History:Paper - Description of a Human Embryo of Twenty-two paired Somites
| Embryology - 12 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Girgis A. Description of a human embryo of twenty-two paired somites. (1926) J Anat. 60(4): 382-410.1. PMID 17104111
| Online Editor |
|---|
| This 1926 paper by Girgis describes a human embryo of 22 somite stage embryo would be similar to a Carnegie stage 12 (26 - 30 days), caudal neuropore closes, Somite Number 21-29.
|
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Description of a Human Embryo of Twenty-two paired Somites
Girgis
Clinical History and Age
A human chorionic vesicle was kindly presented to me in November, 1923, by Dr Mahfouz Bey, Professor of Midwifery at Kasr-el-Eini Hospital,Cairo. He gave me the following short history without dates: "Copulation took place 2weeks after menstruation. Abortion occurred 5 days after the succeeding menstruation was due; haemorrhage two days before the abortion. The period between copulation and abortion is 18 days." Whether that was the only copulation between the two menstrual periods or the last before the abortion is not stated. If it was the only copulation,the fertilisation age of the embryo will be 17 days, or 15 days ifthe embryo ceased to grow after the haemorrhage which occurred 2days before the abortion. If copulation had taken place preceding the one mentioned inthe history, we falback on the menstruation age for estimating the age of the embryo. This would be 37 days, taking the 2 weeks starting from the end of menstruation and adding 5 days, being the average period of flow.
The greatest length of the embryo measures 2-9mm. and according to Mall's age curve, an embryo of 3mm. longhas41daysmenstruationageand 30 days copulation age, which is the probable age of this embryo when compared with other embryos of more definite age. According to His's curve, an embryo of 3mm. long has copulation age of 17 days, which agrees with the age given in the history.
Peter Thompson, in describing an embryo of 23 somites and 2-5 mm. long, says that "it resembles in many ways His's embryo L.G. which was 2-15 mm. in length and estimated to be about 15 days old."
Davis described an embryo of 20 somites in which three copulations have taken place during two menstruations, and from his data he works out three copulation ages, the first 17 days, the second 24 to 27 days and the third 31 to37 days, and he believes that the last is the right age of his embryo. Hesays:
The first copulation age (17 days) is too young to merit serious consideration, for by subtracting one day, as suggested by Triepel (1914) and Bryce and Teacher (1908), the fertilisation age would be 16 days, just one day older than the Bryce-Teacher specimen. Moreover, this coition occurred during the period in which, according to Siegel (1915) and Issmer (1889), intercourse is least likely to be fruitful. The probable fruitful coition, therefore, is reduced to the second or to the third preceding one. From the second we obtain a copulation age of 24 to 27 days, and from the third 31 to 37 days. According to the age curve of Mall (1918) a 3 mm. embryo should have a menstruation age of 40 to 41 days. By comparing the data at hand with this age curve, it is clear that the menstruation age of 42 days is probably the correct one. The copulation age according to Mall's chart would be 30 days. This falls between the possible copulation ages of 27 and 31 days, as shown in Table I, and may, therefore, be regarded as very close to the actual age of the embryo.
Johnson describes an embryo of 24 somites and he says the age of the specimen is uncertain from the data obtained, but it corresponds very closely to an embryo of 28 days as estimated by Mall.
Van den Broek describes an embryo of 22 somites but mentions no age.
The above-mentioned four embryos, Thompson's, Davis', Johnson's and Van den Broek's, resemble very closely this embryo, as willbe shown later,and according to Mall's age curve they range in age between 28 and 30 days, which is more probable than the age of 15 to 18 days given to them by His's age data.
Technique
The vesicle, which had been kept in spirit for some time before I received it, is oval in shape with a broad and a narrow pole.. It measures 18 mm in length and13 mm in diameter. It is evenly covered with vili excepting a small area on one side which is scantily covered. The embryo was attached to the inner surface on the side opposite this area. The vesicle was opened at the slightly villous area and the embryo with the attached part of the vesicle was cut out and transferred to formalin, then graded up through the alcohols, cleared in cedar-wood oil and embedded in paraffin.
The embryo measures 2-9 mm. in greatest length. It was cut transversely into 329 sections of 1Ou each. The sections were stained with haematoxylin and eosin.
The following observations were made in the Institute of Anatomy, University College, London, under the guidance of Professor J. P. Hill, to whom my gratitude is due for his advice and criticism, and kindness in allowing me to work in his Department.
Three models were made by the wax plate method, one of the whole embryo magnified 200 diameters with the left lateral wall removed, another of the brain and a third of the pharynx.
External Form
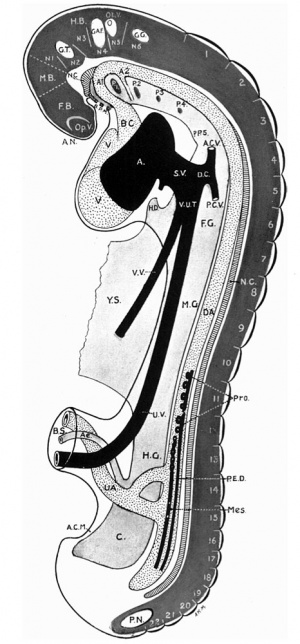
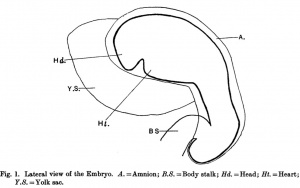
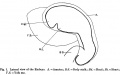
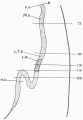
Fig. 1, of the left lateral view of the embryo, shows that the head is large and slightly flattened cephalo-caudally and inclined to be pointed, in which it resembles that of Thompson's embryo.
Caudal to the head there is the large swelling of the heart separated from the head by the cleft-like mouth. The dorsum of the embryo gives the outline of a well-marked curve which can be divided into three portions. The first, the dorsal flexure, is the largest and extends from the head to a level caudal to the yolk stalk, with its convexity directed dorsally; the second (ventral flexure) extends from the caudal limit of the first to a level caudal to the body stalk; it is the smallest and its convexity is directed ventrally. This will place the ventral flexure at a level caudal to that of the yolk stalk, agreeing in this respect with Davis' and Johnson's embryos, and disagreeing with the description of His,who places the ventral flexure at the level of the yolk stalk. The third curve extends from the caudal limit of the second to the tip of the tail; it is intermediate between the other two in size and its convexity is directed dorsally.
The yolk sac isvery large, nearly the same size as the embryo and isdirected cranially and to the right side of the embryo, which appears to lie on it. The body stalk is thick and bends caudal wards. The amnion can be seen enveloping the embryo and there is a space between them, especially at the head end. This space is absent in Van den Broek's embryo, where the amnion is applied closely to the embryo.
In the model the above features can be seen. in the region of the head the optic vesicles are seen as two swellings, one on either side of the cephalic end of the head. Viewed laterally there are to be seen the three ectodermal grooves of bronchial pouches, 1, 2 and 3. They are graduated in size, the 1st having the greatest vertical extent. These grooves mark out three visceral arches which are also graduated in size, the most cephalic or mandibular arch being the largest. The 4th visceral pouch, which is present in this embryo, has no corresponding groove on the surface, a conditional so found in Thompson's embryo.
In apparent continuity with the dorsal ends of the Ist and 2nd arches there are two small swellings, one to each arch. These swellings correspond to the ganglia of the trigeminus and acoustico-facialis, the former related to the 1st andthelattertothe2ndarch. Caudal to the swelling of the acoustico-facial ganglion and close to it, there is the small opening of the otic vesicle which is in line with the second ectodermal groove. The branchial region appears normal and resembles the corresponding region in Johnson's embryo (24 somites), Thompson's embryo (23 somites), Van den Broek's embryo (22 somites), and Davis' embryo (20 somites).
A short distance behind the last ectodermal groove, the 1st somite appears as a bulging from the lateral wall of the embryo near the dorsal middle line. This is followed by regular swellings of the subsequent somites. The first three are large but not distinctly marked out on the surface. The rest become reduced in size towards the tail.
The yolk sac was accidentally detached from the embryo during the process of dehydrating. The yolk stalk is a very wide canal connected with the mid-gut.
At the tail end, the posterior neuropore is widely open. On the ventral surface of the embryo, at the tail end, there is a depression representing the anal pit and at the bottom of it the ano-cloacal membrane.
The body stalk comes out from the caudal third of the embryo. Itisof large size and is bent caudally and to the right side of the embryo.
The embryo is twisted longitudinally with the head turned towards the right and the tail towards the left. The plane of the twist is in the middle of
the mid-gut.
Integument
The integument is generally of one layer of cubical ectodermal cels. It is thin on the dorsal surface over the neural tube where the cells are flattened in shape. There is no indication of the lens primordium opposite the optic vesicles and a thin layer of mesenchyme still intervenes between theopticvesiclesand the ectoderm overlying them. The ectoderm, which has folded in to form the wall of the otic vesicle, is formed of a thick layer of narrow columnar cels, and joins the surface ectoderm round the opening of the otic vesicle.
In the region of the bronchial arches, the ectoderm is thickened and the celsarecolumnarinshape.
In the region of the oral cavity, the ectoderm is slightly thickened, forming the superficial layer of the bucco-pharyngeal membrane, which is perforated in two places. In the roof of the oral cavity,the ectodermis in close apposition with the primitive infundibular recess of the fore-brain.
The ectoderm over the pericardial cavity is composed of one layer of flattened epithelium.
The Nervous System
The nervous system consists of the brain with its three main divisions, the ganglia of the Vth, VIIth, VIIIth, IXth, and Xth cranial nerves, the spinal cord and the ganglionic crest. The whole system lies underneath the ectoderm in the dorsum of the embryo and follows its curvature. Apart from a longitudinal slit which runs through the whole length of the neural tube, obviously an artefact due to shrinkage, the tube is closed except at its caudal end, where the posterior neuropore is still patent. In this feature it agrees with the embryos of Davis (20 somites), Van den Broek (22 somites), Thompson (23 somites) and Johnson (24 somites) , but it disagrees with the Janosik embryo (24 somites), which shows a patent anterior and closed posterior neuropore, and Bremer's embryo of 4mm. long whose neural tube is still open in front and behind. From this it appears that the order of closure of the neuropores varies, but in the majority of embryos the anterior neuropore closes first; and the stage at which closure takes place is at 20 to 24 somites for the anterior, and 30 somites for the posterior neuropore. Whether the delay of closure of the anterior neuropore or the reversal of order of closure is due to some abnormal condition of the embryos in which this occurs, or a variation of a normal condition, cannot be definitely stated. Davis believes that closure of the anterior neuropore first is the normal condition, and he looks upon the failure of its closure as a delay of the normal process or possibly the result of a complete arrest of development.
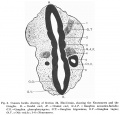
The Brain
The primary divisions of the brain (fig.2) are well marked. The fore-brain is a simple vesicle separated from the mid-brain by a slight groove on the outer surface. A small area at its cephalo-dorsal border is fused with the overlying ectoderm, marking the last point of closure of the anterior neuropore.
The optic vesicles are large somewhat dorso-ventrally flattened outgrowths which project, one from either side of the anterior part of the fore-brain. They occupy practically the entire lateral walls of the anterior region of the fore- brain, and have not yet begun to be constricted off from the latter. Their cephalic borders pass over directly into the front wall of the fore-brain, whilst caudally each vesicle is marked off from the lateral wall of the same by a crescenticgroove. Each vesicle communicates with the fore-brain ventricle by a large opening in front of a crescentic posterior margin.
The ectoderm, which overlies each vesicle, is not thickened, and is separated from the vesicles by a thin sheet of mesenchyme.
The subdivision of the fore-brain into telencephalon and diencephalon is indicated by a slight groove on the dorsal wall of the brain, practically on a level with the hinder borders of the optic vesicles. Corresponding to this groove there is present on the inner surface a slight ridge which runs down on the lateral wall, behind the opening of the optic vesicles. This ridge probably represents the velum transversum.
Below and behind the openings of the optic vesicles, and on the ventral surface of the fore-brain is a well-marked depression, forming the primitive infundibular recess, the projecting floor of which produces a bulging on the roof of the buccal cavity.
The wedge-shaped mid-brain is the shortest of the three divisions and is marked off by two constrictions from the fore and hind-brains, the anterior constriction best marked dorsally, the posterior constriction coinciding with the region of greatest flexure. It shows no evidence of subdivision, as described by Watt, in a younger embryo, and Gage, in an embryo of 29 somites. In this respect it resembles the mid-brain of Johnson's embryo of 24 somites, Thompson's embryo of 23 somites, Van den Broek's embryo of 22 somites and Davis' embryo of 20 somites.
The hind-brain is the largest of the three divisions; it is indeed about one- thirdofthetotallengthofthewholeneuraltube. Itisconnectedwiththe mid-brain by the isthmus and fades gradually into the spinal cord. There is a marked thinning of its roof, but no depression as in Van den Broek's embryo. On its lateral walls six pairs of neuromeres can be seen. They appear in the modelofthebrainaswedge-shapedbulgings,soarrangedthatthebasesofthe 1st, 3rd and 5th, are directed dorsally, and those of the 2nd, 4th and 6th are directed ventrally. The 1st pair has no definite limit in front from the rhombo- mesencephalic groove. The 2nd and 4th pairs of neuromeres correspond with definite down bulgings of the floor of the hind-brain, that related to the 4th pair being more prominent than that of the 2nd pair.
The number of neuromeres recorded in embryos of about the same stage as this is as follows:
Thompson's embryo of 23 somites has 7 neuromeres. Davis' , 20 , 9 VandenBroek's, 22 , 5
Johnson states that the number of neuromeres in his embryo cannot be ascertained.
Lying in relation with the neuromeres are the following structures: The ganglion of the trigeminus in close relation with the 2nd neuromere, the gang- lion of the acoustico-facialis with the 4th neuromere (the acoustico-facialis is the largest ganglion present and can be traced to the 2nd bronchial arch), and the ganglion of the glosso-pharyngeus lies in relation with the 6th neuromere. This arrangement agrees with that described by Gage, Johnson, Peter Thomp- son and Davis, and with Bartelmez, who shows in a table the relation of paretic, otic and postotic primary segments of the hind-brain to the secondary and definitive neuromeres.
The most marked of the neuromeres of this embryo is the 4th or facial which corresponds with the otic neuromere of Bartelmez, and the least distinct is the 6th, which is at a level cranial to the 1st somite.
The ganglion of the vagus is seen in the sections as a small cluster of cels caudal to the ganglion of the glosso-pharyngeus.
The otic vesicle lies in relation with the 5th neuromere and close to the ganglionoftheacoustico-facialis. Itisalargepear-shapedvesicle,withits long axis in the dorso-ventral direction, its broad end being directed dorsally.
Its wall is thick and the vesicle is still open to the exterior; its opening is situated at its dorsal broader end rather on the lateral aspect than on the summit, and the edges of the opening are continuous with the surrounding ectoderm.
The brain is sharply flexed at the junction of the mid-brain and the isthmus, the mid-brain forming the most cephalic part of the embryo. The longitudinal axes of the fore and mid-brains form approximately a straight line which makes an angle of rather more than 90' with the longitudinal axis of the cephalic half of the hind-brain. This flexure corresponds to the primary cephalic flexure in other embryos and involves the anterior end of the notochord. There is a slight flexure of the hind-brain immediately behind the down-bulging related to the 4th pair of neuromeres, and on a level with the otic vesicles.
These two flexures resemble those described by Peter Thompson and Van den Broek in embryos of about the same stage of development as this. Thompson says: "They are so different from those universally regarded as primary cerebral flexures, that one hesitates to go further than simply place them on record." He ends by saying: "Is it not possible that there may be certain flexures of a temporary character, which precede the primary flexures usually described?" Vanden Broek expresses a similar view.
Since the publication of Thompson's and Van den Broek's papers, Low, who described Pfannenstiel III embryo of 13-14 somites, finds a similar condition which agrees very closely with that of Thompson's description. Willin also finds a similar flexure in a brain of an embryo of 13 somites, and states: "The brain flexures do not agree with the His models of this stage but correspond more to the older embryos described by Thompson and Van den Broek."
Hochstetter also finds a similar condition in an embryo of 3.34 mm., somewhat older than Thompson's. He states: " There is a flexure on the ventral aspect of the hind-brain resembling that described by Thompson though not so marked. This disagrees with the condition described by His." He regards His's embryos, L.G., B.B., E.B., as abnormal or else not well preserved.
These flexures are probably due to exaggeration of the usual primary flexures. The flexure between the mid-brain and isthmus corresponds very closely with the primary cephalic flexure, and involves the hinder part of the mid-brain. The slight flexure in the hind-brain is probably due to the exaggerated dorsal flexure of the body of the embryo (as seen in Plate I) extending towards the hind-brain and involving its hinder part.
Spinal Cord
The spinal cord extends from the caudal end of the hind-brain to the tail. The cephalic part of the tube is of the same character as the caudal portion of the hind-brain, where it is compressed laterally and the long axis of its lumen is dorso-ventral in direction. It becomes more cylindrical caudal to the dorsal flexure. It is segmented through most of its length, the segments being separated from each other by circular constrictions which involve its entire circumference, so resembling the segmentation described by Watt and Johnson, and differing from that of Davis' embryo where they only appear on the lateral surface. These segments are very distinct in the region of the dorsal flexure. Eight distinct segments can be seen in the model in that region. Each neural segment lies between two consecutive somites and opposite each segment the dorsal aorta gives off a dorsal segmental artery. The lateral walls of the tube are thicker than its roof and floor; the roof is thinner than the floor and lies in contact with the dorsal ectoderm.
In cross-section (fig. 4), the lateral walls at the level of the dorsal flexure are differentiated into basal and alar laminae and the lumen of the neural canal is lozenge-shaped. This differentiation gets less and less marked towards the tail end where the lumen is circular in shape.
Ganglionic Crest
The ganglionic crest as a definite wedge-shaped structure is seen in the region between the 8th and 13th somites. In this region it is a continuous band of cells situated in the angle between the spinal cord and the somites near the dorsal middle line, and although it is a continuous mass, it shows signs of segmentation by the presence of constrictions at regular intervals. When traced towards the head end, it thins out and becomes less distinguishable; in some places it is only one cel thick. When it is traced towards the tail end, it again thins out and cannot be seen below the 18th somite.
The Digestive System
Oral cavity
The oral cavity is a transverse slit betweentheheadandthemandibulararches. It is wide transversely and narrow cephalo-caudally. The primitive infundibular recess abuts against its cephalic wall and bulges into its cavity. There is a small recess in the dorsal part of the cavity between its roof and the pharyngeal wall; this recesss is the beginning of Rathke's pocket. The bucco-pharyngeal membrane is perforated in two places.
Phaarynx
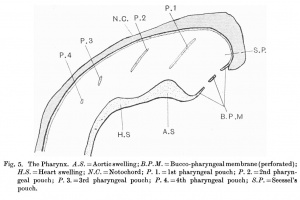
The pharynx (fig. 5) is a large cavity which is flattened dorso-ventrally. Its cephalic limit is the bucco-pharyngeal membrane, but beyond the dorsal limit of the membrane, the cavity extends cranially, forming Seessel's pouch. On each of the lateral walls of the pharynx there are four pharyngeal pouches. The first two are deep long grooves directed dorso-ventrally and inclined towards the head end of the embryo. They are coniIpresed cephalo-caudally, and their lateral walls are in contact with the floors of the firs two ectodermal grooves to forms the closing membranes, the entoderm and ectoderm of which are not yet fused.The third pouch is very small and its lateral wall does not reach the ectoderm of the 3rd groove. Davis and Johnson find the same condition in their embryos; although three ectodermal grooves are present, only the first two pharyngeal pouches reach the corresponding grooves. This disagrees with the view expressed by Grosser, who states: " In man the external grooves first become evident when the pharyngeal pouches have come into contact with the ectoderm." The 4th pouch is a very slight dimple on the lateral wall of the pharynx; the ectoderm opposite it is thickened but is not depressed into a groove.
In the roof of the pharynx there is a shallow groove running along the dorsal middle line parallel to the notochord which bends round the cephalic end of the pharynx.
Projecting into the cavity of the pharynx from its ventral wall in the middle line there are two well-marked swellings. The cephalic one is the larger and is situated on a level with the 1st visceral arch (mandibular arch). This knob-like swelling is moulded over the ventral aorta, just before it divides into the 1st pair of aortic arches. The caudal swelling is much smaller and lies dorsal to the atrium. The two swellings are separated from each other by a groove, which is continuous on either side with the 1st pouch. Kingsbury points out that the contour of the ventral wall of the pharynx is influenced by the pericardial area, which lies ventral to it, and there can be little doubt that the two swellings are produced by the parts underlying them, i.e. the ventral aorta underneath the larger causing an "aortic swelling," and the atrium underneath the smaller causing a "heart swelling." Johnson describes two similar swellings, but he follows Grosser in interpreting them as the tuberculum impar and the copula respectively. He also describes a third swelling caudal to the second one and believes it to be the heart swelling. Davis,on theotherhand,describes the same swellings but follows Kingsbury's interpretation of their being due to the influence of the pericardial area. Watt, in his younger twin embryos of 17-19 somites, describes a swelling due to the underlying ventral aorta.
Projecting from the ventral end of the Ist right pharyngeal pouch into its lumen is a villus-like process of cubical epithelial cells with a core of mesenchyme, which is directed dorsally. On the left side a similar process is present but it is shorter and appears as a slight fold of the entoderm of the cephalic wall of the 1st pouch. There is a small cavity in its core which looks very much like a blood vessel. A similar condition has been described by Watt, Davis and Johnson. Grosser also observed the same condition in a young embryos in which the 1st pharyngeal pouch is well developed. Hestates:"That it is an accidental structure or due to post-mortem changes seems to be excluded by the regularity of its occurrence. It disappears quite early (in embryos of 4.25, 5.0 and5-8mm. and in those still older it is wanting) and may perhaps be interpreted as a rudimentary internal gill."
Caudal to the "heart swelling" which is at the level of the 3rd pouch, the pharynx becomes narrow and its lumen triangular in shape, and its floor bends nearly at right angles. There is no indication of the tuberculum impair or the furcula. The ventral wall of the pharynx is much thicker than the dorsal.
Thyroid
Due to the plane of section which is practically parallel to the floor of the pharynx, the primordial of the thyroid is difficult to determine in this embryo; but there is a definite L.B thickening of the entoderm in the floor of the and5-8mm. and in those still older it is wanting) and may perhaps be interpreted as a rudimentary internal gill."
Caudal to the "heart swelling" which is at the level of the 3rd pouch, the pharynx becomes narrow and its lumen triangular in shape, and its floor bends nearly at right angles. Thereisno indication of the tuberculum impar or the furcula. The ventral wall of the pharynx is much thicker than the dorsal pharynx between the two swellings, which is the usual position of the thyroid primordium. This thickening of the entoderm extends cranially over the aortic swelling.
Lungs
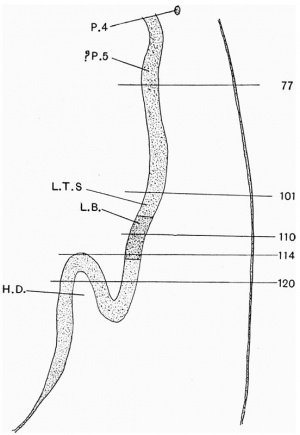
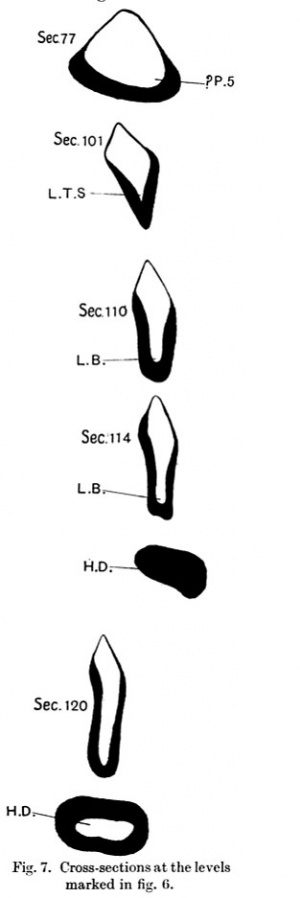
Caudal to the 4th pharyngeal pouch the fore-gut is prismatic in shape, and its base is directed ventrally. In this area there is a rounded swelling (figs. 6 and 7) which is slightly bifid and is situated a short distance from the 4th pouch. This appears to correspond with a similarswellinginThompson's embryo. Grosser interprets this swelling as a "questionable fifth pharyngeal pouch."
Dorsal to the sinus venosus and cephalic to the hepaticdiverticulum,the fore-gutbecomes compressed from side to side, thus forming two lateral walls and ventral and dorsal grooves. As it proceeds caudally the ventral groove becomes deeper and more pointed, and ends in a rounded thickening of the ventral wall. There is a very slight indication of unequal division of the thickenedareaintoalargerrightandasmaller left part. The thickened part is the primordium of the lung and the groove is the laryngo-tracheal sulcus.
Johnson describes two swellings, one of which corresponds to the bifid, and the other to the pointed swellings mentioned above, but he was unable to determine their significance from his specimen alone.
Thompson describes somewhat similar conditions and he interprets the bifid one as the lung bud and the caudal one as the beginning of the stomach. Grosser, however, concludes that the first swelling represents probably a 5th pair of J)haryngeal pouches and the second swelling, the lung bud.
In Davis' embryo the lung bud has already divided into a large right and a smaller left lobe, a condition older than in this embryo. Ileiss states that the lung area is divided into three parts, two lateral and one median, the latter fusing with the right part. The lung bud in this embryo is in too early a stage to show such a tri-partite condition if it occurs.
The primordium of the lung is very near the hepatic diverticulum, in fact the lung bud and the tip of the hepatic diverticulum can be seen in the same section (fig. 7). This is in accord with the descriptions of Davis and Grosser.
The region between the lung area and the hepatic diverticulum is also compressed laterally and according to Lewis is the gastric region.
Liver
Caudal to the lung bud a large hepatic diverticulum (figs. 6 and 7) is present in the ventral wall of the fore-gut and embedded in the transverse septum. It turns cranially and is directed towards the head end of the embryo where it expands transversely. This expansion is divided into two lateral parts by a shallow vertical groove on its ventral wall. From the walls of the distal part of the diverticulum very short processes sprout out giving its contour an irregular appearance. These are the primordia of the hepatic trabeculae; no further extension of them can be seen. Johnson,describing them in his embryo, states that: "Mlost of them (trabeculae) are indistinct and their extent is doubtful, for their cells closely resemble those of the mesenchyme and there is no definite line of separation between the two, such as a basement membrane." In this embryo there is no difficulty in seeing these sprout-like processes ofthe trabeculae, their cels being like those ofthe diverticulum. The proximal (attached) end of the hepatic diverticulum is round and has a thick ventral wall. It does not show the irregularity due to the trabeculae seen in the distal part. The diverticulum seems to be surrounded by blood vessels, and in two or three sections sonic of these vessels are seen to open into the vitelline veins, whichlieon either side of, and dorsal to the diverticulum.
Mid-gut and yolk stalk
A short distance caudal to the hepatic diverticulum the yolk stalk joins the gut; the stalk is a wide short canal. Itiscom- pressed laterally and bends to the right and cranially. It opens into the mid-gut, which is also flattened laterally. The ventral wall thins out as it proceeds from the hepatic diverticulum to theyolkstalk. No signofconstriction of the lateral wall of the yolk stalk is seen except the ridge on the right side due to the sharp bend.
Hind-gut and cloaca
Caudaltotheyolkstalktheshapeofthegutlunmen begins to change; it becomes more or less circular, and as it approaches the cloacal expansion it becomes compressed laterally again. Near the tail end, the hind-gut is greatly dilated to form the cloaca, the ventral wall of which bulges out and abuts against the ectoderm in the anal pit forming the ano-cloacal membrane. There is no thickening of ectoderm or fusion with the entoderm to be seen in the membrane. Caudal to the cloaca the hind-gut narrows again for a short distance, forming the end portion of the gut. The gut between the yolk stalk and cloaca is surrounded with a thick layer of mesenchyme, which is rich in blood vessels and blood islands.
Allantois
The entodermal allantois arises as a funnel-shaped tube from the ventral wall of the cephalic end of the cloaca. It is a very narrow canal directed cranially and ventrally to enter the body stalk where it ends blindly. When traced into the body stalk, it runs first between the two umbilical arteries, then it pierces the anastomosis of the two.
The Vascular System
Heart.
The heart forms a large prominence on the ventral aspect of the embryo, immediately caudal to the head. Its caudal limit is about the level of the middle of the embryo. When the model is viewed from the front, the heart looks like a tilted letter U with the tilt directed from right to left, and from tail to head. The limbs of the U are thick and the space between them is a narrow cleft. They turn dorsally and to the left of the middle line. The right limb is continuous with the ventral aorta, and the left turns sharply dorsal- wards to the sinus venosus. The heart lies free in the pericardial cavity except for a small attachment of the dorsal mesocardium to the dorsal surface of the atrium, and the sinus venosus which is embedded in the transverse septum.
The four divisions of the heart, the bulbus cordis, the ventricle, the atrium and the sinus venosus can be seen. The most dependent partof the heart, "the apex," lies to the left of the middle line.
The sinus venosus consists of two lateral horns and an intermediate part. The right horn is larger than the left and communicates freely with the atrium on its dorsal aspect; the opening is marked on the surface by a slight constriction. The sinus forms the most dorsal part of the heart and is embedded in the tranverse septum. Each horn receives on its lateral aspect the duct of Cuvier cranially and the vitello-umbilical trunk caudally. The duct is directed dorsally and receives cranially the anterior, and caudally the posterior cardinal vein. The vitello-umbilical trunk, a short wide vein lying in the transverse septum, is formed by the union of the vitelline and umbilical veins. The short intermediate part of the sinus venosus lies transversely in the septum, an(l a part of it is attached to the dorsal mesocardium. The mesocardium is attached to the dorsum of the sinus venosus and atrium centrally, and to the dorsal wall of the pericardial cavity dorsally.
There is a short wide vein joining the ventral aspect of the intermediate part of the sinus, and a similar but smaller one joining the right vitello- umbilical trunk; both are cranial to, and near the hepatic diverticulum. Van denBroek finds a vein similar to the former and thinks " it is possibly connected with the liver."
The atrium is placed dorsal to the bulbus cordis and truncus arteriosus. Its concave ventral surface is separated from them by a transverse narrow left. The bulk of the atrium lies on the left side of the ventral middle line and its cranial portion is divided by a notch into two rounded projections, each containing a part of the atrial cavity. The notch corresponds to the attach- mcnt of the septum primum internally. The septum is just beginning in this embryo; it arises from the ventral wall of the atrium at the convexity caused by the pushing in of the ventral wall by the bulbus cordis. The septum divides the cranial part of the cavity into two parts as mentioned above. As the atrium joins the ventricle, its cavity narrows, forming the atrial canal, which is indicated on the outer surface by a constriction separating the atrium from the ventricle.
The ventricle begins on the left side from the atrial canal and is first directed caudally and to the right, crossing the ventral middle line after forming the most caudal part of the heart (the apex), then it turns cranially and to the right side, reaching a level slightly caudal to the atrial projections; then it bends centrally and caudally towards the ventral line to become the bulbus cordis.
The bulbus cordis is well marked and lies between the ventricle and the truncusarteriosus. Its cavity is large at its commencement but narrows near the truncus. On its outer surface it is marked off from the ventricle by a wide shallow groove on its right side; on its left side it is separated by a narrow cleft from the atrium. It forms with the cranial end of the ventricle a V-shaped structure with the open angle directed cranially, with the right limb of the V directed towards the ventricle, and the left limb directed towards the truncus arteriosus (ventral aorta).
According to Keith and Griel, the bulbus cordis is completely incorporated in the right ventricle to form the infundibulum.
The endothelial cardiac tube follows closely the shape of the muscular one. It extends into the projections of the ventricle and the atrium, and is also constricted at the atrial canal. The cavity of the atrium is larger than that of the ventricle, and the opening of the sinus venosus is wide and lies in a trans- verse direction.
There is a large space between the muscular and the endothelial tubes in the ventricle and bulbus cords and,to a small extent, in the truncus arteriosus. In the atrium this space exists in the ventral part only, but in the greater part of its wall the two layers are close together. In this respect it agrees with Davis' embryo, but Mall says that the whole muscular wall of the atrium is in contact with the endothelial lining and he states: " This arrangement is so pronounced in the early heart that it affords away by which we may determine with precision the exact portion of the heart tube from which the atrium arises."
The endothelial tube consists of a single thin syncytial layer; in the space between it and the muscular wall of the heart, the endothelium sends out syncytial processes, some of which are stellate in shape, and others are fork-like. Johnson finds these processes in the bulbus cordis and ventricle, but in this embryo they are also found in the atrium, where the space exists. He says: "The significance of processes of this kind cannot be doubted. They represent the earliest beginnings of the so-called 'heart sinusoids,' which in embryos a little older are numerous. By their growth and anastomoses they give rise to the trabeculae of the heart." It is difficult to imagine the "heart sinusoids" giving rise to trabeculae. Perhaps he means the walls of the "heart sinusoids." The space is also filled with fine, faintly stained fibrils devoid of cells or nuclei forming a meshwork with empty spaces, and here and there are irregular masses faintly stained and devoid of cels. According to Tandler, the space between the two layers isfiledup with serous fluid,which appears as clot-like fibrous masses in sections. Johnson described similar masses and fibrils, which, as in this embryo, are due to coagulation of the serousfluid.
Arteries. The arteries comprise the ventral and dorsal aortae, the aortic arches and the umbilical arteries.
The ventral aorta is a very short vessel and soon divides into the 1st pair ofaorticarches. It lies in contact with the ventral wall of the pharynx, producing a swelling in it as mentioned before.
There are two pairs of aortic arches; they are directed dorsally and lie cranial to the corresponding pharyngeal pouches. The 1st arch is large and joins the beginning of the dorsal aorta, it is situated in the 1st visceral or mandibular arch. The 2nd arch is much smaller than the Ist and does not join the ventral aorta, and is situated in the 2nd visceral arch. There is a 3rd aortic arch on the right side only, which joins the dorsal aorta and is much smaller than the 2nd and only runs for a short distance in the right 3rd visceral arch.
The Ist arch gives off two branches; one of these arises opposite the fore-brain and represents the internal carotid artery. It runs cranially and joins the plexus at the root of the optic vesicle. The other arises some distance caudally, and joins the plexus round the hind-brain.
The dorsal aorta on either side begins from the dorsal end of the 1st aortic arch and ends behind by breaking up round the end of the gut to form the arterial plexus of the tail. It is not of the same size throughout; it is largest caudally and smallest at the level of the mid-gut.
The dorsal aortae run along the whole length of the embryo, lying close to the notochord and dorsal to the gut. They are at first separated by a wide interval, til they reach the level of the 7th somite, where they approach each other and run as two separate vessels with their medial walls very close together. At the level of the 10th somite, the two vessels begin to fuse together. Davis defines the term "fusion" as "implying the disappearance of the approximated endothelial surfaces, thus establishing a communication between the lumina of the vessels, and the fusion istermed complete when the opening is of such a size as to obliterate the original bi-tubular condition." Adopting this definition, the two aortae are fused at seven places (fig. 8); the 1st is complete and at the level of the 10th somite; the 2nd, 3rd and 4th are between the 11th and 13th somites, and are incomplete, i.e. the two vessels still retain their shape and the fusion is an hour-glass in shape; the 5th fusion is the largest and lies between the 14th and 16th somites, and is complete. The 6th and 7th fusions are the smallest and lie at the level of the 17th and 18th somites respectively, and appear as ifthe fusion is just beginning. Caudal to the level of the 18th somite the two dorsal aortae run as separate vessels again.
The dorsal aorta on either side gives off three sets of branches, (a) dorsal, (b) lateral and (c) ventral. These branches are arranged on the whole according LF to the description given by Evans. (a) There are 15 dorsal segmental arteries on the left and 12 on the right side. According to the table givenby Evans, this embryo falls between GrafSpee's embryo (15 somites), which has 11 dorsal segmental arteries and Thompson's embryo (23 somites), which has 21 arteries. Davis' embryo (20 somites) has 14 and Johnson's embryo (24 somites) has 24. Segmental loops are seen in the 1st, 3rd, 4th and 5th branches on the left, and in the 3rd, 5th, 7th and 10th on the right. In the set the dorsal branch divides into two branches,i.e. a short median which reaches the lateral wall of the medullary tube and divides into dorsal and ventral branches and a longlateral branch which joins the cardinal vein.
(b) The lateral branches are very small, and are seen in the nephric region of this embryo between the 11th and 20th somites, except the 1st artery on the left side, which is at the level of the 6th somite. There are five branches on the left and four on the right.They can be traced laterally towards the nephric system but only in two branches on the left side is it possible to make out a connection between the artery and the loop from the posterior cardinal vein.
(c) The ventral branches are not segmental in their arrangement, though they arise in pairs. According to Evans there is originally a ventral artery for each segment, but those of the more cranial body segments degenerate very early. There are 11 arteries on the left and 10 on the right side. The first two on either side are in the pharyngeal region. The rest are given off in the caudal half of the embryo; three branch off at the level of the yolk stalk and five at the level of the hind-gut.
The umbilical arteries leave the dorsal aorta by two large branches on either side. Very shortly after entering the body stalk they unite so as to enclose the allantoic duct, but they very soon separate again. The right vessel is larger than the left.
The plexus round the brain is distinguishable into three parts. The first is arranged round the fore-brain and is connected with the internal carotid artery and with the beginning of the vena capitis medial is.
The second part of the plexus is arranged round the mid-brain and is the smallest of the three.
The third part is the largest of the three and surrounds the hind-brain. It is connected with an artery arising from the 1st aortic arch at the level of the Vth ganglion. This artery divides into two branches, a cephalic one joining the plexus round the mid-brain, and a caudal branch joining the plexus round the hind-brain.
Veins
They comprise the anterior and posterior cardinal, the vitelline and the umbilical veins. The anterior cardinal begins from the caudal end of the vena capitis in the region of the hind-brain; as it proceeds caudally it comes to lie near the surface and on the dorso-lateral corner of the pericardial cavity. Itjoinsthe posterior cardinal to form the common cardinal vein (duct of Cuvier). It receives a few tributaries, one caudal to the otic vesicle and another from the lateral wall of the hind-brain.
The vena capitis medial is has been traced on the left side. It begins from the plexus round the root of the otic vesicle and runs caudally and close to the lateralwallofthebrain,into the anterior cardinal vein. It is freely connected with the plexus round the brain by various capillaries.
The posterior cardinal vein is a discontinuous vessel, which begins at the caudal end of the gut. It proceeds cranially in a position dorso-lateral to the coelomic cavity between it and the somites, and dorsal to the nephric system. It appears in fragments and is connected with the dorsal segmental arteries by the four pairs of loops mentioned above; it is at these connections that the vein is at its best, and this appears to support the view as regard its formation expressed by Evans. He states: "It is probable that lateral loops of the dorsal segmental arteries are instrumental in the formation of these veins, as in the case of the anterior cardinals. This method of formation of the posterior cardinal vein appears fundamental." The vein joins the caudal end of the duct of Cuvier.
The subeardinal veins are seen in the nephric region as disconnected capillaries, situated ventral to the nephric vesicles between them and the coelomic wall and connected with the lateral segmental branches of the dorsal aorta. Two are seen on the left and one on the right side; the fragments are longest at these connections.
The duct of Cuvier is a short medio-laterally flattened tube; it is situated at the level of the 3rd somite. It receives the anterior and posterior cardinal veins and opens medially into the sinus venous.
The umbilical veins begin at the chorionic end of the body stalk by the union of several large veins. These first form one vein which then breaks up in the body stalk into a plexus, which re-uites again into the right and left umbilical veins as they leave the body stalk. The left vein is shown in the model. Through its entire course, it lies in the body wall, from which it receives tributaries. At the level of the 5th somite the umbilical vein unites with the vitelline vein to form the vitello-umbilical trunk. This is short and wide, and is situated in the transverse septum and opens into the caudal aspect of the sinus venous.
The vitelline veins are formed by the plexus round the yolk stalk. They are smaller in cross-section than the umbilical veins, and run on the lateral wall of the mid-gut and enter the transverse septum, where they join the umbilical vein to form the vitello-umbilical trunk.
Blood islands
These are clusters of blood cels varying from one cell to several in diameter. They are distinguished from blood vessels by the entire absence of any sign of vessel wall. They are in contact with the mesenchyme and are chiefly seen in the mesenchyme of the head region, in the mesenchyme surrounding the hind-gut, and in villus-like processes which project from the coelomic wall in the region of the hind-gut.
The Nephric System
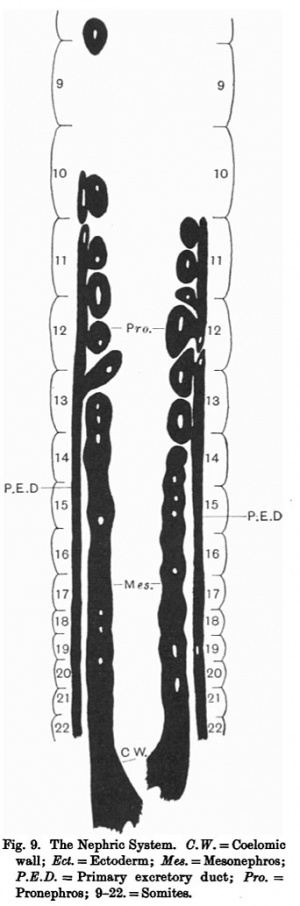
The nephric system (fig. 9) consists of pronephros, mesonephros and a primary excretory duct on either side. The pronephros is more in the form of vesicles than tubules and the mesonephros is in the form of a segmented cord.
The arrangement differs slightly on the two sides. On the right side there are six pronephric vesicles; the 1st is at a level between the 8th and 9th somites. The cranial three-quarter of the 2nd vesicle lies at the level of the 10th somite. The caudal quarter of the 2nd, the whole 3rd and the cranial half of the 4th vesicle lie at the level of the 11th somite. The caudal half of the 4th, the whole of the 5th, and cranial half of the 6th lie at the level of the 12th somite. The caudal half of the 6th lies at the level of the 13th somite. Each of the six vesicles has a lumen, and each is connected with the coelomic wall by a solid strand of cels.This connection is thicker in some than in others. In none of these strands (which represent remains of the nephrostomes) is a lumen seen. There is a gap between the 1st and 2nd vesicles of about 11 somites.
Caudal to the last vesicle, the mesonephros appears as a cord running to the end of the embryo, where it ends in the coelomic wall near the undifferentiated mesodermic mass of the tail. It has an interrupted lumen, and its cranial part is segmented, showing three segments separated by three constrictions, and there is a lumen in each of these segments. The mesonephros is also connected with the wall of the coelomic cavity by the remains of the nephrostomes.
The primary excretory duct begins with the 2nd pronephric tubule and 10 ends in contact with the ectoderm at the level of the last somite. It is interrupted between the 2nd and 3rd pro-nephric vesicles, but from the 3rd to 1 its end it runs as a continuous duct.
It has an interrupted lumen but this does not communicate with the lumen of the pronephros; its connection is 12 only by its wall. The duct is entirely separated from the mesonephros and is situated very close to the ectoderm.
On the left side there are also six pronephric vesicles but they differ in 14 their arrangement from the right side. The 1st, 2nd and cranial half of the 15 3rd are at the level of the 11th somite.
The caudal half of the 3rd, the whole of the 4th and cranial half of the 5th vesicles are at the level of the 12th somite. The caudal half of the 5th and the whole of the 6th vesicles are at the level of the 13th somite. they al possess a lumen. The meso- nephros and the primary excretory duct With the exception of the 1st vesicle, are like those on the right side, except that there appear to be six mesonephric segments instead of three, and the duct is interrupted between the 4th and 5th vesicles, instead of between the 2nd and 3rd on the right side.
The above description differs in one important respect from that given by Felix, and that is the asymmetry and entire absence of metameric arrangement in the pronephric vesicles of the two sides. According to Felix the pronephric duct is formed by the union of the caudal ends of the pronephric tubules, and its subsequent growth caudally. In this embryo the duct has almost become an independent structure, the connection of the pronephric vesicles with the duct being none too complete and in some being only one cell thick.
Somites
There are 22 pairs of somites extending from the caudal part of the hindbrain to the level of the posterior neuropore. The first pair of somites is in the region of the caudal part of the hind-brain. The somites are in different stages of development, more advanced towards the head and less so towards the tail end.
The last pair (22nd) is a solid mass just separating from the tail mass. The 21st, 20th, 19th, 18th and 17th pairs are cubical in shape, with dorsal, lateral, ventral and medial walls composed of epithelial cells about four layers thick and surrounding an empty myocoelic cavity. From the 16th to the17th pair, they become compressed and more or less oval in shape and their walls are arranged in two laminate, i.e. a lateral or dermatome and a medial. The latter divides into two parts, a dorsal (myotome), whose cells are epithelial in character, and a ventral (selerotome), the cells of which are mesenchymatous inappearance. The myocoele isstilfreefrom celsbut inthe 10th and 9th pairs it becomes invaded by cels from the sclerotome. Fromthe8thtothe 4th pair, the sclerotome attains a large size and is wedge-shaped in cross- section. Its apex is directed to the angle between the medullary tube and the notochord, and its base is separated from the dermatome by a slit-like space. The myotome is short and its dorsal end joins the corresponding end of the dermatome. The 3rd, 2nd and 1st pairs are not sharply separated from each other. Each consists of two laminate whose cels are epithelial in character. On cross-section these somites are triangular in shape; the apex is directed dorsally and the base is ill-defined and fuses with the adjacent mesenchyme.
Johnson and Watt found the same condition as described by Felix, though Johnson found that the mesonephros in his embryo is a cord-like structure with irregular swellings (the mesonephric vesicles), a condition similar to that ofthisembryo.
Notochord
The notochord begins caudal to the primitive infundibular recess and ends in the undifferentiated mesodermic mass of the tail. Its tapering cephalic extremity is situated immediately behind the attachment of the bucco- pharyngeal membrane and fuses with the entoderm of the roof of the pharynx for a short distance from that attachment; as it extends caudally from this region it is separately from, but lies close to the roof of the pharynx. Itgradually becomes close to the ventral surface of the medullary tube, a position it retains to its caudal end. Cranially, the notochord is sharply bent over the cephalic end of the pharynx, and there is a distinct swelling in the bent part which fits in the flexure between the mid-brain and isthmus (primary cephalic flexure).
In cross-section the notochord is cylindrical in shape, except at its cephalic end, where it is slightly laterally compressed.
It is invested completely on its lateral sides by mesenchyme; ventrally it is also surrounded by mesenehyme except where it is close to the roof of the pharynx. Dorsally it is surrounded by mesenchyme up to the level of the 7th somite. Caudally to this, the notochord is very closely applied to the ventral surface of the medullary tube.This agrees with the condition found by Johnson and others.
its the notochord is closely applied to the ventral surface of the medullary tube from the 7th somite backwards, itfollows the segmental constrictions and expansions of the tube, thus forming segmental flexures.
The cells forming the notochord are wedge-shaped; their nuclei lie towards theperiphery. In some sections a lumen is seen.The notochord is surrounded by a delicate membrane as described by Van den Broek.
Transverse Septum and Coelom
The transverse septum is a thick plate of mesenchyme with a mesothelial covering. It is placed in an oblique plane, starting from the ventral body wall caudal and ventral to the heart and at the level of the 8th somite. Itpasses obliquely, dorsally and cranially to the level of the 4th somite; its obliquity thus occupies a length of four somites. This corresponds to the position and obliquity of the septum of an embryo between the 2mm. and 4mm. embryos in Mall's scheme of the position of the septum of embryos between 2mm. and 24 mm. long, but Thompson, Davis, Johnson and Watt placethe septum in their respective embryos at a more cranial level than itoccupies in this embryo. Its dorso-cranial border is crescentic in shape with the cornua attached to the lateralbodywall. In the middle it is attached to the dorsal mesocardium, and on either side of this attachment the border is free, so that there is left an aperture of communication between the pericardial and pleuro-peritoneal cavities, and its mesothelial covering is continuous with that of the dorsal mesocardium. The fore-gut invades it dorsally, and in it the hepatic diverticulum is embedded. The part containing the gut and the diverticulum is much thickened, and the thickening bulges into the pericardial cavity, causing a concave impression on the dorsum of the heart, and fills up the space between it and the fore-gut. In cross-section the thickening is pear-shaped with the broad end on the left side where the heart is farthest from the gut. Atits caudal end the vitello-umbilical trunk on either side enters the septum to reach the sinus venosus, which lies across it. At its cephalic free border, the duct of Cuvier is carried medially to the sinus venosus through the corn on eitherside. No sign of Mall's pulmonary ridge is yet seen.
The Pericardial Cavity
The transverse septum divides the body cavity transversly into two unequal divisions; the cephalic division (pericardial cavity) is the smaller. It is a single cavity except for a short distance where it is divided dorsally by the dorsal mesocardium in the middle dorsal line. Its cranial limit is at the bulbus cordis and it is bounded dorsally by the pharynx and transverse septum, and centrally and laterally by the thin body wall. Its floor is formed by the transverse septum which is deficient dorso-laterally where the cavity communicates freely with the pleuro-peritoneal cavity.
The Pleuro-peritoneal Cavity
It begins at the level of the 5th somite and for some distance it lies dorsal to the pericardial cavity. Itisdividedbythe gut into two lateral divisions, i.e. the pleuro-peritoneal passages. The cephalic extremity of each division communicates with the pericardial cavity; cranial to the yolk stalk where the amnion is reflected from the caudal end of the pericardium, the two divisions communicate transversely and ventral to the gut. At the level of the attachment of the yolk stalk with the mid-gut and for some distance cranially and caudally to that level the two divisions communicate with the extra-embryonic cavity. At the region of the hind-gut the two divisions join ventral to the gut. Just in front of the level of the cloaca the two divisions of the pleuro-peritoneal cavity separate again where the umn- bilical vessels and allantois leave and enter the embryo. They remain separated up to their caudal termination so that in this region there is a ventral mesentery as well as a dorsal one.
The Amnion
The amnion forms a closed sac in which the embryo lies; it is formed by two layers of flattened epithelium with a thin layer of mesenchynme between them. The inner layer is continuous with the ectodermal integument of the embryo and the outer layer is continuous with the mesodermal lining of the body cavity.
The line of attachment of the amnion to the embryo is as follows: eranially the amnion is reflected from the caudal border of the ventral wall of the peri- cardial cavity, so that the whole heart, head and the region of the embryo cranial to the apex of the heart lie freely in the amniotic cavity. From the pericardial reflection the amnion sweeps round caudally and is reflected from the ventral border of the lateral body wall and proceeds to the body stalk. At the region of the stalk the two lateral lines of reflection begin to converge, leaving the body of the embryo and proceeding on the surface of the body stalk that faces the embryo. There they meet, forming a V-shaped attachment with the angle of the V directed towards the chorionic end of the body stalk. It will be seen that the line of reflection of the amnion is an elliptical opening on the ventral aspect of the embryo; through this opening the yolk stalk and body stalk emerge. The line of demarcation between the amnion and the lateral wall of the embryo is very marked, due to the sudden thickening of the bodv wall.
Summary
Dimensions
- Chorionic vesicle, 18x13mm.
- Embryo, 2-9mm.
Age
- About 30 days.
External Form
- Head is slightly flattened and flexed.
- Cervical and caudal flexures.
- Ventral flexure caudal to yolk stalk.
- Longitudinal twist; head to the right and tail to the left.
Somites
- 22. The last pair is still attached to the tail mass.
Notochord
- The thickened cranial end is bent and fused to the cephalic wall of the pharynx.
- Caudal end terminates in the tail mass.
- It is surrounded by a delicate membrane.
Nervous System
- Two flexures (Thompson's flexures) in the brain, one at the junction of the mid-brain and the isthmus, and the other in the hind-brain.
- The three main divisions of the brain (fore, mid, and hind-brains) are present.
- Fore-brain. Optic vesicles are separated from the ectodermn; no lens primordium. Primitive infundibular recess present. Slight indication of sub-division into telencephalon and diencephalon.
- Mid-brain. Shortest division.
- Hind-brain. Thinning of the roof. Six neuromeres. Ganglia of V, VII, VIII, IX and X. Otic vesicle still open to the exterior.
- Spinal Cord. Segmented for the most part.
- Ganglionic Crest. Present between 8th and 13th somites.
DiGestive System
- Oral Cavity. Oral plate perforated in two places. Beginning of Rathke's pouch.
- Pharynx. Four pharyngeal pouches; the first two reach the ectoderm. The ectoderm is thickened opposite the other two. Two swellings in the floor of the pharynx, the cranial one the larger and due to the ventral aorta, the caudal one small and due to the pericardium. Two processes ("internal gills" of Grosser) are seen in the 1st pair of pouches, one in each pouch.
- Thyroid. There is a thickening of the entoderm between the two swellings in the floor of the pharynx.
- Lung. Laryngo-tracheal sulcus and bifid bud lung are present caudal to the 4th pharyngeal pouch.
- Liver. A large hepatic diverticulum and trabeculae lie caudal and close to thelungbud.
- Yolk Stalk. Is very wide.
- Cloacal membrane and Anal pit are present.
Vascular System
- Heart. S-shaped stage. Four divisions well marked.
- Sinus venosus, atrium, ventricle and bulbus cordis. The latter is well marked. A large venous cavity appears on the ventral aspect of the sinus venosus. The sinus has two horns and an intermediate part. Each horn receives the duct of Cuvier and the vitello-umbilical trunk. Atrium shows the beginning of the septum primum. The endothelial cardiac tube follows the shape of the myo- cardial heart, and is separated from it by a space, except in the greater part of the atrium where it is in contact. The space contains coagulated serous fluid and cardiac trabeculae.
- Aortic Arches are two on either side; the 2nd is incomplete. A 3rd is just beginning on the right side.
- Dorsal Aortae fuse between the 10th and 18th somites. Dorsal segmental branches are 15 on the left and 12 on the right side. Ventral branches are 11 on the left and 10 on the right side. Lateral branches are 6 on the left and 2 on the right side.
- Umbilical arteries unite at the beginning of the body stalk. Each joins the aorta of the corresponding side by two branches. The right is larger than the left.
- Veins. Complete anterior and incomplete posterior cardinals. Beginning of the sub cardinal. Umbilical vein is larger than the vitelline.
Nephric System
- Six pronephric vesicles are present on either side. Each has a lumen except the 1st left. Mesonephros is a segmented cord and ends in the coelomic wall at the tail end.
- Primary excretory duct interrupted once on either side, and ends in the ectoderm at the level of the last somite. Connected by its wall only to the pronephric vesicles.
Transverse Septum
- Crescentic in shape; the middle part is much thickened. It lies obliquely between the 4th and 8th somites. Its dorsal border is attached to the dorsal body wall by the cornua and middle part of the crescent only, leaving two openings on either side of the middle line, through which the pericardial and the pleuro-peritoneal cavities communicate.
Conclusion
This embryo fals into a group of embryos of about the same stage of development whose descriptions have already been published.
Peter Thompson (1907) described Robert Meyer's embryo 300 of 23 somites. Van den Broek (1911) published an account of two embryos of 22-23 somites. Bujard (1915) gave an account of Eternod-Delaf's embryo of 20 somites, which he regarded as pathological. Johnson (1917) described an embryo of24 somites, and Davis (1923) described an embryo of 20 somites. All these embryos very closelyresembletheonedescribedinthispaper,andonthewholemy observa- tions confirm those made by the above-named authors, though there are certain pointsofdifference.
The flexures of the brain in this embryo agree with those first described by Thompson in 1907, and later by different authors. Van den Broek found "Thompson's flexures" (as I venture to call them) in his 22-somite embryo. Low, in 1908, described Pfannenstiel's embryo III of 13-14 somites and he states: " The brain flexures do not agree with the His models of this stage, but correspond more to the older embryos described by Thompson and Van den Broek." In 1913 Willin reported an embryo of 13 somites with "Thompson's
flexures." In1919 Hochestetter described a somewhat older embryo than Thompson's, and found similar flexures though less marked. He regarded His's embryo L.G., B.B., E.B., as abnormal or else not well preserved.
Thompson's view was that these two flexures are special flexures of a temporary character which precede the definitive ones. That may hold good for the more caudal of the two but the more cephalic appears to be none other than the usual primary cephalic flexure. The small size of the mid-brain and the narrowness of the fore-brain found in this embryo, and observed also in the figures of the brains of the embryos described by Thompson, Van den Broek and I-Lochstetter would appear to be features characteristic of the early human brain.
In the nephric system the pronephric tubules are in the form of vesicles, and the primary excretory duct is well formed, with one interruption only. This appears to be an older stage than that described by Felix, Johnson and Watt in embryos more or less of the same age as this. There is also another difference in the nephric system of this embryo from others and this is the entire absence of metamerism in the system.
While reviewing the literature on the subject I noticed the chief criteria usedforclassifyingearlyembryos arelengthandage. When comparing embryos of the same length or nearly so, a great difference is found in the stage of their development. For example, Broman's embryo measured 3mm. and has 35 somites, limb buds and other characteristics much older than Watt's embryo of 3:37mm. and Bremer's embryo of 4mm. Waterson described an embryo measuring 2-9mm. with 27 somites, and possessing other characteristics older than the 2-9mm. embryo described in this paper. Many other examples can be quoted to show the fallacy of the length of embryos as a criterion for classification. There are many factors which influence the length of an early embryo, i.e. the different degrees of shrinkage and flexures in different embryos, the different methods of measuring used by different observers and possibly also the normal variations between embryos such as are found between adults. As regards the determination of the age of embryos, ifwe could know the fertilization age that would be the best and most decisive criterion, but the fertilisation age will be always a hypothetical one worked out by varying data by different authors, and the margin of error, though it may be counted in a very few days, or even hours, makes a great deal of difference in the stage of development reached. Embryos in their early life grow very rapidly and a few days nay a few hours may transfer an embryo from one stage to another.
Davis advocates the closure of the neuropores as a reliable criterion. He says: "The closure of the neuropore is perhaps the best criterion we possess for the classification of this particular stage of embryonic development." The variation in the closure of the neuropore has been pointed out in this paper (p. 386), though it is not so frequent and marked as are variations in length andage. I find the number of somites is the best criterion we possess up to the present, especially as the somites appear very early and are easily demon- started.
Bibliography
DELMANN, H.B. (1925). "The development of the neural folds and cranial ganglia of the rat." Journ. Comp. Neurol. vol. XXXIX.
BARTELMEZ, G.W. (1923). "Thesub-divisionsoftheneuralfoldsinman." Journ.Comp.Neurol. vol. xxxv.
BREMER, J. L. (1906). "Description of a 4 mm. human embryo." Amer. Journ. Anat. vol. v. (1914). "The earliest blood-vessels in man." Amer. Journ.Anat.vol.xvi.
BUJARD, E. (1915). "Description d'un embryon humain (Etemod-Delaf) de 20 somites, avec flexiondorsale." Internat.Monatsschr.f.Anat.u.Phys.Bd.XXXI.
CHIARUGI, G. (1888). "Anatomia di un Embrione Umano della lunghezza di mm. 2-6 in linea retta." Inst.Anat.diSiena,Pisa.
DAVIS, C. L. (1923). "Description of a human embryo having 20 paired somites." Contrib. to Embryology Carnegie Inst. Wash. vol. xv.
Evans HM. The development of the vascular system. In Keibel F. and Mall FP. Manual of Human Embryology II. (1912) J. B. Lippincott Company, Philadelphia. pp570-708.
EVANS, H.M. (1912). "The development of the vascular system." Manual of Human Embryology (Keibel and Mall), vol. I.
FELIX, W. (1912). "The development of the uro-genital organs." Manual of Human Embryology (Keibel and Mall), vol. I.
FLINT, I. M. (1906). "The development of the lungs." Amer. Journ. Anat. vol. vi. GAGE,S.P.(1906). "A three weeks' human embryo with special reference to the brain and nephric system." Amer. Journ. Anat. vol. iv.
GROSSER, 0. (1912). "The development of the pharynx and of the organs of respiration." Manual of Human Embryology (Keibel and Mall).
HEISS, R. (1912). "Ueberderfriihe EntwicklungdermenschlichenLunge, nebsteinem Versuch einermechanischenBegriindungde rLappen. Anat.Anz.Bd.XLI. (1919). ZurEntwicklungund Anatomic der menschlichen Lunge. Arch.f.Anat.u.Physiol. Anat.Abth.
HIS,W. (1880-1885). Anatomiremenschlicher Embryonen.
HOCHSTETTER, F. (1919). Beitrdgezur Entwicklungs geschichte des menschlicher Gehirns. INGALLS,N.W.(1920). "A human embryo at the beginning of segmentation, with special reference to the vascular system." Contrib. to Embryology,vol.I.
JANOSIK ,J. (1887). "ZweijungemenschlicheEmbryonen." Arch.f.mikr.Anat.Bd.xxx. JOHNSON, F. P. (1917). "A human embryo of twenty-four pairs of somites." Contrib. to Embry- ology,vol.VI. (1916). "Notes on the neuromeres of brain and spinal cord." Anat.Record,vol.x.
KEIBEL, F. and ELZE (1908). Normentafel zur Entwicklungs geschichte des Menschen. Jena. KEITH,A.(1921). Human Embryology and Morphology.
KINGSBURY, B. F. (1915). "The development of the human pharynx." Amer. Journ. Anat. vol.XVIII.
Low A. Description of a human embryo of 13-14 mesodermic somites. (1908) J Anat Physiol. 42(3): 237-51. PMID 17232769 | PMC1289161
LOW, A. (1908). "Description of a human embryo of 13-14 mesodermic somites." Journ. Anat. and Phys. vol. XLII.
MALL, F. P. (1912). "On the development of the human heart." Amer. Journ. Anat. vol. XXIII.
(1918). "On the age of human embryos." Amer. Journ. Anat. vol. xxv.
(1910). "Coelom and diaphragm." Manual of Human Embryology (Keibel and Mall), vol.I.
MINOT, C.S. (1907). "The segmental flexures of the notochord." Anat. Record,vol.I.
NORRIS, E.H. (1918). "The early morphogenesis of the human thyroid." Amer.Journ.Anat.vol.XXIV.
STREETER, G.L. (1912). "The development of the nervous system." Manual of Human Embryology (Keibeland Mall), vol.I. Anatomy LX
TANDLER, J. (1912). "The development of the heart." Manual of Human Embryology (Keibel and Mall), vol. I.
Thompson P. Description of a human embryo of twenty-three paired somites. (1907) J Anat Physiol, 41(3):159-71. PMID 17232726
THOMPSON, PETER (1907). "Description of a human embryo of 23 paired somites." Journ. Anat. Phys. vol. XLI. (1908). "A note on the development of the Transverse Septum and the Liver." Journ. Anat. and Phys. vol. XLII.
TRIEPEL, H. (1914). "AlterbestimmungbeimenschlichenEmbrvonen." Anat.Anz.Bd.XLIX.
VANDENBROEK ,A.J.P. (1911). "ZurKasuistikjungermenschlicherEmbryonen." Anat.I Hefte, vol.XLTV.
VEIT, 0. and ESCH, P. (1922). "Untersuchung eines in situ fixierten, operativ gewonnenen menschlichenEiesderviertenWoche." Zeitschr. fur Anat. u. Entwicklungsges.Bd.LXIII.
WATERSON, DAVID (1914). "A human embryo of twenty-seven pairs of somites embedded in decidua." Journ. Anat. vol .XLIX.
WATT, J.C. (1915). "Description of two young twin embryos with 17-19 paired somites." Contrib. to Embryology, CarnegieInst. Wash. vol.I.
WILLIN, I. E. (1913). "A human embryo of 13 somites." Amer. Journ. Anat. vol. xv.
Wilson JT. Observations upon young human embryos. (1914) J Anat Physiol., 48(3): 315-51 PMID 17233002 PMC1288949
WILSON, J. T. (1914). "Observations upon young human embryos." Journ. Anat. and Phys. vol. XLVIII.
Description of Plate
Plate I
A. = Atrium; A 1, A 2 = Aortic arches; A.C.M.= Ano-cloacal membrane; A.C.V. = Anterior cardinal vein; Al. = Allantois; A N. = Anterior neuropore; B.C. = Bulbus cordis; B.P.M. = Bucco-pharyngeal membrane; B.S. =Body stalk; D.A. = Dorsal aorta; D.C. =Duct of Cuvier; F.B.=Fore-brain; F.G.= Fore-gut; G.A.F.=Ganglion acoustic-facialis; G.G.=Ganglion glosso-pharyngeus; G.T. = Ganglion trigeminus; H.B. = Hind-brain; H.D. =Hepatic diverticulum; H.G.=Hind-gut; M.B.= Mid-brain; Mes.=Mesonephric cord; M.G.=Mid-gut; NI-N6.= Neuromeres; N.C. = Notochord; Op.V.= Optic vesicle; Ot.V. = Otic vesicle;P -P5 = Pharyngeal pouches; P.C.V. = Posterior cardinal vein; P.E.D = Primary excretory duct; P.N.= Posterior neuropore; Pro.= Pronephric vesicles; S.V.=Sinus venosus; U.A. Um- bilical artery; U.V.=Umbilical vein; V.=Ventricle; V.U.T.=Vitello-umbilical trunk; V.V.= Vitelline vein; Y.S.= Yolkstalk; 1-22 = Somites.
Images
- Links: Fig 1 | Fig 2 | Fig 3 | Fig 4 | Fig 5 | Fig 6 | Fig 7 | Fig 8 | Fig 9 | Plate 1 | Girgis 1926 | Carnegie stage 12
Reference
Girgis A. Description of a human embryo of twenty-two paired somites. (1926) J Anat. 60(4): 382-410.1. PMID 17104111
Cite this page: Hill, M.A. (2026, Mayıs 12) Embryology Paper - Description of a Human Embryo of Twenty-two paired Somites. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/History:Paper_-_Description_of_a_Human_Embryo_of_Twenty-two_paired_Somites
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G
- Fig.4. Cross sections of the Spinal Cord at three levels: A. (sec- level of the 12th somite; B. (section 218) at the somite. C. (section 245) at the level of the 16th
A.L.= Alar lamina; B.L. = Basal lamina;F.P. = Floorplate; R.P. Roofplate.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Reference
<pubmed>17104111</pubmed>| PMC1249865
This is a slightly edited version of the original 1926 paper published in Journal of Anatomy. The full paper is still available as a PDF document through PubMed Central.
The information below was not part of the above original historic article.
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
| Week: | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Carnegie stage: | 1 2 3 4 | 5 6 | 7 8 9 | 10 11 12 13 | 14 15 | 16 17 | 18 19 | 20 21 22 23 |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 12) Embryology Paper - Description of a Human Embryo of Twenty-two paired Somites. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/History:Paper_-_Description_of_a_Human_Embryo_of_Twenty-two_paired_Somites
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G