BGDA Lecture - Development of the Embryo/Fetus 1
| Embryology - 21 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
In medicine foundations you were given a broad overview of human development. Now in BGDA we will be working through the human development process in more detail, focussing on key events.
2018 Lecture PDF
- Begin by reviewing the recent Foundations Lecture and Practical.
- This BGDA lecture covers conceptus development from fertilization to implantation to trilaminar embryo formation.
- Note that fertilization and week 1 concepts have already been covered in an earlier BGDA lecture.
- The lecture will also introduce early fetal membranes and placentation.
1 Minute Embryology | UNSW theBox
2019 Lecture PDF
| Lecture Archive |
|---|
| 2018 PDF
2017 PDF | 2016 |
2015 | 2015 PDF | 2014 | 2014 PDF | 2013 |
|
BGDA Practical Classes
| Practical 3 - Fertilization to Implantation | Practical 6 - Implantation to 8 Weeks | Practical 12 - Fetal Period |
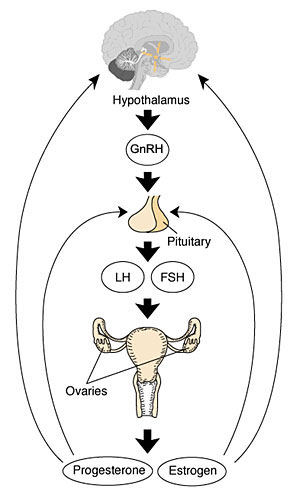
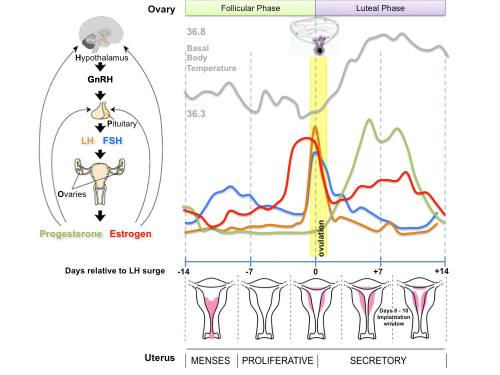
Human Reproductive Cycle
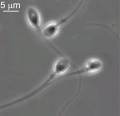
- meiosis in gonad produces haploid gametes (oocyte, egg) and (spermatozoa, sperm)
| Female | Male |
|---|---|
|
|
Gametogenesis
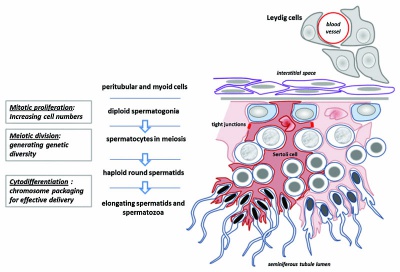
Male
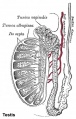
The testes have two functions.
- produce the male gametes or spermatozoa
- produce male sexual hormone, testosterone (internal and external genitalia, sex characteristics)
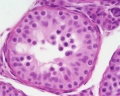
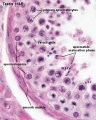
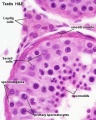
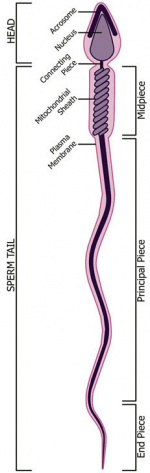
Human spermatozoa take about 48 days from entering meiosis until morphologically mature spermatozoa.
sertoli cells (support cells) Interstitial cells or Leydig cells (produce hormone testosterone) |
|
| Spermatozoa Development (expand to see terms) | ||
|---|---|---|
|
Note there are additional glossaries associated with genital, spermatozoa, oocyte and renal.
See also: Spermatozoa Terms collapse table
|
Female
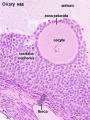
The ovary has two main functions.
- produce the female gametes or oocytes
- produce female hormones, estrogen and progesterone (secondary sex characteristics, menstrual cycle)
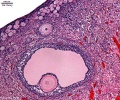
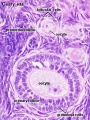
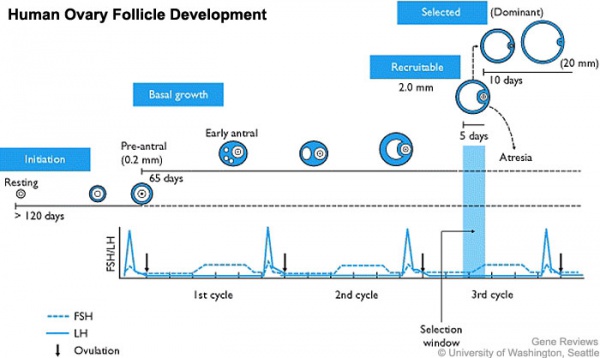
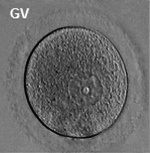
In an adult human female the development of a primordial follicle containing an oocyte to a preovulatory follicle takes in excess of 120 days.
Human ovary follicle development
- Ovarian Follicle Stages: primordial follicle - primary follicle - secondary follicle - preovulatory follicle
Follicle cells (support cells) Theca cells (produce hormone)
| Oocyte Development (expand to see terms) | ||
|---|---|---|
|
Note there are additional specific term glossaries available listed at bottom of this table.
|
- Links: spermatozoa | oocyte | MBoC - Figure 20-18. Influence of Sry on gonad development | Endocrinology - Comparative anatomy of male and female reproductive tracts
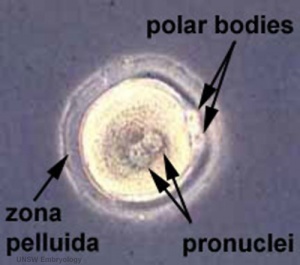
Fertilization
- oogenesis - 1 gamete produced/meiosis + 3 polar bodies, meiosis is slow, 1 egg produced and released at ovulation
- spermatogenesis - 4 gametes produced/meiosis, meiosis is fast, 200-600 million sperm released at ejaculation
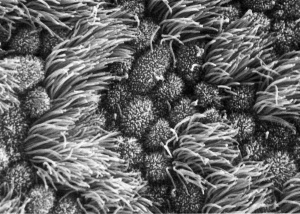
Fertilization Site
- Fertilization usually occurs in first 1/3 of uterine tube (oviduct, Fallopian tube)
- Fertilization can also occur outside uterine tube associated with Assisted Reproductive Technologies (ART, IVF, GIFT, ZIFT...) and ectopic pregnancy
- The majority of fertilized eggs do not go on to form an embryo
Fertilization - Spermatozoa
- Capacitation - alteration of the spermatozoa metabolism and surface proteins
- Sperm Binding - zona pellucida protein ZP3 acts as receptor for sperm
- Acrosome Reaction - exocytosis of acrosome contents (Calcium mediated) MBoC - Figure 20-31. The acrosome reaction that occurs when a mammalian sperm fertilizes an egg
- enzymes to digest the zona pellucida
- exposes sperm surface proteins to bind ZP2
- Membrane Fusion - between sperm and egg, allows sperm nuclei passage into egg cytoplasm
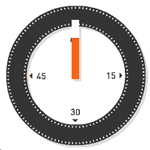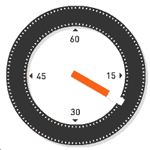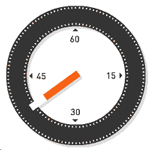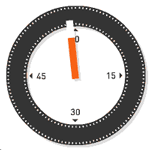
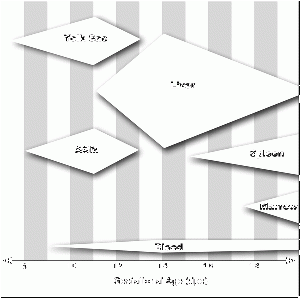
| Approximate Timing of Early Human Events (in vitro) | ||||
|---|---|---|---|---|

|

|

| ||
| 20 min - components | 28 min - spermatozoa penetrates zone pellucida | 31 min - spermatozoa penetrates oocyte - fertilization | ||
| See also clock in lower righthand corner for the approximate timing of events. | ||||
| Reference: PMID 22695746 J Assist Reprod Genet. | ||||
Fertilization - Oocyte
- Membrane Depolarization - caused by sperm membrane fusion, primary block to polyspermy
- Cortical Reaction - IP3 pathway elevates intracellular Calcium, exocytosis of cortical granules MBoC - Figure 20-32. How the cortical reaction in a mouse egg is thought to prevent additional sperm from entering the egg
- enzyme alters ZP3 so it will no longer bind sperm plasma membrane
- Meiosis 2 - completion of 2nd meiotic division
- forms second polar body (a third polar body may be formed by meiotic division of the first polar body)
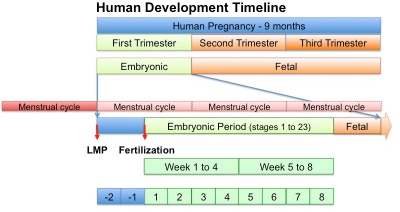
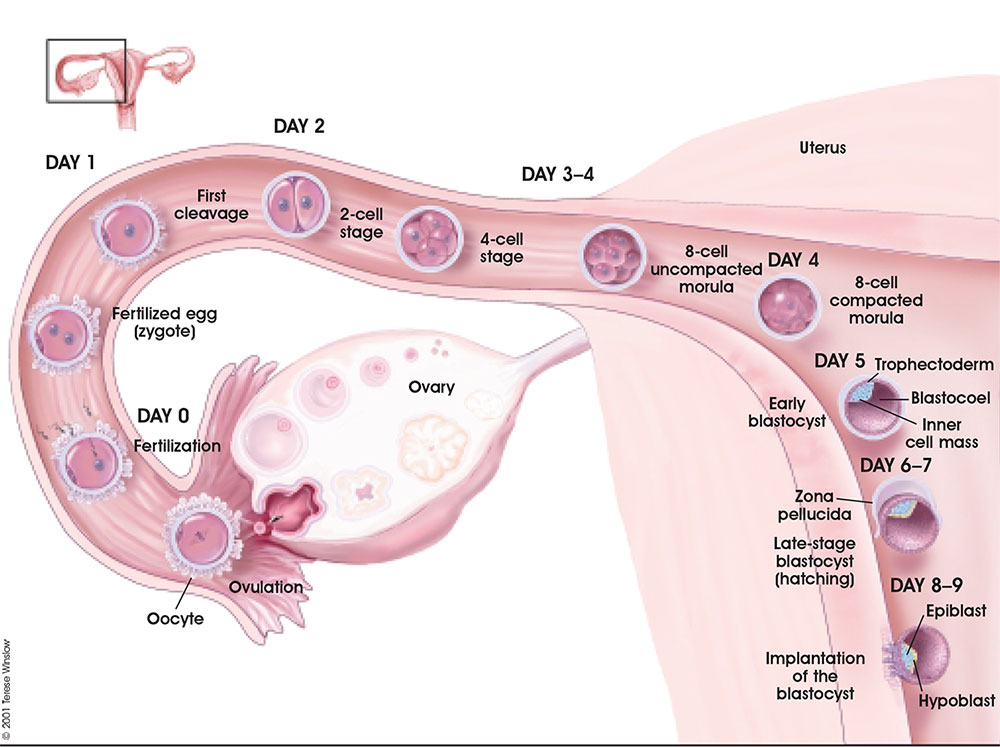
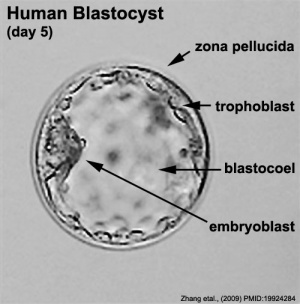
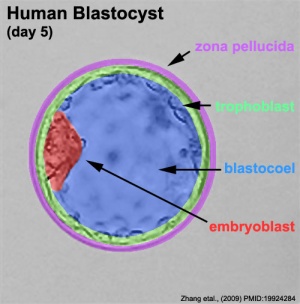
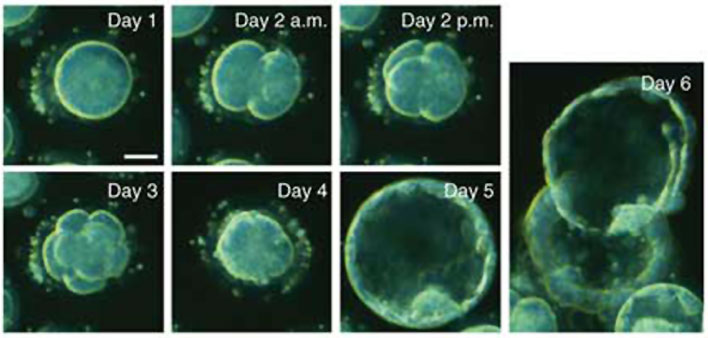
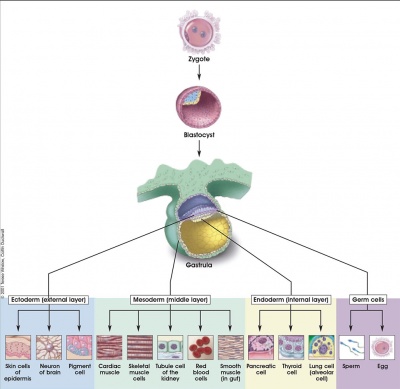
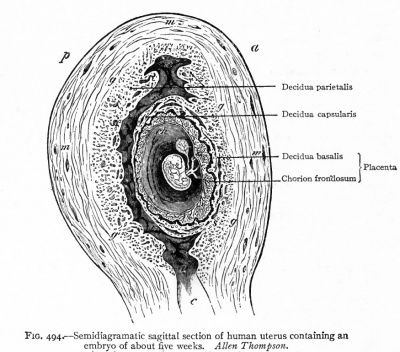
Week 1 and 2
Week 2 Implantation
- Bilaminar embryo - epiblast and hypoblast
- Bilaminar trophoblast - Cytotrophoblast and Syncytiotrophoblast
| Uterine Implantation | Ectopic Implantation |
|---|---|
|
|
|
|
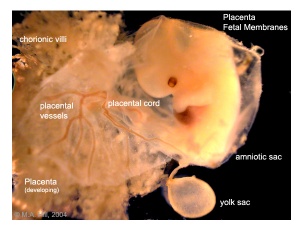
Early Placenta
|
|
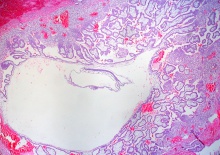
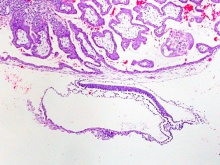
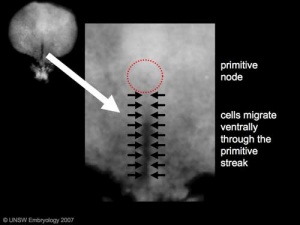
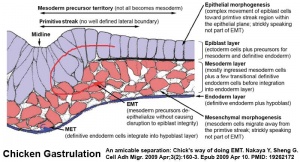
Week 3 Gastrulation
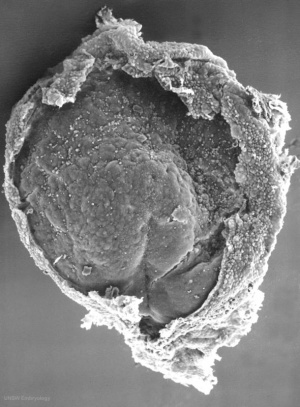
|
|
|
| Embryonic disc | Primitive Streak | Gastrulation |
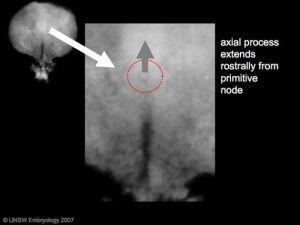
- Primitive node - region in the middle of the early embryonic disc epiblast from which the primitive streak extends caudally (tail)
- nodal cilia establish the embryo left/right axis
- axial process extends from the nodal epiblast
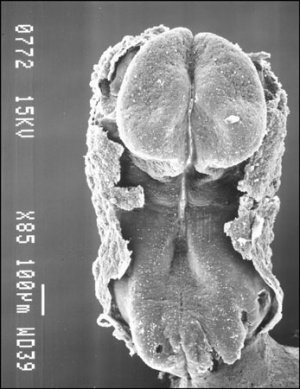
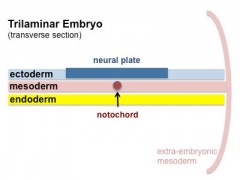
- Primitive streak - region of cell migration (gastrulation) from the epiblast layer forming sequentially the two germ cell layers (endoderm and mesoderm)
| Gastrulation, (Greek = belly)
Means the formation of gut, but has been used in a more looser sense to to describe the formation of the trilaminar embryo. The epiblast layer, consisting of totipotential cells, derives all 3 embryo layers: The primitive streak is the visible feature which represents the site of cell migration to form the additional layers. Historically, gastrulation was one of the earliest observable morphological event occurring in the frog embryo. |
|

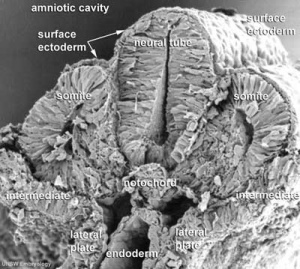
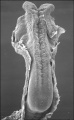
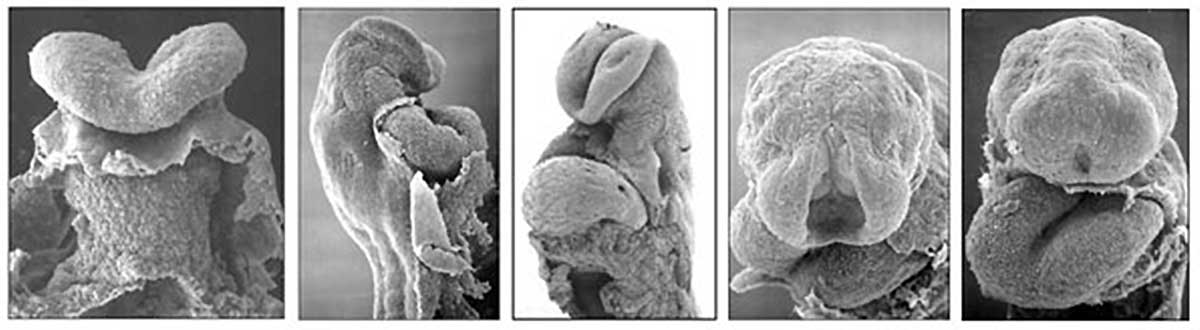
Trilaminar embryo (SEM) |
| Virtual Slides - Human Embryo (stage 7) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Notochord
The notochord is a structure which has an early mechanical role in embryonic disc folding and a major signaling role in patterning surrounding embryonic tissue development. This signaling role patterns many different tissues (neural plate, neural tube, somites, endodermal organs). It has its own sequence of development from a primitive axial process and is a developmental feature not present in the adult anatomy.
- axial process an initial epiblast hollow epithelial tube which extends in the midline from the primitive pit, cranially in the embryonic disc (toward the oral membrane).
- neuroenteric canal is a transient communication between the amnionic cavity and the yolk sac cavity formed by the axial process.
- notochordal plate forms from the axial process merging with the endoderm layer.
- notochord forms from the notochordal plate which then separates back into the mesoderm layer as a solid column of cells lying in the midline of the embryonic disc and running rostro-caudally (head to tail).
- An alternate name for the notochord is "axial mesoderm".
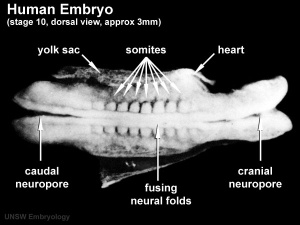
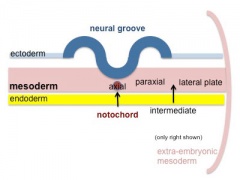
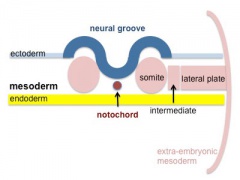
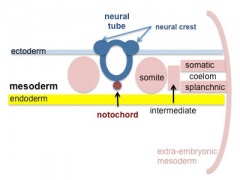
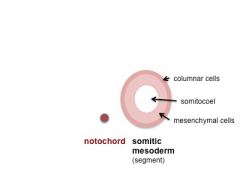
Somitogenesis
Mesoderm means the "middle layer" and it is from this layer that nearly all the bodies connective tissues are derived. In early mesoderm development a number of transient structures will form and then be lost as tissue structure is patterned and organised. Humans are vertebrates, with a "backbone", and the first mesoderm structure we will see form after the notochord will be somites.
- Mesoderm and Ectoderm Cartoons
Coelom, meaning "cavity", and major fluid-filled cavities can be seen to form both within the embryo (|intraembryonic coelom) and outside the embryo (extraembryonic coelom).
The intraembryonic coelom is the single primitive cavity that lies within the mesoderm layer that will eventually form the 3 major anatomical body cavities (pericardial, pleural, peritoneal).
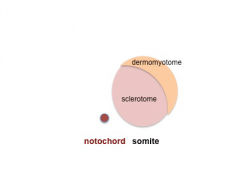
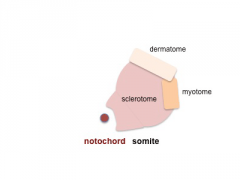
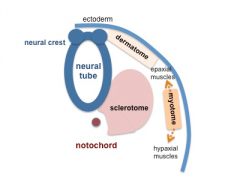
Somite initially forms 2 main components
- sclerotome - (ventromedial) forms axial skeleton - vertebral body and intervertebral disc
- dermomyotome - (dorsolateral) forms dermis and skeletal muscle
- Somite Cartoons
Neural
Week 3 ectoderm - 2 parts
- midline - neural plate (columnar cells)
- central nervous system (CNS)
- edge of neural plate - neural crest
- peripheral nervous system (PNS), and many other structures.
- lateral - surface ectoderm (cuboidal cells)
- epidermis of skin, hair, glands, anterior pituitary, teeth enamel
- head region - sensory placodes
- Ectoderm
- extends from buccopharyngeal membrane to primitive node
- forms above notochord and paraxial mesoderm
- neuroectodermal cells
- broad brain plate
- narrower spinal cord
- 3 components form: floor plate,neural plate, neural crest
|
|
|
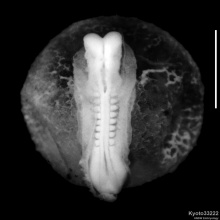
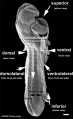
| Week 4 Embryo (dorsal view) | |||
|---|---|---|---|
| |||
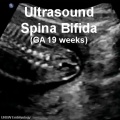
| Neural Tube Defects | |||||
|---|---|---|---|---|---|
|
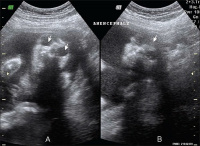
|
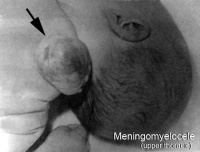
| |||
| Spina-Bifida | Anencephaly | Meningomyelocele |
Cardiogenesis
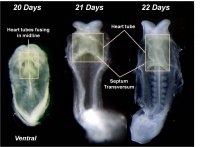
Early Development of Heart Tube
The Human Heart from day 10 to 25 (scanning electron micrograph)
- forms initially in splanchnic mesoderm of prechordal plate region - cardiogenic region
- growth and folding of the embryo moves heart ventrallly and downward into anatomical position
- week 3 begins as paired heart tubes that fuse to form single heart tube
- begins to beat in Humans- day 22-23
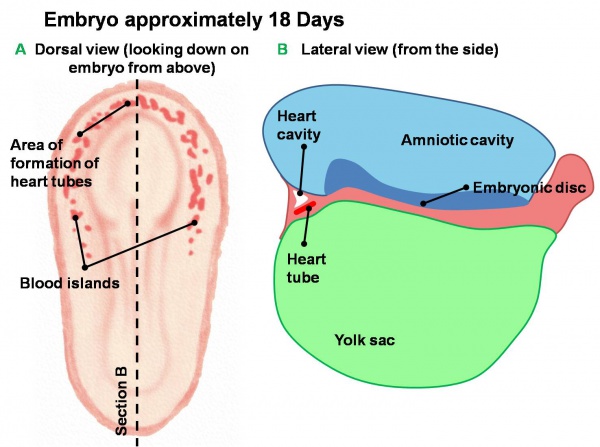
Blood Islands
- 2 populations of cells
- peripheral- form endothelial cells that form the lining of all blood vessels
- core- form blood cells (haemocytoblasts)
- all vessels (arteries and veins) appear initially the same
Blood Formation
- blood formation from stem cells occurs initially in the extra-embryonic mesoderm of the yolk sac
- then later (week 5) throughout embryonic mesenchyme
- blood stem cells then migrate into the liver
- then spleen, bone marrow, lymph nodes
Red Blood Cells
The only cells in the blood are initially entirely fetal red blood cells (RBC).
These red blood cells differ from adult red blood cells in:
- may remaining nucleated
- contain fetal haemoglobin - has different oxygen and carbon dioxide binding characteristics
- Links: Basic Cardiac Embryology
Early Placentation
The trophoblast layer has now differentiated into two morphologically distinct cellular layers.
- Syncitiotrophoblasts - form a multinucleated cytoplasmic mass by cytotrophoblast cell fusion and both invade the decidua and secrete hCG
- Cytotrophoblasts - form a cellular layer around the blastocyst, proliferates and extends behind syncitiotrophoblasts
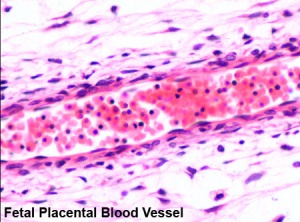
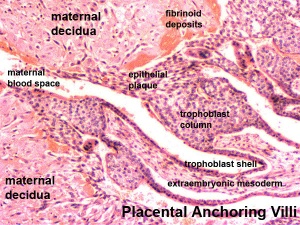
Early Utero-Placental exchange - transfer of nutrition from maternal lacunae filled with secretions from uterine glands and maternal blood from blood vessels. The development of trophoblast villi extending into the uterine decidua.
There are three stages of villi development:
- Primary Villi - cytotrophoblast
- Secondary Villi - cytotrophoblast + extraembryonic mesoderm
- Tertiary Villi - cytotrophoblast + extraembryonic mesoderm + blood vessels
There are two main types of early villi:
- Anchoring villi - attached to decidua
- Floating villi - not attached to decidua, floating in maternal lacunae.
Abnormalities
Critical periods, Genetic and Environmental factors leading to abnormal development will be covered in the associated practical classes.
BGDA: Lecture 1 | Lecture 2 | Practical 3 | Practical 6 | Practical 12 | Lecture Neural | Practical 14 | Histology Support - Female | Male | Tutorial
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 21) Embryology BGDA Lecture - Development of the Embryo/Fetus 1. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/BGDA_Lecture_-_Development_of_the_Embryo/Fetus_1
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G