Cardiovascular System - Double Outlet Right Ventricle
| Embryology - 25 Mar 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
LA85.2 Double Outlet Right Ventricle
| ICD-11 |
|---|
LA85.2 Double outlet right ventricle
|
Introduction
- 1-1.5% of Congenital Heart Disease
- Both large arteries arise wholly or mainly from the right ventricle.
- Arrangement of the atrioventricular valves and the ventriculoarterial connections are variable.
- Clinical manifestations variable.
If the pulmonary trunk retains its location above the morphologically right ventricle, then the end result will be yet another variant of double outlet right ventricle, specifically with a subpulmonary interventricular communication. This variation is known as the Taussig-Bing malformation.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Double Outlet Right Ventricle | Taussig-Bing malformation |
Anatomy
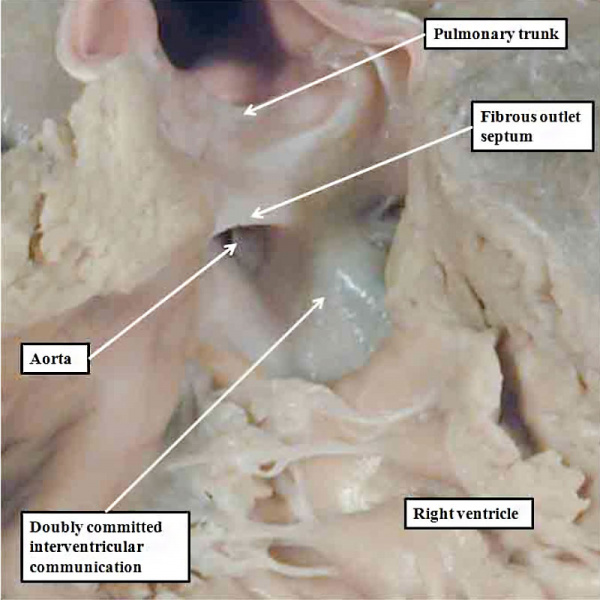
A human heart with double outlet from the right ventricle, but with a doubly committed interventricular communication. The proximal outflow cushions have fused, but have failed to muscularise. Because of this, the outlet septum, derived from the fused cushions, is fibrous and hypoplastic.
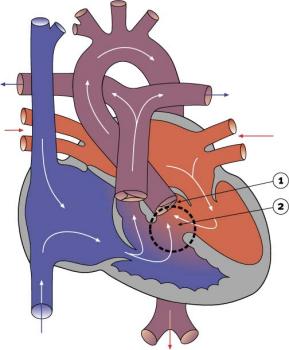
Double Outlet from Right Ventricle
Genetics
HAND1
Heart-And Neural Crest Derivatives-Expressed 1 (HAND1) gene located at 5q32, is a 215-amino acid protein transcription factor that contains a bHLH region and several potential phosphorylation sites. The mouse and human sequences share 93% overall sequence identity.
- Links: OMIM - HAND1
References
- ↑ Chaudhry B, Henderson D & Anderson R. (2019). Double-outlet right ventricle is not hypoplastic left heart syndrome. Nat. Genet. , 51, 198. PMID: 30617254 DOI.
- ↑ Liu X, Yagi H, Saeed S, Bais AS, Gabriel GC, Chen Z, Peterson KA, Li Y, Schwartz MC, Reynolds WT, Saydmohammed M, Gibbs B, Wu Y, Devine W, Chatterjee B, Klena NT, Kostka D, de Mesy Bentley KL, Ganapathiraju MK, Dexheimer P, Leatherbury L, Khalifa O, Bhagat A, Zahid M, Pu W, Watkins S, Grossfeld P, Murray SA, Porter GA, Tsang M, Martin LJ, Benson DW, Aronow BJ & Lo CW. (2017). The complex genetics of hypoplastic left heart syndrome. Nat. Genet. , 49, 1152-1159. PMID: 28530678 DOI.
- ↑ Priya S, Nagpal P, Sharma A, Pandey NN & Jagia P. (2019). Imaging Spectrum of Double-Outlet Right Ventricle on Multislice Computed Tomography. J Thorac Imaging , , . PMID: 30801451 DOI.
- ↑ Li L, Wang J, Liu XY, Liu H, Shi HY, Yang XX, Li N, Li YJ, Huang RT, Xue S, Qiu XB & Yang YQ. (2017). HAND1 loss-of-function mutation contributes to congenital double outlet right ventricle. Int. J. Mol. Med. , 39, 711-718. PMID: 28112363 DOI.
Reviews
Fraser CD. (2017). The Neonatal Arterial Switch Operation: Technical Pearls. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu , 20, 38-42. PMID: 28007063 DOI.
Spicer DE, Hsu HH, Co-Vu J, Anderson RH & Fricker FJ. (2014). Ventricular septal defect. Orphanet J Rare Dis , 9, 144. PMID: 25523232 DOI.
Articles
Aherrera JA, Magno JD, Uy CC, Abrahan LL, Maria HF, Buitizon RR & Jara RD. (2015). The Triply Twisted Heart: Cyanosis in an Adult With Situs Inversus, Levocardia, Double Outlet Right Ventricle, and Malposition of the Great Arteries. Cardiol Res , 6, 362-366. PMID: 28197259 DOI.
Search Pubmed
Search Pubmed: Search PubMed
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.