Mesoderm
| Embryology - 26 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
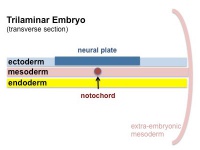
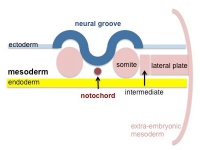
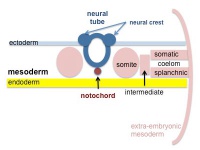
The mesoderm forms the middle layer of the early trilaminar embryo germ layers (ectoderm, mesoderm and endoderm) formed by gastrulation. The segmentation of the initial mesoderm into somites, and their regular addition, is often used to stage embryonic development (23 somite embryo).
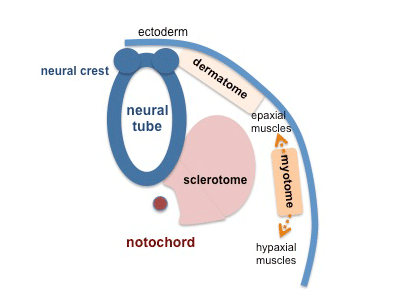
This middle germ layer forms connective tissues and muscle throughout the body, with the exception of in the head region where some of these structures have a neural crest (ectoderm) origin.
- connective tissues - cartilage, bone, blood, blood vessel endothelium, dermis, etc.
- muscle - cardiac, skeletal, smooth.
Students often mix-up the terms mesoderm (middle layer) with mesenchyme (embryonic connective tissue). It is true that mesoderm initially does have a mesenchymal cellular organisation, but can also form a range of epithelial structures (surrounding somites, mesothelium lining of body cavities).
| Mesoderm Links: endoderm | mesoderm | ectoderm | Lecture - Mesoderm | Lecture - Musculoskeletal | 2016 Lecture | notochord | somitogenesis | somite | splanchnic mesoderm | skeletal muscle | smooth muscle | heart | Notochord Movie | musculoskeletal | cartilage | bone | sonic hedgehog | Category:Mesoderm | ||||
|
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Mesoderm Development | Extra-embryonic Mesoderm Development | Axial Mesoderm | Paraxial Mesoderm | Intermediate Mesoderm | Splanchnic Mesoderm | Somatic Mesoderm |Mesoderm Images |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Overview
|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Overview: Ectoderm | Mesoderm | Endoderm Layers: ectoderm | mesoderm | endoderm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hierarchical View | ||
|---|---|---|
|
| Mesoderm Structures | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Mesoderm Movies
|
|
|
|
| |||||||||||||||
|
|
|
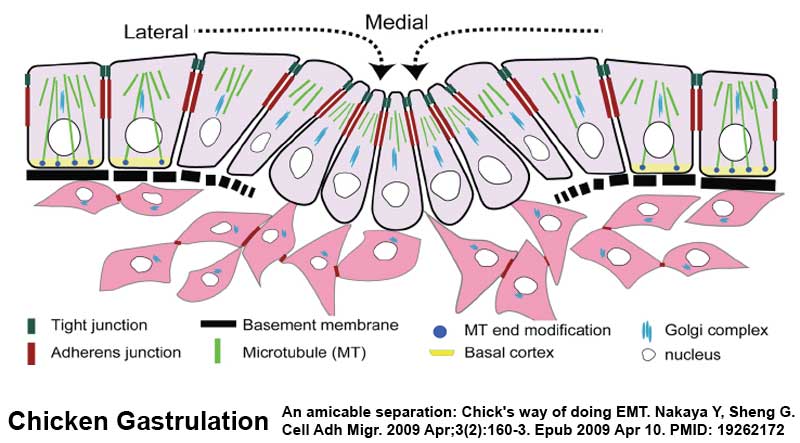
Mesoderm Formation during Gastrulation
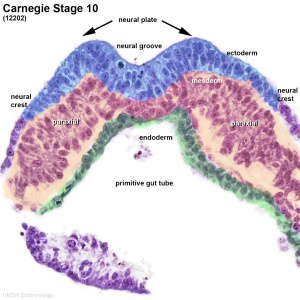
- Links: gastrulation
Patterning
Somite patterning
Embryonic Mesoderm
|
| Germ Layers: ectoderm | mesoderm | endoderm |
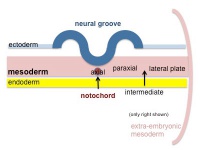
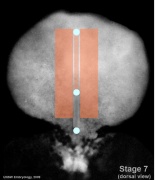
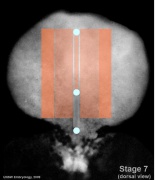
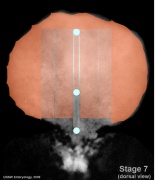
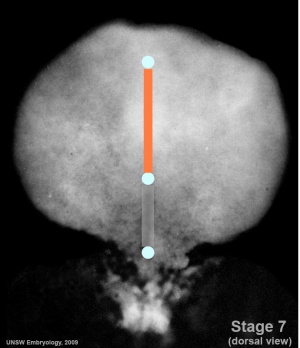
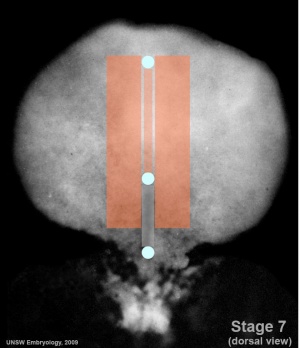
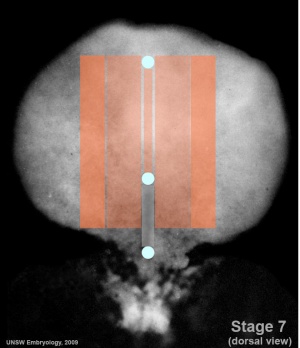
Mesoderm Regions (week 3)
- Embryo Stage 7 (dorsal)
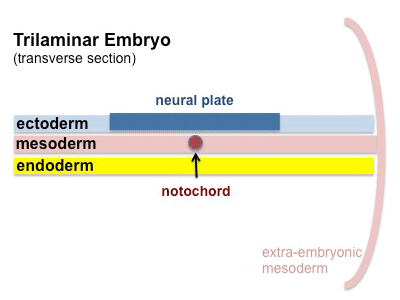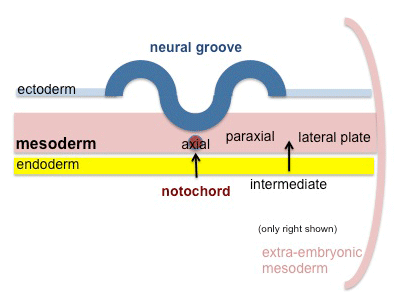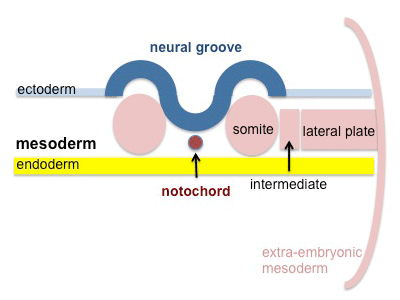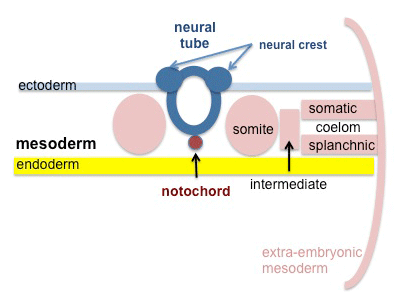
Axial Mesoderm
Paraxial Mesoderm
Intermediate Mesoderm
Somatic Mesoderm
Splanchnic Mesoderm
Extra-embryonic Mesoderm
The origin of extra-embryonic mesoderm (EEM) has been extensively discussed in the literature. This mesoderm lies outside the embryo, associated with fetal membrane and placenta development. This mesoderm formed at gastrulation along with the embryonic mesoderm from the proximal side of the primitive streak. A recent study in mouse, has shown that the primitive streak absence and excessive epiblast Nodal activity in pre-gastrulation stage, but not in the primitive streak cells during gastrulation, disrupts extraembryonic mesoderm development. [9]
- Links: placenta | placental membranes
Molecular Factors
References
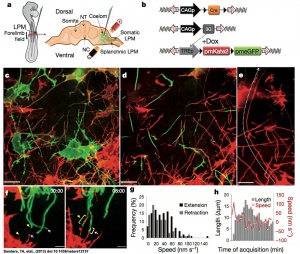
- ↑ Sanders TA, Llagostera E & Barna M. (2013). Specialized filopodia direct long-range transport of SHH during vertebrate tissue patterning. Nature , 497, 628-32. PMID: 23624372 DOI.
- ↑ Row RH, Pegg A, Kinney B, Farr GH, Maves L, Lowell S, Wilson V & Martin BL. (2018). BMP and FGF signaling interact to pattern mesoderm by controlling basic helix-loop-helix transcription factor activity. Elife , 7, . PMID: 29877796 DOI.
- ↑ Beisaw A, Tsaytler P, Koch F, Schmitz SU, Melissari MT, Senft AD, Wittler L, Pennimpede T, Macura K, Herrmann BG & Grote P. (2018). BRACHYURY directs histone acetylation to target loci during mesoderm development. EMBO Rep. , 19, 118-134. PMID: 29141987 DOI.
- ↑ Bazzi H, Soroka E, Alcorn HL & Anderson KV. (2017). STRIP1, a core component of STRIPAK complexes, is essential for normal mesoderm migration in the mouse embryo. Proc. Natl. Acad. Sci. U.S.A. , 114, E10928-E10936. PMID: 29203676 DOI.
- ↑ Koh PW, Sinha R, Barkal AA, Morganti RM, Chen A, Weissman IL, Ang LT, Kundaje A & Loh KM. (2016). An atlas of transcriptional, chromatin accessibility, and surface marker changes in human mesoderm development. Sci Data , 3, 160109. PMID: 27996962 DOI.
- ↑ Fleming BM, Yelin R, James RG & Schultheiss TM. (2013). A role for Vg1/Nodal signaling in specification of the intermediate mesoderm. Development , 140, 1819-29. PMID: 23533180 DOI.
- ↑ Aulehla A & Pourquié O. (2010). Signaling gradients during paraxial mesoderm development. Cold Spring Harb Perspect Biol , 2, a000869. PMID: 20182616 DOI.
- ↑ Shapiro IM & Risbud MV. (2010). Transcriptional profiling of the nucleus pulposus: say yes to notochord. Arthritis Res. Ther. , 12, 117. PMID: 20497604 DOI.
- ↑ Jin JZ, Zhu Y, Warner D & Ding J. (2016). Analysis of extraembryonic mesodermal structure formation in the absence of morphological primitive streak. Dev. Growth Differ. , 58, 522-9. PMID: 27273137 DOI.
Reviews
Risbud MV, Schaer TP & Shapiro IM. (2010). Toward an understanding of the role of notochordal cells in the adult intervertebral disc: from discord to accord. Dev. Dyn. , 239, 2141-8. PMID: 20568241 DOI.
Burke AC. (2007). Development and evolution of the vertebrate mesoderm. Dev. Dyn. , 236, 2369-70. PMID: 17705304 DOI.
Articles
Martin BL & Kimelman D. (2010). Brachyury establishes the embryonic mesodermal progenitor niche. Genes Dev. , 24, 2778-83. PMID: 21159819 DOI.
Korecki CL, Taboas JM, Tuan RS & Iatridis JC. (2010). Notochordal cell conditioned medium stimulates mesenchymal stem cell differentiation toward a young nucleus pulposus phenotype. Stem Cell Res Ther , 1, 18. PMID: 20565707 DOI.
Trainor PA, Tan SS & Tam PP. (1994). Cranial paraxial mesoderm: regionalisation of cell fate and impact on craniofacial development in mouse embryos. Development , 120, 2397-408. PMID: 7956820
Historic
Florian J. (1933). The Early Development of Man, with Special Reference to the Development of the Mesoderm and Cloacal Membrane. J. Anat. , 67, 263-76. PMID: 17104422
Search PubMed
Search NLM Online Textbooks: "Mesoderm" : Developmental Biology | The Cell- A molecular Approach | Molecular Biology of the Cell | Endocrinology
Search Pubmed: Mesoderm | Notochord
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Take the Quiz
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, April 26) Embryology Mesoderm. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Mesoderm
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G