Introduction

All-trans retinoic acid (atRA)
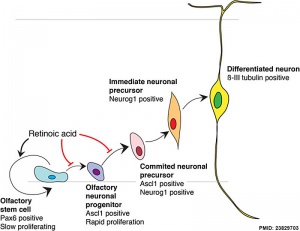
Model for retinoid acid in olfactory development
[1]
All-trans retinoic acid (atRA) is the transcriptionally active product of vitamin A and is known to play many roles in regulating embryo development, controlling patterning, cell fate decisions and differentiation. In the adult retinoid acid has additional signalling roles including spermatozoa maturation. Transient RA signaling gradients are formed by the RA local degradation by enzymes of the Cyp26a gene family.
The compound has been used extensively postnatally in therapeutic treatments, for example in skin disease. As this compound also acts as a developmental signal, it has known teratogenic effects[2][3] following maternal to conceptus transfer.
- Retinoic Acid Links: retinoic acid | limb | Category:Retinoic acid | Molecular Development
Category:Retinoic acid | Abnormal Development
Some Recent Findings
- Review - RA Signaling in limb Development and Regeneration in Different Species[4] "This chapter brings together data on the role of retinoic acid (RA) in the embryonic development of fins in zebrafish , limbs in amphibians , chicks , and mice, and regeneration of the amphibian limb. The intention is to determine whether there is a common set of principles by which we can understand the mode of action of RA in both embryos and adults. What emerges from this synthesis is that there are indeed commonalities in the involvement of RA in processes that ventralize, posteriorize, and proximalize the developing and regenerating limb . Different axes of the limb have historically been studied independently; as for example, the embryonic development of the anteroposterior (AP) axis of the chick limb bud versus the regeneration of the limb bud proximodistal (PD) axis . But when we take a broader view, a unifying principle emerges that explains why RA administration to embryos and regenerating limbs results in the development of multiple limbs in both cases. As might be expected, different molecular pathways govern the development of different systems and model organisms, but despite these differences, the pathways involve similar RA signaling genes, such as tbx5, meis, shh, fgfs and hox genes. Studies of developing and regenerating systems have highlighted that RA acts by being synthesized in one embryonic location while acting in another one, exactly as embryonic morphogens do, although there is no evidence for the presence of an RA gradient within the limb. What also emerges is that there is a paucity of information on the involvement of RA in development of the dorsoventral (DV) axis."
- Review - Retinoic Acid and Germ Cell Development in the Ovary and Testis[5] "Retinoic acid (RA), a derivative of vitamin A, is critical for the production of oocytes and sperm in mammals. These gametes derive from primordial germ cells, which colonize the nascent gonad, and later undertake sexual differentiation to produce oocytes or sperm. During fetal development, germ cells in the ovary initiate meiosis in response to RA, whereas those in the testis do not yet initiate meiosis, as they are insulated from RA, and undergo cell cycle arrest. After birth, male germ cells resume proliferation and undergo a transition to spermatogonia, which are destined to develop into haploid spermatozoa via spermatogenesis. Recent findings indicate that RA levels change periodically in adult testes to direct not only meiotic initiation, but also other key developmental transitions to ensure that spermatogenesis is precisely organized for the prodigious output of sperm."
- Retinoic acid signaling reduction recapitulates the effects of alcohol on embryo size[6] "Intrauterine growth restriction (IUGR) is commonly observed in human pregnancies and can result in severe clinical outcomes. IUGR is observed in Fetal Alcohol Syndrome (FAS) fetuses as a result of alcohol (ethanol) exposure during pregnancy. To further understand FAS, the severe form of Fetal Alcohol Spectrum Disorder, we performed an extensive quantitative analysis of the effects of ethanol on embryo size utilizing our Xenopus model. Ethanol-treated embryos exhibited size reduction along the anterior-posterior axis. This effect was evident primarily from the hindbrain caudally, while rostral regions appeared refractive to ethanol-induced size changes, also known as asymmetric IUGR. Interestingly, some embryo batches in addition to shortening from the hindbrain caudally also exhibited an alcohol-dependent reduction of the anterior head domain, known as symmetric IUGR. To study the connection between ethanol exposure and reduced retinoic acid levels we treated embryos with the retinaldehyde dehydrogenase inhibitors, DEAB and citral. Inhibition of retinoic acid biosynthesis recapitulated the growth defects induced by ethanol affecting mainly axial elongation from the hindbrain caudally. To study the competition between ethanol clearance and retinoic acid biosynthesis we demonstrated that, co-exposure to alcohol reduces the teratogenic effects of treatment with retinol (vitamin A), the retinoic acid precursor. These results further support the role of retinoic acid in the regulation of axial elongation."
- Retinoic acid temporally orchestrates colonization of the gut by vagal neural crest cells[7] "The enteric nervous system arises from neural crest cells that migrate as chains into and along the primitive gut, subsequently differentiating into enteric neurons and glia. Little is known about the mechanisms governing neural crest migration en route to and along the gut in vivo. Here, we report that Retinoic Acid (RA) temporally controls zebrafish enteric neural crest cell chain migration. In vivo imaging reveals that RA loss severely compromises the integrity and migration of the chain of neural crest cells during the window of time window when they are moving along the foregut. After loss of RA, enteric progenitors accumulate in the foregut and differentiate into enteric neurons, but subsequently undergo apoptosis resulting in a striking neuronal deficit. Moreover, ectopic expression of the transcription factor meis3 and/or the receptor ret, partially rescues enteric neuron colonization after RA attenuation. Collectively, our findings suggest that retinoic acid plays a critical temporal role in promoting enteric neural crest chain migration and neuronal survival upstream of Meis3 and RET in vivo." neural crest | zebrafish
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Embryo Retinoic acid | Images
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Cyp26 Enzymes Facilitate Second Heart Field Progenitor Addition and Maintenance of Ventricular Integrity[8] "Although retinoic acid (RA) teratogenicity has been investigated for decades, the mechanisms underlying RA-induced outflow tract (OFT) malformations are not understood. Here, we show zebrafish embryos deficient for Cyp26a1 and Cyp26c1 enzymes, which promote RA degradation, have OFT defects resulting from two mechanisms: first, a failure of second heart field (SHF) progenitors to join the OFT, instead contributing to the pharyngeal arch arteries (PAAs), and second, a loss of first heart field (FHF) ventricular cardiomyocytes due to disrupted cell polarity and extrusion from the heart tube." Zebrafish Development | Cardiovascular Abnormalities
- Retinoic acid signaling and neuronal differentiation[9] "The identification of neurological symptoms caused by vitamin A deficiency pointed to a critical, early developmental role of vitamin A and its metabolite, retinoic acid (RA). The ability of RA to induce post-mitotic, neural phenotypes in various stem cells, in vitro, served as early evidence that RA is involved in the switch between proliferation and differentiation. In vivo studies have expanded this "opposing signal" model, and the number of primary neurons an embryo develops is now known to depend critically on the levels and spatial distribution of RA. The proneural and neurogenic transcription factors that control the exit of neural progenitors from the cell cycle and allow primary neurons to develop are partly elucidated, but the downstream effectors of RA receptor (RAR) signaling (many of which are putative cell cycle regulators) remain largely unidentified. The molecular mechanisms underlying RA-induced primary neurogenesis in anamniote embryos are starting to be revealed; however, these data have been not been extended to amniote embryos. There is growing evidence that bona fide RARs are found in some mollusks and other invertebrates, but little is known about their necessity or functions in neurogenesis. One normal function of RA is to regulate the cell cycle to halt proliferation, and loss of RA signaling is associated with dedifferentiation and the development of cancer. Identifying the genes and pathways that mediate cell cycle exit downstream of RA will be critical for our understanding of how to target tumor differentiation. Overall, elucidating the molecular details of RAR-regulated neurogenesis will be decisive for developing and understanding neural proliferation-differentiation switches throughout development."
- Visualization of an endogenous retinoic acid gradient across embryonic development[10] "In vertebrate development, the body plan is determined by primordial morphogen gradients that suffuse the embryo. Retinoic acid (RA) is an important morphogen involved in patterning the anterior-posterior axis of structures, including the hindbrain and paraxial mesoderm. RA diffuses over long distances, and its activity is spatially restricted by synthesizing and degrading enzymes. ...Live imaging of endogenous concentration gradients across embryonic development will allow the precise assignment of molecular mechanisms to developmental dynamics and will accelerate the application of approaches based on morphogen gradients to tissue engineering and regenerative medicine." zebrafish
|
Organ Expression
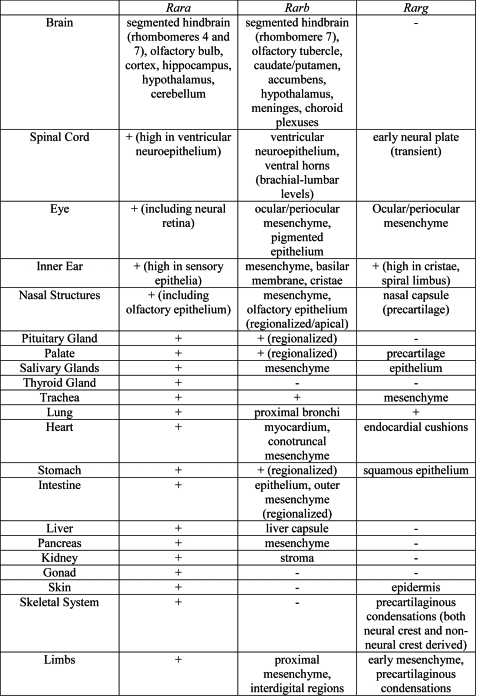
Summary of Rar gene expression patterns in mouse developing organ systems.[11]
Endoderm
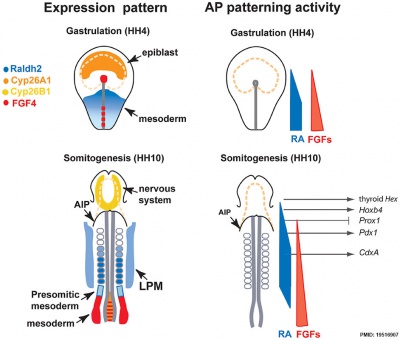
Chicken antero-posterior endoderm patterning[12]
- Links: endoderm | chicken
Fetal Gonad

Immunohistochemical localisation of retinoid receptor expression in the human fetal gonad[13]
Adult Gonad
Retinoic acid "waves" are required for the cycle of the seminiferous epithelium, influencing both Sertoli cells and germ cells, the pulse serves as a trigger to commit undifferentiated progenitor cells to the timed pathway into meiosis and spermatid differentiation.[14]
- Links: spermatozoa | sertoli cell
Neural
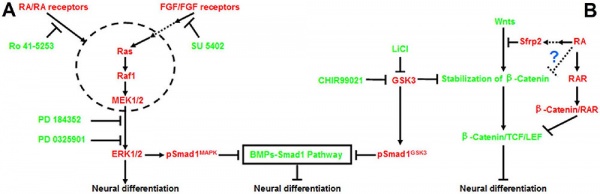
Model retinoic acid extracellular signal-regulated kinase and Wnt pathway interactions[15]

RA Is Not Required for Radial Expansion of the Embryonic Cortex
References
- ↑ Paschaki M, Cammas L, Muta Y, Matsuoka Y, Mak SS, Rataj-Baniowska M, Fraulob V, Dollé P & Ladher RK. (2013). Retinoic acid regulates olfactory progenitor cell fate and differentiation. Neural Dev , 8, 13. PMID: 23829703 DOI.
- ↑ Collins MD & Mao GE. (1999). Teratology of retinoids. Annu. Rev. Pharmacol. Toxicol. , 39, 399-430. PMID: 10331090 DOI.
- ↑ Tzimas G & Nau H. (2001). The role of metabolism and toxicokinetics in retinoid teratogenesis. Curr. Pharm. Des. , 7, 803-31. PMID: 11375780
- ↑ Maden M. (2020). RA Signaling in Limb Development and Regeneration in Different Species. Subcell Biochem , 95, 87-117. PMID: 32297297 DOI.
- ↑ Endo T, Mikedis MM, Nicholls PK, Page DC & de Rooij DG. (2019). Retinoic Acid and Germ Cell Development in the Ovary and Testis. Biomolecules , 9, . PMID: 31771306 DOI.
- ↑ Shukrun N, Shabtai Y, Pillemer G & Fainsod A. (2019). Retinoic acid signaling reduction recapitulates the effects of alcohol on embryo size. Genesis , , e23284. PMID: 30672660 DOI.
- ↑ Uribe RA, Hong SS & Bronner ME. (2018). Retinoic acid temporally orchestrates colonization of the gut by vagal neural crest cells. Dev. Biol. , 433, 17-32. PMID: 29108781 DOI.
- ↑ Rydeen AB & Waxman JS. (2016). Cyp26 Enzymes Facilitate Second Heart Field Progenitor Addition and Maintenance of Ventricular Integrity. PLoS Biol. , 14, e2000504. PMID: 27893754 DOI.
- ↑ Janesick A, Wu SC & Blumberg B. (2015). Retinoic acid signaling and neuronal differentiation. Cell. Mol. Life Sci. , 72, 1559-76. PMID: 25558812 DOI.
- ↑ Shimozono S, Iimura T, Kitaguchi T, Higashijima S & Miyawaki A. (2013). Visualization of an endogenous retinoic acid gradient across embryonic development. Nature , 496, 363-6. PMID: 23563268 DOI.
- ↑ Dollé P. (2009). Developmental expression of retinoic acid receptors (RARs). Nucl Recept Signal , 7, e006. PMID: 19471585 DOI.
- ↑ Bayha E, Jørgensen MC, Serup P & Grapin-Botton A. (2009). Retinoic acid signaling organizes endodermal organ specification along the entire antero-posterior axis. PLoS ONE , 4, e5845. PMID: 19516907 DOI.
- ↑ Childs AJ, Cowan G, Kinnell HL, Anderson RA & Saunders PT. (2011). Retinoic Acid signalling and the control of meiotic entry in the human fetal gonad. PLoS ONE , 6, e20249. PMID: 21674038 DOI.
- ↑ Griswold MD. (2016). Spermatogenesis: The Commitment to Meiosis. Physiol. Rev. , 96, 1-17. PMID: 26537427 DOI.
- ↑ Lu J, Tan L, Li P, Gao H, Fang B, Ye S, Geng Z, Zheng P & Song H. (2009). All-trans retinoic acid promotes neural lineage entry by pluripotent embryonic stem cells via multiple pathways. BMC Cell Biol. , 10, 57. PMID: 19642999 DOI.
Reviews
Ghyselinck NB & Duester G. (2019). Retinoic acid signaling pathways. Development , 146, . PMID: 31273085 DOI.
Feneck E & Logan M. (2020). The Role of Retinoic Acid in Establishing the Early Limb Bud. Biomolecules , 10, . PMID: 32079177 DOI.
Draut H, Liebenstein T & Begemann G. (2019). New Insights into the Control of Cell Fate Choices and Differentiation by Retinoic Acid in Cranial, Axial and Caudal Structures. Biomolecules , 9, . PMID: 31835881 DOI.
Dubey A, Rose RE, Jones DR & Saint-Jeannet JP. (2018). Generating retinoic acid gradients by local degradation during craniofacial development: One cell's cue is another cell's poison. Genesis , 56, . PMID: 29330906 DOI.
Articles
Bozzo P, Chua-Gocheco A & Einarson A. (2011). Safety of skin care products during pregnancy. Can Fam Physician , 57, 665-7. PMID: 21673209
Search Pubmed
Search Bookshelf Retinoic acid
Search Pubmed Now: Retinoic acid
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 3) Embryology Developmental Signals - Retinoic acid. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Signals_-_Retinoic_acid
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G







