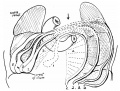
Introduction
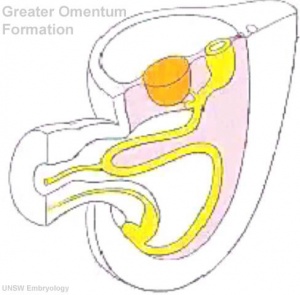
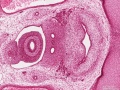
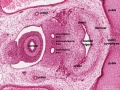
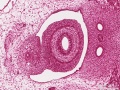
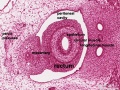
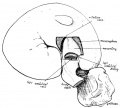
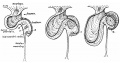
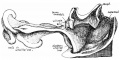
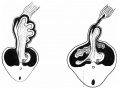
Early embryonic mesentery (pink)
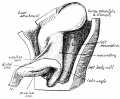
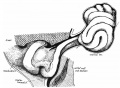
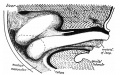
The adult gastrointestinal tract (GIT) is attached along its length by a dorsal mesentery between the stomach (oesophagogastric) and anus (anorectal junctions) arising from splanchnic mesoderm. This differentiates into a mesothelium, an epithelium derived from mesoderm. The majority of the ventral mesentery is developmentally lost at the level of the midgut and only the dorsal mesentery remains in the adult, through which blood vessels, nerves and lymph connects to the gastrointestinal wall.
Recently mouse mesentery has been used as a transplantation site for human intestinal organoid study.[1]
- Historic Embryology: 1884 great omentum and transverse mesocolon | 1912 Small Intestine | 1912 large Intestine | 1915 Intestinal Rotation
Some Recent Findings
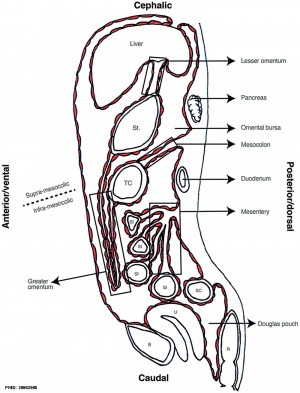
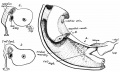
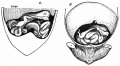
Sagittal view of abdominal cavity
[2]
- The development of the dorsal mesentery in human embryos and fetuses[3] "The vertebrate intestine has a continuous dorsal mesentery between pharynx and anus that facilitates intestinal mobility. Based on width and fate the dorsal mesentery can be subdivided into that of the caudal foregut, midgut, and hindgut. The dorsal mesentery of stomach and duodenum is wide and topographically complex due to strong and asymmetric growth of the stomach. The associated formation of the lesser sac partitions the dorsal mesentery into the right-sided "caval fold" that serves as conduit for the inferior caval vein and the left-sided mesogastrium. The thin dorsal mesentery of the midgut originates between the base of the superior and inferior mesenteric arteries, and follows the transient increase in intestinal growth that results in small-intestinal looping, intestinal herniation and, subsequently, return. The following fixation of a large portion of the abdominal dorsal mesentery to the dorsal peritoneal wall by adhesion and fusion is only seen in primates and is often incomplete. Adhesion and fusion of mesothelial surfaces in the lesser pelvis results in the formation of the "mesorectum".
- Digestive Tract in Human Embryos Between Carnegie Stages 11 and 13[4] "We selected 37 human embryos at Carnegie Stage (CS) 11-13 (28-33 days after fertilization) and three-dimensionally analyzed the morphology and positioning of the digestive tract and derived primordia in all samples, using images reconstructed from histological serial sections. The digestive tract was initially formed by a narrowing of the yolk sac, and then several derived primordia such as the pharynx, lung, stomach, liver, and dorsal pancreas primordia differentiated during 12 (21-29 somites) and CS13 (≥ 30 somites). The differentiation of four pairs of pharyngeal pouches was complete in all 13 embryos. The respiratory primordium was recognized in ≥ 26-somite embryos and it flattened and then branched at CS13. The trachea formed and then elongated in ≥ 35-somite embryos. The stomach adopted a spindle shape in all ≥ 34-somite embryos, and the liver bud was recognized in ≥ 27-somite embryos. The dorsal pancreas appeared as definitive buddings in all but three CS13 embryos, and around these buddings, the small intestine bent in ≥ 33-somite embryos. In ≥ 35-somite embryos, the small intestine rotated around the cranial-caudal axis and had begun to form a primitive intestinal loop, which led to umbilical herniation."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Mesentery Embryology | Mesentery Development |
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Growth of the colon and rectum throughout gestation: evaluation with fetal MRI[5] "Congenital abnormalities of the gastrointestinal tract are increasingly being evaluated by prenatal magnetic resonance imaging (MRI). However, there is a paucity of reports describing the normal quantitative development of the fetal colon and rectum on MRI. This study provides normal ranges of the prenatal colon and rectum as a function of gestational age. They may serve as reference values when interpreting fetal MRI."
- Review - How to make an intestine[6] With the high prevalence of gastrointestinal disorders, there is great interest in establishing in vitro models of human intestinal disease and in developing drug-screening platforms that more accurately represent the complex physiology of the intestine. We will review how recent advances in developmental and stem cell biology have made it possible to generate complex, three-dimensional, human intestinal tissues in vitro through directed differentiation of human pluripotent stem cells.
|
Movies
Regions
The following are anatomical sub-regions of the continuous mesentery.
- dorsal mesogastrium
- mesoduodenum
- mesenteric root region
- small intestine region
- right mesocolon
- left mesocolon
- mesosigmoid
- mesorectum
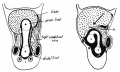
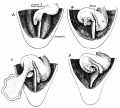
Dorsal Mesogastrium
Greater Omentum
| <html5media height="520" width="490">File:Greater omentum 001.mp4</html5media>
Click Here to play on mobile device
|
This animation shows the development of the greater omentum associated with the greater curvature of the stomach.
The greater omentum begins as an extended fold of the dorsal mesogastrium that continues to grow and extend down into the peritoneal cavity and eventually lies anterior to the small intestines. This fold of mesentery will also fuse to form a single sheet.
Legend
- liver
- gastrointestinal tract
- mesentery
|
Small Intestine
Intestinal Functions
Small Intestine
- absorption of nutrients and minerals found in food
- Duodenum -principal site for iron absorption
Colon
- absorbs fluid, water and salts, from solid wastes
Embryonic Development
Week 4
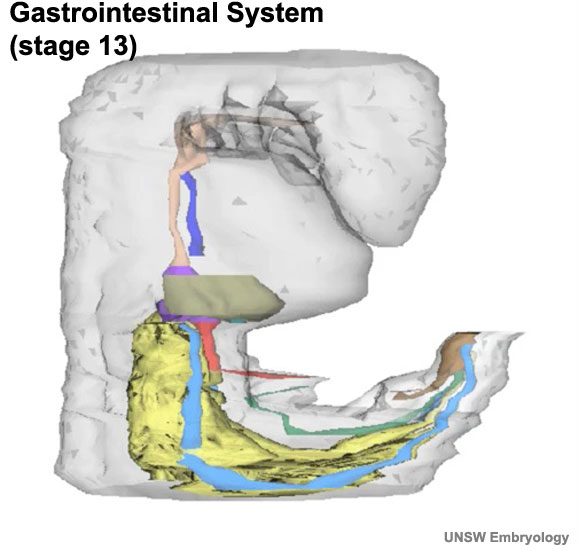
|
Colour code:
- Foregut - oropharyngeal membrane, oesophagus, pharynx (dark salmon - Foregut, oropharyngeal membrane, oesophagus, pharynx)
- Trachea and Lung Buds (dark blue - Trachea and Lung Buds)
- Mesonephros (yellow - mesonephros)
- Mesonephric Ducts (light blue - mesonephric ducts)
- Hindgut and Cloaca (brown - hindgut and cloaca)
|
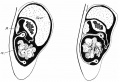
Week 7
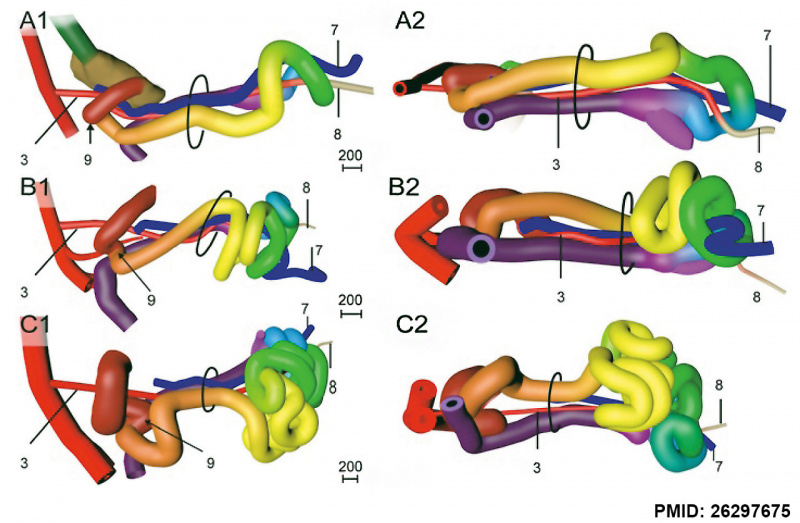
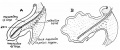
Human embryo small intestine secondary loops (week 7 to 8).[7]
Week 8
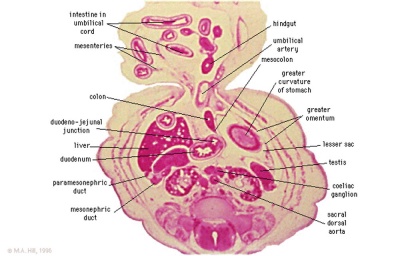
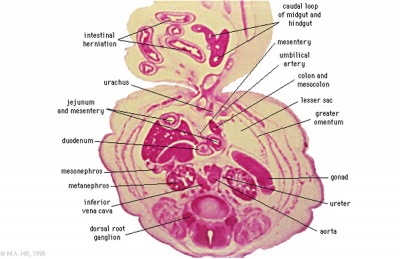
Late embryonic small intestine commencing at the duodenum, continuing as ventrally herniated and returning to join the colon.
rectum and bladder labeled
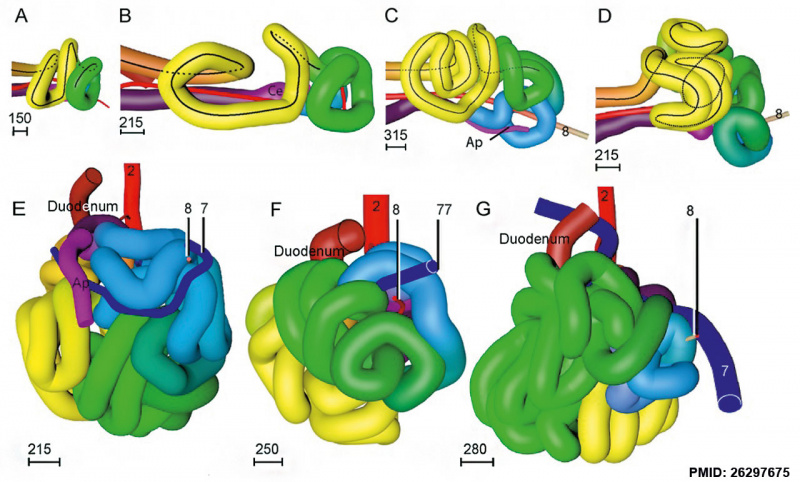
Small intestine tertiary loops week 8.[7]
- Links: Carnegie stage 22 | Week 8
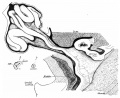
Rotation
A recent 3 dimensional study[7] has suggested a modified “en-bloc rotation” of the small intestine, compared the the earlier simplified description of 270 degree rotation.
Fetal Intestine Length
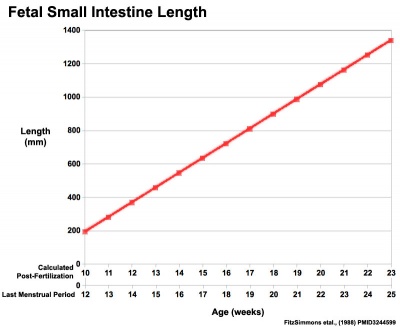
|
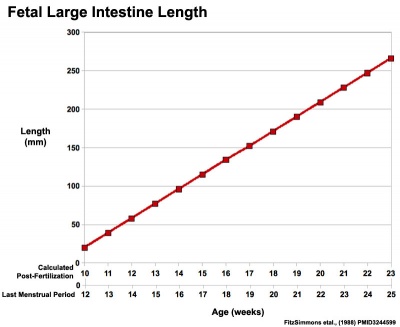
|
| Fetal small Intestine length growth
|
Fetal Large Intestine length growth
|
Data from[8][9]
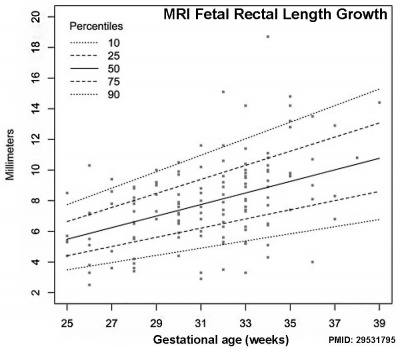
Data from[5]
Small Intestine Length
Small intestine growth in length is initially linear (first half pregnancy to 32 cm CRL), followed by rapid growth in the last 15 weeks doubling the overall length. Growth continues postnatally but after 1 year slows again to a linear increase to adulthood.[10]
Small Intestine Length
| Age (weeks GA gestational age)
|
Average Length (cm)
|
| 20
|
125
|
| 30
|
200
|
| term
|
275
|
| 1 year postnatal
|
380
|
| 5 years
|
450
|
| 10 years
|
500
|
| 20 years
|
575
|
| Table data based upon 8 published reports of necropsy measurement of 1010 guts.[10]
|
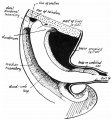
Hindgut
Anatomically the distal third of the transverse colon and the splenic flexure, the descending colon, sigmoid colon and rectum. The developmental timing of the anus and rectum formation[11] in human embryos of the Carnegie Collection has been previously carried out (1974). A more recent study[12] has also been made of the Kyoto Collection embryos.
There has been some recent controversy over the "anal membrane" formation.
A recent study hindgut and anorectum development in human embryos shows that WNT5a is active in this region prior to anus formation, when it is down-regulated.[13]
Other studies - [14][15][16][17][18] (rat)
Abnormalities
Intestinal malrotation[19]
Midgut Volvulus[20]
Molecular Factors
Histology
The adult mesentery consists of loose irregular connective tissue, areolar connective tissue, collagen fibres, elastic fibres.
There are also numerous mast cells (granulocytes) containing histamine and other substances released during inflammatory and allergic reactions.
Adult Mesentery Histology
| Low power (x10)
|
High power (x40)
|
|
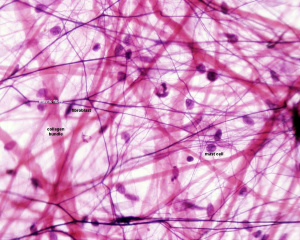
|
References
- ↑ Cortez AR, Poling HM, Brown NE, Singh A, Mahe MM & Helmrath MA. (2018). Transplantation of human intestinal organoids into the mouse mesentery: A more physiologic and anatomic engraftment site. Surgery , 164, 643-650. PMID: 30072255 DOI.
- ↑ Isaza-Restrepo A, Martin-Saavedra JS, Velez-Leal JL, Vargas-Barato F & Riveros-Dueñas R. (2018). The Peritoneum: Beyond the Tissue - A Review. Front Physiol , 9, 738. PMID: 29962968 DOI.
- ↑ Hikspoors JPJM, Kruepunga N, Mommen GMC, Peeters JPWU, Hülsman CJM, Eleonore Köhler S & Lamers WH. (2018). The development of the dorsal mesentery in human embryos and fetuses. Semin. Cell Dev. Biol. , , . PMID: 30142441 DOI.
- ↑ Ueno S, Yamada S, Uwabe C, Männer J, Shiraki N & Takakuwa T. (2016). The Digestive Tract and Derived Primordia Differentiate by Following a Precise Timeline in Human Embryos Between Carnegie Stages 11 and 13. Anat Rec (Hoboken) , 299, 439-49. PMID: 26995337 DOI.
- ↑ 5.0 5.1 Ben-Nun MS, Ben-Shlush A & Raviv Zilka L. (2018). Growth of the colon and rectum throughout gestation: evaluation with fetal MRI. Acta Radiol Open , 7, 2058460118761206. PMID: 29531795 DOI.
- ↑ Wells JM & Spence JR. (2014). How to make an intestine. Development , 141, 752-60. PMID: 24496613 DOI.
- ↑ 7.0 7.1 7.2 Soffers JH, Hikspoors JP, Mekonen HK, Koehler SE & Lamers WH. (2015). The growth pattern of the human intestine and its mesentery. BMC Dev. Biol. , 15, 31. PMID: 26297675 DOI.
- ↑ FitzSimmons J, Chinn A & Shepard TH. (1988). Normal length of the human fetal gastrointestinal tract. Pediatr Pathol , 8, 633-41. PMID: 3244599
- ↑ Archie JG, Collins JS & Lebel RR. (2006). Quantitative standards for fetal and neonatal autopsy. Am. J. Clin. Pathol. , 126, 256-65. PMID: 16891202 DOI.
- ↑ 10.0 10.1 Weaver LT, Austin S & Cole TJ. (1991). Small intestinal length: a factor essential for gut adaptation. Gut , 32, 1321-3. PMID: 1752463
- ↑ de Vries PA. and Friedland GW. The staged sequential development of the anus and rectum in human embryos and fetuses. (1974) J. Pediatr. Surg., 9(5): 755-69 PMID 4424274
- ↑ Hashimoto R. (2013). Development of the human tail bud and splanchnic mesenchyme. Congenit Anom (Kyoto) , 53, 27-33. PMID: 23480355 DOI.
- ↑ Li FF, Zhang T, Bai YZ, Yuan ZW & Wang WL. (2011). Spatiotemporal expression of Wnt5a during the development of the hindgut and anorectum in human embryos. Int J Colorectal Dis , 26, 983-8. PMID: 21431850 DOI.
- ↑ van der Putte SC. (2009). The development of the human anorectum. Anat Rec (Hoboken) , 292, 951-4. PMID: 19496155 DOI.
- ↑ Kromer P. (1999). Further study of the urorectal septum in staged human embryos. Folia Morphol. (Warsz) , 58, 53-63. PMID: 10504783
- ↑ Nievelstein RA, van der Werff JF, Verbeek FJ, Valk J & Vermeij-Keers C. (1998). Normal and abnormal embryonic development of the anorectum in human embryos. Teratology , 57, 70-8. PMID: 9562679 <70::AID-TERA5>3.0.CO;2-A DOI.
- ↑ Kromer P. (1996). Development of the urorectal septum and differentiation of the urogenital sinus in human embryos of stages 13 to 19. Folia Morphol. (Warsz) , 55, 362-3. PMID: 9243909
- ↑ Kluth D, Fiegel HC & Metzger R. (2011). Embryology of the hindgut. Semin. Pediatr. Surg. , 20, 152-60. PMID: 21708335 DOI.
- ↑ Ezer SS, Oguzkurt P, Temiz A, Ince E, Gezer HO, Demir S & Hicsonmez A. (2016). Intestinal malrotation needs immediate consideration and investigation. Pediatr Int , 58, 1200-1204. PMID: 27353636 DOI.
- ↑ Coste AH & Bhimji SS. (2018). Midgut Volvulus. , , . PMID: 28722991
Reviews
Byrnes KG, Walsh D, Lewton-Brain P, McDermott K & Coffey JC. (2019). Anatomy of the mesentery: Historical development and recent advances. Semin. Cell Dev. Biol. , 92, 4-11. PMID: 30316830 DOI.
Byrnes KG, McDermott KW & Coffey JC. (2019). Mesenteric organogenesis. Semin. Cell Dev. Biol. , 92, 1-3. PMID: 30336281 DOI.
Byrnes KG, Walsh D, Lewton-Brain P, McDermott K & Coffey JC. (2019). Anatomy of the mesentery: Historical development and recent advances. Semin. Cell Dev. Biol. , 92, 4-11. PMID: 30316830 DOI.
Yoshino T & Saito D. (2019). Epithelial-to-mesenchymal transition-based morphogenesis of dorsal mesentery and gonad. Semin. Cell Dev. Biol. , 92, 105-112. PMID: 30193994 DOI.
Hen G & Sela-Donenfeld D. (2019). "A narrow bridge home": The dorsal mesentery in primordial germ cell migration. Semin. Cell Dev. Biol. , 92, 97-104. PMID: 30153479 DOI.
Hikspoors JPJM, Kruepunga N, Mommen GMC, Peeters JPWU, Hülsman CJM, Eleonore Köhler S & Lamers WH. (2018). The development of the dorsal mesentery in human embryos and fetuses. Semin. Cell Dev. Biol. , , . PMID: 30142441 DOI.
Byrnes KG, McDermott K & Coffey JC. (2018). Development of mesenteric tissues. Semin. Cell Dev. Biol. , , . PMID: 30347243 DOI.
Byrnes KG, Walsh D, Dockery P, McDermott K & Coffey JC. (2018). Anatomy of the mesentery: Current understanding and mechanisms of attachment. Semin. Cell Dev. Biol. , , . PMID: 30316831 DOI.
Isaza-Restrepo A, Martin-Saavedra JS, Velez-Leal JL, Vargas-Barato F & Riveros-Dueñas R. (2018). The Peritoneum: Beyond the Tissue - A Review. Front Physiol , 9, 738. PMID: 29962968 DOI.
Wells JM & Spence JR. (2014). How to make an intestine. Development , 141, 752-60. PMID: 24496613 DOI.
Noah TK, Donahue B & Shroyer NF. (2011). Intestinal development and differentiation. Exp. Cell Res. , 317, 2702-10. PMID: 21978911 DOI.
Burns AJ, Roberts RR, Bornstein JC & Young HM. (2009). Development of the enteric nervous system and its role in intestinal motility during fetal and early postnatal stages. Semin. Pediatr. Surg. , 18, 196-205. PMID: 19782301 DOI.
Articles
Wozniak S, Florjanski J, Kordecki H, Podhorska-Okolow M & Domagala Z. (2018). Fetal sigmoid colon mesentery - In relevance in fetal ultrasound application. A pilot study. Ann. Anat. , 216, 152-158. PMID: 29292173 DOI.
Cho BH, Kim JH, Jin ZW, Wilting J, Rodríguez-Vázquez JF & Murakami G. (2018). Topographical anatomy of the intestines during in utero physiological herniation. Clin Anat , 31, 583-592. PMID: 29044646 DOI.
Nerurkar NL, Mahadevan L & Tabin CJ. (2017). BMP signaling controls buckling forces to modulate looping morphogenesis of the gut. Proc. Natl. Acad. Sci. U.S.A. , 114, 2277-2282. PMID: 28193855 DOI.
Soffers JH, Hikspoors JP, Mekonen HK, Koehler SE & Lamers WH. (2015). The growth pattern of the human intestine and its mesentery. BMC Dev. Biol. , 15, 31. PMID: 26297675 DOI.
Coffey JC, Culligan K, Walsh LG, Sehgal R, Dunne C, McGrath D, Walsh D, Moore M, Staunton M, Scanlon T, Dewhurst C, Kenny B, O'Riordan C, O'Brien JM, Quondamatteo F & Dockery P. (2016). An appraisal of the computed axial tomographic appearance of the human mesentery based on mesenteric contiguity from the duodenojejunal flexure to the mesorectal level. Eur Radiol , 26, 714-21. PMID: 26186959 DOI.
Davis NM, Kurpios NA, Sun X, Gros J, Martin JF & Tabin CJ. (2008). The chirality of gut rotation derives from left-right asymmetric changes in the architecture of the dorsal mesentery. Dev. Cell , 15, 134-45. PMID: 18606147 DOI.
Cascio S, Tien AS, Agarwal P & Tan HL. (2006). Dorsal mesenteric agenesis without small bowel atresia: a rare cause of midgut volvulus in children. J. Pediatr. Surg. , 41, E5-7. PMID: 16952580 DOI.
Search Pubmed
Search Bookshelf Mesentery Development
Search Pubmed Now: Mesentery Embryology | Mesentery Development
Terms
| Gastrointestinal Tract Terms
|
- allantois - An extraembryonic membrane, endoderm in origin extension from the early hindgut, then cloaca into the connecting stalk of placental animals, connected to the superior end of developing bladder. In reptiles and birds, acts as a reservoir for wastes and mediates gas exchange. In mammals is associated/incorporated with connecting stalk/placental cord fetal-maternal interface.
- amnion - An extra-embryonic membrane, ectoderm and extraembryonic mesoderm in origin, also forms the innermost fetal membrane, that produces amniotic fluid. This fluid-filled sac initially lies above the trilaminar embryonic disc and with embryoic disc folding this sac is drawn ventrally to enclose (cover) the entire embryo, then fetus. The presence of this membrane led to the description of reptiles, bird, and mammals as amniotes.
- amniotic fluid - The fluid that fills amniotic cavity totally encloses and cushions the embryo. Amniotic fluid enters both the gastrointestinal and respiratory tract following rupture of the buccopharyngeal membrane. The late fetus swallows amniotic fluid.
- atresia - is an abnormal interruption of the tube lumen, the abnormality naming is based upon the anatomical location.
- buccal - (Latin, bucca = cheek) A term used to relate to the mouth (oral cavity).
- bile salts - Liver synthesized compounds derived from cholesterol that function postnatally in the small intestine to solubilize and absorb lipids, vitamins, and proteins. These compounds act as water-soluble amphipathic detergents. liver
- buccopharyngeal membrane - (oral membrane) (Latin, bucca = cheek) A membrane which forms the external upper membrane limit (cranial end) of the early gastrointestinal tract. This membrane develops during gastrulation by ectoderm and endoderm without a middle (intervening) layer of mesoderm. The membrane lies at the floor of the ventral depression (stomodeum) where the oral cavity will open and will breakdown to form the initial "oral opening" of the gastrointestinal tract. The equivilent membrane at the lower end of the gastrointestinal tract is the cloacal membrane.
- celiac artery - (celiac trunk) main blood supply to the foregut, excluding the pharynx, lower respiratory tract, and most of the oesophagus.
- cholangiocytes - epithelial cells that line the intra- and extrahepatic ducts of the biliary tree. These cells modify the hepatocyte-derived bile, and are regulated by hormones, peptides, nucleotides, neurotransmitters, and other molecules. liver
- cloaca - (cloacal cavity) The term describing the common cavity into which the intestinal, genital, and urinary tracts open in vertebrates. Located at the caudal end of the embryo it is located on the surface by the cloacal membrane. In many species this common cavity is later divided into a ventral urogenital region (urogenital sinus) and a dorsal gastrointestinal (rectal) region.
- cloacal membrane - Forms the external lower membrane limit (caudal end) of the early gastrointestinal tract (GIT). This membrane is formed during gastrulation by ectoderm and endoderm without a middle (intervening) layer of mesoderm. The membrane breaks down to form the initial "anal opening" of the gastrointestinal tract.
- coelomic cavity - (coelom) Term used to describe a space. There are extra-embryonic and intra-embryonic coeloms that form during vertebrate development. The single intra-embryonic coelom forms the 3 major body cavities: pleural cavity, pericardial cavity and peritoneal cavity.
- crypt of Lieberkühn - (intestinal gland, intestinal crypt) intestinal villi epithelia extend down into the lamina propria where they form crypts that are the source of epithelial stem cells and immune function.
- duplication - is an abnormal incomplete tube recanalization resulting in parallel lumens, this is really a specialized form of stenosis. (More? Image - small intestine duplication)
- esophageal - (oesophageal)
- foregut - first embryonic division of gastrointestinal tract extending from the oral (buccopharyngeal) membrane and contributing oesophagus, stomach, duodenum (to bile duct opening), liver, biliary apparatus (hepatic ducts, gallbladder, and bile duct), and pancreas. The forgut blood supply is the celiac artery (trunk) excluding the pharynx, lower respiratory tract, and most of the oesophagus.
- galactosemia - Metabolic abnormality where the simple sugar galactose (half of lactose, the sugar in milk) cannot be metabolised. People with galactosemia cannot tolerate any form of milk (human or animal). Detected by the Guthrie test.
- gastric transposition - clinical term for postnatal surgery treatment for esophageal atresia involving esophageal replacement. Typically performed on neonates between day 1 to 4. (More? gastrointestinal abnormalities | PMID 28658159
- gastrointestinal divisions - refers to the 3 embryonic divisions contributing the gastrointestinal tract: foregut, Midgut and hindgut.
- gastrula - (Greek, gastrula = little stomach) A stage of an animal embryo in which the three germ layers (endoderm/mesoderm/ectoderm) have just formed. All of these germ layers have contributions to the gastrointestinal tract.
- gastrulation - The process of differentiation forming a gastrula. Term means literally means "to form a gut" but is more in development, as this process converts the bilaminar embryo (epiblast/hypoblast) into the trilaminar embryo (endoderm/mesoderm/ectoderm) establishing the 3 germ layers that will form all the future tissues of the entire embryo. This process also establishes the the initial body axes. (More? gastrulation)
- Guthrie test - (heel prick) A neonatal blood screening test developed by Dr Robert Guthrie (1916-95) for determining a range of metabolic disorders and infections in the neonate. (More? Guthrie test)
- heterotaxia - (Greek heteros = different; taxis = arrangement) is the right/left transposition of thoracic and/or abdominal organs.
- hindgut - final embryonic division of gastrointestinal tract extending to the cloacal membrane and contributing part of the transverse colon (left half to one third), descending colon, sigmoid colon, rectum, part of anal canal (superior), urinary epithelium (bladder and most urethra). The hindgut blood supply is the inferior mesenteric artery.
- inferior mesenteric artery - main blood supply to the hindgut
- intestine - (bowel) part of the gastrointestinal tract (GIT) lying between the stomach and anus where absorption of nutrients and water occur. This region is further divided anatomically and functionally into the small intestine or bowel (duodenum, jejunum and ileum) and large intestine or bowel (cecum and colon).
- intestinal perforation - gastrointestinal abnormality identified in neonates can be due to necrotizing enterocolitis, Hirschsprung’s disease or meconium ileus.
- intraembryonic coelom - The "horseshoe-shaped" space (cavity) that forms initially in the third week of development in the lateral plate mesoderm that will eventually form the 3 main body cavities: pericardial, pleural, peritoneal. The intraembryonic coelom communicates transiently with the extraembryonic coelom.
- meconium ileus intestine obstruction within the ileum due to abnormal meconium properties.
- mesentery - connects gastrointestinal tract to the posterior body wall and is a double layer of visceral peritoneum.
- mesothelium - The mesoderm derived epithelial covering of coelomic organs and also line their cavities.
- Midgut - middle embryonic division of gastrointestinal tract contributing the small intestine (including duodenum distal bile duct opening), cecum, appendix, ascending colon, and part of the transverse colon (right half to two thirds). The midgut blood supply is the superior mesenteric artery.
- neuralation - The general term used to describe the early formation of the nervous system. It is often used to describe the early events of differentiation of the central ectoderm region to form the neural plate, then neural groove, then neural tube. The nervous system includes the central nervous system (brain and spinal cord) from the neural tube and the peripheral nervous system (peripheral sensory and sympathetic ganglia) from neural crest. In humans, early neuralation begins in week 3 and continues through week 4.
- neural crest - region of cells at the edge of the neural plate that migrates throughout the embryo and contributes to many different tissues. In the gastrointestinal tract it contributes mainly the enteric nervous system within the wall of the gut responsible for peristalsis and secretion.
- peritoneal stomata - the main openings forming the pathways for drainage of intra-peritoneal fluid from the peritoneal cavity into the lymphatic system.
- pharynx - uppermost end of gastrointestinal and respiratory tract, in the embryo beginning at the buccopharyngeal membrane and forms a major arched cavity within the phrayngeal arches.
- recanalization - describes the process of a hollow structure becoming solid, then becoming hollow again. For example, this process occurs during GIT, auditory and renal system development.
- retroperitoneal - (retroperitoneum) is the anatomical space (sometimes a potential space) in the abdominal cavity behind (retro) the peritoneum. Developmentally parts of the GIT become secondarily retroperitoneal (part of duodenum, ascending and descending colon, pancreas)
- somitogenesis The process of segmentation of the paraxial mesoderm within the trilaminar embryo body to form pairs of somites, or balls of mesoderm. A somite is added either side of the notochord (axial mesoderm) to form a somite pair. The segmentation does not occur in the head region, and begins cranially (head end) and extends caudally (tailward) adding a somite pair at regular time intervals. The process is sequential and therefore used to stage the age of many different species embryos based upon the number visible somite pairs. In humans, the first somite pair appears at day 20 and adds caudally at 1 somite pair/4 hours (mouse 1 pair/90 min) until on average 44 pairs eventually form.
- splanchnic mesoderm - Gastrointestinal tract (endoderm) associated mesoderm formed by the separation of the lateral plate mesoderm into two separate components by a cavity, the intraembryonic coelom. Splanchnic mesoderm is the embryonic origin of the gastrointestinal tract connective tissue, smooth muscle, blood vessels and contribute to organ development (pancreas, spleen, liver). The intraembryonic coelom will form the three major body cavities including the space surrounding the gut, the peritoneal cavity. The other half of the lateral plate mesoderm (somatic mesoderm) is associated with the ectoderm of the body wall.
- stomodeum - (stomadeum, stomatodeum) A ventral surface depression on the early embryo head surrounding the buccopharyngeal membrane, which lies at the floor of this depression. This surface depression lies between the maxillary and mandibular components of the first pharyngeal arch.
- stenosis - abnormal a narrowing of the tube lumen, the abnormality naming is based upon the anatomical location.
- superior mesenteric artery - main blood supply to the Midgut.
- viscera - the internal organs in the main cavities of the body, especially those in the abdomen, for example the Template:Intestines.
- visceral peritoneum - covers the external surfaces of the intestinal tract and organs within the peritoneum. The other component (parietal peritoneum) lines the abdominal and pelvic cavity walls.
- yolk sac - An extraembryonic membrane which is endoderm origin and covered with extraembryonic mesoderm. Yolk sac lies outside the embryo connected initially by a yolk stalk to the midgut with which it is continuous with. The endodermal lining is continuous with the endoderm of the gastrointestinal tract. The extra-embryonic mesoderm differentiates to form both blood and blood vessels of the vitelline system. In reptiles and birds, the yolk sac has a function associated with nutrition. In mammals the yolk sac acts as a source of primordial germ cells and blood cells. Note that in early development (week 2) a structure called the "primitive yolk sac" forms from hypoblast, this is an entirely different structure.
- yolk stalk - (vitelline duct, omphalomesenteric duct, Latin, vitellus = yolk of an egg) The endodermal connection between the midgut and the yolk sac. See vitelline duct.
|
|
|
Additional Images
Historic Images
| Historic Disclaimer - information about historic embryology pages
|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers)
|
Frazer JE. and Robbins RH. On the factors concerned in causing rotation of the intestine in man. (1915) J Anat. 50(1): 75-110. PMID 17233053
- Intestine Development
3 vitello-umbilical anastomosis
13 returning proximal limb of loop
15 before and after bowel return
18 colon at different periods
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 24) Embryology Gastrointestinal Tract - Mesentery Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Gastrointestinal_Tract_-_Mesentery_Development
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G