Paper - On the development of the aortae cardinal and umbilical veins and the other blood vessels of vertebrate embryos from capillaries (1909)
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Evans HM. On the development of the aortae, cardinal and umbilical veins, and the other blood vessels of vertebrate embryos from capillaries. (1909) Anat. Rec. 3: 498-518.
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
On the Development of the Aortae, Cardinal and Umbilical Veins, and the Other Blood Vessels of Vertebrate Embryos from Capillaries
By
From the Anatomical Laboratory of the Johns Hopkins University.
With Twenty-one Figures.
- Read at the Twenty fourth Session of the Association of American Anatomists, December, 1908.
Introduction
In explanation of the somewhat comprehensive title of this report, I would say that I have omitted entirely, for the present at least, the development of the heart, first arch and cephalic aortae. But, on the other hand, the injections which I shall describe speak with great clearness on the primitive form and development of all that portion of the aorta below the omphalo-mesenteric arteries, of the entire upper portions of the anterior cardinal veins, and quite fully, of the posterior cardinal and umbilical veins. In addition to these primary trunks, I have explored other branches of the vascular tree in embryos of different ages and in various portions of the body and find everywhere the same story.
The patient and thoroughgoing analysis of serial sections through vertebrate embryos has, in the last twenty years, given us a fairly accurate idea of the position, size, and relations of all the chief vascular trunks present in successive stages. But such knowledge, even though complete, can tell us little or nothing about the method of development of the vascular system. When a large trunk is described as having extended to a certain level in the body wall or on a viscus we have still no idea of how it reached its position. Many processes could be concerned. if the vessel in question was derived from neighboring vessels, and in some instances even this has been disputed, have we then to conceive of it as having grown out as such to its future territory? Certainly such a conception dominated the descriptive anatomy of a century ago and even to-day has not altogether lost its influence. Arteries are still described as dividing dichotomously and otherwise in accordance with the unconscious simile afforded by plant growth. To such an idea many of the vascular anomalies‘ proved an utter enigma. Double vessels in place of the usual single one were disposed of by considering one vessel as the normal one and its brother as the interloper or “aberrant.” It is easy to understand the curiosity with which anastomoses between strong and usually separate arterial channels were looked upon. Why indeed should vessels which had grown out to their destination, send communicating branches to their neighbors? Thus the surprise with which Hunter discovered the phenomenon of a collateral circulation. Such phenomena in the adult body are themselves splendid evidences for quite another conception of the development of the vascular system, a conception which was first partially expressed by the anatomist Aeby in his “Der Bau des menschlichen Körpers,” in 1868. Aeby contributed a mere hypothesis, but it has only recently been recalled and can now be convincingly supported by actual studies on the early developing vessels.
Both arteries and veins, said Aeby, arise from netlike anlagen; the veins retain this the more, whence the numerous venous anastomoses; the arteries hardly at all, whence their greater rarity here. He conceived of the whole question as one of functional adaptation. Not all the members of the vascular net are retained; the victors in the struggle are the trunks as we know them in the adult.
Such an illuminating conception explained adequately all vascular anomalies. From the preceding network, the persisting vessel could course in practically any direction and with practically any connections. Though Krause, who wrote the admirable section on the variation of the vascular system in Henle’s Anatomy,[1] readily saw the advantages of Aeby’s explanation and adopted it, the conception was not recalled in any form for many years.
In the anatomy of the adult vascular system nothing approaching a netlike condition exists until we reach the capillary bed, where it forms a characteristic feature. Is it not indeed a capillary net, this primitive net postulated by Aeby?
The answer to this was given by the research of R. Thoma[2] in 1893. In his attempt to solve the question of ancestry of arteries and veins Thoma selected the chick’s yolk vessels where he soon observed stages so early that only a plexus of capillaries existed. Here no channels of the net were conspicuously larger or smaller than their neighbors, but from among them, in later stages, the chief vitelline arteries and veins are formed. Thoma’s idea of how this transformation is brought about, his conception of the determining influence of the velocity of the circulation, is of the greatest interest, though it has not been adequately tested, nor does it concern us here. On anatomical grounds alone, however, it should be possible to establish clearly or disprove the more general statement that arteries and veins exist originally in the form of a capillary mesh. If this is a fact of general- value, it will apply to the developing vascular system within the body of the embryo quite as well as to the extraembryonic circulation which Thoma had studied. The work of three recent investigators has indicated that this is indeed the case. I refer to the papers of Müller, Rabl, and Sterzi.[3] The latter observer has studied the vascularization of the spinal cord and some of his descriptions are splendid evidence of the capillary plexus ancestry of these vessels, though Sterzi himself has not realized their wider significance in this light. As I shall show later, the vessels of the cord and brain illustrate particularly well the method of origin and growth of the vascular system. The work of Müller and of Rabl has been referred to in a previous communication‘ in which I also called attention to Curt Elze’s emphatic criticism of their contention. Elze,[4] in a research done chiefly in Hochstetter’s laboratory, has seen fit to deny emphatically such an origin for the blood vessels. One were as justified, he says, in the absurdity of considering the aortic arches as arising from capillaries, and yet many of the injections in this series show exactly this origin for the arches.
Moreover, it has now been possible to show that the main vessels of the limbs, the subclavian and the sciatic arteries, are the single persisting channels of a distinct plexus of capillaries which springs directly from the lateral aortic wall opposite the earliest indication of the limb buds.[5]
Surely however, the whole question can be put to the most convincing test possible if the primary vessels themselves, the aorta-. and the cardinal veins, can be shown to be formed in this way. I have accordingly set to work to decide this question through injections of very young embryos. The injection method had already shown itself to be by far the most efiicient way to demonstrate the form and entire extent of the vascular system in any area or organ. The methods employed in the injection of these minute vessels have already been indicated in a previous publication and a portion of the injected specimens shown at the last two annual meetings of this association, at New York, 1906, and at Chicago, 1907.
Chick embryos were selected for the study, not only for the convenience in securing and controlling material but also because 1he presence of early and easily accessible vitelline vessels furnishes a good portal of entry for the injection.
Development of the Lower Aortae
In embryos of the chick and duck possessing from twelve to fifteen somites, the aortae begin to be in free communication with the extraeinbryonic vitelline capillary net near the most caudal of the series of somites. Here also the character of the aortae becomes entirely changed. They are no longer the distinct and fairly straight tubes which they constitute in the upper portion of the body, but instead begin to be most irregular, connecting with the vitelline capillaries at as many points as would obtain in any capillary mesh and soon becoming resolved completely into the general extra-embryonic net from which they are entirely indistinguishable. In but a short distance, corresponding to the length of some five or six somites (were they present here), the vitelline capillaries no longer gain the median region of the embryo but surround the latter in a wide detour which always characteristically encirclesthe caudal extremity—the region of the primitive streak.
By the time the embryo possesses some twenty-four somites, the two aortae extend entirely through it, fusing with the vitelline capillaries only when the caudal tip has been reached. In what manner were the lower aortas developed?
A study of the intermediate stages in injected specimens makes it possible to give a very clear answer to this question.
By the stage of twenty somites marked changes have occurred from the earlier condition described. The aortae no longer appear to terminate in the region from the twelfth somite on, but are continued as strong vessels to the level of the twentieth segment, the caudal limit formerly reached by the most medial portion of the extra-embryonic plexus. Evidently the plexus formerly here has given place to the stronger single channel, but it has also continued to grow caudally in the tissues of the embryo, for it now reaches a considerable distance further caudad, to a point corresponding in position with the future twenty-fifth somite. In other words, there has been a continuous caudad invasion of the embryo by a plexus continuous always laterally with the extra-embryonic or vitelline net.
|
|
|
| Fig. 1. Ventral view of the posterior part of an injected chick embryo of 17 somites, showing plexiform character of the aorta: opposite the 17th somite. | Fig. 2. Ventral view of the posterior part of an injected chick embryo of 20 somites, showing an extension of the plexus out of which the aorta develops. The large nonvascular area surrounding the region of the primitive steak is here much reduced in extent. | Fig. 3. Ventral view of the posterior part of an injected chick embryo of 23 somites, showing the completion of the down growth of the capillary plexus out of which the aortae are formed. |
That portion of the plexus formerly occurring in the region from the fifteenth to the twentieth somites has had its most medial margin enlarged into the continuation of the aorta on each side, but this has involved also the elimination of its former frequent connections in this region with the remainder of the mesh. Some of the steps in this process are to be seen beginning in the mesh now present below the level of the twentieth somite, for in the upper portions of this the medial margin of the plexus is distinctly accentuated above its fellows, with which, however, it is still in continuous communication.
From now on, in successive stages, there is continued caudally this invasion of capillaries and the conversion of the innermost strand of the plexus into the continuation of the aortae.
Such a conception of the development of the aorta, I find was clearly indicated by His and especially by Vitalleton. The single figure of His showing one of the stages in this process gave the clue to the story, but, doubtless due to the difficulty -in recognizing the limit of capillaries by ordinary methods, Riickert and others have not accepted these views. Our preparations, however, have given in all its details this method of the formation of the lower aortaa.
It is interesting to note that the many connections of the aorta with the plexus from which they have been formed persist for a long time in the area below the level of origin of the main vitelline arteries. They may be said to form the primary circulation of this portion of the intestine, for, when the caudal vitelline vein is established and comes to encircle the posterior intestinal portal, these tiny vessels, coursing in the splanchnoplenre, connect the aortae with this vein.
Anterior Cardinal Veins
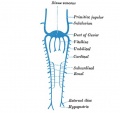
At the same time that capillary growth begins to more actively extend the aortae caudally, i.e., at the stage of fifteen somites, significant changes also begin in the region of the head. Here the earliest capillaries to grow out independently into the tissues of the embryos have arisen from the cephalic convexity of the first aortic arch and, extending dorso-laterally, formed a few meshes opposite the constriction between ‘fore and mid-brain. From here the capillaries spread forwards and backwards, growing somewhat more rapidly in the latter direction, so that a small plexus is soon formed at the side of the mid-brain. Anteriorly, the sprouts tend to encircle the stalk of the optic vesicles. At other points more caudad the dorsal aortas give rise to capillary sprouts which grow forwards and join those just mentioned and growing also in the opposite direction, coalesce with the vitelline veins near the junction of the latter trunks with the heart. Thus a slender but continuous chain of capillary vessels extends from the head region to the vitelline veins. Evidently enough of a circulation exists through this minute head plexus, fed as it is at several points from the aortas, to fashion a venule from the more caudal capillaries, i.e., those opposite the hind brain, so that at a very early date we have the picture of a long slender venule leading back from the plexus at about the region of the isthmus between mid and hind-brain to the vitelline veins near the heart.
It is out of this capillary plexus which has begun to grow up about the mid and fore brain vesicles that the head veins are all ultimately formed. These veins are the chief tributaries of the anterior cardinal trunk and consequently extend the latter vessel much forward into the region of the head.
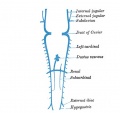
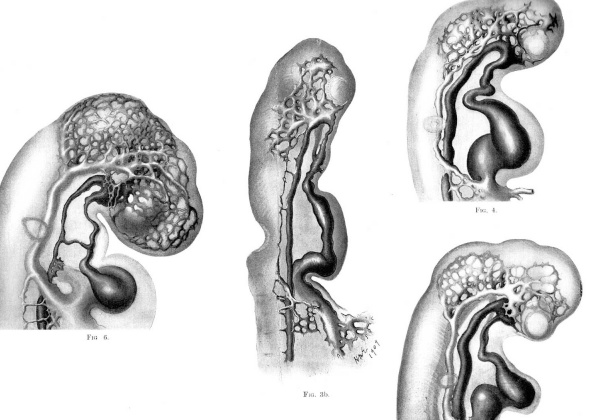
In all vertebrate embryos which I have studied, a portion of this capillary plexus opposite the mid-brain soon lies more superficially than the remainder and it is from these capillaries, enlarging soon, that the main vein is destined to be continued. This interesting stage in the development of the head vessels is seen in figs. 4, 5, and 6.
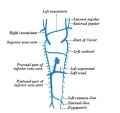
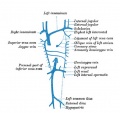
It is thus possible to trace the history of all the head tributaries of the anterior cardinal. Out of the capillaries connecting the more superficial ones just mentioned with those surrounding the sides of the optic vesicle, are formed the ophthalmic veins. And at the same time the caudal margin of the plexus covering the mid-brain is enlarged to form a prominent drainage channel, a vein which is thus situated at the isthmus between hind and mid-brain or at the caudal edge of the latter vesicle.
Posterior Cardinal Vein
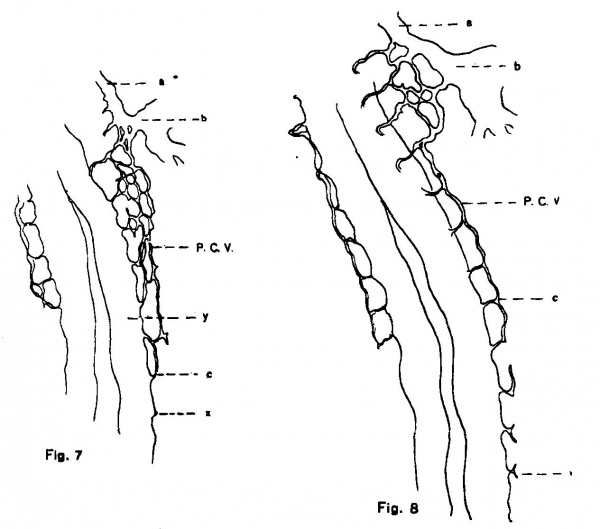
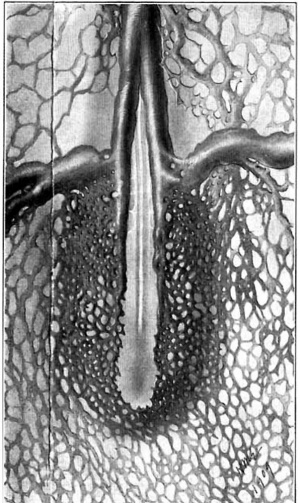
The origin and development of the posterior cardinal vein whose entire history can be followed, is the result of the activities of two systems of capillaries, a chain of capillaries arising from the duct of Cuvier and growing caudally in the splanchnopleure, and a row of capillaries, the intersegmental vessels, which in simple loops spring from the aorta and annex themselves successively to the former chain. What guides the course of these particular capillaries—the segmental vessels—so accurately into these loops is at present unknown, but it is no doubt a direct influence of the segmental structure of the neighboring mesenchyme which only favors endothelial proliferation at the inter«somitic spaces. Hence it is that at these intervals, the segmental capillaries (to become later the segmental arteries) grow out at right angles to the main axis of the embryo and after a dorsolateral loop or bend are free to extend longitudinally. Such a longitudinal extension involves their union with the capillary chain which has grown down from Cuvier’s duct and thus this channel is extended. Beyond the first four or five segments, this channel consists often of a single longitudinally coursing capillary and it is now further extended caudally solely by the longitudinal sprouts of the segmental capillaries, the cephalic sprout of the last of these joining the caudal sprout of the next preceding one as figs. 7 and 8 show. Coincident With~the extension of this vessel its upper section becomes larger, for the increased number of segmental afferents gives a considerable drainage territory; thus it is that it soon becomes a vessel of more than capillary size and recognizable as the posterior cardinal vein.
Fig. 3b. Lateral view of head of injected chick embryo of 15 somites, showing primary head capillary plexus. The plexus takes origin from the convexity of the first aortic arch at several points and is continued posteriorly as a slender capillary chain which eventually joins the main vitelline vein near its junction with the heart. This slender capillary chain has arisen at several points from the dorsal aorta on each side, and two of these points of origin are still preserved, opposite the region of the hind brain. The delicate capillary path from the head plexus to the vitelline vein is destined to form the anterior cardinal vein.
Fig. 4. Lateral view of head of injected chick embryo of 17 somites. showing the primary head capillary plexus partially covering the lateral sides of the fore and mid-brain vesicles. It will be seen that a‘ portion of the plexus lies more superficially than the remainder, and it is this superficial portion which is destined to become the main trunk of the vein in this region. The artery is shown darker than the vein.
Fig. 5. Lateral view of head of injected chick embryo of 20 omites. showing the further development of the anterior cardinal vein out of the primary head capillary plexus. The capillaries bordering the groove between mid and bind brain have formed a prominent tributary of the main vein.
Fig. 6. Lateral view of head of injected chick embryo of 25 somites. The lateral surfaces of the fore and mid brain vesicles are now completely covered by the capillary net, which is extending dorsally but is still far from the middorsal line. There is seen a corresponding great growth of the anterior cardinal vein and its system of tributaries.
Fig. 7. View or a total mount of an injected chick embryo of 17 somites, showing the duct of Cuvier and subadjacent region. (X 80.) a = anterior cardinal vein; b = duct of Cuvier; c = 10th segmental vessel; P. C. V. = capillaries from which the poterior cardinal vein is formed; x = endothelial sprout representing the 11th segmental vessel.
Fig. 8. View or a total mount of an injected chick embryo of 21 somites. showing the duct of Cuvier and subjacent region. (x 80.) The lettering is the same as in the preceding figure, with the exception of x, which represents the 14th segmental vessel. One sees the segmental capillaries biturcate often into anterior and posterior sprouts. the union of which makes the continuation of the vein.
Umbilical Vein
Those irregular capillary meshes which border the duct of Cuvier in embryos of from fifteen to seventeen somites and which aid in the formation of the upper end of the posterior cardinal vein, after a considerable interval, again sprout caudally, this time in the somatopleure, and are developed later into the veins which We can recognize as the umbilicals. The history of these veins in the chick is fraught with the greatest interest, for by the injections we can follow them in the assumption of several roles in the embryonic circulation, long before the establishment of their ultimate function in connection with the allantois. They are successively the drainage channels of the arm, thebody wall, and the leg before the allantois has arisen. The latter sac indeed attains some little size before its vessels are in connection with the umbilical veins.
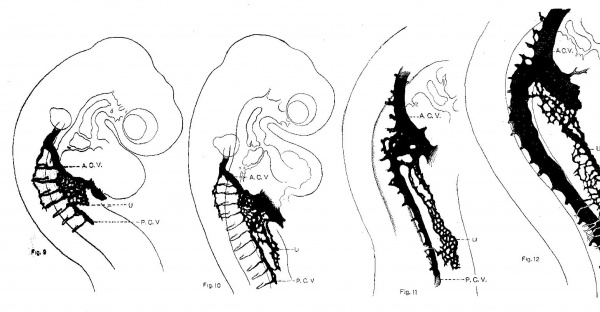
By the stage of twenty-three somites the first capillaries of the later umbilical vein form a simple mesh Work in the uppermost portion of the somatopleure. Soon the cell mass constituting the future anterior limb becomes evident and its growth stimulates the outgrowth from the aorta of a whole series of capillaries which unite to form a delicate plexus. These capillaries find and unite with those which have grown down from the duct of Cuvier and thus is established the earliest circulation in the limb bud, a circulation consisting of many afferent capillaries streaming from the lateral aortic wall, forming in the limb tissue a few simple meshes and draining headwards into the capillary chain, already somewhat enlarged and venous in character, which is the later umbilical vein.
Fig. 9. Injected chick embryo of 23 somites to show the origin of the umbilical vein from a capillary plexus situated in the angle between the posterior cardinal vein and the duct of Cuvier. A. C. V. = anterior cardinal vein; P. C. V. = posterlor cardinal vein; U = capiliaries destined to form the umbilical vein.
Fig. 10. Injected chick embryo of 24 somites to show the extension in the somatopleure, of capillary plexus forming the umbilical vein. The lettering is the same as fig. 9.
Fig. 11. Injected chick embryo of 30 somites. The capillaries destined to form the umbilical vein have reached the region of the future arm bud where they are joined by a direct capillary sprout from the aorta (subclavian artery).
Fig. 12. Injected chick embryo of 35 somites, showing establishment of umbilical vein as the main drainage channel of the anterior limb.
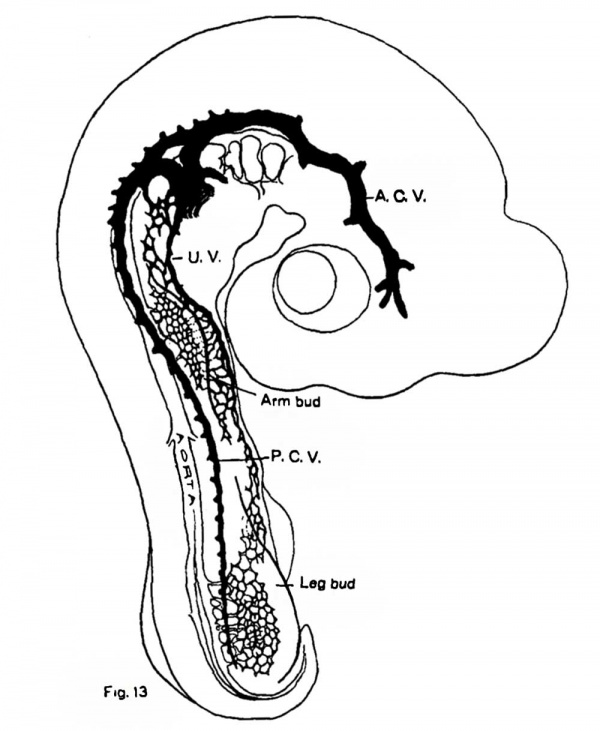
Fig. 13. Injected chick embryo of the third day showing extension of capillaries from which the umbilical vein is formed, as far as the posterior limb bud. The reduction is much greater than in the preceding figures in order that the entire embryo can be shown.
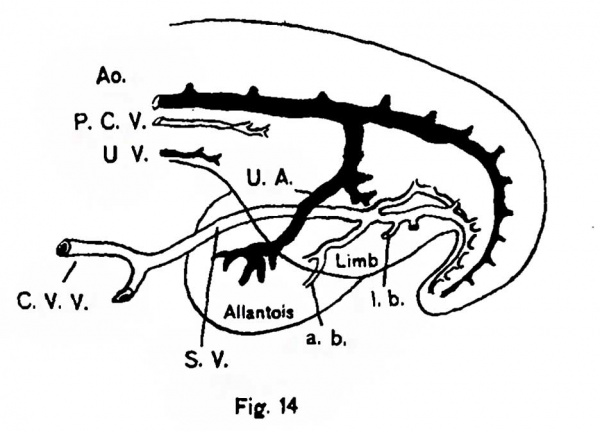
Fig. 14. Cuudal end of an injected chick embryo showing the subintestinal vein draining the tail, allantois and posterior limbs. a. b. = allantoic branches; l. b. = limb branches; S. V. = subintestinal vein; P. C. V. = posterior cardinal vein; Ao. = aorta; C. V. V. = caudal vitelline vein; U. A. = umbilical artery; U. V. = umbilical vein.
This capillary net still continues to grow caudally in the somatopleure, below the level of the upper limbs. At the same time another mesh of capillaries, that which has arisen in the posterior limb buds, has begun to grow upwards and the union of these two plexuses establishes a narrow continuous mesh in the somatopleure, into which the vessels of both limb buds and the body wall now drain. The capillaries of the hind limbs have also acquired connections with a more ventrally placed vein - the sub-intestinal vein, which has arisen in connection with the drainage of the tail and the allantois. This vein has hitherto been entirely overlooked and its presence in any of the embryos of the higher vertebrates is entirely unknown save for a few sentences announcing its occurrence in the ninety-six hour chick in the recent work by Lillie.[6] His remarks on the discovery of this vessel can be very appreciably extended now from the injections. As has just been indicated, the sub—intestinal vein in Aves forms the primary drainage channel for the tail, hind limbs and allantois. Its position and chief tributaries can be seen from fig. 14. Somewhat later, and at about the time the allantois approaches a millimeter in diameter, the umbilical system of capillaries has united with its vessels and begins to function as a means of drainage for the allantoic circulation. The uppermost portions of the umbilical have now enlarged appreciably, its connections with both limb buds are eventually lost and its last territory supplies it with such a volume that it becomes a relatively huge channel, the allantoic or umbilical vein.
In the Mammalia, although the umbilical veins precede the limb buds in time of appearance, nevertheless here also, when the limbs arise, they are at first drained into the umbilical veins.[5]
Dr. F. T. Lewis informs me that he had observed and demonstrated this drainage of the mammalian limbs into the umbilical veins at the meeting of this association in 1903. Unfortunately no record of this was made in the proceedings for that session, but Dr. Lewis has been kind enough to send me sketches and notes made at the time, showing this fact for rabbit embryos: I have recently been able to confirm these findings on the human embryo also, so that there is little reason to doubt its general applicability for the Mammalia.
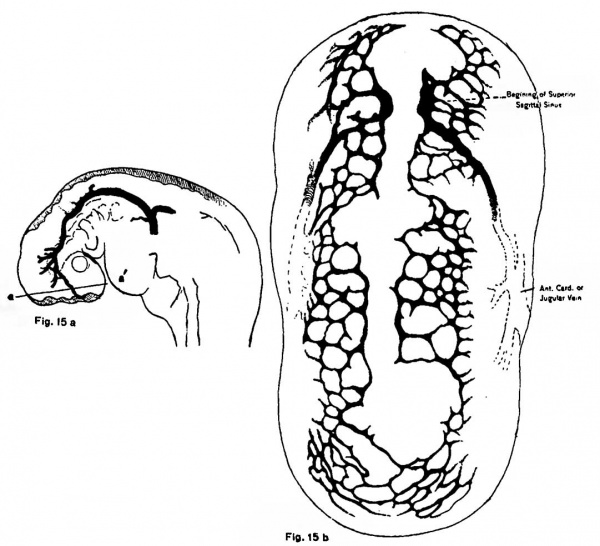
Many other prominent vessels in the body have been traced to a similar origin from a capillary plexus, but time will now permit the mention of only a few of these. In reconstructions of the vessels of the head which have been made by various investigators, it appears as if the tip of the anterior cardinal veins grew forwards in a dorsomedial position, in place to form the future sagittal sinus. It has been possible to trace the formation of this vein quite completely in injected mammalian embryos. In pig embryos five and six millimeters in length the primitive capillary plexus which grows up over the sides of the mid and fore brain has not yet reached the dorsal surface, though sprouts can be seen along its upper margin. By the time the embryo has attained a length of nine millimeters, the capillary mesh has covered the top of fore and ’tween-brain vesicles save in the median line. Here the two meshes are as yet unfused, and confront each other along two parallel lines, which thus bound a median dorsal non-vascular strip, across which no connecting capillaries have ventured to grow. It is these two medial margins of the plexus which, in the region of the cerebral hemispheres, are enlarged to form the superior sagittal sinus, thus originally paired. (figs. 15a and 15b.)
Fig. 15 a. Lateral view of upper portion of pig embryo 8 mm. long, showing location of mid dorsal non-vascular area, the extent of which has been purposely exaggerated laterally. It will be noted that the capillaries have fused dorsally over the mid brain and upper portion of the hind brain.
Fig. 15 b. Dorsal view of fore and mid brain region of the pig of 8 mm. shown in fig. 15 a, showing the limit of extension of the capillary plexus here. The mesh work which has grown dorsally from either side halts sharply in two parallel lines between which is the narrow non-vascular strip. Anteriorly is seen the earliest indication of the superior sagittal sinus, which is formed from either margin of the capillary mesh, and consequently at this stage paired.
One of the most beautiful and evident instances of the conversion of a capillary mesh into an arterial channel is afiorded in the history of the anterior spinal artery. Here too we have the best posible axis of reference, for the mid-ventral line of the spinal cord is constant. On the ventral surface of the cord we can observe all the steps in the first invasion of a plexus of capillaries there, their later coalescence and enlargement in the mid-line as an irregular, illdefined channel, and eventually, the further conversion of this into the very definite artery of regular contour and calibre —the anterior spinal. fig. 18 illustrates the development of this vessel in the pig.
I may be permitted to instance one more vessel, in this case one of the very largest in the body, though not the earliest to develop, which can easily be seen in the young embryo in the form of a capillary mesh. I refer to the pulmonary artery. The endothelial sprouts which later form this trunk spring from the sixth aortic arch as true capillaries. In fact they reach the lung bud as a chain of capillary meshes and retain this character for some time, as fig. 21 shows.
Besides the history of many individual blood vessels of the body, these specimens have given weighty evidence towards a number of general laws or phenomena of blood vessel development and these will be briefly mentioned. They concern
- The presence always in the embryo, of a united vascular system, so that the blood vessels form a single though irregularly branched endothelial tree whose branches are in no case added after an independent formation but arise by sprouting from the parent trunks.
- The place and manner of spread of the first capillaries through the body.
In discussing these briefly, we may say
(1) Serial sections of perfectly injected embryos show no evidences of vessels which have not received the injection mass and are hence unconnected with the general system. Investigators, working with uninjected material, have repeatedly reported such vessels. Their findings are in all probability to be explained by a collapse of the connecting vessels, since
- a, injected specimens show these connecting vessels and
- b, injected specimens fill other vessels previously unrevealed by ordinary methods, thus furnishing a far more complete picture than is otherwise obtainable.
The recent accounts by Riickert and Mollier in Hertwig’s Handbuch, on the subject of the first blood vessels are perhaps the most conspicuous of the claims of vessel origin in situ. Their evidence has come from Riickert’s studies of serial sections through selachian embryos. His statements can doubtless be successfully attacked by injecting selachian embryos and studying carefully the areas in question.
With the light which such specimens have shed, the statement that any vessels in the embryo arise at first unconnected with the vessels in that region can be now challenged. If an instance be given it can doubtless be speedily disproven, providing complete injections of that area can be secured.
(2) The spread of the first vessels through the body. Whatever may be the first source of the endothelium in the body of the embryo, after the earliest stages, the injections have furnished a complete history of the further capillary proliferation and outgrowth into the tissues of the embryo.
Inasmuch as the first vessels lie somewhat centrally in the embryonic body, the general direction of growth is from center to periphery. The center consists of the upper dorsal aortas together with the first arch and Cuvier’s duct; the periphery comprises the various viscera and central nervous system as well as the body wall, but the ultimate periphery, the skin, is supplied late.
In spreading outward, the capillaries do not grow uniformly in all directions, thus successively invading various zones, but are apparently governed by the character and needs of the various tissues, reaching some of them early and some remarkably late. Hence there are present during all the early stages in the embryo’s growth, vascular and non—vascular areas.
The method of injection reveals such a wealth of small vessels whose existence we had not hitherto known, that at first thought one is inclined to suspect the universal presence of the vascular net, throughout the tissues of the embryo. This, however, is as much an error as was the former notion of the scant extent of the embryonic vessels. Injections made under the best possible conditions and afterwards explored in serial section have all shown the existence of definite non-vascular areas bordered by a margin of true capillary sprouts. The position of such non-vascular areas is as constant as is that of any vascular channel in the body and the more fundamental of them are probably represented at homologous stages in all vertebrate embryos.
Among the tissues, the central nervous system receives the first investing capillary net, but even here the capillaries do not at once surround the neural tube but occupy only the lateral aspect, gradually growing ventrally and dorsally. At the top of the brain, the capillary mesh is some time in fusing from either side, so that there exists here relatively late the narrow non-vascular strip in the middorsal line already mentioned. (fig. 15.) In the case of the hind brain there is an especially conspicuous lack of much capillary proliferation in a dorsal direction, so that in comparatively late stages of all vertebrate embryos the roof of the hind brain presents a characteristic large non-vascular zone. Indeed, while in pig embryos ten millimeters in length the lateral capillary beds have completely fused dorsally, in the fore and mid-brain region, the nonvascular area on the top of the hind-brain persists until the embryo has attained a length of over twenty millimeters. (figs. 16, 17a, and 17b.)
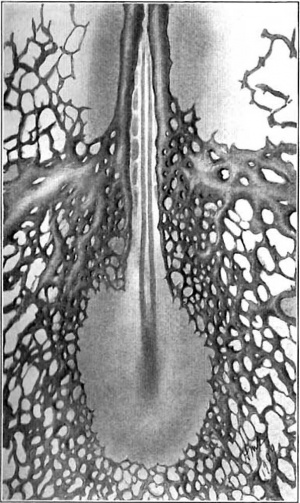
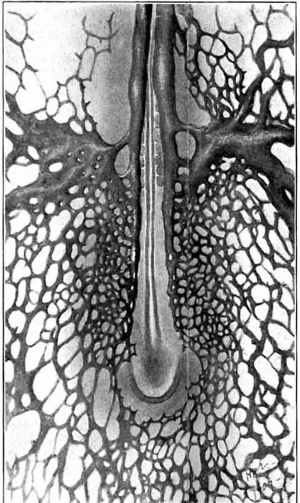
In the cord also the ventral and dorsal surfaces are invaded only secondarily and are at first entirely non-vascular. The dorsal surface is bridged last of all and so the spinal axis presents for a time the remarkable sight of a close capillary investment everywhere save on its upper aspect, at the margins of which the two long parallel borders of invading capillaries and their sprouts have halted sharply in their spread. (fig. 19.) This narrow non-vascular zone is maintained for a long time, but when the time comes for its obliteration, quite suddenly, capillary sprouts push out and bridge the gap. This bridging occurs successively from above downwards and embryos which have the dorsal surface thickly covered with capillaries in the upper half will show the first bridging capillaries: in the caudal region, as fig. 20 shows.
Other examples of vascular and non-vascular areas may be mentioned. The center of each sclerotome is, on its upper surface, supplied by a sheet of closely anastomosed capillaries; but the outer divisions of the sclerotome are not so supplied. There capillaries are absent for a considerable time, so that the vertebral column presents a succession of vascular and non-vascular zones, the former areas in each case overlying the segmental vessels.
Furthermore, in the growth of the embryo, tissue at one time permeated with a quite uniform capillary mesh may in its further growth show a later difierentiation into vascular and non-vascular areas.. This arrangement of its vascular mesh is of course coincident With corresponding changes in the nature of the tissue at various areas and it is often the most positive evidence of these changes. Thus pre-cartilage and pre-muscle tissue are characteristically non-vascular and wherever these condensations of the mesenchyme occur, they form islands of tissue sharply circumscribed by capillaries but uninvaded by the latter for a considerable interval. Of this the limb buds furnish splendid examples, for the blastema of the arm, which is at first furnished with a uniformly distributed capillary net, later begins to exhibit areas which the capillaries appear to avoid—areas corresponding to the later masses of cartilage or muscle groups.
Fig. 16. Dorsal surface of bind and mid brain of a pig embryo 8.5 mm. long, showing fusion\ of the primary head plexus across the mid line, except the three non-vascular areas shown.
Fig. 17 a. Dorsal surface of fore and mid-brain vesicles of injected chick embryo of 32 omites.
Fig. 17 b. Dorsal surface of fore and mid-brain vesicles of injected chick embryo at _the end of the 3d day. c. h.=cerebral hemisphere; th.=thalainencephalon; m. b.=mid brain. In the earlier stage (fig. 17 a) the primary head capillary plexus has fused across the mid dorsal line only at one point, between the two divisions of the primitive _fore brain. In the later stage the mesh quite completely invests the mid dorsal surface of the head, but the cleft between the cerebral hemispheres is non-vascular, as is also the zone surrounding the pineal organ. At the mesial margins of the two prominent lobes of the mid brain are seen the two mesencephalic veins which have been formed from the plexus.
Fig. 18. The ventral surface of the spinal cord in the region of the first three thoracic segments in a series of injected pig embryos, showing the origin and development at the anterior spinal artery. )4 331,9. a, from an embryo 6 mm. long. No capillaries appear on the ventral surface. those seen invest the lateral surface and the ganglia. b. from an embryo 8.5 mm. in length. Capillary sprouts are seen invading the ventral surface of the cord. c, from an embryo 9 mm. long. The sprouts shown in the preceding figure have new extended as delicate straight parallel capillaries which bridge the mid line. so that the non-vascular area there is now obliterated. e, from an embryo 14 mm. long. The processes of endothelial coalescence suggested in the preceding stage have resulted in the formation of an irregular fenestrated mid-ventral channel, in freer communication at intervals with the ventral branches of the segmental vessels; f, from an embryo 15.5 mm. long, the mid~ventral channel has become narrower and its segmental aflerents much stronger; g, from an embryo 28 mm. in length. The arterial character of the mid-ventral channel is now apparent, some of the segmental aflerents have disappeared and others have been much exaggerated in growth. The capillnries everywhere are of smaller caliber than those found in the earliest stages.
Fig. 19. A series of views of the dorsal surface of the spinal cord at the level of the lower cervical segments in injected pig embryos. a, from an embryo 8 mm. in length. All the capillaries seen are limited to the lateral surfaces of the cord and the spinal ganglia. b, from an embryo 10.5 mm. long, showing the first capillary bridges across the cord in this region. c, from an embryo 15 mm. long, showing the type of plexus established here. d, from an embryo 20 mm. long, illustrating the farther growth of the plexus.
Fig. 20. Tail of an injected pig embryo, 13.5 mm. long. In the cervical and thoracic regions, the plexus investing the dorsal surface of the cord has become a close mesh; in this region, however, the flrst capillary bridges. and, at the end, the first sprouts, are shown.
Fig. 21. Camera lucida tracing of the fourth and sixth aortic arches of an injected pig embryo 12 mm. long to show the early character of the pulmonary arteries. P. A.=pulmonary arteries; L.=lungs; P. V.=pulmonary vein; VI =sixth arch; IV: fourth arch; S. A.=subclavian artery.
In most cases the capillaries tend to anastomose at greater or lesser intervals forming a loose or close mesh and this plexus formation is doubtless one of their most characteristic and fundamental properties. It has perhaps been better termed their tendency to grow in every direction, yet influences often check this tendency successfully and in some areas permit their growth from the very first only in a certain definite direction. The best example of this is furnished by the dorsal segmental vessels which, as is well known, are rigidly governed in position by the presence of the primitive segments, between which they course.
All of these examples clearly indicate that the behavior and character of the capillaries is even from the very first intimately influenced by the tissues into which they grow. A new set of problems confronts us, problems which can aim more than ever before at the causes at work in the developing organism, for now that We may recognize with certainty vascular and non—vascular areas and the relation of each of these to the tissues, there come up at once questions concerning the difierences in chemical nature of the tissues and a closer determination of the real stimulant for vascular growth.
The story of the development of the vascular system is primarily the story of the spread of the capillaries, the form and relation of their plexuses and the role of these in the elaboration of the trunks of the adult.
We have to do always with the extension of a functioning system not the blind outgrowth of vessels to their ultimate territory. Such a system extends itself by capillary sprouts and as the capillary bed increases, its supplying and draining channels, the arteries and the veins, grow and rearrange themselves concordantly.
Received for publication July 6, 1909.
References
- ↑ Henle. J. "Handbuch der Gcfiisslehre des Menschen," Braunschweig, 1876.
- ↑ Thoma. R. “Untersuchungen fiber die Histogenese und Hlstomechanlk des Gefiisssystems,” 1893.
- ↑ Evans HM. On an instance of two subclavian arteries of the early arm bud of man and its fundamental significance. (1908) Anat. Rec.
- ↑ Elze. “Beschreibung eines menschlichen Embryo," Anat. Hefte, Bd. 35, 1907.
- ↑ 5.0 5.1 Evans HM. On the earliest blood-vessels in the anterior limb-buds of birds and their relation to the primary subclavian artery. (1909) Amer. J Anat. 9: 281-319.
- ↑ Lillie. "The Development of the Chick.” 1909.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - On the development of the aortae cardinal and umbilical veins and the other blood vessels of vertebrate embryos from capillaries (1909). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_On_the_development_of_the_aortae_cardinal_and_umbilical_veins_and_the_other_blood_vessels_of_vertebrate_embryos_from_capillaries_(1909)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G