 9, 46. PMID: 25941476 DOI.
Santos-Durán GN, Menuet A, Lagadec R, Mayeur H, Ferreiro-Galve S, Mazan S, Rodríguez-Moldes I & Candal E. (2015). Prosomeric organization of the hypothalamus in an elasmobranch, the catshark Scyliorhinus canicula. Front Neuroanat , 9, 37. PMID: 25904850 DOI.
Puelles L & Rubenstein JL. (2015). A new scenario of hypothalamic organization: rationale of new hypotheses introduced in the updated prosomeric model. Front Neuroanat , 9, 27. PMID: 25852489 DOI.
Cajal M, Creuzet SE, Papanayotou C, Sabéran-Djoneidi D, Chuva de Sousa Lopes SM, Zwijsen A, Collignon J & Camus A. (2014). A conserved role for non-neural ectoderm cells in early neural development. Development , 141, 4127-38. PMID: 25273086 DOI.
Domínguez L, González A & Moreno N. (2014). Characterization of the hypothalamus of Xenopus laevis during development. II. The basal regions. J. Comp. Neurol. , 522, 1102-31. PMID: 24122702 DOI.
Lauter G, Söll I & Hauptmann G. (2013). Molecular characterization of prosomeric and intraprosomeric subdivisions of the embryonic zebrafish diencephalon. J. Comp. Neurol. , 521, 1093-118. PMID: 22949352 DOI.
Domínguez L, Morona R, González A & Moreno N. (2013). Characterization of the hypothalamus of Xenopus laevis during development. I. The alar regions. J. Comp. Neurol. , 521, 725-59. PMID: 22965483 DOI.
Beccari L, Marco-Ferreres R & Bovolenta P. (2013). The logic of gene regulatory networks in early vertebrate forebrain patterning. Mech. Dev. , 130, 95-111. PMID: 23111324 DOI.
Rash BG & Grove EA. (2011). Shh and Gli3 regulate formation of the telencephalic-diencephalic junction and suppress an isthmus-like signaling source in the forebrain. Dev. Biol. , 359, 242-50. PMID: 21925158 DOI.
Villar-Cerviño V, Barreiro-Iglesias A, Mazan S, Rodicio MC & Anadón R. (2011). Glutamatergic neuronal populations in the forebrain of the sea lamprey, Petromyzon marinus: an in situ hybridization and immunocytochemical study. J. Comp. Neurol. , 519, 1712-35. PMID: 21452205 DOI.
Martínez-de-la-Torre M, Pombal MA & Puelles L. (2011). Distal-less-like protein distribution in the larval lamprey forebrain. Neuroscience , 178, 270-84. PMID: 21185911 DOI.
Moreno N & González A. (2011). The non-evaginated secondary prosencephalon of vertebrates. Front Neuroanat , 5, 12. PMID: 21427782 DOI.
Domínguez L, González A & Moreno N. (2011). Ontogenetic distribution of the transcription factor nkx2.2 in the developing forebrain of Xenopus laevis. Front Neuroanat , 5, 11. PMID: 21415915 DOI.
Sugahara F, Aota S, Kuraku S, Murakami Y, Takio-Ogawa Y, Hirano S & Kuratani S. (2011). Involvement of Hedgehog and FGF signalling in the lamprey telencephalon: evolution of regionalization and dorsoventral patterning of the vertebrate forebrain. Development , 138, 1217-26. PMID: 21343370 DOI.
Yamamoto K, Ruuskanen JO, Wullimann MF & Vernier P. (2011). Differential expression of dopaminergic cell markers in the adult zebrafish forebrain. J. Comp. Neurol. , 519, 576-98. PMID: 21192085 DOI.
Suda Y, Kokura K, Kimura J, Kajikawa E, Inoue F & Aizawa S. (2010). The same enhancer regulates the earliest Emx2 expression in caudal forebrain primordium, subsequent expression in dorsal telencephalon and later expression in the cortical ventricular zone. Development , 137, 2939-49. PMID: 20667915 DOI.
Garcia-Lopez R & Martinez S. (2010). Oligodendrocyte precursors originate in the parabasal band of the basal plate in prosomere 1 and migrate into the alar prosencephalon during chick development. Glia , 58, 1437-50. PMID: 20648637 DOI.
Abellán A, Vernier B, Rétaux S & Medina L. (2010). Similarities and differences in the forebrain expression of Lhx1 and Lhx5 between chicken and mouse: Insights for understanding telencephalic development and evolution. J. Comp. Neurol. , 518, 3512-28. PMID: 20589911 DOI.
Osório J, Mueller T, Rétaux S, Vernier P & Wullimann MF. (2010). Phylotypic expression of the bHLH genes Neurogenin2, Neurod, and Mash1 in the mouse embryonic forebrain. J. Comp. Neurol. , 518, 851-71. PMID: 20058311 DOI.
Pritz MB. (2010). Forebrain and midbrain fiber tract formation during early development in Alligator embryos. Brain Res. , 1313, 34-44. PMID: 19968970 DOI.
García-López R, Soula C & Martínez S. (2009). Expression analysis of Sulf1 in the chick forebrain at early and late stages of development. Dev. Dyn. , 238, 2418-29. PMID: 19653319 DOI.
Hoch RV, Rubenstein JL & Pleasure S. (2009). Genes and signaling events that establish regional patterning of the mammalian forebrain. Semin. Cell Dev. Biol. , 20, 378-86. PMID: 19560042 DOI.
Quinn JC, Molinek M, Mason JO & Price DJ. (2009). Gli3 is required autonomously for dorsal telencephalic cells to adopt appropriate fates during embryonic forebrain development. Dev. Biol. , 327, 204-15. PMID: 19121302 DOI.
Mueller T & Wullimann MF. (2009). An evolutionary interpretation of teleostean forebrain anatomy. Brain Behav. Evol. , 74, 30-42. PMID: 19729894 DOI.
Pombal MA, Megías M, Bardet SM & Puelles L. (2009). New and old thoughts on the segmental organization of the forebrain in lampreys. Brain Behav. Evol. , 74, 7-19. PMID: 19729892 DOI.
Morona R & González A. (2008). Calbindin-D28k and calretinin expression in the forebrain of anuran and urodele amphibians: further support for newly identified subdivisions. J. Comp. Neurol. , 511, 187-220. PMID: 18781620 DOI.
Takahashi K, Liu FC, Hirokawa K & Takahashi H. (2008). Expression of Foxp4 in the developing and adult rat forebrain. J. Neurosci. Res. , 86, 3106-16. PMID: 18561326 DOI.
van den Akker WM, Brox A, Puelles L, Durston AJ & Medina L. (2008). Comparative functional analysis provides evidence for a crucial role for the homeobox gene Nkx2.1/Titf-1 in forebrain evolution. J. Comp. Neurol. , 506, 211-23. PMID: 18022953 DOI.
Poitras L, Ghanem N, Hatch G & Ekker M. (2007). The proneural determinant MASH1 regulates forebrain Dlx1/2 expression through the I12b intergenic enhancer. Development , 134, 1755-65. PMID: 17409112 DOI.
Menuet A, Alunni A, Joly JS, Jeffery WR & Rétaux S. (2007). Expanded expression of Sonic Hedgehog in Astyanax cavefish: multiple consequences on forebrain development and evolution. Development , 134, 845-55. PMID: 17251267 DOI.
Kitambi SS & Hauptmann G. (2007). The zebrafish orphan nuclear receptor genes nr2e1 and nr2e3 are expressed in developing eye and forebrain. Gene Expr. Patterns , 7, 521-8. PMID: 17127102 DOI.
Jeong JY, Einhorn Z, Mathur P, Chen L, Lee S, Kawakami K & Guo S. (2007). Patterning the zebrafish diencephalon by the conserved zinc-finger protein Fezl. Development , 134, 127-36. PMID: 17164418 DOI.
Hirata T, Nakazawa M, Muraoka O, Nakayama R, Suda Y & Hibi M. (2006). Zinc-finger genes Fez and Fez-like function in the establishment of diencephalon subdivisions. Development , 133, 3993-4004. PMID: 16971467 DOI.
García-Calero E, de Puelles E & Puelles L. (2006). EphA7 receptor is expressed differentially at chicken prosomeric boundaries. Neuroscience , 141, 1887-97. PMID: 16844303 DOI.
Ribes V, Wang Z, Dollé P & Niederreither K. (2006). Retinaldehyde dehydrogenase 2 (RALDH2)-mediated retinoic acid synthesis regulates early mouse embryonic forebrain development by controlling FGF and sonic hedgehog signaling. Development , 133, 351-61. PMID: 16368932 DOI.
Junghans D, Hack I, Frotscher M, Taylor V & Kemler R. (2005). Beta-catenin-mediated cell-adhesion is vital for embryonic forebrain development. Dev. Dyn. , 233, 528-39. PMID: 15844200 DOI.
Kimura J, Suda Y, Kurokawa D, Hossain ZM, Nakamura M, Takahashi M, Hara A & Aizawa S. (2005). Emx2 and Pax6 function in cooperation with Otx2 and Otx1 to develop caudal forebrain primordium that includes future archipallium. J. Neurosci. , 25, 5097-108. PMID: 15917450 DOI.
Whitlock KE. (2005). Origin and development of GnRH neurons. Trends Endocrinol. Metab. , 16, 145-51. PMID: 15860410 DOI.
Piñuela C, Rendón C, González de Canales ML & Sarasquete C. (2004). Development of the Senegal sole, Solea senegalensis forebrain. Eur J Histochem , 48, 377-84. PMID: 15718204
Villablanca JR. (2004). Counterpointing the functional role of the forebrain and of the brainstem in the control of the sleep-waking system. J Sleep Res , 13, 179-208. PMID: 15339255 DOI.
Kage T, Takeda H, Yasuda T, Maruyama K, Yamamoto N, Yoshimoto M, Araki K, Inohaya K, Okamoto H, Yasumasu S, Watanabe K, Ito H & Ishikawa Y. (2004). Morphogenesis and regionalization of the medaka embryonic brain. J. Comp. Neurol. , 476, 219-39. PMID: 15269967 DOI.
Puelles L & Rubenstein JL. (2003). Forebrain gene expression domains and the evolving prosomeric model. Trends Neurosci. , 26, 469-76. PMID: 12948657 DOI.
Zhao Y, Marín O, Hermesz E, Powell A, Flames N, Palkovits M, Rubenstein JL & Westphal H. (2003). The LIM-homeobox gene Lhx8 is required for the development of many cholinergic neurons in the mouse forebrain. Proc. Natl. Acad. Sci. U.S.A. , 100, 9005-10. PMID: 12855770 DOI.
Brox A, Puelles L, Ferreiro B & Medina L. (2003). Expression of the genes GAD67 and Distal-less-4 in the forebrain of Xenopus laevis confirms a common pattern in tetrapods. J. Comp. Neurol. , 461, 370-93. PMID: 12746875 DOI.
Hébert JM, Hayhurst M, Marks ME, Kulessa H, Hogan BL & McConnell SK. (2003). BMP ligands act redundantly to pattern the dorsal telencephalic midline. Genesis , 35, 214-9. PMID: 12717732 DOI.
González A, López JM & Marín O. (2002). Expression pattern of the homeobox protein NKX2-1 in the developing Xenopus forebrain. Brain Res. Gene Expr. Patterns , 1, 181-5. PMID: 12638129
Meléndez-Ferro M, Pérez-Costas E, Villar-Cheda B, Abalo XM, Rodríguez-Muñoz R, Rodicio MC & Anadón R. (2002). Ontogeny of gamma-aminobutyric acid-immunoreactive neuronal populations in the forebrain and midbrain of the sea lamprey. J. Comp. Neurol. , 446, 360-76. PMID: 11954035
Levers TE, Tait S, Birling MC, Brophy PJ & Price DJ. (2002). Etr-r3/mNapor, encoding an ELAV-type RNA binding protein, is expressed in differentiating cells in the developing rodent forebrain. Mech. Dev. , 112, 191-3. PMID: 11850193
Alonso A & Trujillo CM. (2002). Continuity and discontinuity of the radial scaffolding in the forebrain of a lizard embryo. Brain Res. Bull. , 57, 505-8. PMID: 11923019
Hauptmann G, Söll I & Gerster T. (2002). The early embryonic zebrafish forebrain is subdivided into molecularly distinct transverse and longitudinal domains. Brain Res. Bull. , 57, 371-5. PMID: 11922991
Marín O, Baker J, Puelles L & Rubenstein JL. (2002). Patterning of the basal telencephalon and hypothalamus is essential for guidance of cortical projections. Development , 129, 761-73. PMID: 11830575
Nelson PA, Sutcliffe JG & Thomas EA. (2002). A new UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferase mRNA exhibits predominant expression in the hypothalamus, thalamus and amygdala of mouse forebrain. Brain Res. Gene Expr. Patterns , 1, 95-9. PMID: 15018805
Ohkubo Y, Chiang C & Rubenstein JL. (2002). Coordinate regulation and synergistic actions of BMP4, SHH and FGF8 in the rostral prosencephalon regulate morphogenesis of the telencephalic and optic vesicles. Neuroscience , 111, 1-17. PMID: 11955708
Bachy I, Vernier P & Retaux S. (2001). The LIM-homeodomain gene family in the developing Xenopus brain: conservation and divergences with the mouse related to the evolution of the forebrain. J. Neurosci. , 21, 7620-9. PMID: 11567052
Puelles L. (2001). Brain segmentation and forebrain development in amniotes. Brain Res. Bull. , 55, 695-710. PMID: 11595354
Ekström P, Johnsson CM & Ohlin LM. (2001). Ventricular proliferation zones in the brain of an adult teleost fish and their relation to neuromeres and migration (secondary matrix) zones. J. Comp. Neurol. , 436, 92-110. PMID: 11413549
McCarthy M, Na E, Neyt C, Langston A & Fishell G. (2001). Calcium-dependent adhesion is necessary for the maintenance of prosomeres. Dev. Biol. , 233, 80-94. PMID: 11319859 DOI.
Nery S, Wichterle H & Fishell G. (2001). Sonic hedgehog contributes to oligodendrocyte specification in the mammalian forebrain. Development , 128, 527-40. PMID: 11171336
Rohr KB, Barth KA, Varga ZM & Wilson SW. (2001). The nodal pathway acts upstream of hedgehog signaling to specify ventral telencephalic identity. Neuron , 29, 341-51. PMID: 11239427
Crossley PH, Martinez S, Ohkubo Y & Rubenstein JL. (2001). Coordinate expression of Fgf8, Otx2, Bmp4, and Shh in the rostral prosencephalon during development of the telencephalic and optic vesicles. Neuroscience , 108, 183-206. PMID: 11734354
Corbin JG, Gaiano N, Machold RP, Langston A & Fishell G. (2000). The Gsh2 homeodomain gene controls multiple aspects of telencephalic development. Development , 127, 5007-20. PMID: 11060228
Dávila JC, Guirado S & Puelles L. (2000). Expression of calcium-binding proteins in the diencephalon of the lizard Psammodromus algirus. J. Comp. Neurol. , 427, 67-92. PMID: 11042592
Shanmugalingam S, Houart C, Picker A, Reifers F, Macdonald R, Barth A, Griffin K, Brand M & Wilson SW. (2000). Ace/Fgf8 is required for forebrain commissure formation and patterning of the telencephalon. Development , 127, 2549-61. PMID: 10821754
Pombal MA & Puelles L. (1999). Prosomeric map of the lamprey forebrain based on calretinin immunocytochemistry, Nissl stain, and ancillary markers. J. Comp. Neurol. , 414, 391-422. PMID: 10516604
Hidalgo-Sánchez M, Simeone A & Alvarado-Mallart RM. (1999). Fgf8 and Gbx2 induction concomitant with Otx2 repression is correlated with midbrain-hindbrain fate of caudal prosencephalon. Development , 126, 3191-203. PMID: 10375509
Tuttle R, Nakagawa Y, Johnson JE & O'Leary DD. (1999). Defects in thalamocortical axon pathfinding correlate with altered cell domains in Mash-1-deficient mice. Development , 126, 1903-16. PMID: 10101124
Braisted JE, Tuttle R & O'leary DD. (1999). Thalamocortical axons are influenced by chemorepellent and chemoattractant activities localized to decision points along their path. Dev. Biol. , 208, 430-40. PMID: 10191056 DOI.
Wullimann MF & Puelles L. (1999). Postembryonic neural proliferation in the zebrafish forebrain and its relationship to prosomeric domains. Anat. Embryol. , 199, 329-48. PMID: 10195307
Wullimann MF, Puelles L & Wicht H. (1999). Early postembryonic neural development in the zebrafish: a 3-D reconstruction of forebrain proliferation zones shows their relation to prosomeres. Eur J Morphol , 37, 117-21. PMID: 10342441
Alcántara S, Ruiz M, D'Arcangelo G, Ezan F, de Lecea L, Curran T, Sotelo C & Soriano E. (1998). Regional and cellular patterns of reelin mRNA expression in the forebrain of the developing and adult mouse. J. Neurosci. , 18, 7779-99. PMID: 9742148
Hatanaka Y & Jones EG. (1998). Early region-specific gene expression during tract formation in the embryonic rat forebrain. J. Comp. Neurol. , 395, 296-309. PMID: 9596525
Wong CJ. (1997). Connections of the basal forebrain of the weakly electric fish, Eigenmannia virescens. J. Comp. Neurol. , 389, 49-64. PMID: 9390759
Shimamura K & Rubenstein JL. (1997). Inductive interactions direct early regionalization of the mouse forebrain. Development , 124, 2709-18. PMID: 9226442
Parmantier E, Braun C, Thomas JL, Peyron F, Martinez S & Zalc B. (1997). PMP-22 expression in the central nervous system of the embryonic mouse defines potential transverse segments and longitudinal columns. J. Comp. Neurol. , 378, 159-72. PMID: 9120057
Alvarez-Bolado G, Rosenfeld MG & Swanson LW. (1995). Model of forebrain regionalization based on spatiotemporal patterns of POU-III homeobox gene expression, birthdates, and morphological features. J. Comp. Neurol. , 355, 237-95. PMID: 7608343 DOI.
Papalopulu N. (1995). Regionalization of the forebrain from neural plate to neural tube. Perspect Dev Neurobiol , 3, 39-52. PMID: 8542255
de la Llave R S & Tompaidis. (1994). Nature of singularities for analyticity domains of invariant curves. Phys. Rev. Lett. , 73, 1459-1463. PMID: 10056799 DOI.
Price M. (1993). Members of the Dlx- and Nkx2-gene families are regionally expressed in the developing forebrain. J. Neurobiol. , 24, 1385-99. PMID: 7901324 DOI.
Bulfone A, Puelles L, Porteus MH, Frohman MA, Martin GR & Rubenstein JL. (1993). Spatially restricted expression of Dlx-1, Dlx-2 (Tes-1), Gbx-2, and Wnt-3 in the embryonic day 12.5 mouse forebrain defines potential transverse and longitudinal segmental boundaries. J. Neurosci. , 13, 3155-72. PMID: 7687285
Terms
| Neural Terms |
|---|
Neural Development
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 25) Embryology Neural - Prosomere. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Prosomere
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G