Neural - Spinal Cord Development
| Embryology - 26 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The spinal cord is the central nervous system part that extends into the axial skeleton and provides the two-way traffic required to interact with our environment. During pregnancy, early development of the spinal cord is influenced by the maternal dietary requirement for folate for closure of the neural tube. Later development requires the contribution of neural crest associating with the cord to form the dorsal root ganglia and ventral sympathetic ganglia. The animal models of spinal cord development has also been a key models of patterning, establishing ventral and dorsal compartments based upon surrounding signals.
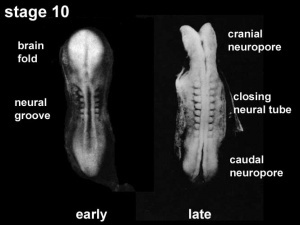
The early central nervous system begins as a simple neural plate that folds to form a groove then tube, open initially at each end. Failure of these opening to close contributes a major class of neural abnormalities (neural tube defects).
Neural development is one of the earliest systems to begin and the last to be completed after birth. This development generates the most complex structure within the embryo and the long time period of development means in utero insult during pregnancy may have consequences to development of the nervous system.
Within the neural tube stem cells generate the 2 major classes of cells that make the majority of the nervous system: neurons and glia. Both these classes of cells differentiate into many different types generated with highly specialized functions and shapes. This section covers the establishment of neural populations, the inductive influences of surrounding tissues and the sequential generation of neurons establishing the layered structure seen in the brain and spinal cord.
- Neural development beginnings quite early, therefore also look at notes covering Week 3- neural tube and Week 4-early nervous system.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Spinal Cord Development | Spinal Cord Embryology | |
| Older papers |
|---|
|
Neural Development Overview
Neuralation begins at the trilaminar embryo with formation of the notochord and somites, both of which underly the ectoderm and do not contribute to the nervous system, but are involved with patterning its initial formation. The central portion of the ectoderm then forms the neural plate that folds to form the neural tube, that will eventually form the entire central nervous system.
- Early developmental sequence: Epiblast - Ectoderm - Neural Plate - Neural groove and Neural Crest - Neural Tube and Neural Crest
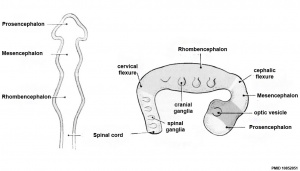
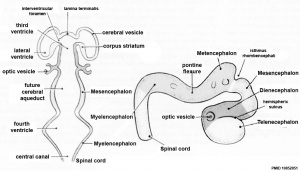
| Neural Tube | Primary Vesicles | Secondary Vesicles | Adult Structures |
|---|---|---|---|
| week 3 | week 4 | week 5 | adult |
| prosencephalon (forebrain) | telencephalon | Rhinencephalon, Amygdala, hippocampus, cerebrum (cortex), hypothalamus, pituitary | Basal Ganglia, lateral ventricles | |
| diencephalon | epithalamus, thalamus, Subthalamus, pineal, posterior commissure, pretectum, third ventricle | ||
| mesencephalon (midbrain) | mesencephalon | tectum, Cerebral peduncle, cerebral aqueduct, pons | |
| rhombencephalon (hindbrain) | metencephalon | cerebellum | |
| myelencephalon | medulla oblongata, isthmus | ||
| spinal cord, pyramidal decussation, central canal | |||
Early Brain Vesicles
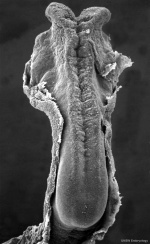
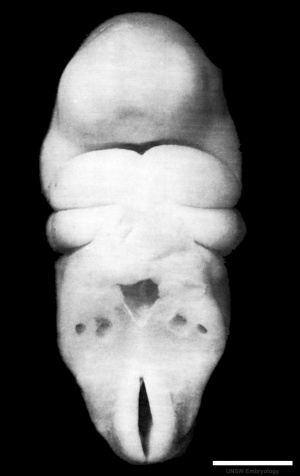
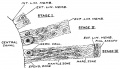
In week 3, the neural plate forms and the caudal end of the neural plate remains narrow compared to the cranial end which rapidly expands.
In week 4, when the plate folds to form the neural tube, the cranial end of the tube then forms a series of enlarged cavities (vesicles) that will eventually form the brain. The caudal end of the tube forms a narrower tube of relatively the same size along its length that will eventually form the spinal cord.
| Primary Vesicles | Secondary Vesicles |
|---|---|
|
|
| early embryonic | late embryonic |
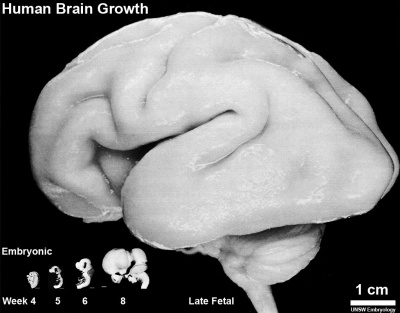
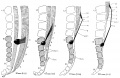
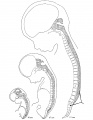
Direct comparison of brain growth embryonic and fetal period. Note the relative size of the spinal cord seen at the lower end of each image.
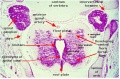
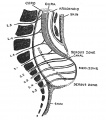
Spinal Cord Regions
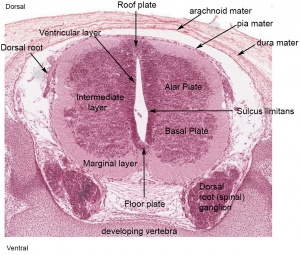
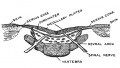
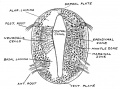
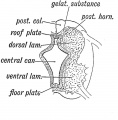
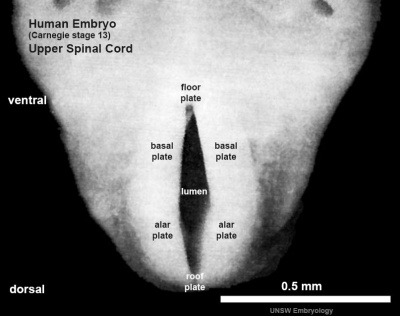
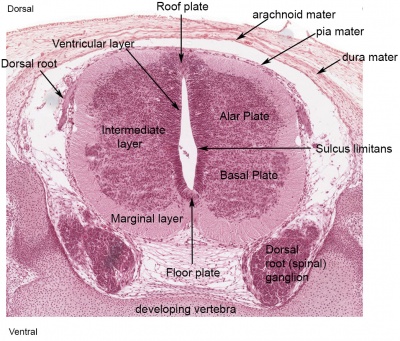
The neural tube forms similar regions around the wall along its length, including the spinal cord. The floor and roof plate are specialised developmental regions, important embryonic "patterning" regions.[8]
- Floor plate - thin wall region that overlies the notochord. Ventral patterns the spinal cord, both floor plate and notochord produce Sonic hedgehog (Shh) (see also notochord)
- Basal plate - thick wall region lying either side of the floor floor plate. The ventral horn motor neurons develop here and extend axons out of the spinal cord to innervate developing skeletal muscle. Tracts formed by axons surround these horns and project both up and down the spinal cord.
- Alar plate - thick wall region lying either side of the roof floor plate. The sensory dorsal horn develops there and receives axons from the sensory structures outside the spinal cord. The adult horn is divided into 6 laminae (I to VI). Tracts formed by axons surround these horns and project both up and down the spinal cord.
- Roof plate - thin wall region that underlies the dorsal ectoderm epithelium. Dorsal patterns the spinal cord, the roof plate produces Bone morphogenetic proteins (BMPs).[9][10]
- Lumen - neuroepithelium lined fluid-filled space continuous with the brain ventricular system.
| Week 4 | Week 8 |
|---|---|
|
|
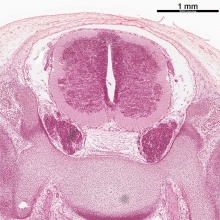
| Stage 13 Spinal cord cross-section (upper part of cord). labeled image | unlabeled image |
Stage 22 Spinal cord cross-section (ventral is at bottom of image) labeled image | unlabeled image |
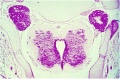
Embryonic Development
Week 4
| Spinal cord cross-section (upper part of cord) (Carnegie Stage 13) | |
|---|---|
|
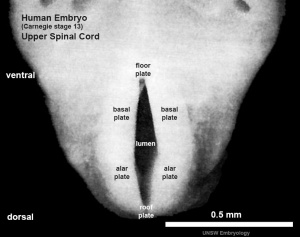
|
Week 8
Virtual Slide
|
These listed features link to zoomed views of the virtual slide with the named feature generally in the centre of the view.
Use the (-) at the top left of the screen to see where this feature is located. | ||||
| Spinal Cord Features | Other Features
| ||||
Fetal Development
- Links: fetal
Conus Medullaris
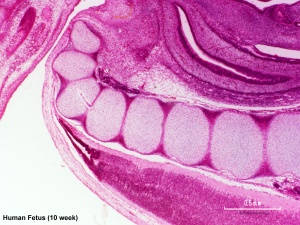
The conus medullaris (Latin, "medullary cone") is the tapered, lower end of the spinal cord. An ultrasound study of the position of the spinal cord conus medullaris at 18-22 weeks (20 to 24 weeks ((GA}}) showed that it ended adjacent to vertebrae L2, L2-3 vertebral space, and L3 (73/78, 93%).[11]
Plexus Development
The spinal nerves initially leave the spinal cord at each individual segmental levels. At various levels they then form an intersecting network of nerves, a plexus, from which mixed segmental nerves emerge.
Cervical Plexus
Search PubMed: cervical plexus embryology
Brachial Plexus
- Search PubMed: brachial plexus embryology
Lumbar Plexus
| Adult Lumbar Plexus | |
|---|---|
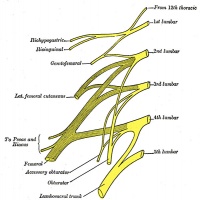
plexus lumbalis |
|
- Search PubMed: lumbar plexus embryology
Sacral Plexus
Dermatomes
A dermatome represents the area of skin that is mainly supplied by a single spinal nerve. Therefore each spinal nerve can be "mapped" to a region of the external body surface and that this "map" is established before embryonic limb rotation.
Molecular
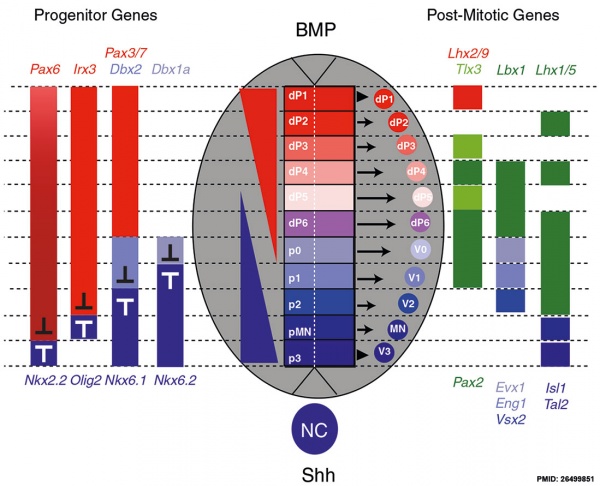
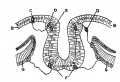
Neural tube Dorsoventral Patterning by SHH BMP[12]
Dorsoventral domains are established by opposing concentration gradients of Sonic Hedgehog (Shh) and Bone Morphogenic Protein (BMP).
- left - These regulate progenitor gene expression. The progenitor genes cross-repress each other to establish domain boundaries.
- right - Each domain will give rise to a specific cell type that expresses various post-mitotic differentiation genes.
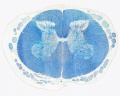
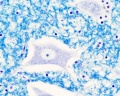
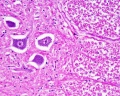
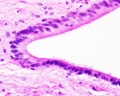
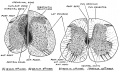
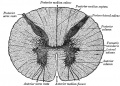
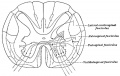
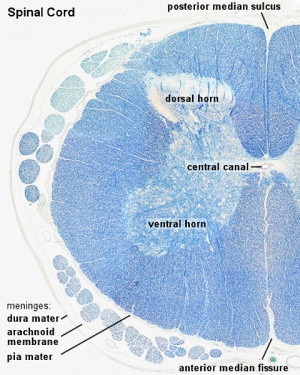
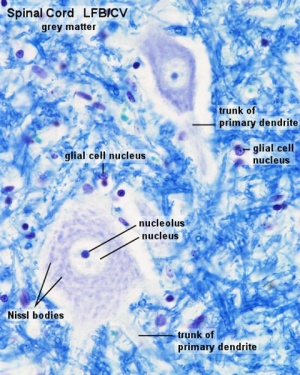
Spinal Cord Histology
Identify gray and white matter, central canal (surrounded by ependymal cells), dorsal and ventral horns, meninges (pia, arachnoid and dura mater), subarachnoid space with dorsal and ventral rootlets, blood vessels, a motor neurone with a cell body (soma), nucleus, nucleolus, Nissl granules, an axon with axon hillock area, dendrites, glial cells (oligodendrocytes, astrocytes).
| Spinal cord (Luxol Fast Blue) | |
|---|---|
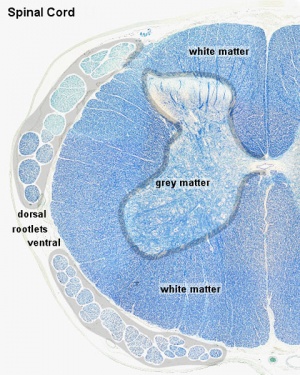
|
|
| Spinal cord - Grey and white matter | |
|---|---|
|
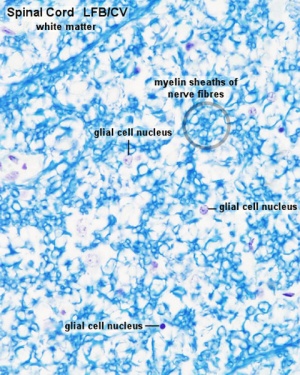
|
| Spinal cord - Grey matter | |
|---|---|
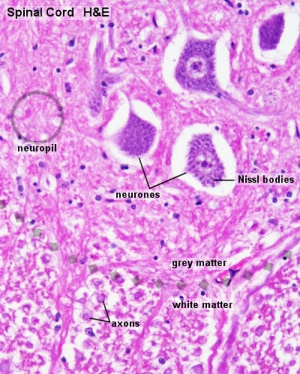
Grey matter (HE) |
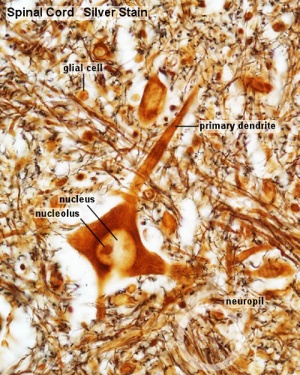
Grey matter (silver) |
- Spinal Cord: Overview 1 | Overview 2 | Overview animation | Grey matter | Grey matter | Grey matter | White matter | Overview unlabeled | Grey matter unlabeled 1 | Grey matter unlabeled 2 | White matter unlabeled 1 | Ependymal cells unlabeled
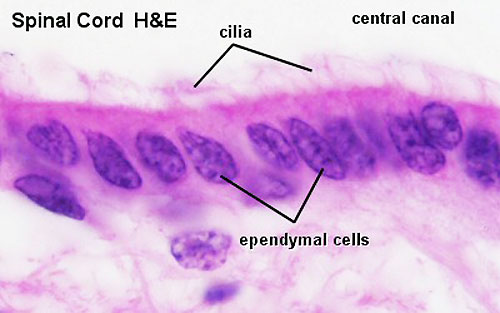
|
| |||
| <mediaplayer width='600' height='230' image="http://php.med.unsw.edu.au/embryology/images/5/5f/Mouse_ependymal_cilia_01-icon.jpg">File:Mouse_ependymal_cilia_01.mp4</mediaplayer> |
Abnormalities
Spinal Muscular Atrophy
ICD-11 8B61 Spinal muscular atrophy (ICD-10-CM Diagnosis Code G12.9)
Spinal muscular atrophy (SMA) is a rare neural autosomal recessive genetic condition that leads to a loss of motor neurons and then progressive muscle wasting. A new drug treatment using nusinersen[13] (trade name Spinraza®), see reviews[14][15], has led to the recent inclusion of SMA in the newborn heel-prick test (Guthrie test). Clinical classification of SMA type depends upon the age of onset and highest level of motor function achieved.
This genetic disease exists in several different forms related to SMN1 gene on chromosome 5q13:
|
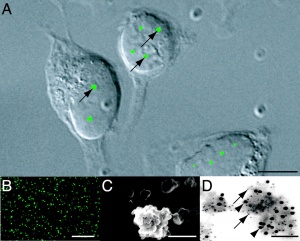 Cajal bodies panel (D) EM purified CB, SMN (arrowheads).[16] |
The Hammersmith Functional Motor Scale (HFMS), was first developed in 2003 and recently revised by international collaboration[17] as a tool for assessment of the physical abilities of SMA type 2 and type 3 patients with limited ambulation. (More? Hammersmith Functional Motor Scale - 2015 revision)
References
- ↑ Gray de Cristoforis A, Ferrari F, Clotman F & Vogel T. (2020). Differentiation and localization of interneurons in the developing spinal cord depends on DOT1L expression. Mol Brain , 13, 85. PMID: 32471461 DOI.
- ↑ Iulianella A & Stanton-Turcotte D. (2019). The Hedgehog receptor Patched1 regulates proliferation, neurogenesis, and axon guidance in the embryonic spinal cord. Mech. Dev. , 160, 103577. PMID: 31634536 DOI.
- ↑ Ohmura Y, Morokuma S, Kato K & Kuniyoshi Y. (2018). Species-specific Posture of Human Foetus in Late First Trimester. Sci Rep , 8, 27. PMID: 29311655 DOI.
- ↑ Diez Del Corral R & Morales AV. (2017). The Multiple Roles of FGF Signaling in the Developing Spinal Cord. Front Cell Dev Biol , 5, 58. PMID: 28626748 DOI.
- ↑ Kicheva A, Bollenbach T, Ribeiro A, Valle HP, Lovell-Badge R, Episkopou V & Briscoe J. (2014). Coordination of progenitor specification and growth in mouse and chick spinal cord. Science , 345, 1254927. PMID: 25258086 DOI.
- ↑ Demireva EY, Shapiro LS, Jessell TM & Zampieri N. (2011). Motor neuron position and topographic order imposed by β- and γ-catenin activities. Cell , 147, 641-52. PMID: 22036570 DOI.
- ↑ Pyrgaki C, Trainor P, Hadjantonakis AK & Niswander L. (2010). Dynamic imaging of mammalian neural tube closure. Dev. Biol. , 344, 941-7. PMID: 20558153 DOI.
- ↑ Wilson L & Maden M. (2005). The mechanisms of dorsoventral patterning in the vertebrate neural tube. Dev. Biol. , 282, 1-13. PMID: 15936325 DOI.
- ↑ Chizhikov VV & Millen KJ. (2004). Mechanisms of roof plate formation in the vertebrate CNS. Nat. Rev. Neurosci. , 5, 808-12. PMID: 15378040 DOI.
- ↑ Chizhikov VV & Millen KJ. (2005). Roof plate-dependent patterning of the vertebrate dorsal central nervous system. Dev. Biol. , 277, 287-95. PMID: 15617675 DOI.
- ↑ Perlitz Y, Izhaki I & Ben-Ami M. (2010). Sonographic evaluation of the fetal conus medullaris at 20 to 24 weeks' gestation. Prenat. Diagn. , 30, 862-4. PMID: 20582935 DOI.
- ↑ Zannino DA & Sagerström CG. (2015). An emerging role for prdm family genes in dorsoventral patterning of the vertebrate nervous system. Neural Dev , 10, 24. PMID: 26499851 DOI.
- ↑ Finkel RS, Mercuri E, Darras BT, Connolly AM, Kuntz NL, Kirschner J, Chiriboga CA, Saito K, Servais L, Tizzano E, Topaloglu H, Tulinius M, Montes J, Glanzman AM, Bishop K, Zhong ZJ, Gheuens S, Bennett CF, Schneider E, Farwell W & De Vivo DC. (2017). Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. N. Engl. J. Med. , 377, 1723-1732. PMID: 29091570 DOI.
- ↑ Gidaro T & Servais L. (2018). Nusinersen treatment of spinal muscular atrophy: current knowledge and existing gaps. Dev Med Child Neurol , , . PMID: 30221755 DOI.
- ↑ Vukovic S, McAdam L, Zlotnik-Shaul R & Amin R. (2018). Putting our best foot forward: Clinical, treatment-based and ethical considerations of nusinersen therapy in Canada for spinal muscular atrophy. J Paediatr Child Health , , . PMID: 30246272 DOI.
- ↑ Ogg SC & Lamond AI. (2002). Cajal bodies and coilin--moving towards function. J. Cell Biol. , 159, 17-21. PMID: 12379800 DOI.
- ↑ Ramsey D, Scoto M, Mayhew A, Main M, Mazzone ES, Montes J, de Sanctis R, Dunaway Young S, Salazar R, Glanzman AM, Pasternak A, Quigley J, Mirek E, Duong T, Gee R, Civitello M, Tennekoon G, Pane M, Pera MC, Bushby K, Day J, Darras BT, De Vivo D, Finkel R, Mercuri E & Muntoni F. (2017). Revised Hammersmith Scale for spinal muscular atrophy: A SMA specific clinical outcome assessment tool. PLoS ONE , 12, e0172346. PMID: 28222119 DOI.
Reviews
Borodinsky LN. (2017). Xenopus laevis as a Model Organism for the Study of Spinal Cord Formation, Development, Function and Regeneration. Front Neural Circuits , 11, 90. PMID: 29218002 DOI.
Welniarz Q, Dusart I & Roze E. (2017). The corticospinal tract: Evolution, development, and human disorders. Dev Neurobiol , 77, 810-829. PMID: 27706924 DOI.
Greene ND & Copp AJ. (2009). Development of the vertebrate central nervous system: formation of the neural tube. Prenat. Diagn. , 29, 303-11. PMID: 19206138 DOI.
Ulloa F & Martí E. (2010). Wnt won the war: antagonistic role of Wnt over Shh controls dorso-ventral patterning of the vertebrate neural tube. Dev. Dyn. , 239, 69-76. PMID: 19681160 DOI.
Dasen JS & Jessell TM. (2009). Hox networks and the origins of motor neuron diversity. Curr. Top. Dev. Biol. , 88, 169-200. PMID: 19651305 DOI.
Dessaud E, McMahon AP & Briscoe J. (2008). Pattern formation in the vertebrate neural tube: a sonic hedgehog morphogen-regulated transcriptional network. Development , 135, 2489-503. PMID: 18621990 DOI.
Molyneaux BJ, Arlotta P & Macklis JD. (2007). Molecular development of corticospinal motor neuron circuitry. Novartis Found. Symp. , 288, 3-15; discussion 15-20, 96-8. PMID: 18494249
Glenn OA & Barkovich AJ. (2006). Magnetic resonance imaging of the fetal brain and spine: an increasingly important tool in prenatal diagnosis, part 1. AJNR Am J Neuroradiol , 27, 1604-11. PMID: 16971596
Glenn OA & Barkovich J. (2006). Magnetic resonance imaging of the fetal brain and spine: an increasingly important tool in prenatal diagnosis: part 2. AJNR Am J Neuroradiol , 27, 1807-14. PMID: 17032846
Sadler TW. (2005). Embryology of neural tube development. Am J Med Genet C Semin Med Genet , 135C, 2-8. PMID: 15806586 DOI.
Placzek M & Briscoe J. (2005). The floor plate: multiple cells, multiple signals. Nat. Rev. Neurosci. , 6, 230-40. PMID: 15738958 DOI.
Articles
Saitsu H & Shiota K. (2008). Involvement of the axially condensed tail bud mesenchyme in normal and abnormal human posterior neural tube development. Congenit Anom (Kyoto) , 48, 1-6. PMID: 18230116 DOI.
Books
- Bayer S.A and Altman J. The Spinal Cord from Gestational Week 4 to the 4th Postnatal Month CRC Press 2002 Print ISBN: 978-0-8493-1420-9 eBook ISBN: 978-1-4200-4018-0 http://www.crcnetbase.com/doi/book/10.1201/9781420040180
Search PubMed
November 2010 search "Spinal Cord Embryology" All (7631) Review (641) Free Full Text (1562)
Search Pubmed: Spinal Cord Embryology | Spinal Cord Development
Additional Images
Historic
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
| Filum Terminale (1919) |
|---|
| Streeter GL. Factors involved in the formation of the filum terminale. (1919) Amer. J Anat. 22(1): 1-11.
|
| Human Embryology And Morphology (1921) |
|---|
| Keith, A. Human Embryology And Morphology (1921) Longmans, Green & Co.:New York.
|
| Anatomy of the Human Body (1918) |
|---|
| Gray, H. Anatomy of the Human Body. Philadelphia: Lea & Febiger, 1918.
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 26) Embryology Neural - Spinal Cord Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Neural_-_Spinal_Cord_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G