Abnormal Development - Bacterial Infection
| Embryology - 25 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
| Educational Use Only - Embryology is an educational resource for learning concepts in embryological development, no clinical information is provided and content should not be used for any other purpose. |
Introduction
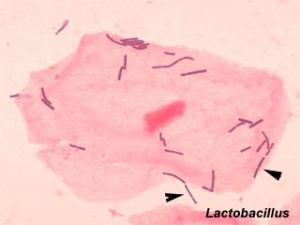
The variety of bacterial infections that can occur during pregnancy is as variable as the potential developmental effects, from virtually insignificant to major developmental, abortive or fatal in outcome. Some bacteria are common and are part of the normal genital tract flora (Lactobacillus sp), while other bacterial infections are less common or even rare and initially infect/transmit by air or fluids through the different epithelia (genital tract, lungs, gastrointestinal tract).
Note that some infections may have historic or alternative common names, for example Pertussis "whooping cough".
Maternal uterine tubal damage by bacterial infection (Chlamydia trachomatis) can also lead to Ectopic Implantation.
Many common pregnancy related bacterial infections, such as maternal urinary tract infection (UTI), are safely treated with antibiotics that can also cross the placenta. Most antibiotics have good data available on their teratogenic and toxic risks, while some antibiotics have limited data available.[1] This should also be considered postnatally, with transfer from mother to infant through lactation and breast milk. Note that pregnant and breastfeeding women are generally excluded from drug clinical trials and most data comes from animal studies.[2]
| Bacterial Links: bacterial infection | syphilis | gonorrhea | tuberculosis | listeria | salmonella | TORCH | Environmental | Category:Bacteria |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Bacteria Teratogen |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Neisseria Gonorrhea
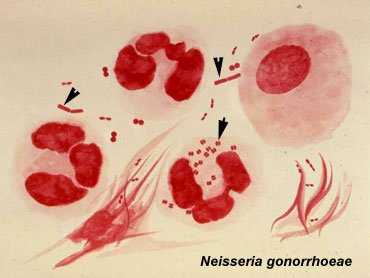 |
The gram-negative bacterium Neisseria gonorrhoeae causes the disease Gonorrhea which is a sexually transmitted disease (STD). Maternal infection increases the risk of premature birth and ophthalmia neonatorum (infantile purulent conjunctivitis).
A recent paper has described in the rat mother to the fetus during pregnancy, suggesting this as a model for human transmission.[9]
|
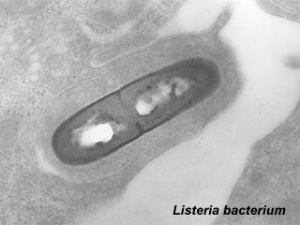
Listeria Monocytogenes
The bacterium Listeria monocytogenes is the pathogenic form of the 7 listeria species. Infection is generally through ingestion of organisms in contaminated food. Maternal symptoms may be mild, fetal effects can range from insignificant through to major abnormalities. Maternal treatment relates to potential developmental effects. Pregnancy greatly increases the risk of listeriosis, with pregnant women about 60% of all cases (male and female) aged 10 to 40 years. Similar effects are seem in other mammalian species.[10] and the Guinea pig placenta listeria model[11] Generalized suppression of immunity during pregnancy is suggest to have a role in susceptibility, though recent results in a mouse model suggest that susceptibility can occur very early in a pregnancy and may relate to enteric carriage rate.[10]
Infection
- ingestion of contaminated food
- colonization of the intestine
- intestinal translocation
- replication in the liver and spleen
- either the resolution of infection or spread to other organs resulting in a systemic infection
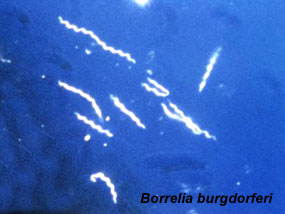
Lyme Disease
The bacterium spirochete Borrelia Burgdorferi causes Lyme disease. Infection can be through the blood by tick bite.
- Links: CDC (USA) - Lyme Disease
Mycoplasma
Mycoplasmas come in many different varieties, occur as part of the normal human flora, and lack a bacterial cell wall. Infection is generally through the female genital tract.
- Links: NCBI Bookshelf Medical Microbiology - Mycoplasma | CDC Mycoplasmas: Sophisticated, Reemerging, and Burdened by Their Notoriety
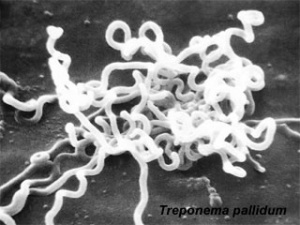
Syphilis
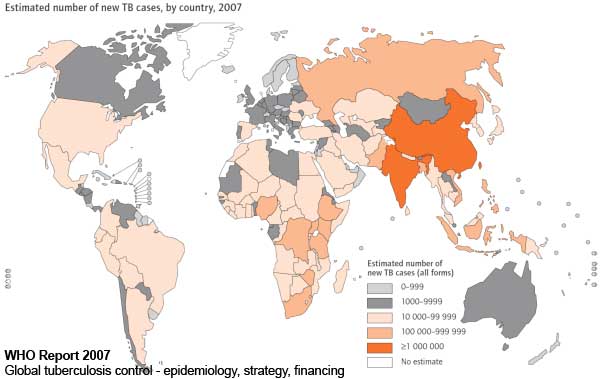
Mycobacterium Tuberculosis
Australian Recommendations
BCG vaccination is not recommended for general use in the Australian population.
BCG is recommended for:
- Aboriginal neonates in areas of high incidence of TB (e.g. Northern Territory, Far North Queensland, northern areas of Western Australia and South Australia)
- neonates and children 5 years and under who will be travelling or living in countries or areas with a high prevalence of TB for extended periods
- neonates born to parents with leprosy or a family history of leprosy
In addition to these recommendations BCG may be considered in the following:
- Children over 5 years who will be travelling or living in countries or areas with a high prevalence of TB for extended periods
- Health care workers (HCWs) who may be at high risk of exposure to drug resistant cases.
(Text Source: Communicable Diseases Intelligence Volume 30 Number 1, March 2006 - The BCG vaccine: information and recommendations for use in Australia)
Melioidosis
(Greek, melis = distemper of asses, oeidēs, resemblance, and osis, a suffix indicating an abnormal condition or disease. The bacteria Burkholderia pseudomallei is normally found in soil surface layers and in muddy surface waters.
- 1912 - Alfred Whitmore, a British pathologist serving in Burma, and his assistant C. S. Krishnaswami first described melioidosis. The infection became known as Whitmore’s disease.
- 1925 - Ambrose T. Stanton and William Fletcher identified Burkholderia pseudomallei as the infection’s causative agent, renamed the infection melioidosis because of its clinical resemblance to glanders. Glanders is an infectious disease that is caused by another bacterium Burkholderia mallei.
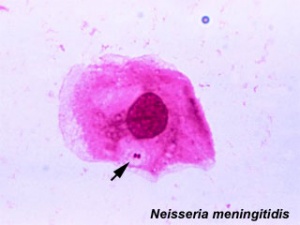
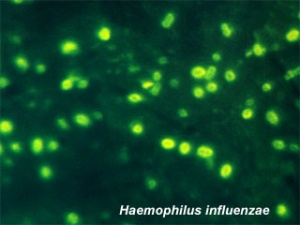
Bacterial Meningitis
The bacterium Neisseria meningitidis or Haemophilus influenzae type B (Hib) can cause the disease bacterial meningitis.
Hib immunization for infants and children are generally recommended.
Recently a universal vaccine for serogroup B meningococcus has been developed (See meningococcal vaccine 2001[12])
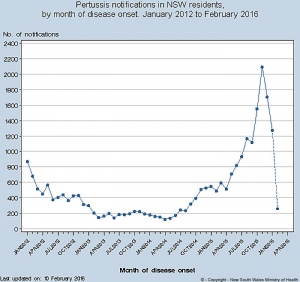
Pertussis
The bacterium Bordetella pertussis can cause the disease Pertussis (Whooping Cough) can lead to infant mortality.
May 2005 - First Combination Vaccine Approved to Help Protect Adolescents Against Whooping Cough USA Food and Drug Administration has approved booster immunization against pertussis (whooping cough) in combination with tetanus and diphtheria for adolescents. Pertussis is a highly contagious bacterial disease. (FDA 03 May 2005)
Links: CDC (USA) - Pertussis | Medline Plus - Pertussis | Pertussis Vaccination: Use of Acellular Pertussis Vaccines Among Infants and Young Children Recommendations of the Advisory Committee on Immunization Practices (ACIP) |
Salmonella
Salmonellosis is the infection caused bacterial genus Salmonella, these are mainly associated with foodborne transmission from contaminated animal–derived meat and dairy products. Though infection can also occur after handling pets, particularly reptiles like snakes, turtles and lizards.
Staphylococcus aureus
Staphylococcus aureus a gram-positive bacterium commonly present (25% of healthy people and animals) on the skin and nasal surfaces, no vaccines are available. Staphylococcus aureus bacteraemia (SAB), commonly known as ‘golden staph’ and has risk factors including injectable drug use, haemodialysis, indwelling vascular catheters and immunosuppression.[13] Strains of this bacteria can produce toxins related to food poisoning and be resistant to various antibiotics.
Methicillin-resistant Staphylococcus aureus (MRSA) bacteria are resistant to various antibiotics including Methicillin, there are other strains which are resistant to specific antibiotics (vancomycin).
About 2% of Staphylococcus aureus produce a toxin Panton-Valentine leucocidin (PVL) which can be fatal in neonates.
- Links: CDC (USA) - Staphylococcus | CDC (USA) - emerging infectious diseases | Medline Plus - Staph aureus food poisoning | Medical Microbiology - Staphylococcus
Cholera
Cholera is an infection of the small intestine caused by the bacterium Vibrio cholerae. The cholera bacterium is usually found in water or food contaminated by faeces and the incubation period ranges from less than one day to five days. The bacteria produces an enterotoxin that causes a watery diarrhoea, leading to severe dehydration and death if treatment is not promptly given. Vomiting also occurs in most patients. Outbreaks are generally associated with poor sanitation and water supply. Recent outbreaks have been identified in Haiti and Mexico.
Chlamydia
Chlamydia trachomatis
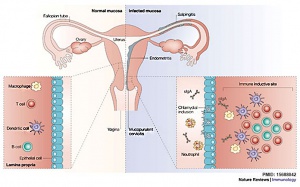
[[File:Female_genital_tract_chlamydia_trachomatis_infection_02.jpg|thumb|Chlamydia trachomatis developmental cycle[14]
A genus of bacteria commonly as a sexually transmitted disease. Chlamydia trachomatis is a a gram-negative bacterium associated with uterine tube damage and infections are a risk factor for ectopic implantation. An infected mother can during childbirth also pass chlamydia to the neonate.
Female genital tract chlamydia trachomatis infection[14]
Chlamydophila abortus
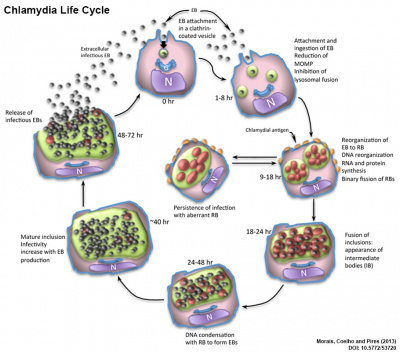
|
Originally called Chlamydia psittaci A Chlamydiae species (C. abortus) gram-negative bacteria associated with genital tract infections that causes abortion (chlamydiosis in pregnancy) and fetal death in mammals, including humans, and a major cause of fetal loss in sheep (ovine enzootic abortion) and cattle.
Chlamydia life cycle Infection begins with the attachment of the elementary bodies (EB) to the surface of target epithelial cells. These cells promote a pseudopod formation to engulf the EB. Inside the cytoplasm this bacterium inhibits the fusion of the vesicle with the cell lysosomes. The nascent inclusion is accompanied by the transition from EBs to reticulate bodies (RB). Late in the cycle, RBs replicate by binary fission to generate both RBs and intermediate bodies (IB). At this stage, antigenic proteins are exposed into the cell surface. An elongated, aberrant RB could be formed at this time with an arrest on chlamydia cycle originating a persistent infection, or continuing the cycle. The various intracytoplasmic inclusions with bacterium inside, can also be fused in this phase, and the agent develop into intermediate bodies (IB), before DNA condensation and RB transformation into a newly EB. The mature inclusion increases in size with EB formation, until becoming infectious and released into the extracellular space to continue a new intracellular cycle. N – nucleus; G – Golgi apparatus: EB – elementary bodies; RB – reticulate bodies; IB – intermediate bodies. (modified text from figure legend) |
- Links: Ectopic Implantation
Bacterial Vaginosis
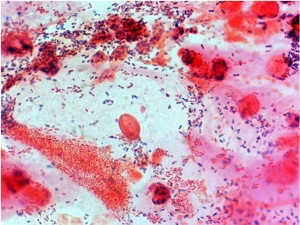
- Bacterial vaginosis imbalance of the normal vaginal flora (more anaerobic bacteria and less normal gram-positive bacteria Lactobacillus sp).
- Maternal infection is associated with a variety of pregnancy abnormalities including preterm birth and poor perinatal outcome.[16]
- Smear Image Links: L. crispatus | L. crispatus | non-L. crispatus with thin lactobacilli | non-L. crispatus with thin lactobacilli | mixture non-L. crispatus with L. crispatus | mixture non-L. crispatus with L. crispatus | irregular-shaped Gram positive rod | irregular-shaped Gram positive rod | mixture Lactobacillus and bacterial vaginosis-associated | mixture Lactobacillus and bacterial vaginosis-associated | bacterial vaginosis | bacterial vaginosis
- Links: Menstrual Cycle - Histology | Histology - Gram Stain | Bacterial Vaginosis | CDC (USA) Fact Sheet - Bacterial Vaginosis
Prokaryote Mycoplasmas
- smallest self-replicating organisms, with the smallest genomes (about 500 to 1000 genes)
- spherical to filamentous cells, no cell walls
- surface parasites of the human respiratory and urogenital tracts
- Mycoplasma pneumoniae infect the upper and lower respiratory tract
- Mycoplasma genitalium a prevalent sexually transmitted infection
- Mycoplasma hyorhinis found in patients with AIDS
- Search PubMed: Mycoplasma pneumoniae Pregnancy | Mycoplasma genitalium Pregnancy | Mycoplasma hyorhinis Pregnancy
Gram Stain
Bacterial staining procedure named after Hans Christian Gram (1853 - 1938). Generally divides bacteria into either:
- Gram-positive bacteria - purple crystal violet stain is trapped by layer of peptidoglycan (forms outer layer of the cell).
- Gram-negative bacteria - outer membrane prevents stain from reaching peptidoglycan layer in the periplasm, outer membrane then permeabilized and pink safranin counterstain is trapped by peptidoglycan layer.
Antibiotics
Many common pregnancy related bacterial infections, such as maternal urinary tract infection (UTI), are safely treated with antibiotics that can also cross the placenta. Most antibiotics have good data available on their teratogenic and toxic risks, while some antibiotics have limited data available. Teratogenic potential below is based upon the availability of published data from a 2006 literature study.[1]
- none - penicillin G and VK
- unlikely - amoxicillin, chloramphenicol, ciprofloxacin, doxycycline, levofloxacin, and rifampin
- undetermined - clindamycin, gentamicin, and vancomycin
Assessments were based on "good data" (penicillin G and VK), "fair data" (amoxicillin, chloramphenicol, ciprofloxacin, doxycycline, levofloxacin, and rifampin), "limited data" (clindamycin and gentamicin), and "very limited data" (vancomycin).
Antibiotic use should also be considered postnatally, with transfer from mother to infant through lactation and breast milk.[17] There has also been recent public concern about the persistence of antibiotics in animals used for human food production, with very little teratogenic data available for this concept.
Note that pregnant and breastfeeding women are generally excluded from drug clinical trials and most data comes from animal studies.[2]
- Links: Abnormal Development - Drugs | Milk
Australian NHMRC Guidelines
NHMRC Guidelines "are intended to promote health, prevent harm, encourage best practice and reduce waste" have replaced the earlier historic 1988 NHMRC recommendations.
- Links: Clinical Practice Guidelines - Pregnancy Care (2019) | PDF | Related Documents | Australian Clinical Practice Guidelines | NHMRC guidelines
| Historic - NHMRC (1988) Recommendations | ||
|---|---|---|
Neonates be assessed for follow-up care under the following conditions:
|
References
- ↑ 1.0 1.1 Nahum GG, Uhl K & Kennedy DL. (2006). Antibiotic use in pregnancy and lactation: what is and is not known about teratogenic and toxic risks. Obstet Gynecol , 107, 1120-38. PMID: 16648419 DOI.
- ↑ 2.0 2.1 Illamola SM, Bucci-Rechtweg C, Costantine MM, Tsilou E, Sherwin CM & Zajicek A. (2018). Inclusion of pregnant and breastfeeding women in research - efforts and initiatives. Br J Clin Pharmacol , 84, 215-222. PMID: 28925019 DOI.
- ↑ McHugh L, Marshall HS, Perrett KP, Nolan T, Wood N, Lambert SB, Richmond P, Ware RS, Binks P, Binks MJ & Andrews RM. (2019). The Safety of Influenza and Pertussis Vaccination in Pregnancy in a Cohort of Australian Mother-Infant Pairs, 2012-2015: The FluMum Study. Clin. Infect. Dis. , 68, 402-408. PMID: 30475988 DOI.
- ↑ Fricke EM, Elgin TG, Gong H, Reese J, Gibson-Corley KN, Weiss RM, Zimmerman K, Bowdler NC, Kalantera KM, Mills DA, Underwood MA & McElroy SJ. (2018). Lipopolysaccharide-induced maternal inflammation induces direct placental injury without alteration in placental blood flow and induces a secondary fetal intestinal injury that persists into adulthood. Am. J. Reprod. Immunol. , 79, e12816. PMID: 29369434 DOI.
- ↑ Jana N, Barik S & Arora N. (2011). Tuberculosis in pregnancy--a major maternal and perinatal challenge. BJOG , 118, 1145-6; author reply 1146. PMID: 21749618 DOI.
- ↑ Knight M, Kurinczuk JJ, Nelson-Piercy C, Spark P & Brocklehurst P. (2009). Tuberculosis in pregnancy in the UK. BJOG , 116, 584-8. PMID: 19250368 DOI.
- ↑ Giuliani MM, Adu-Bobie J, Comanducci M, Aricò B, Savino S, Santini L, Brunelli B, Bambini S, Biolchi A, Capecchi B, Cartocci E, Ciucchi L, Di Marcello F, Ferlicca F, Galli B, Luzzi E, Masignani V, Serruto D, Veggi D, Contorni M, Morandi M, Bartalesi A, Cinotti V, Mannucci D, Titta F, Ovidi E, Welsch JA, Granoff D, Rappuoli R & Pizza M. (2006). A universal vaccine for serogroup B meningococcus. Proc. Natl. Acad. Sci. U.S.A. , 103, 10834-9. PMID: 16825336 DOI.
- ↑ Colombo DF, Lew JL, Pedersen CA, Johnson JR & Fan-Havard P. (2006). Optimal timing of ampicillin administration to pregnant women for establishing bactericidal levels in the prophylaxis of Group B Streptococcus. Am. J. Obstet. Gynecol. , 194, 466-70. PMID: 16458647 DOI.
- ↑ Nowicki S, Selvarangan R & Anderson G. (1999). Experimental transmission of Neisseria gonorrhoeae from pregnant rat to fetus. Infect. Immun. , 67, 4974-6. PMID: 10456962
- ↑ 10.0 10.1 Suyemoto MM, Spears PA, Hamrick TS, Barnes JA, Havell EA & Orndorff PE. (2010). Factors associated with the acquisition and severity of gestational listeriosis. PLoS ONE , 5, e13000. PMID: 20885996 DOI.
- ↑ Bakardjiev AI, Stacy BA & Portnoy DA. (2005). Growth of Listeria monocytogenes in the guinea pig placenta and role of cell-to-cell spread in fetal infection. J. Infect. Dis. , 191, 1889-97. PMID: 15871123 DOI.
- ↑ Morley SL, Cole MJ, Ison CA, Camaraza MA, Sotolongo F, Anwar N, Cuevas I, Carbonero M, Campa HC, Sierra G & Levin M. (2001). Immunogenicity of a serogroup B meningococcal vaccine against multiple Neisseria meningitidis strains in infants. Pediatr. Infect. Dis. J. , 20, 1054-61. PMID: 11734711
- ↑ Mitchell DH & Howden BP. (2005). Diagnosis and management of Staphylococcus aureus bacteraemia. Intern Med J , 35 Suppl 2, S17-24. PMID: 16271058 DOI.
- ↑ 14.0 14.1 Brunham RC & Rey-Ladino J. (2005). Immunology of Chlamydia infection: implications for a Chlamydia trachomatis vaccine. Nat. Rev. Immunol. , 5, 149-61. PMID: 15688042 DOI.
- ↑ Verhelst R, Verstraelen H, Claeys G, Verschraegen G, Van Simaey L, De Ganck C, De Backer E, Temmerman M & Vaneechoutte M. (2005). Comparison between Gram stain and culture for the characterization of vaginal microflora: definition of a distinct grade that resembles grade I microflora and revised categorization of grade I microflora. BMC Microbiol. , 5, 61. PMID: 16225680 DOI.
- ↑ Guerra B, Ghi T, Quarta S, Morselli-Labate AM, Lazzarotto T, Pilu G & Rizzo N. (2006). Pregnancy outcome after early detection of bacterial vaginosis. Eur. J. Obstet. Gynecol. Reprod. Biol. , 128, 40-5. PMID: 16460868 DOI.
- ↑ Brucker MC & King TL. (2017). The 2015 US Food and Drug Administration Pregnancy and Lactation Labeling Rule. J Midwifery Womens Health , 62, 308-316. PMID: 28556499 DOI.
Reviews
Gantert M, Been JV, Gavilanes AW, Garnier Y, Zimmermann LJ & Kramer BW. (2010). Chorioamnionitis: a multiorgan disease of the fetus?. J Perinatol , 30 Suppl, S21-30. PMID: 20877404 DOI.
Donders GG. (2006). Management of genital infections in pregnant women. Curr. Opin. Infect. Dis. , 19, 55-61. PMID: 16374219
Boggess KA. (2005). Pathophysiology of preterm birth: emerging concepts of maternal infection. Clin Perinatol , 32, 561-9. PMID: 16085020 DOI.
Hirsch E & Wang H. (2005). The molecular pathophysiology of bacterially induced preterm labor: insights from the murine model. J. Soc. Gynecol. Investig. , 12, 145-55. PMID: 15784499 DOI.
Cretekos CJ, Weatherbee SD, Chen CH, Badwaik NK, Niswander L, Behringer RR & Rasweiler JJ. (2005). Embryonic staging system for the short-tailed fruit bat, Carollia perspicillata, a model organism for the mammalian order Chiroptera, based upon timed pregnancies in captive-bred animals. Dev. Dyn. , 233, 721-38. PMID: 15861401 DOI.
Doganay M. (2003). Listeriosis: clinical presentation. FEMS Immunol. Med. Microbiol. , 35, 173-5. PMID: 12648833
Goldenberg RL, Hauth JC & Andrews WW. (2000). Intrauterine infection and preterm delivery. N. Engl. J. Med. , 342, 1500-7. PMID: 10816189 DOI.
Ross SM. (1982). Sexually transmitted diseases in pregnancy. Clin Obstet Gynaecol , 9, 565-92. PMID: 6293753
Articles
Guerra B, Ghi T, Quarta S, Morselli-Labate AM, Lazzarotto T, Pilu G & Rizzo N. (2006). Pregnancy outcome after early detection of bacterial vaginosis. Eur. J. Obstet. Gynecol. Reprod. Biol. , 128, 40-5. PMID: 16460868 DOI.
Giuliani MM, Adu-Bobie J, Comanducci M, Aricò B, Savino S, Santini L, Brunelli B, Bambini S, Biolchi A, Capecchi B, Cartocci E, Ciucchi L, Di Marcello F, Ferlicca F, Galli B, Luzzi E, Masignani V, Serruto D, Veggi D, Contorni M, Morandi M, Bartalesi A, Cinotti V, Mannucci D, Titta F, Ovidi E, Welsch JA, Granoff D, Rappuoli R & Pizza M. (2006). A universal vaccine for serogroup B meningococcus. Proc. Natl. Acad. Sci. U.S.A. , 103, 10834-9. PMID: 16825336 DOI.
Colombo DF, Lew JL, Pedersen CA, Johnson JR & Fan-Havard P. (2006). Optimal timing of ampicillin administration to pregnant women for establishing bactericidal levels in the prophylaxis of Group B Streptococcus. Am. J. Obstet. Gynecol. , 194, 466-70. PMID: 16458647 DOI.
Goffinet F, Maillard F, Mihoubi N, Kayem G, Papiernik E, Cabrol D & Paul G. (2003). Bacterial vaginosis: prevalence and predictive value for premature delivery and neonatal infection in women with preterm labour and intact membranes. Eur. J. Obstet. Gynecol. Reprod. Biol. , 108, 146-51. PMID: 12781402
Morley SL, Cole MJ, Ison CA, Camaraza MA, Sotolongo F, Anwar N, Cuevas I, Carbonero M, Campa HC, Sierra G & Levin M. (2001). Immunogenicity of a serogroup B meningococcal vaccine against multiple Neisseria meningitidis strains in infants. Pediatr. Infect. Dis. J. , 20, 1054-61. PMID: 11734711
Nowicki S, Selvarangan R & Anderson G. (1999). Experimental transmission of Neisseria gonorrhoeae from pregnant rat to fetus. Infect. Immun. , 67, 4974-6. PMID: 10456962
Pubmed
Bookshelf
- Approved Lists of Bacterial Names Edited by VBD Skerman, Vicki McGowan, and PHA Sneath. Washington (DC): ASM Press; 1989. ISBN-13: 978-1-55581-014-6 http://www.ncbi.nlm.nih.gov/books/NBK814 PMID 20806452
- Search Jan2006 "bacterial infection" 547,445 reference articles of which 45,020 were reviews.
Search PubMed: embryonic bacterial infection | prenatal bacterial infection | maternal bacterial infection | Chorioamnionitis
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- CDC (USA) Public Health Training Network Epidemiology and Prevention of Vaccine-Preventable Diseases (viewable Webcasts requires Media Player) | Advisory Committee on Immunization Practices (ACIP) Recommendations
- Royal College of Obstetricians and Gynaecologists (UK) Infection and Pregnancy - study group recommendations (Jun 2001)
- REPROTOX - contains summaries on the effects of medications, chemicals, infections, and physical agents on pregnancy, reproduction, and development.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 25) Embryology Abnormal Development - Bacterial Infection. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Abnormal_Development_-_Bacterial_Infection
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G