Implantation: Difference between revisions
| Line 39: | Line 39: | ||
* Trophinin is a membrane protein expressed on blastocyst trophectoderm cells and on uterine endometrium epithelial cells. | * Trophinin is a membrane protein expressed on blastocyst trophectoderm cells and on uterine endometrium epithelial cells. | ||
* Adhesion is thought to occur through trophinin-trophinin binding. | * Adhesion is thought to occur through trophinin-trophinin binding. | ||
* Adhesion also triggers two effects: | * Adhesion also triggers two trophinin mediated effects: | ||
# trophectoderm cells activate for implantation (invasion) | # trophectoderm cells activate for implantation (invasion) | ||
# maternal endometrial epithelial cells induced programmed cell death (apoptosis).<ref>Tamura N, Sugihara K, Akama TO, Fukuda MN. '''Trophinin-mediated cell adhesion induces apoptosis of human endometrial epithelial cells through PKC-δ.''' Cell Cycle. 2011 Jan 1;10(1):135-43 [http://www.ncbi.nlm.nih.gov/pubmed/21191175 PMID21191175]</ref> | # maternal endometrial epithelial cells induced programmed cell death (apoptosis).<ref>Tamura N, Sugihara K, Akama TO, Fukuda MN. '''Trophinin-mediated cell adhesion induces apoptosis of human endometrial epithelial cells through PKC-δ.''' Cell Cycle. 2011 Jan 1;10(1):135-43 [http://www.ncbi.nlm.nih.gov/pubmed/21191175 PMID21191175]</ref> | ||
Revision as of 17:53, 30 July 2011
Introduction
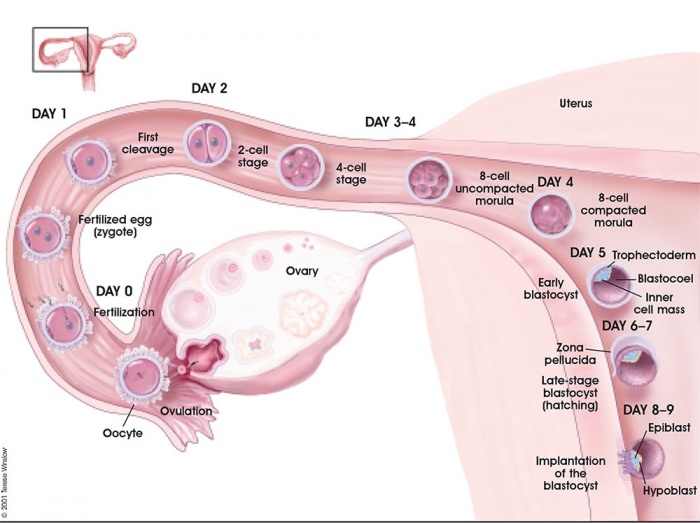
The term "implantation" is used to describe process of attachment and invasion of the uterus endometrium by the blastocyst (conceptus) in placental animals. In humans, this process begins at the end of week 1, with most successful human pregnancies the conceptus implants 8 to 10 days after ovulation, and early pregnancy loss increases with later implantation.[2] The implantation process continues through the second week of development.
The initial phase of the implantation process is "adplantation". This first phase requires the newly hatched blastocyst to loosely adhere to the endometrial epithelium, often "rolling" to the eventual site of implantation where it is firmly adhered. This process requires both the blastocyst adhesion interaction with the endometrium during the "receptive window".
Subsequent development of the placenta allows maternal support of embryonic and fetal development. If implantation has not proceeded sufficiently during the menstrual cycle to allow hormonal feedback to the ovary, then the next cycle may commence leading to conceptus loss. There is also evidence, from animal models, that a conceptus with major genetic does not develop or implant correctly leading to their loss during the first and second weeks of development.
In recent years with the development or Assisted Reproductive Technologies (ART or IVF) there is a growing interest in this process, with techniques that introduce the blastocyst into the uterus to allow normal implantation to occur.
Abnormal implantation is where this process does not occur in the body of the uterus (ectopic) or where the placenta forms incorrectly. In addition implantation can occur normally but with an abnormal conceptus, as in a hydatiform mole development.
Some Recent Findings
|
Week 1 and 2 Human Development Overview
Endometrial Receptivity
In humans, receptivity occurs 6 days after the post-ovulatory progesterone surge and lasts about 2 to 4 days. A similar "receptivity window" occurs following fertilization in other species: rat day 5 and mouse day 4.5. Many studies have looked into identifying markers for this receptivity period both to optimise and to block this process.
Implantation Factors
Trophinin
- Trophinin is a membrane protein expressed on blastocyst trophectoderm cells and on uterine endometrium epithelial cells.
- Adhesion is thought to occur through trophinin-trophinin binding.
- Adhesion also triggers two trophinin mediated effects:
- trophectoderm cells activate for implantation (invasion)
- maternal endometrial epithelial cells induced programmed cell death (apoptosis).[5]
- Links: OMIM
Cytokines
In mice, endometrial secretion of two IL-6 family cytokines, leukemia inhibitory factor (LIF) and Interleukin-11 (IL-11), are key requirements for implantation. A recent human study suggests that there is a similar requirement for human conceptus implantation.[6]
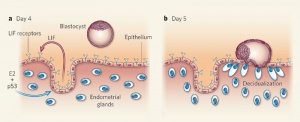
Uterine Leukemia Inhibitory Factor (LIF) Expression[1]
| a - At day 4 of pregnancy, oestrogen E2 induces LIF expression in the endometrial glands, leading to LIF secretion into the uterine lumen. There, LIF binds to its receptors on the surface of epithelial cells. | b - This makes the uterus receptive to the blastocyst, which implants by day 5 of pregnancy. Hu et al. find that LIF expression in the endometrial glands also depends on the regulatory activity of p53. In the absence of p53, insufficient LIF is produced, the uterus does not become adequately receptive, and fewer blastocysts implant. |
Galectin 9
Galectin 9, a protein that binds galactosides and has many different roles, has been identified as a marker for the mid- and late-secretory phases of human endometrium and decidua. The high expression at uterodomes during the period of implantation, suggests that it may also mark endometrial receptivity.[7]
Pinopods
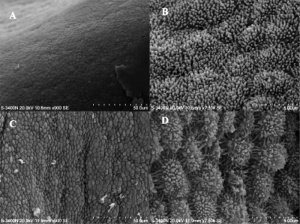
(uterodomes) Cellular feature seen on the apical uterine epithelium surface. The presence of these structures is thought in many species to be a marker for endometrial receptivity. In humans though, recent studies have shown pinopodes are also present throughout the luteal phase of the menstrual cycle.[9] It has also been suggested that their role is not primarily pinocytotic, hence the alternate suggested name "uterodomes" based upon their appearance when imaged by electron microscopy.[10]
These transient microprotrusions inter-digitate with microvilli on the apical syncytiotrophoblast surface of the blastocyst during initial adplantation and implantation process.
Implantation Cartoon
| width=250px|height=240px|controller=true|autoplay=false</qt> | This animation shows the process of implantation, occurring during week 2 of development in humans.
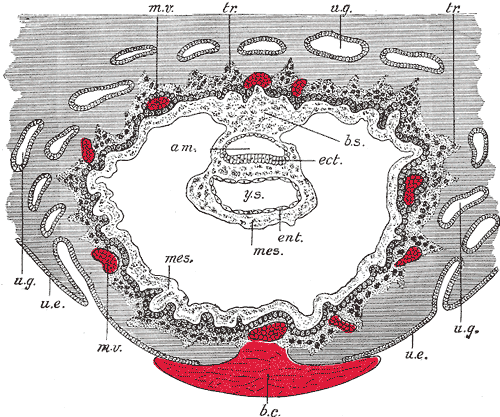
The beginning of the animation shows: the uterus lining (endometrium epithelium), the hatched blastocyst with a flat outer layer of trophoblast cells (green), the inner cell mass which has formed into the bilaminar embryo (epiblast and hypoblast) and the large fluid-filled space (blastocoel). green cells - trophoblast layer of the conceptus blue cells - epiblast layer of the bilaminar embryo yellow cells - hypoblast layer of the bilaminar embryo white cells - uterine endometrium epithelium red - maternal blood vessel Links: Quicktime version | Flash Version | Quicktime movie |
The second week of human development is concerned with the process of implantation and the differentiation of the blastocyst into early embryonic and placental forming structures.
- implantation commences about day 6 to 7
- Adplantation - begins with initial adhesion to the uterine epithelium
- blastocyst then slows in motility, "rolls" on surface, aligns with the inner cell mass closest to the epithelium and stops
- Implantation - migration of the blastocyst into the uterine epithelium, process complete by about day 9
- coagulation plug - left where the blastocyst has entered the uterine wall day 12
Normal Implantation Sites - in uterine wall superior, posterior, lateral
Day 8 to 9
Decidual Reaction
During pregnancy, at implantation the endometrium is altered by the maternal steroid hormones, estrogen and progesterone and in response to the implanting conceptus and renamed the "decidua". This process of signaling is called the decidual reaction or decidualization, and commences at the site of implantation and spreads throughout the uterine lining.
- occurs within the uterus wall
- initially at site of implantation and includes both cellular and matrix changes
- reaction spreads throughout entire uterus, not at cervix
- promoted by the maternal steroid hormones, estrogen and progesterone
- extensive proliferation and differentiation of uterine stromal cells
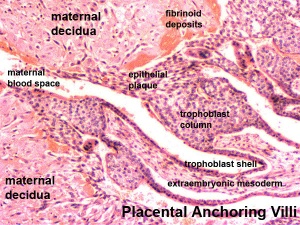
- deposition of fibrinoid and glycogen and epithelial plaque formation (at anchoring villi)
- presence of decidual cells are indicative of pregnancy
In uterine stromal decidualization, bone morphogenetic protein 2 (BMP2) nonactive precursor protein is cleaved by proprotein convertase 5/6 (PC6) to produce the active form. Deletion or knockdown of either BMP2 or PC6 inhibits decidualization leads to implantation failure and female infertility.[11]
A recent human histological study has shown that endometrial stromal cell decidualization leads to a loss of lymphatics particularly apparent around the uterine spiral arteries.[12]
Cervical Mucus Plug
Along with the decidualization, estrogen also stimulates the production of mucus from glands at the opening of the uterus, the cervix, where it joins the vagina. This secreted mucus then forms a plug/barrier (CMP) acting in a mechanical and antibacterial manner.
Abnormal Implantation
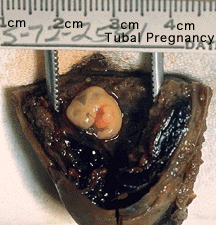
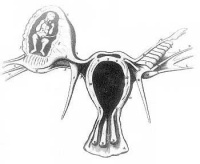
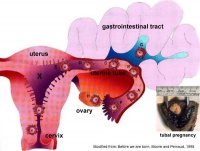
Tubal Pregnancy
Abnormal implantation sites or Ectopic Pregnancy occurs if implantation is in uterine tube or outside the uterus.
- sites - external surface of uterus, ovary, bowel, gastrointestinal tract, mesentry, peritoneal wall
- If not spontaneous then, embryo has to be removed surgically
Tubal pregnancy - 94% of ectopic pregnancies
- if uterine epithelium is damaged (scarring, pelvic inflammatory disease)
- if zona pellucida is lost too early, allows premature tubal implantation
- embryo may develop through early stages, can erode through the uterine horn and reattach within the peritoneal cavity
|
|
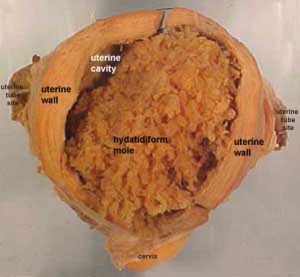
Hydatidiform Mole
Another type of abnormality is when only the conceptus trophoblast layers proliferates and not the embryoblast, no embryo develops, this is called a "hydatidiform mole", which is due to the continuing presence of the trophoblastic layer, this abnormal conceptus can also implant in the uterus. The trophoblast cells will secrete human chorionic gonadotropin (hCG), as in a normal pregnancy, and may appear maternally and by pregnancy test to be "normal". Prenatal diagnosis by ultrasound analysis demonstrates the absence of a embryo.
There are several forms of hydatidiform mole: partial mole, complete mole and persistent gestational trophoblastic tumor. Many of these tumours arise from a haploid sperm fertilizing an egg without a female pronucleus (the alternative form, an embryo without sperm contribution, is called parthenogenesis). The tumour has a "grape-like" placental appearance without enclosed embryo formation. Following a first molar pregnancy, there is approximately a 1% risk of a second molar pregnancy.
This topic is also covered in Placenta - Abnormalities
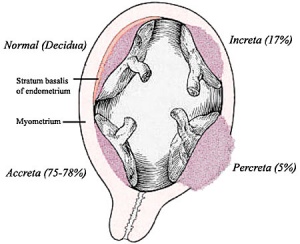
Abnormal Placentation
Abnormalities can range from anatomical associated with degree or site of inplantation, structure (as with twinning), to placental function, placento-maternal effects (pre-eclampsia, fetal erythroblastosis) and finally mechanical abnormalities associated with the placental (umbilical) cord.
This topic is also covered in Placenta - Abnormalities
References
- ↑ 1.0 1.1 <pubmed>18046411</pubmed>
- ↑ <pubmed>10362823</pubmed>
- ↑ <pubmed>20729534</pubmed>
- ↑ <pubmed>20708167</pubmed>
- ↑ Tamura N, Sugihara K, Akama TO, Fukuda MN. Trophinin-mediated cell adhesion induces apoptosis of human endometrial epithelial cells through PKC-δ. Cell Cycle. 2011 Jan 1;10(1):135-43 PMID21191175
- ↑ <pubmed>19213836</pubmed>
- ↑ <pubmed>18506087</pubmed>
- ↑ <pubmed>20640155</pubmed>| PMC2904919
- ↑ <pubmed>18997181</pubmed>
- ↑ <pubmed>11098008</pubmed>
- ↑ <pubmed>20555025</pubmed>
- ↑ <pubmed>20729537</pubmed>
Reviews
<pubmed>18412772</pubmed>
Articles
<pubmed>17495530</pubmed>| PDF
Search PubMed
Search Pubmed: Embryo Adplantation | Embryo Implantation | tubal pregnancy | Placenta Abnormalities | Pinopods | decidualization
Embryo Week: Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | Week 9
- Carnegie Stages: 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | About Stages | Timeline
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, June 10) Embryology Implantation. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Implantation
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G