Zygote
| Embryology - 8 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The zygote is the first diploid cell that forms following fertilization by fusion of the haploid oocyte (egg) and spermatozoa (sperm) resulting in the combination of their separate genomes. The zygote will therefore form the conceptus, the embryonic (embryo, fetus) and extra-embryonic (fetal membranes, fetal component of the placenta) cellular products of fertilisation.
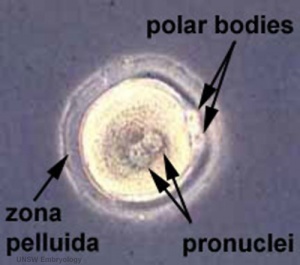
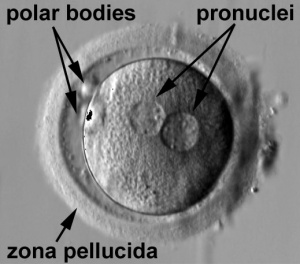
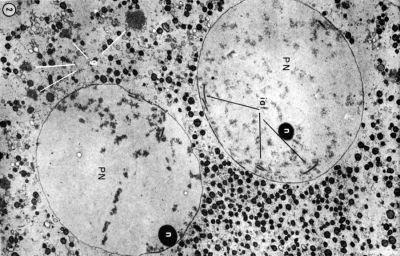
The first image shows the cell with the 2 pronuclei still present before fusion enclosed within the zona pellucida. These two pronuclei contain the parental genomes and are reprogrammed separately, and also have different epigenetic changes at this early zygote stage.[1][2]
In humans, zygote stage of development occurs during the first day in week one following fertilization (GA week 3) and is described as Carnegie stage 1. This stage is followed by mitosis to form 2 blastomeres and then a solid cell mass called the morula.
| Zygote Links: zygote Carnegie stage 1 | fertilization | Week 1 | morula | blastocyst | Assisted Reproductive Technology | SCNT | epigenetics | Category:Zygote |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search PubMed: Zygote Development | Zygotic Genome Activation |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Movies
|
|
|
| ||||||||||||||||
|
|
|
|
|
Human Zygote Size
Parental Pronuclei
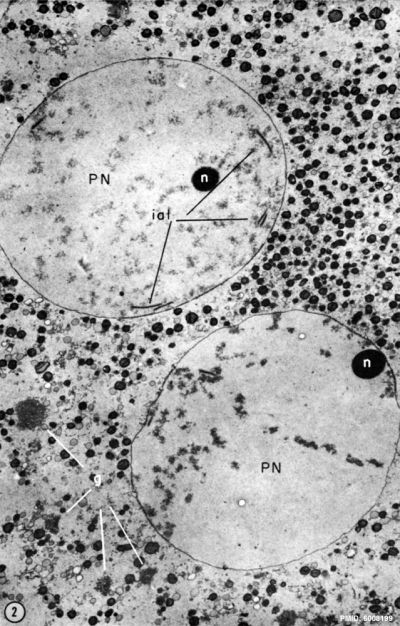
The pronuclei (singular: pronucleus) are the initial male and female nuclei, containing their respective genetic material, that occur in the early zygote. These two haploid nuclei fuse to form the diploid nucleus of the late zygote.
- Maternal pronuclei - (oocyte) genome is inherited from the meiosis II oocyte where the chromosomes are condensed in a mitotic‐like state.
- Paternal pronuclei - (spermatozoa) genome is contributed by a compacted sperm chromatin that is remodeled upon fertilization. Chiefly by protamine removal and the original nucleosomal chromatin is established. (More? Male Pronucleus Reprogramming)
|
|
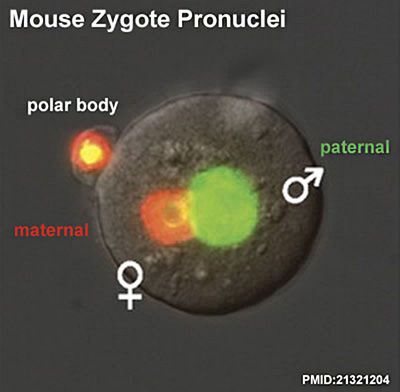
Parental Genomes
Animation based upon individual images of mouse maternal and paternal genomes.
| Mouse zygote pronuclei | |
|---|---|
|

|
| Fluorescence[7] | DIC Optics showing pronuclear bodies[8] |
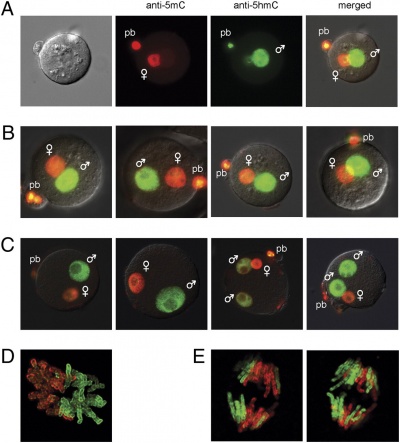
Male Pronucleus Reprogramming
| Mouse zygote paternal genome reprogramming[7]
Mouse zygotes male pronucleus contains 5-hydroxymethylcytosine (5hmC) thought to be formed by enzymatic oxidation of 5-methylcytosine (5mC).
|
|
Mouse zygote mitosis[7]
Subcortical Maternal Complex
The Subcortical Maternal Complex (SCMC) is peripheral cellular region identified in mouse oogenesis, but also present in human, required for determining the first cleavage-stages of division. The SCMA functions in: zygotic genome activation, F-actin dynamics, genome stability, organelle organization, and DNA methylation maintenance at imprinted loci.[9]
Identified Proteins in mouse - Mater, Filia, Floped, Tle6, Zbed3, Nlrp2, possibly Padi6, and other proteins.
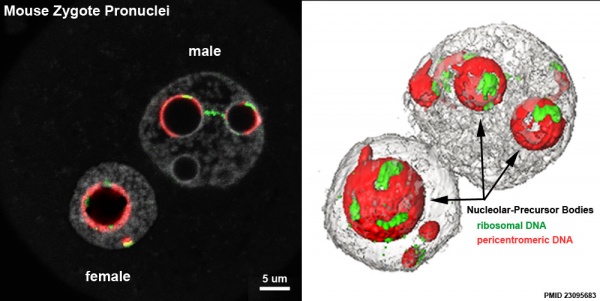
Nucleolar-Precursor Bodies
Most ribosomal DNA located around the Nucleolar-Precursor Bodies (NPBs), with some associated with pericentromeric filaments (extending from the NPBs towards the nuclear periphery) as well as rDNA signals joining two NPBs.[10]
Formation of Zygote
- male and female pronuclei, 2 nuclei approach each other and nuclear membranes break down
- DNA replicates, first mitotic division
- sperm contributes centriole which organizes mitotic spindle
Movie - Pronuclear Fusion | Movie - Parental Genomes
Conceptus - term refers to all material derived from this fertilized zygote and includes both the embryo and the non-embryonic tissues (placenta, fetal membranes).
Zygotic Genome Activation
Zygotic Genome Activation (ZGA) is a slight misnomer, as the zygote has initially low expression which increases to activation occurring at the 4 to 8 cell transition in the human, the early morula stage. In the mouse ZGA occurs earlier during the 2 cell blastomere stage. A recent study in mouse and human has identified Pleomorphic Adenoma Gene 1 (OMIM PLAG1) is a transcription factor that regulates many genes (1,089 genes) involved ribosome biogenesis and protein synthesis.[3] In the adult, PLAG1 is involved in growth and also cancer development. Dux transcription factor is also associated with ZGA and is positively regulated in the mouse by developmental pluripotency-associated 2 (Dppa2) and Dppa4.[11]
- Links: GHR PLAG1 | OMIM PLAG1
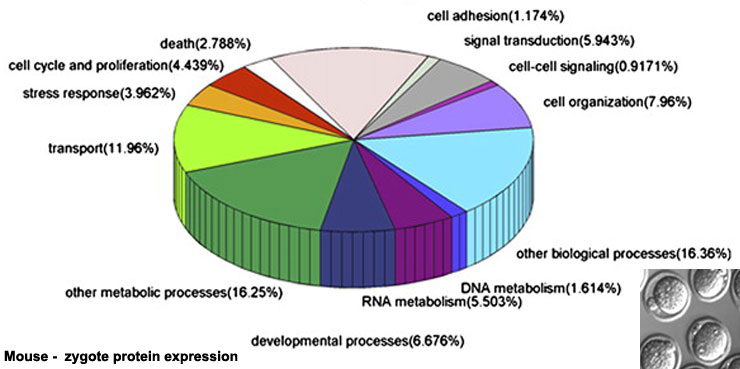
Zygote Protein Expression
Maternally inherited Yes-associated protein (Yap), a co-activator of TEAD family transcription factors, plays a key role in activating embryonic transcription following fertilization in the mouse. Lysophosphatidic acid (LPA) in the mouse tubal fluid binds to its G-protein coupled receptor at the plasma membrane, and induces the activation of YAP by inhibiting LATS1/2.[12] Hippo
The table above shows the pattern of protein expression (as percentages of total) in the mouse zygote according to 14 molecular function categories.[13]
- Links: Germinal vesicle oocyte protein expression | MII oocyte protein expression | Zygote Protein Expression | mouse | oocyte | zygote
References
- ↑ Ladstätter S & Tachibana-Konwalski K. (2016). A Surveillance Mechanism Ensures Repair of DNA Lesions during Zygotic Reprogramming. Cell , 167, 1774-1787.e13. PMID: 27916276 DOI.
- ↑ Mackay DJG & Temple IK. (2017). Human imprinting disorders: Principles, practice, problems and progress. Eur J Med Genet , 60, 618-626. PMID: 28818477 DOI.
- ↑ 3.0 3.1 Madissoon E, Damdimopoulos A, Katayama S, Krjutškov K, Einarsdottir E, Mamia K, De Groef B, Hovatta O, Kere J & Damdimopoulou P. (2019). Pleomorphic Adenoma Gene 1 Is Needed For Timely Zygotic Genome Activation and Early Embryo Development. Sci Rep , 9, 8411. PMID: 31182756 DOI.
- ↑ Niakan KK & Eggan K. (2013). Analysis of human embryos from zygote to blastocyst reveals distinct gene expression patterns relative to the mouse. Dev. Biol. , 375, 54-64. PMID: 23261930 DOI.
- ↑ Silvestre F, Gallo A, Cuomo A, Covino T & Tosti E. (2011). Role of cyclic AMP in the maturation of Ciona intestinalis oocytes. Zygote , 19, 365-71. PMID: 20810008 DOI.
- ↑ Li CB, Wang ZD, Zheng Z, Hu LL, Zhong SQ & Lei L. (2011). Number of blastomeres and distribution of microvilli in cloned mouse embryos during compaction. Zygote , 19, 271-6. PMID: 20735894 DOI.
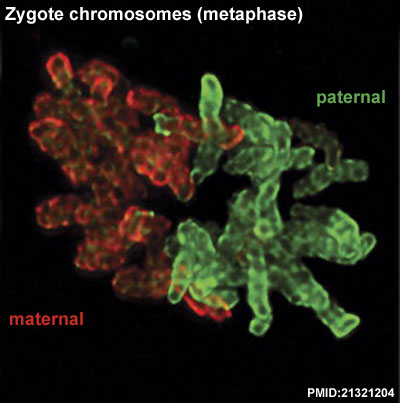
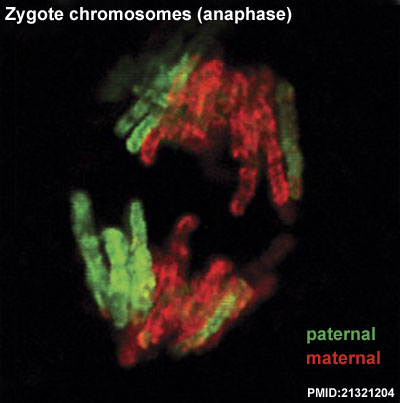
- ↑ 7.0 7.1 7.2 Iqbal K, Jin SG, Pfeifer GP & Szabó PE. (2011). Reprogramming of the paternal genome upon fertilization involves genome-wide oxidation of 5-methylcytosine. Proc. Natl. Acad. Sci. U.S.A. , 108, 3642-7. PMID: 21321204 DOI.
- ↑ Singer AB & Gall JG. (2011). An inducible nuclear body in the Drosophila germinal vesicle. Nucleus , 2, 403-9. PMID: 21941118 DOI.
- ↑ Lu X, Gao Z, Qin D & Li L. (2017). A Maternal Functional Module in the Mammalian Oocyte-To-Embryo Transition. Trends Mol Med , 23, 1014-1023. PMID: 28993030 DOI.
- ↑ Aguirre-Lavin T, Adenot P, Bonnet-Garnier A, Lehmann G, Fleurot R, Boulesteix C, Debey P & Beaujean N. (2012). 3D-FISH analysis of embryonic nuclei in mouse highlights several abrupt changes of nuclear organization during preimplantation development. BMC Dev. Biol. , 12, 30. PMID: 23095683 DOI.
- ↑ Eckersley-Maslin M, Alda-Catalinas C, Blotenburg M, Kreibich E, Krueger C & Reik W. (2019). Dppa2 and Dppa4 directly regulate the Dux-driven zygotic transcriptional program. Genes Dev. , 33, 194-208. PMID: 30692203 DOI.
- ↑ Yu C, Ji SY, Dang YJ, Sha QQ, Yuan YF, Zhou JJ, Yan LY, Qiao J, Tang F & Fan HY. (2016). Oocyte-expressed yes-associated protein is a key activator of the early zygotic genome in mouse. Cell Res. , 26, 275-87. PMID: 26902285 DOI.
- ↑ Wang S, Kou Z, Jing Z, Zhang Y, Guo X, Dong M, Wilmut I & Gao S. (2010). Proteome of mouse oocytes at different developmental stages. Proc. Natl. Acad. Sci. U.S.A. , 107, 17639-44. PMID: 20876089 DOI.
Journals
- Zygote An international journal dedicated to the rapid publication of original research in early embryology, Zygote covers interdisciplinary studies in animals and humans, from gametogenesis through fertilization to gastrulation.
Reviews
Lu X, Gao Z, Qin D & Li L. (2017). A Maternal Functional Module in the Mammalian Oocyte-To-Embryo Transition. Trends Mol Med , 23, 1014-1023. PMID: 28993030 DOI.
Jukam D, Shariati SAM & Skotheim JM. (2017). Zygotic Genome Activation in Vertebrates. Dev. Cell , 42, 316-332. PMID: 28829942 DOI.
Destouni A & Vermeesch JR. (2017). How can zygotes segregate entire parental genomes into distinct blastomeres? The zygote metaphase revisited. Bioessays , 39, . PMID: 28247957 DOI.
Chaigne A, Terret ME & Verlhac MH. (2017). Asymmetries and Symmetries in the Mouse Oocyte and Zygote. Results Probl Cell Differ , 61, 285-299. PMID: 28409310 DOI.
Yartseva V & Giraldez AJ. (2015). The Maternal-to-Zygotic Transition During Vertebrate Development: A Model for Reprogramming. Curr. Top. Dev. Biol. , 113, 191-232. PMID: 26358874 DOI.
Articles
Eckersley-Maslin M, Alda-Catalinas C, Blotenburg M, Kreibich E, Krueger C & Reik W. (2019). Dppa2 and Dppa4 directly regulate the Dux-driven zygotic transcriptional program. Genes Dev. , 33, 194-208. PMID: 30692203 DOI.
Yuan P, Zheng L, Liang H, Li Y, Zhao H, Li R, Lai L, Zhang Q & Wang W. (2018). A novel mutation in the TUBB8 gene is associated with complete cleavage failure in fertilized eggs. J. Assist. Reprod. Genet. , , . PMID: 29704226 DOI.
Conti M & Franciosi F. (2018). Acquisition of oocyte competence to develop as an embryo: integrated nuclear and cytoplasmic events. Hum. Reprod. Update , 24, 245-266. PMID: 29432538 DOI.
Ho JR, Arrach N, Rhodes-Long K, Salem W, McGinnis LK, Chung K, Bendikson KA, Paulson RJ & Ahmady A. (2018). Blastulation timing is associated with differential mitochondrial content in euploid embryos. J. Assist. Reprod. Genet. , , . PMID: 29353449 DOI.
Gassler J, Brandão HB, Imakaev M, Flyamer IM, Ladstätter S, Bickmore WA, Peters JM, Mirny LA & Tachibana K. (2017). A mechanism of cohesin-dependent loop extrusion organizes zygotic genome architecture. EMBO J. , 36, 3600-3618. PMID: 29217590 DOI.
Search Pubmed
June 2010 "zygote" All (6213) Review (651) Free Full Text (1543)
Search Pubmed: zygote
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 8) Embryology Zygote. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Zygote
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G