User:Z5015014: Difference between revisions
No edit summary |
|||
| Line 113: | Line 113: | ||
==Lab 7 Assessment== | ==Lab 7 Assessment== | ||
Duchenne muscular dystrophy (DMD) is the most common muscular dystrophy affecting | Duchenne muscular dystrophy (DMD) is the most common muscular dystrophy, affecting 1 in 3,500-5,000 newborn males globally. It is caused by recessive loss-of-function mutations in the dystrophin gene on the X chromosome.<ref name="PMID26140716"><pubmed>26140716</pubmed></ref> Frame-shift mutations in the gene lead to failed expression of the dystrophin mutation and hence the manifestation of DMD. One-third of DMD cases can occur by spontaneous mutation, meaning that a positive family history is not required for diagnosis. Physical examinations, molecular biopsies, genetic testing, and review of clinical history are all involved when diagnosing DMD. Serum creatine kinase (CK) is used as an initial diagnostic tool to measure the levels of muscle enzymes.<ref name="PMID27524897"><pubmed>27524897</pubmed></ref> DMD is defined at the molecular level as producing no dystrophin and clinically as the loss of ambulation at 12 years of age or younger.<ref name="PMID26140716"/> A less severe mutation of the dystrophin gene, such as an in-frame deletion, can instead produce a truncated version of the protein which results in Becker muscular dystrophy (BMD), a milder form of DMD.<ref name="PMID25740330"><pubmed>25740330</pubmed></ref> | ||
< | Dystrophin is a major muscle protein which connects the cytoskeleton (cytoskeletal actin, microtubules, and intermediate filaments) and the extracellular matrix (ECM).<ref name="PMID25740330"/><ref name="PMID26295289"><pubmed>26295289</pubmed></ref> Dystrophin is anchored to the plasma membrane by phospholipids and β-dystroglycan. The dystrophin complex is found in costameres of skeletal and heart muscle, which are located at the periphery of the muscle fibers. Costameres are involved in transmitting forces from the cytosol to the ECM and therefore prevent the collapse of the cell membrane during muscle contraction. Hence, in DMD and BMD the impaired function of dystrophin results in frequent cell damage when the muscle contracts. The rupture of the plasma membrane causes leakage of the intracellular components; for example, creatine kinase can be released from the damaged cell which explains the high concentrations of serum creatine kinase in DMD patients.<ref name="PMID26295289"/> | ||
The most common mutation implicated in DMD and BMD is a deletion of one or more exons, accounting for 60-70% of DMD cases and 80-85% of BMD cases. Point mutations are have been found in approxiamtely 26% of DMD cases and 13% of BMD cases, while exonic duplications are involved in 10-15% of DMD cases and 5-10% of BMD cases. Other possible mutations include subexonic insertions, deletions, splice mutations, and missense mutations.<ref name="PMID26140716"/> | |||
Animal models used to investigate DMD and BMD include the ''mdx'' mouse and the ''grmd'' dog.<ref name="PMID26295289"/><ref name="PMID25740330"/> Due to the size of the dystrophin gene, it is difficult to treat DMD or BMD with gene therapy.<ref name="PMID27594988"><pubmed>27594988</pubmed></ref> The idea of producing a smaller version of the protein, micro-dystrophin, has been attempted, but due to its smaller size it cannot act as a perfect functional substitute for dystrophin. In the case of DMD, exon skipping therapy has been investigated as a way of altering the mutation of the dystrophin gene. Instead of the frame-shift mutation which results in no functional dystrophin being produced, the splicing of the mRNA can be modified to exclude one or more additional exons which therefore restores the reading frame and results in production of a BMD-like dystrophin (truncated, but still partly functional). This has been accomplished by introducing small antisense oligonucleotides (AONs) sequences, which bind to exon splice junctions and inhibit the action of the spliceosome.<ref name="PMID26295289"/> However, this ultimately only lessens the severity of the disease and still presents the problem of treating the BMD-like symptoms. There is still no effective cure for either DMD or BMD. Most available treatments are focused on slowing the progression of the diseases and continued research is being done into ways of converting a severe phenotype of disease to a more mild phenotype.<ref name="PMID27594988"/><ref name="PMID26140716"/> | |||
<br><br> | <br><br> | ||
==References== | ==References== | ||
<references/> | <references/> | ||
Revision as of 14:34, 6 October 2016
| Student Information (expand to read) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Individual Assessments | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Please leave this template on top of your student page as I will add your assessment items here. Beginning your online work - Working Online in this course
Click here to email Dr Mark Hill | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 1 Assessment - Researching a Topic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In the lab I showed you how to find the PubMed reference database and search it using a topic word. Lab 1 assessment will be for you to use this to find a research reference on "fertilization" and write a brief summary of the main finding of the paper.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 2 Assessment - Uploading an Image | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
OK you are now in a group
Initially the topic can be as specific or as broad as you want. Chicken embryo E-cad and P-cad gastrulation[1] References
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 4 Assessment - GIT Quiz | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
ANAT2341 Quiz Example | Category:Quiz | ANAT2341 Student 2015 Quiz Questions | Design 4 quiz questions based upon gastrointestinal tract. Add the quiz to your own page under Lab 4 assessment and provide a sub-sub-heading on the topic of the quiz. An example is shown below (open this page in view code or edit mode). Note that it is not just how you ask the question, but also how you explain the correct answer. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 5 Assessment - Course Review | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complete the course review questionnaire and add the fact you have completed to your student page. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 6 Assessment - Cleft Lip and Palate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 7 Assessment - Muscular Dystrophy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 8 Assessment - Quiz | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A brief quiz was held in the practical class on urogenital development. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 9 Assessment - Peer Assessment | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 10 Assessment - Stem Cells | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
As part of the assessment for this course, you will give a 15 minutes journal club presentation in Lab 10. For this you will in your current student group discuss a recent (published after 2011) original research article (not a review!) on stem cell biology or technology.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lab 11 Assessment - Heart Development | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Read the following recent review article on heart repair and from the reference list identify a cited research article and write a brief summary of the paper's main findings. Then describe how the original research result was used in the review article.
<pubmed>26932668</pubmed>Development | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lab Attendance
Z5015014 (talk) 14:35, 5 August 2016 (AEST)
Z5015014 (talk) 14:40, 12 August 2016 (AEST)
Z5015014 (talk) 13:13, 19 August 2016 (AEST)
Z5015014 (talk) 13:03, 26 August 2016 (AEST)
Z5015014 (talk) 2 September 2016 (AEST)
Z5015014 (talk) 13:07, 9 September 2016 (AEST)
Z5015014 (talk) 13:03, 16 September 2016 (AEST)
Week 1 Homework
Team Role
I studied the Belbin Team Roles in a previous course and identified myself as a Teamworker, which I still definitely agree with, but I'd probably also add that I have the tendencies of a Complete Finisher. In group assignments I always prefer not to take charge and rather to provide support by facilitating discussions and encouraging good communication within the group. In the past, I've often mediated between my real life friends when conflict happens so I'm comfortable in doing that for any group situation I find myself in. I'm also very indecisive as a person and I know this reflects into any group work that I do because I find it difficult if I have to lead or make choices for the rest of the team. I would also say that because I don't particularly look forward to group assignments in the first place, because I'd much rather complete a task by myself, I can sometimes be a Complete Finisher. I know that I hesitate having to rely on other people because I worry that they won't do something the way I would have chosen to do it.
Lecture 1: Fertilisation (2/8/16)
I've always found fertilisation to be an exciting and interesting topic. Even though I've studied it before in previous courses I still find it so fascinating just how much goes into the 'simple' union of a sperm and an egg cell. The way that so many processes and reactions and biological design come together to allow fertilisation to happen really amazes me. But I think the most interesting thing I heard in today's lecture was about the zona pellucida and the ZP2 protein, and how it's used as a signal to attract the sperm to bind to the ovum, and then modified to prevent polyspermy by stopping additional sperm from binding. I'd never learnt about this before so it was really interesting to understand the mechanism involved.
| Mark Hill (talk) 12:36, 5 August 2016 (AEST) Very good. maybe a little to many sub-sub-sub- headings, this will get messy. I will discuss in today's lab online formatting etc. |
Lab 1 Assessment
IVF culture medium affects post-natal weight in humans during the first 2 years of life.[1]
Sander H.M. Kleijkers, Aafke P.A. van Montfoort, Luc J.M. Smits, Wolfgang Viechtbauer, Tessa J. Roseboom, Ewka C.M. Nelissen, Edith Coonen, Josien G. Derhaag, Lobke Bastings, Inge E.L. Schreurs, Johannes L.H. Evers & John C.M. Dumoulin
- In this study, Kleijkers et al. investigated the relationship between the culture media used for embryos during the process of IVF and the subsequent development of the IVF children in early life. This followed on from previous research in which it was observed that the choice of culture medium had an impact on birthweight as well as development of the fetus during pregnancy. 1432 IVF treatments were initially involved and each was randomly allocated to either grow in Cook medium or Vitrolife medium. From this, 126 embryos from the Cook group and 168 embryos from the Vitrolife group were successfully birthed and used for further study. Kleijkers et al. contacted all of the parents two years after the initial birth for records of the postnatal weight, height, and head circumference of their child over the two year period. Statistical analysis and different analytical models were used to compare the collected data. It was observed that the weight of singletons whose embryos had been cultured in Cook medium was consistently lower than that of the Vitrolife group; this confirms that the effect of culture media on fetal growth, as had been previously researched, persists in early postnatal development. Kleijkers et al. propose that these results show the sensitivity of the human embryo to its environment, whether in vivo or in vitro. Hence because the chosen media for the embryo is the environment it grows in before implantation, the adaptation of this embryo to the media, possibly by epigenetic modification, is seen to have consequences for later development. This emphasises the need for further research into the growth media used for IVF treatment to ensure an optimal environment for the preimplantation growth of the embryo, and therefore both prenatal and postnatal development of an IVF child.
| Mark Hill 18 August 2016 - You have added the citation correctly and written a good brief summary of the article findings. You also seem to have practiced using some of the Wiki formatting tools. So the very conditions of initial growth may have long-term developmental ramifications. You can see why research in this area is important. These days we have a reasonably large IVF cohort to begin to get some useful data.
You have to be careful though when commercial products are being compared. Cook medium | Vitrolife medium |
Assessment 5/5 |
Lab 2 Assessment
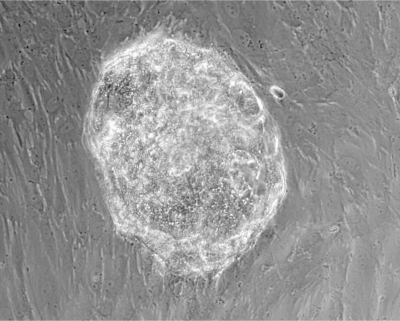
Human blastocyst attached to decidualized human endometrial stromal cells after 72 hours of co-culture.[2]
| Mark Hill 29 August 2016 - All information Reference, Copyright and Student Image template correctly included with the file and referenced on your page here. | Assessment 5/5 |
Lab 3 Assessment
| Mark Hill 31 August 2016 - Lab 3 Assessment Quiz - Mesoderm and Ectoderm development. All correct, well done! | Assessment 5/5 |
Lab 4 Assessment
Gastrointestinal Development Quiz
Lab 5 Assessment
I completed the course review questionnaire!
Lab 6 Assessment
Meta-analysis Reveals Genome-Wide Significance at 15q13 for Nonsyndromic Clefting of Both the Lip and the Palate, and Functional Analyses Implicate GREM1 As a Plausible Causative Gene.[3]
Kerstin U Ludwig, Syeda Tasnim Ahmed, Anne C Böhmer, Nasim Bahram Sangani, Sheryil Varghese, Johanna Klamt, Hannah Schuenke, Pinar Gültepe, Andrea Hofmann, Michele Rubini, Khalid Ahmed Aldhorae, Regine P Steegers-Theunissen, Augusto Rojas-Martinez, Rudolf Reiter, Guntram Borck, Michael Knapp, Mitsushiro Nakatomi, Daniel Graf, Elisabeth Mangold & Heiko Peters
- In their research, Ludwig et al. looked at findings from previous studies and confirmed that the 15q13 region is a risk locus for developing nonsyndromic cleft lip and palate (nsCL/P) but not nonsyndromic cleft lip only (nsCLO). By experimenting on mice, they also associated genetic variation in a 5 kb region downstream from the Gremlin-1 (Grem1) transcription start site with developing nsCL/P. They observed localised expression of Grem1 in the soft palate and in the processes forming the lip during mouse embryogenesis. While Grem1-deficient mice did not develop lip or palate defects, the addition of ectopic Grem1 was shown to have a pathogenic effect on the morphogenesis of the palatal shelves during secondary palate development. Grem1 is known to be critical for limb bud development, but not lip and palate development. Its role in limb bud development is to establish a positive feedback loop between Shh and Fgf via inhibition of Bmp signalling (a negative regulator of SHH expression in limb mesenchyme). However, previous research determined that Bmp signalling is a positive regulator of Shh expression in the palatal shelf epithelium and cell proliferation in the anterior palatal shelf mesenchyme. Bmp4 and Bmp2 are involved in the normal development of the anterior palate and Bmp4 and Bmp7 are expressed in the posterior palatal shelves. Therefore, it was concluded that an abnormality in Grem1 expression could interrupt normal development of the secondary palate due to the inhibition of Bmp signalling.
Lab 7 Assessment
Duchenne muscular dystrophy (DMD) is the most common muscular dystrophy, affecting 1 in 3,500-5,000 newborn males globally. It is caused by recessive loss-of-function mutations in the dystrophin gene on the X chromosome.[4] Frame-shift mutations in the gene lead to failed expression of the dystrophin mutation and hence the manifestation of DMD. One-third of DMD cases can occur by spontaneous mutation, meaning that a positive family history is not required for diagnosis. Physical examinations, molecular biopsies, genetic testing, and review of clinical history are all involved when diagnosing DMD. Serum creatine kinase (CK) is used as an initial diagnostic tool to measure the levels of muscle enzymes.[5] DMD is defined at the molecular level as producing no dystrophin and clinically as the loss of ambulation at 12 years of age or younger.[4] A less severe mutation of the dystrophin gene, such as an in-frame deletion, can instead produce a truncated version of the protein which results in Becker muscular dystrophy (BMD), a milder form of DMD.[6]
Dystrophin is a major muscle protein which connects the cytoskeleton (cytoskeletal actin, microtubules, and intermediate filaments) and the extracellular matrix (ECM).[6][7] Dystrophin is anchored to the plasma membrane by phospholipids and β-dystroglycan. The dystrophin complex is found in costameres of skeletal and heart muscle, which are located at the periphery of the muscle fibers. Costameres are involved in transmitting forces from the cytosol to the ECM and therefore prevent the collapse of the cell membrane during muscle contraction. Hence, in DMD and BMD the impaired function of dystrophin results in frequent cell damage when the muscle contracts. The rupture of the plasma membrane causes leakage of the intracellular components; for example, creatine kinase can be released from the damaged cell which explains the high concentrations of serum creatine kinase in DMD patients.[7]
The most common mutation implicated in DMD and BMD is a deletion of one or more exons, accounting for 60-70% of DMD cases and 80-85% of BMD cases. Point mutations are have been found in approxiamtely 26% of DMD cases and 13% of BMD cases, while exonic duplications are involved in 10-15% of DMD cases and 5-10% of BMD cases. Other possible mutations include subexonic insertions, deletions, splice mutations, and missense mutations.[4]
Animal models used to investigate DMD and BMD include the mdx mouse and the grmd dog.[7][6] Due to the size of the dystrophin gene, it is difficult to treat DMD or BMD with gene therapy.[8] The idea of producing a smaller version of the protein, micro-dystrophin, has been attempted, but due to its smaller size it cannot act as a perfect functional substitute for dystrophin. In the case of DMD, exon skipping therapy has been investigated as a way of altering the mutation of the dystrophin gene. Instead of the frame-shift mutation which results in no functional dystrophin being produced, the splicing of the mRNA can be modified to exclude one or more additional exons which therefore restores the reading frame and results in production of a BMD-like dystrophin (truncated, but still partly functional). This has been accomplished by introducing small antisense oligonucleotides (AONs) sequences, which bind to exon splice junctions and inhibit the action of the spliceosome.[7] However, this ultimately only lessens the severity of the disease and still presents the problem of treating the BMD-like symptoms. There is still no effective cure for either DMD or BMD. Most available treatments are focused on slowing the progression of the diseases and continued research is being done into ways of converting a severe phenotype of disease to a more mild phenotype.[8][4]