Introduction
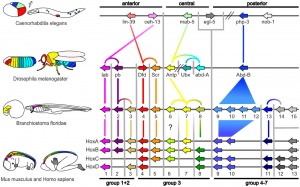
Proposed Hox protein classification
[1]The family of homeobox (Hox) proteins has been a focus of research for over 30 years. In humans, the homeobox gene family contains 300 human homeobox loci, which we divide into 235 probable functional genes and 65 probable pseudogenes.[2]
This family of genes were also the basis of the embryo patterning studies that led to the Nobel Prize in Medicine 1995. We now know that in addition to whole embryo axes patterning, this family of genes has many roles in establishing pattern throughout the embryo in different tissues and organs.
There has recently been a revival of an earlier theory[3] that Hox expression during vertebrate pattern formation is linked to the process of segmentation of paraxial mesoderm during somitogenesis.[4]
This signalling pathway has also been implicated in many developmental abnormalities and diseases.
| Category:Hox
Some Recent Findings
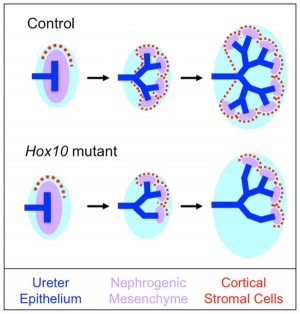
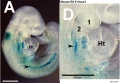
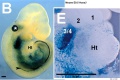
Model Hox10 kidney development
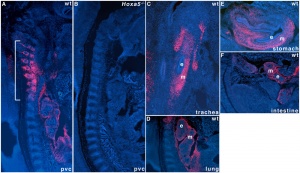
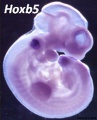
Mouse Hoxa5 expression (E12.5)
[6]
- The Hox Gene egl-5 Acts as a Terminal Selector for VD13 Development via Wnt Signaling[7] "Nervous systems are comprised of diverse cell types that differ functionally and morphologically. During development, extrinsic signals, e.g., growth factors, can activate intrinsic programs, usually orchestrated by networks of transcription factors. Within that network, transcription factors that drive the specification of features specific to a limited number of cells are often referred to as terminal selectors. While we still have an incomplete view of how individual neurons within organisms become specified, reporters limited to a subset of neurons in a nervous system can facilitate the discovery of cell specification programs. We have identified a fluorescent reporter that labels VD13, the most posterior of the 19 inhibitory GABA (γ-amino butyric acid)-ergic motorneurons, and two additional neurons, LUAL and LUAR. Loss of function in multiple Wnt signaling genes resulted in an incompletely penetrant loss of the marker, selectively in VD13, but not the LUAs, even though other aspects of GABAergic specification in VD13 were normal. The posterior Hox gene, egl-5, was necessary for expression of our marker in VD13, and ectopic expression of egl-5 in more anterior GABAergic neurons induced expression of the marker. These results suggest egl-5 is a terminal selector of VD13, subsequent to GABAergic specification." worm neural
- The formation of the thumb requires direct modulation of Gli3 transcription by Hoxa13[8] "In the tetrapod limb, the digits (fingers or toes) are the elements most subject to morphological diversification in response to functional adaptations. However, despite their functional importance, the mechanisms controlling digit morphology remain poorly understood. Here we have focused on understanding the special morphology of the thumb (digit 1), the acquisition of which was an important adaptation of the human hand. To this end, we have studied the limbs of the Hoxa13 mouse mutant that specifically fail to form digit 1. We show that, consistent with the role of Hoxa13 in Hoxd transcriptional regulation, the expression of Hoxd13 in Hoxa13 mutant limbs does not extend into the presumptive digit 1 territory, which is therefore devoid of distal Hox transcripts, a circumstance that can explain its agenesis. The loss of Hoxd13 expression, exclusively in digit 1 territory, correlates with increased Gli3 repressor activity, a Hoxd negative regulator, resulting from increased Gli3 transcription that, in turn, is due to the release from the negative modulation exerted by Hox13 paralogs on Gli3 regulatory sequences. Our results indicate that Hoxa13 acts hierarchically to initiate the formation of digit 1 by reducing Gli3 transcription and by enabling expansion of the 5'Hoxd second expression phase, thereby establishing anterior-posterior asymmetry in the handplate. Our work uncovers a mutual antagonism between Gli3 and Hox13 paralogs that has important implications for Hox and Gli3 gene regulation in the context of development and evolution." limb
- Evolutionary selection and morphological integration in the vertebral column of modern humans[9] "The main objective is to quantify integration, modularity, and response to selection in the presacral vertebral column of modern humans. MATERIALS AND METHODS: Seventeen linear variables on each presacral vertebra were collected in 108 modern humans producing a total of ~39,000 measurements. Then, we studied patterns and magnitudes of integration at regional, vertebral, and intra-vertebral levels. Additionally, we calculated the ability of vertebrae to respond to selection by quantifying differences in evolvability, flexibility, and constraint throughout the spine. The results indicate that caudal vertebrae are more evolvable than those located more cranially in the presacral vertebral column, following an increasing pattern of evolvability from the cervical to the lumbar region. Additionally, the atlas and fifth lumbar vertebra show the lowest values of integration, while central thoracic vertebrae display the highest magnitudes of integration. These results could be related to three main factors: body plan organization expressed by the Hox genes, the strong developmental constraints that determine the number of mammalian vertebrae, and, finally, the functional requirements of an adaptation to bipedal locomotion in the human lineage."
- Hox13 is essential for formation of a sensory organ at the terminal end of the sperm duct in Ciona[10] "Species-specific traits are thought to have been acquired by natural selection. Transcription factors play central roles in the evolution of species-specific traits. Hox genes encode a set of conserved transcription factors essential for establishing the anterior-posterior body axis of animals. Changes in the expression or function of Hox genes can lead to the diversification of animal-body plans. The tunicate ascidian Ciona intestinalis Type A has an orange-colored structure at the sperm duct terminus. This orange-pigmented organ (OPO) is the characteristic that can distinguish this ascidian from other closely related species. The OPO is formed by the accumulation of orange-pigmented cells (OPCs) that are present throughout the adult body. We show that Hox13 is essential for formation of the OPO. Hox13 is expressed in the epithelium of the sperm duct and neurons surrounding the terminal openings for sperm ejection, while OPCs themselves do not express this gene. OPCs are mobile cells that can move through the body vasculature by pseudopodia, suggesting that the OPO is formed by the accumulation of OPCs guided by Hox13-positive cells. Another ascidian species, Ciona savignyi, does not have an OPO. Like Hox13 of C. intestinalis, Hox13 of C. savignyi is expressed at the terminus of its sperm duct; however, its expression domain is limited to the circular area around the openings. The genetic changes responsible for the acquisition or loss of OPO are likely to occur in the expression pattern of Hox13."
- Analysis of a limb-specific regulatory element in the promoter of the link protein gene[11] "Link protein is encoded by the Hapln1 gene and is a prototypical protein found in the cartilage matrix. It acts as an important component of the endochondral skeleton during early development. To study its transcriptional regulation, promoter fragments derived from the link protein gene were coupled to the β-galactosidase reporter and used to study in vivo transgene expression in mice. In day 15.5 mouse embryos, a link promoter fragment spanning -1020 to +40 nucleotides demonstrated highly specific β-galactosidase staining of skeletal structures, including the appendicular and axial cartilaginous tissues. Two shorter promoter fragments, spanning -690 to +40 and -315 to +40 nucleotides, demonstrated limb- and genitalia-specific expression resembling that of homeodomain-regulated tissues. Bioinformatic analysis revealed a highly conserved, Hox-like binding site (HLBS) at approximately -220 bp of the promoter, shared by both constructs, which contained the Hox-core consensus sequence TAATTA. Electromobility shift assays demonstrated binding of Hox-B4 recombinant protein to the HLBS, which was eliminated with nucleotide substitutions within the core-binding element. Co-transfection analysis of the HLBS demonstrated a 22-fold transcriptional activation by HoxA9 expression, which was ablated with a substitution within the core HLBS element. Together these findings establish promoter regions within the link protein gene that are important for in vivo expression and identify the potential role of homeodomain-containing proteins in controlling cartilage and limb gene expression."
- Hox genes limit germ cell formation in the short germ insect Gryllus bimaculatus[12] "Hox genes are conserved transcription factor-encoding genes that specify the identity of body regions in bilaterally symmetrical animals. In the cricket Gryllus bimaculatus, a member of the hemimetabolous insect group Orthoptera, the induction of a subset of mesodermal cells to form the primordial germ cells (PGCs) is restricted to the second through the fourth abdominal segments (A2 to A4). In numerous insect species, the Hox genes Sex-combs reduced (Scr), Antennapedia (Antp), Ultrabithorax (Ubx), and abdominal-A (abd-A) jointly regulate the identities of middle and posterior body segments, suggesting that these genes may restrict PGC formation to specific abdominal segments in G. bimaculatus Here we show that reducing transcript levels of some or all of these Hox genes results in supernumerary and/or ectopic PGCs, either individually or in segment-specific combinations, suggesting that the role of these Hox genes is to limit PGC development with respect to their number, segmental location, or both. These data provide evidence of a role for this ancient group of genes in PGC development."
|
| More recent papers
|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
- This search now requires a manual link as the original PubMed extension has been disabled.
- The displayed list of references do not reflect any editorial selection of material based on content or relevance.
- References also appear on this list based upon the date of the actual page viewing.
References listed on the rest of the content page and the associated discussion page (listed under the publication year sub-headings) do include some editorial selection based upon both relevance and availability.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References
Search term: Hox | antennapedia
|
| Older papers
|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
- Hox genes in the pharyngeal region: how Hoxa3 controls early embryonic development of the pharyngeal organs[13] "The pharyngeal organs, namely the thyroid, thymus, parathyroids, and ultimobranchial bodies, derive from the pharyngeal endoderm during embryonic development. The pharyngeal region is a segmented structure comprised of a series of reiterated structures: the pharyngeal arches on the exterior surface, the pharyngeal pouches on the interior, and a mesenchymal core. It is well known that Hox genes control spatial identity along the anterior-posterior axis of the developing vertebrate embryo, and nowhere is this is more evident than in the pharyngeal region. Each of the distinct segmented regions has a unique pattern of Hox expression, which conveys crucial positional information to the cells and tissues within it. In the context of pharyngeal organ development, molecular data suggest that HOXA3 is responsible for specifying organ identity within the third pharyngeal pouch, and in its absence, thymus and parathyroid organogenesis fails to proceed normally"
- A genome-wide assessment of the ancestral neural crest gene regulatory network[14] "The neural crest (NC) is an embryonic cell population that contributes to key vertebrate-specific features including the craniofacial skeleton and peripheral nervous system. Here we examine the transcriptional and epigenomic profiles of NC cells in the sea lamprey, in order to gain insight into the ancestral state of the NC gene regulatory network (GRN). Transcriptome analyses identify clusters of co-regulated genes during NC specification and migration that show high conservation across vertebrates but also identify transcription factors (TFs) and cell-adhesion molecules not previously implicated in NC migration. ATAC-seq analysis uncovers an ensemble of cis-regulatory elements, including enhancers of Tfap2B, SoxE1 and Hox-α2 validated in the embryo. Cross-species deployment of lamprey elements identifies the deep conservation of lamprey SoxE1 enhancer activity, mediating homologous expression in jawed vertebrates. Our data provide insight into the core GRN elements conserved to the base of the vertebrates and expose others that are unique to lampreys."
- A single three-dimensional chromatin compartment in amphioxus indicates a stepwise evolution of vertebrate Hox bimodal regulation[15] "The HoxA and HoxD gene clusters of jawed vertebrates are organized into bipartite three-dimensional chromatin structures that separate long-range regulatory inputs coming from the anterior and posterior Hox-neighboring regions. This architecture is instrumental in allowing vertebrate Hox genes to pattern disparate parts of the body, including limbs. ...We find that, in contrast to the architecture in vertebrates, the amphioxus Hox cluster is organized into a single chromatin interaction domain that includes long-range contacts mostly from the anterior side, bringing distant cis-regulatory elements into contact with Hox genes."
- Homeobox Genes of Caenorhabditis elegans and Spatio-Temporal Expression[16] "We show that, out of 103 homeobox genes, 70 are co-orthologous to human homeobox genes. 14 are highly divergent, lacking an obvious ortholog even in other Caenorhabditis species. One of these homeobox genes encodes 12 homeodomains, while three other highly divergent homeobox genes encode a novel type of double homeodomain, termed HOCHOB. To understand how transcription factors regulate cell fate during development, precise spatio-temporal expression data need to be obtained. Using a new imaging framework that we developed, Endrov, we have generated spatio-temporal expression profiles during embryogenesis of over 60 homeobox genes, as well as a number of other developmental control genes using GFP reporters." Worm Development
- A Hox regulatory network of hindbrain segmentation is conserved to the base of vertebrates[17] "Here, using a novel tool that allows cross-species comparisons of regulatory elements between jawed and jawless vertebrates, we report deep conservation of both upstream regulators and segmental activity of enhancer elements across these distant species. Regulatory regions from diverse gnathostomes drive segmental reporter expression in the lamprey hindbrain and require the same transcriptional inputs (for example, Kreisler (also known as Mafba), Krox20 (also known as Egr2a)) in both lamprey and zebrafish. We find that lamprey hox genes display dynamic segmentally restricted domains of expression; we also isolated a conserved exonic hox2 enhancer from lamprey that drives segmental expression in rhombomeres 2 and 4. Our results show that coupling of Hox gene expression to segmentation of the hindbrain is an ancient trait with origin at the base of vertebrates that probably led to the formation of rhombomeric compartments with an underlying Hox code."
- Hoxb1b controls oriented cell division, cell shape and microtubule dynamics in neural tube morphogenesis[18] "Hox genes are classically ascribed to function in patterning the anterior-posterior axis of bilaterian animals; however, their role in directing molecular mechanisms underlying morphogenesis at the cellular level remains largely unstudied. ...Hoxb1b regulates mitotic spindle rotation during the oriented neural keel symmetric mitoses that are required for normal neural tube lumen formation in the zebrafish."
- Evolution of anterior Hox regulatory elements among chordates[19] "The Hox family of transcription factors has a fundamental role in segmentation pathways and axial patterning of embryonic development and their clustered organization is linked with the regulatory mechanisms governing their coordinated expression along embryonic axes. Among chordates, of particular interest are the Hox paralogous genes in groups 1-4 since their expression is coupled to the control of regional identity in the anterior nervous system, where the highest structural diversity is observed. ...Together, our results indicate that during chordate evolution, cis-elements dependent upon Hox/Pbx regulatory complexes, are responsible for key aspects of segmental Hox expression in neural tissue and appeared with urochordates after cephalochordate divergence."
- Hox10 Genes Function in Kidney Development in the Differentiation and Integration of the Cortical Stroma [20] "Consistent with loss of cortical stromal cell function, Hox10 mutant kidneys display reduced and aberrant ureter branching, decreased nephrogenesis. These data therefore provide critical novel insights into the cellular and genetic mechanisms governing cortical cell development during kidney organogenesis. These results, combined with previous evidence demonstrating that Hox11 genes are necessary for patterning the metanephric mesenchyme, support a model whereby distinct populations in the nephrogenic cord are regulated by unique Hox codes, and that differential Hox function along the AP axis of the nephrogenic cord is critical for the differentiation and integration of these cell types during kidney organogenesis."
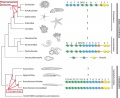
- Proposed Hox protein classification[1] "Our classification scheme offers a higher-resolution classification that is in accordance with phylogenetic as well as experimental data and, thereby, provides a novel basis for experiments, such as comparative and functional analyses of Hox-proteins."
- Homeobox A7 up-regulation of epidermal growth factor receptor expression in human granulosa cells[21] "Our present study reveals a novel mechanistic role for HOXA7 in modulating granulosa cell proliferation via the regulation of EGFR. This finding contributes to the knowledge of the pro-proliferation effect of HOXA7 in granulosa cell growth and differentiation."
- Hoxa5 transcriptional complexity in the mouse embryo[6] "Our observation that the Hoxa5 larger transcripts possess a developmentally-regulated expression combined to the increasing sum of data on the role of long noncoding RNAs in transcriptional regulation suggest that the Hoxa5 larger transcripts may participate in the control of Hox gene expression."
|
Classification
Commonly recognized HOX groupings include: ANTP, PRD, LIM, POU, HNF, SINE, TALE, CUT, PROS and ZF.
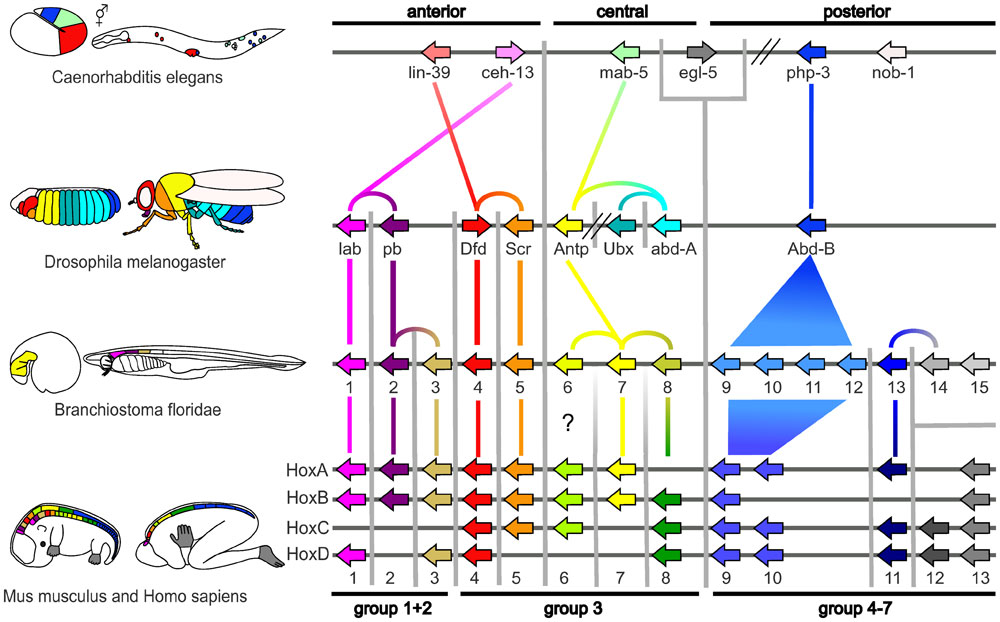
|
| Proposed Hox protein classification[1]
|
- PAX2, PAX5 and PAX8 have a partial homeobox
- PAX1 and PAX9 lack a homeobox.
Human Hox

Chromosomal Distribution of Human Homeobox Genes[2]
Functions
Developmental patterning signal.
Neural
Segmentation
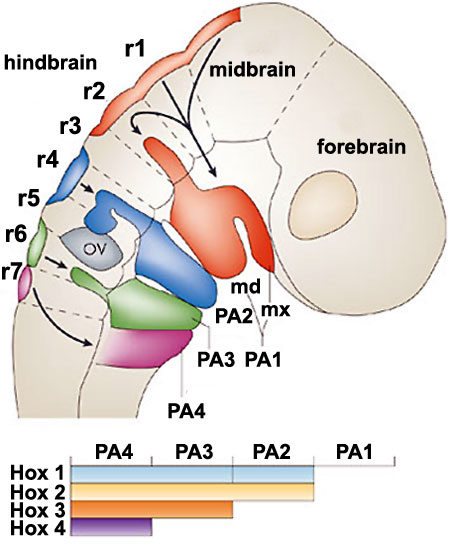
|
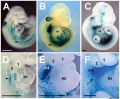
Hindbrain neural crest migration and Hox expression pattern[22]
A schematic diagram of a chick head at embryonic day two (Hamburger Hamilton Stages), showing pathways of neural crest migration in the chick and mouse embryo and patterns of Hox gene expression in the pharyngeal arches. Hox genes are expressed in neural crest cells, which emigrate predominantly from even-numbered rhombomeres into the pharyngeal (branchial) arches generating skeletal tissues and cranial ganglia.
Note that the first pharyngeal arch is free of Hox expression.
Legend
- PA - pharyngeal arch
- Md - mandibular part of pharyngeal arch 1
- Mx - maxillary part of pharyngeal arch 1
- OV - otic vesicle
- r - rhombomere
Adapted by permission from Macmillan Publishers Ltd: Nature Reviews Neuroscience[22], copyright (2007)
|
- Links: Neural System Development | Neural Crest Development
Phrenic Motor Neurons
Hox5 (Hoxa5 and Hoxc5) required for phrenic motor column (PMC) development that form the respiratory motor neurons driving the diaphragm for respiration.[23]
- Links: Respiratory System Development
Axial Skeleton
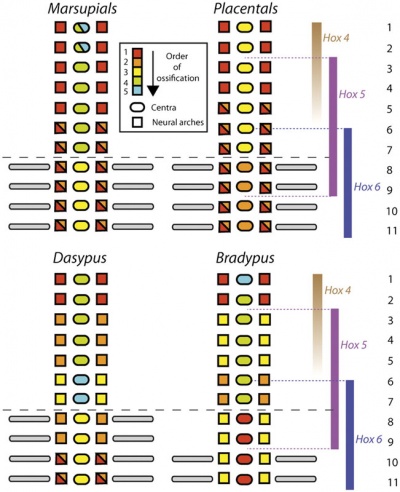
Vertebral element ossification between species.[24]
|
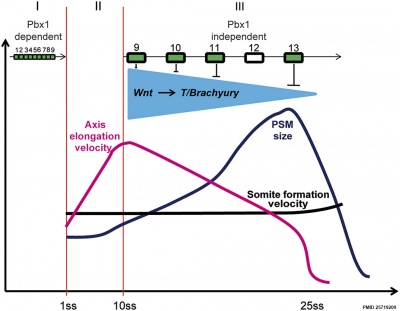
Chicken model of Hox in paraxial mesoderm precursors in the epiblast/tail-bud during axis elongation[25]
|
- Links: Somitogenesis | Axial Skeleton Development
Limb
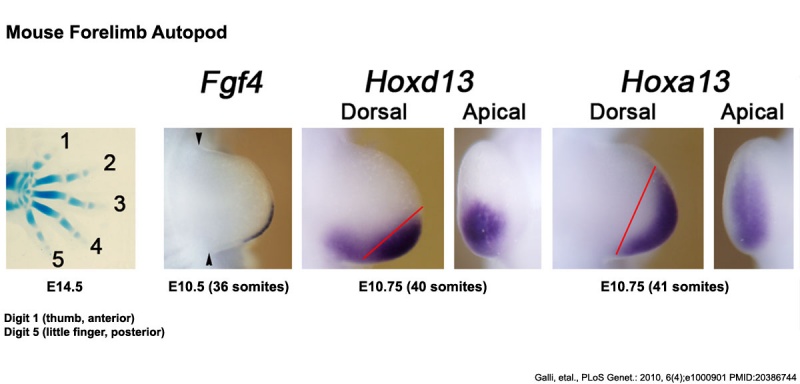
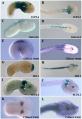
Mouse Limb Patterning Fgf and Hox Expression[26]
Fgf and Hox expression in E10.5 to 10.75 wild-type embryonic forelimb autopod, compared to future E14.5 digit arrangement.
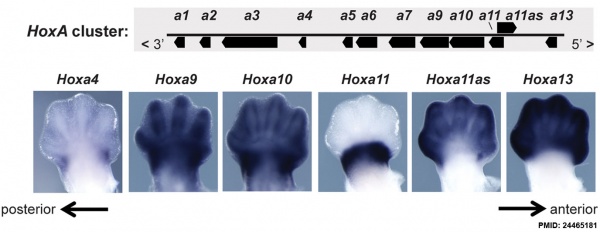
Expression of Hoxa4, Hoxa9, Hoxa10, Hoxa11, Hoxa11 antisense (Hoxa11as), and Hoxa13 in E12.5 limb buds.[27]
- Links: Limb Development
Other
Signaling Pathway
Otx
Otx is a txanscription factor essential for the normal development of the brain, cerebellum, pineal gland, and eye.
OTX is a homeobox family gene related to a gene expressed in the developing Drosophila head termed 'orthodenticle.' OTX transcription factors bind with high affinity to TAATCC/T elements on DNA.
- OMIM: OTX1 | OTX2 | OTX3
- Links: Pineal | Vision | PubMed - Otx
Additional Images
Hox deuterostomes phylogenetic tree PMID 23819519
Mouse forelimb bud Fgf4-Hoxd13-Hoxa13
Mouse spleen Hox11 expression
References
- ↑ 1.0 1.1 1.2 Hueber SD, Weiller GF, Djordjevic MA & Frickey T. (2010). Improving Hox protein classification across the major model organisms. PLoS ONE , 5, e10820. PMID: 20520839 DOI.
- ↑ 2.0 2.1 2.2 2.3 Holland PW, Booth HA & Bruford EA. (2007). Classification and nomenclature of all human homeobox genes. BMC Biol. , 5, 47. PMID: 17963489 DOI.
- ↑ Meinhardt H. Models For Biological Pattern Formation. Academic Press; 1982.
- ↑ Gu S, Gu W, Shou J, Xiong J, Liu X, Sun B, Yang D & Xie R. (2017). The Molecular Feature of HOX Gene Family in the Intramedullary Spinal Tumors. Spine , 42, 291-297. PMID: 25785959 DOI.
- ↑ 5.0 5.1 Turner FR & Mahowald AP. (1979). Scanning electron microscopy of Drosophila melanogaster embryogenesis. III. Formation of the head and caudal segments. Dev. Biol. , 68, 96-109. PMID: 108157
- ↑ 6.0 6.1 Coulombe Y, Lemieux M, Moreau J, Aubin J, Joksimovic M, Bérubé-Simard FA, Tabariès S, Boucherat O, Guillou F, Larochelle C, Tuggle CK & Jeannotte L. (2010). Multiple promoters and alternative splicing: Hoxa5 transcriptional complexity in the mouse embryo. PLoS ONE , 5, e10600. PMID: 20485555 DOI.
- ↑ Kurland M, O'Meara B, Tucker DK & Ackley BD. (2020). The Hox Gene egl-5 Acts as a Terminal Selector for VD13 Development via Wnt Signaling. J Dev Biol , 8, . PMID: 32138237 DOI.
- ↑ Bastida MF, Pérez-Gómez R, Trofka A, Zhu J, Rada-Iglesias A, Sheth R, Stadler HS, Mackem S & Ros MA. (2020). The formation of the thumb requires direct modulation of Gli3 transcription by Hoxa13. Proc. Natl. Acad. Sci. U.S.A. , 117, 1090-1096. PMID: 31896583 DOI.
- ↑ Arlegi M, Veschambre-Couture C & Gómez-Olivencia A. (2019). Evolutionary selection and morphological integration in the vertebral column of modern humans. Am. J. Phys. Anthropol. , , . PMID: 31675109 DOI.
- ↑ Tajima Y, Hozumi A, Yoshida K, Treen N, Sakuma T, Yamamoto T & Sasakura Y. (2019). Hox13 is essential for formation of a sensory organ at the terminal end of the sperm duct in Ciona. Dev. Biol. , , . PMID: 31682808 DOI.
- ↑ Rhodes CS, Matsunobu T & Yamada Y. (2019). Analysis of a limb-specific regulatory element in the promoter of the link protein gene. Biochem. Biophys. Res. Commun. , 518, 672-677. PMID: 31470976 DOI.
- ↑ Barnett AA, Nakamura T & Extavour CG. (2019). Hox genes limit germ cell formation in the short germ insect Gryllus bimaculatus. Proc. Natl. Acad. Sci. U.S.A. , 116, 16430-16435. PMID: 31346080 DOI.
- ↑ Gordon J. (2018). Hox genes in the pharyngeal region: how Hoxa3 controls early embryonic development of the pharyngeal organs. Int. J. Dev. Biol. , 62, 775-783. PMID: 30604847 DOI.
- ↑ Hockman D, Chong-Morrison V, Green SA, Gavriouchkina D, Candido-Ferreira I, Ling ITC, Williams RM, Amemiya CT, Smith JJ, Bronner ME & Sauka-Spengler T. (2019). A genome-wide assessment of the ancestral neural crest gene regulatory network. Nat Commun , 10, 4689. PMID: 31619682 DOI.
- ↑ Acemel RD, Tena JJ, Irastorza-Azcarate I, Marlétaz F, Gómez-Marín C, de la Calle-Mustienes E, Bertrand S, Diaz SG, Aldea D, Aury JM, Mangenot S, Holland PW, Devos DP, Maeso I, Escrivá H & Gómez-Skarmeta JL. (2016). A single three-dimensional chromatin compartment in amphioxus indicates a stepwise evolution of vertebrate Hox bimodal regulation. Nat. Genet. , 48, 336-41. PMID: 26829752 DOI.
- ↑ Hench J, Henriksson J, Abou-Zied AM, Lüppert M, Dethlefsen J, Mukherjee K, Tong YG, Tang L, Gangishetti U, Baillie DL & Bürglin TR. (2015). The Homeobox Genes of Caenorhabditis elegans and Insights into Their Spatio-Temporal Expression Dynamics during Embryogenesis. PLoS ONE , 10, e0126947. PMID: 26024448 DOI.
- ↑ Parker HJ, Bronner ME & Krumlauf R. (2014). A Hox regulatory network of hindbrain segmentation is conserved to the base of vertebrates. Nature , 514, 490-3. PMID: 25219855 DOI.
- ↑ Zigman M, Laumann-Lipp N, Titus T, Postlethwait J & Moens CB. (2014). Hoxb1b controls oriented cell division, cell shape and microtubule dynamics in neural tube morphogenesis. Development , 141, 639-49. PMID: 24449840 DOI.
- ↑ Natale A, Sims C, Chiusano ML, Amoroso A, D'Aniello E, Fucci L, Krumlauf R, Branno M & Locascio A. (2011). Evolution of anterior Hox regulatory elements among chordates. BMC Evol. Biol. , 11, 330. PMID: 22085760 DOI.
- ↑ Yallowitz AR, Hrycaj SM, Short KM, Smyth IM & Wellik DM. (2011). Hox10 genes function in kidney development in the differentiation and integration of the cortical stroma. PLoS ONE , 6, e23410. PMID: 21858105 DOI.
- ↑ Zhang Y, Huang Q, Cheng JC, Nishi Y, Yanase T, Huang HF & Leung PC. (2010). Homeobox A7 increases cell proliferation by up-regulation of epidermal growth factor receptor expression in human granulosa cells. Reprod. Biol. Endocrinol. , 8, 61. PMID: 20540809 DOI.
- ↑ 22.0 22.1 Guthrie S. (2007). Patterning and axon guidance of cranial motor neurons. Nat. Rev. Neurosci. , 8, 859-71. PMID: 17948031 DOI.
- ↑ Philippidou P, Walsh CM, Aubin J, Jeannotte L & Dasen JS. (2012). Sustained Hox5 gene activity is required for respiratory motor neuron development. Nat. Neurosci. , 15, 1636-44. PMID: 23103965 DOI.
- ↑ Hautier L, Weisbecker V, Sánchez-Villagra MR, Goswami A & Asher RJ. (2010). Skeletal development in sloths and the evolution of mammalian vertebral patterning. Proc. Natl. Acad. Sci. U.S.A. , 107, 18903-8. PMID: 20956304 DOI.
- ↑ Denans N, Iimura T & Pourquié O. (2015). Hox genes control vertebrate body elongation by collinear Wnt repression. Elife , 4, . PMID: 25719209 DOI.
- ↑ Galli A, Robay D, Osterwalder M, Bao X, Bénazet JD, Tariq M, Paro R, Mackem S & Zeller R. (2010). Distinct roles of Hand2 in initiating polarity and posterior Shh expression during the onset of mouse limb bud development. PLoS Genet. , 6, e1000901. PMID: 20386744 DOI.
- ↑ Woltering JM, Noordermeer D, Leleu M & Duboule D. (2014). Conservation and divergence of regulatory strategies at Hox Loci and the origin of tetrapod digits. PLoS Biol. , 12, e1001773. PMID: 24465181 DOI.
Reviews
Mallo M, Wellik DM & Deschamps J. (2010). Hox genes and regional patterning of the vertebrate body plan. Dev. Biol. , 344, 7-15. PMID: 20435029 DOI.
Narita Y & Rijli FM. (2009). Hox genes in neural patterning and circuit formation in the mouse hindbrain. Curr. Top. Dev. Biol. , 88, 139-67. PMID: 19651304 DOI.
Deschamps J & van Nes J. (2005). Developmental regulation of the Hox genes during axial morphogenesis in the mouse. Development , 132, 2931-42. PMID: 15944185 DOI.
Schilling TF & Knight RD. (2001). Origins of anteroposterior patterning and Hox gene regulation during chordate evolution. Philos. Trans. R. Soc. Lond., B, Biol. Sci. , 356, 1599-613. PMID: 11604126 DOI.
Articles
Brauchle M, Bilican A, Eyer C, Bailly X, Martínez P, Ladurner P, Bruggmann R & Sprecher SG. (2018). Xenacoelomorpha Survey Reveals That All 11 Animal Homeobox Gene Classes Were Present in the First Bilaterians. Genome Biol Evol , 10, 2205-2217. PMID: 30102357 DOI.
Rodrigues MF, Esteves CM, Xavier FC & Nunes FD. (2016). Methylation status of homeobox genes in common human cancers. Genomics , 108, 185-193. PMID: 27826049 DOI.
Dunwell TL & Holland PW. (2016). Diversity of human and mouse homeobox gene expression in development and adult tissues. BMC Dev. Biol. , 16, 40. PMID: 27809766 DOI.
Search Pubmed
Search Bookshelf hox
Search Pubmed Now: Hox Homeobox
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 28) Embryology Developmental Signals - Homeobox. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Developmental_Signals_-_Homeobox
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G