Genital System Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (25 intermediate revisions by the same user not shown) | |||
| Line 9: | Line 9: | ||
<br> | <br> | ||
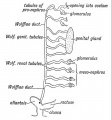
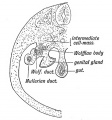
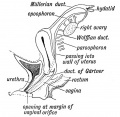
The paired '''mesonephric | The paired '''{{mesonephric duct}}s''' ([[History_-_Embryologists#Caspar_Friedrich_Wolff_.281733_-_1794.29|Wolffian ducts]]) and '''paramesonephric ducts''' ([[History_-_Embryologists#Johannes_Peter_M.C3.BCller_.281801_-_1858.29|Müllerian ducts]]) contribute the majority of male and female internal genital tract respectively. | ||
Development of this system commences in the embryo, continues through the fetal period then with key changes around birth, only completes functional development postnatally at puberty. The mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while. | Development of this system commences in the embryo, continues through the fetal period then with key changes around birth, only completes functional development postnatally at puberty. The mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while. The term "{{gonad}}" is used to refer to both the {{ovary}} and {{testis}}. | ||
| Line 28: | Line 28: | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | * '''{{Fox}}a1 and {{Fox}}a2 orchestrate development of the {{urethra}}l tube and division of the embryonic cloaca through an autoregulatory loop with {{Shh}}'''{{#pmid:32645357|PMID32645357}} "Congenital anomalies of external genitalia affect approximately 1 in 125 live male births. Development of the genital tubercle, the precursor of the {{penis}} and clitoris, is regulated by the urethral plate epithelium, an endodermal signaling center. Signaling activity of the urethral plate is mediated by Sonic hedgehog (SHH), which coordinates outgrowth and patterning of the genital tubercle by controlling cell cycle kinetics and expression of downstream genes. The mechanisms that govern Shh transcription in urethral plate cells are largely unknown. Here we show that deletion of {{Fox}}a1 and {{Fox}}a2 results in persistent cloaca, an incomplete separation of urinary, genital, and anorectal tracts, and severe {{hypospadias}}, a failure of urethral tubulogenesis. Loss of Foxa2 and only one copy of Foxa1 results in urethral fistula, an additional opening of the penile urethra. Foxa1/a2 participate in an autoregulatory feedback loop with Shh, in which {{FOX}}A1 and {{FOX}}A2 positively regulate transcription of {{Shh}} in the urethra, and {{SHH}} feeds back to negatively regulate Foxa1 and Foxa2 expression. These findings reveal novel roles for Foxa genes in development of the urethral tube and in division of the embryonic cloaca." | ||
* ''' | * '''Bmp4 is an essential growth factor for the initiation of genital tubercle (GT) outgrowth'''{{#pmid:30714224|PMID30714224}} "The external genitalia are appendage organs outgrowing from the posterior body trunk. Murine genital tubercle (GT), anlage of external genitalia, initiates its outgrowth from embryonic day (E) {{ME10.5}} as a bud structure. Several growth factors such as fibroblast growth factor (FGF), Wnt and Sonic hedgehog ({{Shh}}) are essential for the GT outgrowth. However, the mechanisms of initiation of GT outgrowth are poorly understood. We previously identified bone morphogenetic protein ({{Bmp}}) signaling as a negative regulator for GT outgrowth. We show here novel aspects of Bmp4 functions for GT outgrowth. We identified the Bmp4 was already expressed in cloaca region at E9.5, before GT outgrowth. To analyze the function of Bmp4 at early stage for the initiation of GT outgrowth, we utilized the Hoxa3-Cre driver and Bmp4 flox/flox mouse lines. Hoxa3 Cre/+ ; Bmp4 flox/flox mutant mice showed the hypoplasia of GT with reduced expression of outgrowth promoting genes such as Wnt5a, Hoxd13 and p63, whereas Shh expression was not affected. Formation of distal urethral epithelium (DUE) marked by the Fgf8 expression is essential for controlling mesenchymal genes expression in GT and subsequent its outgrowth. Furthermore, Fgf8 expression was dramatically reduced in such mutant mice indicating the defective DUE formation. Hence, current results indicate that Bmp4 is an essential growth factor for the initiation of GT outgrowth independent of Shh signaling. Thus, Bmp4 positively regulates for the formation of DUE. The current study provides new insights into the function of Bmp signaling at early stage for the initiation of GT outgrowth." {{BMP}} | ||
* ''' | * '''Differences of sex development: the road to diagnosis'''{{#pmid:30803928|PMID30803928}} "The diagnosis and management of children born with ambiguous genitalia is challenging for clinicians. Such differences of sex development (DSDs) are congenital conditions in which chromosomal, gonadal, or anatomical sex is atypical. The aetiology of DSDs is very heterogenous and a precise diagnosis is essential for management of genetic, endocrine, surgical, reproductive, and psychosocial issues. In this Review, we outline a step-by-step approach, compiled in a diagnostic algorithm, for the clinical assessment and molecular diagnosis of a patient with ambiguity of the external genitalia on initial presentation. We appraise established and emerging technologies and their effect on diagnosis, and discuss current controversies." | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 41: | Line 40: | ||
| [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | | [[File:Mark_Hill.jpg|90px|left]] {{Most_Recent_Refs}} | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Genital+Embryology ''Genital Embryology''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Genital+Embryology ''Genital Embryology''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Genital+Development ''Genital Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Female+Genital+Development ''Female Genital Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Male+Genital+Development ''Male Genital Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Female+Urogenital+Development ''Female Urogenital Development''] | [http://www.ncbi.nlm.nih.gov/pubmed/?term=Male+Urogenital+Development ''Male Urogenital Development''] | ||
|} | |||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''Immunohistochemical expression analysis of the human fetal lower urogenital tract'''{{#pmid:30287094|PMID30287094}} "We have studied the ontogeny of the developing human {{male}} and {{female}} urogenital tracts from 9 weeks (indifferent stage) to 16 weeks (advanced sex differentiation) of gestation by immunohistochemistry on mid-sagittal sections. Sixteen human fetal pelvises were serial sectioned in the sagittal plane and stained with antibodies to epithelial, muscle, nerve, proliferation and hormone receptor markers. Key findings are: (1) The corpus cavernosum in males and females extends into the glans penis and clitoris, respectively, during the ambisexual stage (9 weeks) and thus appears to be an androgen-independent event. (2) The entire human male (and female) urethra is endodermal in origin based on the presence of {{FOX}}A1, KRT 7, uroplakin, and the absence of KRT10 staining. The endoderm of the urethra interfaces with ectodermal epidermis at the site of the urethral meatus. (3) The surface epithelium of the verumontanum is {{endoderm}}al in origin (FOXA1-positive) with a possible contribution of Pax2-positive epithelial cells implying additional input from the Wolffian duct epithelium. (4) Prostatic ducts arise from the endodermal (FOXA1-positive) urogenital sinus epithelium near the verumontanum. (5) Immunohistochemical staining of mid-sagittal and para-sagittal sections revealed the external anal sphincter, levator ani, bulbospongiosus muscle and the anatomic relationships between these developing skeletal muscles and organs of the {{male}} and {{female}} reproductive tracts." | |||
* '''Expression analysis identifies cascades of activation and repression and maps a putative regulator of mammalian sex determination'''{{#pmid:23874228|PMID23874228}} "In vertebrates, primary sex determination refers to the decision within a bipotential organ precursor to differentiate as a testis or ovary. Bifurcation of organ fate begins between embryonic day (E) 11.0-E12.0 in mice and likely involves a dynamic transcription network that is poorly understood. ...We provide strong evidence that Lmo4 (Lim-domain only 4) is a novel regulator of sex determination upstream of SF1 (Nr5a1), {{Sox}}9, {{Fgf}}9, and Col9a3. This approach can be readily applied to identify regulatory interactions in other systems." | |||
* '''Male reproductive tract abnormalities: More common after assisted reproduction?'''{{#pmid:20674196|PMID20674196}} "IVF and ICSI, by increasing the risks of prematurity, low birthweight, and multiple gestation, are indirect risk factors for developing male genital malformations. In infants with normal birhtweight or from singleton pregnancies, ICSI is a specific risk factor for hypospadias." | |||
* '''Temporal and spatial dissection of Shh signaling in genital tubercle development.'''{{#pmid:19906863|PMID19906863}} "Genital tubercle (GT) initiation and outgrowth involve coordinated morphogenesis of surface ectoderm, cloacal mesoderm and hindgut endoderm. GT development appears to mirror that of the limb. Although Shh is essential for the development of both appendages, its role in GT development is much less clear than in the limb. Here, by removing Shh at different stages during GT development in mice, we demonstrate a continuous requirement for Shh in GT initiation and subsequent androgen-independent GT growth." | |||
* '''Bmp7 expression and null phenotype in the urogenital system suggest a role in re-organization of the urethral epithelium.'''{{#pmid:19159697|PMID19159697}} "Signaling by Bone morphogenetic proteins (Bmps) has multiple and diverse roles in patterning and morphogenesis of the kidney, eye, limbs and the neural tube. ...Together, our analysis of Bmp7 expression and the null phenotype, indicates that Bmp7 may play an important role in re-organization of the epithelium during cloacal septation and morphogenesis of the genital tubercle." | |||
|} | |} | ||
== Textbooks == | == Textbooks == | ||
| Line 78: | Line 89: | ||
{{Genital cartoons}} | {{Genital cartoons}} | ||
== Development Overview == | == Development Overview == | ||
| Line 99: | Line 102: | ||
==Gender by Ultrasound== | ==Gender by Ultrasound== | ||
A 2012 Czech ultrasound study | A 2012 Czech ultrasound study{{#pmid:22660228|PMID22660228}} of 1222 singleton pregnancies has attempted to determine the earliest gestational age {{GA}} that fetal gender may reliably be determined. Their study concluded "when CRL ≥ 60 mm (gestational age ≥ 12+2). Male gender may already be reliably determined when CRL ≥ 55 mm (gestational age ≥ 12+0). If CRL < 50 mm (gestational age < 11+4) the gender cannot be reliably predicted." | ||
:'''Links:''' | :'''Links:''' {{ultrasound}} | ||
==Sexual Development Genes== | ==Sexual Development Genes== | ||
Table below modified from [http://physrev.physiology.org/content/87/1/1/T1.expansion.html Table 1. Genes implicated in sexual development in mammals] in | Table below modified from [http://physrev.physiology.org/content/87/1/1/T1.expansion.html Table 1. Genes implicated in sexual development in mammals] in a review article.{{#pmid:17237341|PMID17237341}} | ||
{{Sex development genes table}} | {{Sex development genes table}} | ||
===Orphan Nuclear Receptors=== | |||
Orphan nuclear receptors are proteins with a nuclear receptor domain structure that lack identified signaling ligands. | |||
Steroidogenic factor-1 (NR5A1, SF-1) and liver receptor homolog-1 (NR5A2, LRH-1) both bind to the same DNA sequences, with different and non-overlapping effects on targets (see review{{#pmid:30810078|PMID30810078}}). | |||
* SF-1 is expressed mainly in steroidogenic tissues - adrenal development, sexual differentiation, and Leydig cell function | |||
* LRH-1 in tissues of endodermal origin and gonads - development beyond gastrulation | |||
==Human Genital== | |||
Studies of human genital development are covered in several early historical studies<ref name=Pohlman1911>{{Ref-Pohlman1911}}</ref><ref name=Wood-Jones1914>{{Ref-Wood-Jones1914}}</ref><ref name=Spaulding1921>{{Ref-Spaulding1921}}</ref><ref name=Wilson1926a>{{Ref-Wilson1926a}}</ref><ref name=Wilson1926b>{{Ref-Wilson1926b}}</ref><ref name=Koff1933>{{Ref-Koff1933}}</ref> and a later 1980's review article based on the [[Carnegie Collection]] embryos.{{#pmid:6846859|PMID6846859}} | |||
<br> | |||
{{Human Embryo Genital table}} | |||
<br> | |||
==Animal Models== | ==Animal Models== | ||
| Line 114: | Line 133: | ||
[[File:Mouse gonad development timeline.jpg|600px]] | [[File:Mouse gonad development timeline.jpg|600px]] | ||
Mouse E11.0 to E12.0 shows the critical transition in the gonad from a bipotential to sexually-differentiated state. Based upon transcriptome analysis. | Mouse E11.0 to E12.0 shows the critical transition in the gonad from a bipotential to sexually-differentiated state. Based upon transcriptome analysis.{{#pmid:23874228|PMID23874228}} | ||
==Historic== | ==Historic== | ||
| Line 199: | Line 218: | ||
===Reviews=== | ===Reviews=== | ||
{{#pmid:30810078}} | |||
{{#pmid:27893151}} | |||
{{#pmid:21465625}} | |||
{{#pmid:18460940}} | |||
===Articles=== | ===Articles=== | ||
| Line 214: | Line 238: | ||
{{Genital terms}} | {{Genital terms}} | ||
{{ | {{Glossary}} | ||
{{Systems}} | {{Systems}} | ||
{{Footer}} | {{Footer}} | ||
[[Category:System Development]] | [[Category:System Development]] | ||
Latest revision as of 23:37, 24 August 2020
| Embryology - 19 Jun 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The male and female reproductive systems develop initially embryonically "indifferent", it is the product of the Y chromosome SRY gene that makes the "difference".
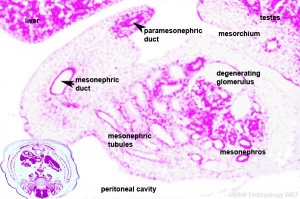
The paired mesonephric ducts (Wolffian ducts) and paramesonephric ducts (Müllerian ducts) contribute the majority of male and female internal genital tract respectively.
Development of this system commences in the embryo, continues through the fetal period then with key changes around birth, only completes functional development postnatally at puberty. The mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while. The term "gonad" is used to refer to both the ovary and testis.
There are many different issues to consider in the development of the genital system. Importantly its sex chromosome dependence, late embryonic/fetal differential development, complex morphogenic changes, long time-course, hormonal sensitivity and hormonal influences make it a system prone to many different abnormalities.
This current page provides only a general introduction to the topic, use the links listed below to read about specific developmental topics.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Genital Embryology | Genital Development | Female Genital Development | Male Genital Development | Female Urogenital Development | Male Urogenital Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 10 p261-306
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 13 p303-346
- Before We Are Born (5th ed.) Moore and Persaud Chapter 14 p289-326
- Essentials of Human Embryology, Larson Chapter 10 p173-205
- Human Embryology, Fitzgerald and Fitzgerald Chapter 21-22 p134-152
- Developmental Biology (6th ed.) Gilbert Chapter 14 Intermediate Mesoderm
| UNSW Students | |
|---|---|

|
You have access the following online Embryology textbooks through the UNSW Library. |

|
Moore, K.L. & Persuad, T.V.N. (2008). The Developing Human: clinically oriented embryology (8th ed.). Philadelphia: Saunders. |

|
Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R. and Francis-West, P.H. (2009). Larsen’s Human Embryology (4th ed.). New York; Edinburgh: Churchill Livingstone. |
Objectives
- Understand the role of the Y chromosome in sex determination.
- Understand the differences in male/female duct develpoment (mesonephric/paramesonephric).
- Compare the development of the cloaca in the male and female.
- Understand the developmental abnormalities in male and female development.
Movies
| Genital Movies | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
| |||||||||||||||
|
|
| |||||||||||||||||
| Mouse Primordial Germ Cell Migration | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
| |||||||||
Development Overview
Three main stages during development, mesonephric/paramesonephric duct changes are one of the first male/female differences that occur in development, while external genitaila remain indeterminate in appearance for quite a while.
- Differentiation of gonad (Sex determination)
- Differentiation of internal genital organs
- Differentiation of external genital organs
The 2nd and 3rd stages dependent on endocrine gonad. Reproductive development has a long maturation timecourse, begining in the embryo and finishing in puberty. (More? Puberty Development)
Gender by Ultrasound
A 2012 Czech ultrasound study[9] of 1222 singleton pregnancies has attempted to determine the earliest gestational age GA that fetal gender may reliably be determined. Their study concluded "when CRL ≥ 60 mm (gestational age ≥ 12+2). Male gender may already be reliably determined when CRL ≥ 55 mm (gestational age ≥ 12+0). If CRL < 50 mm (gestational age < 11+4) the gender cannot be reliably predicted."
- Links: ultrasound
Sexual Development Genes
Table below modified from Table 1. Genes implicated in sexual development in mammals in a review article.[10]
| Gene (OMIM) | Protein Function | Gonad Phenotype of Null Mice | Human Syndrome | |
| Bipotential gonad | ||||
| Wt1 | Transcription factor | Blockage in genital ridge development | Denys-Drash, WAGR, Frasier syndrome | |
| Sf1 | Nuclear receptor | Blockage in genital ridge development | Embryonic testicular regression syndrome | |
| Lhx9 | Transcription factor | Blockage in genital ridge development | a | |
| Emx2 | Transcription factor | Blockage in genital ridge development | a | |
| M33 | Transcription factor | Gonadal dysgenesis | a | |
| Testis-determining pathway | ||||
| Gata4/Fog2 | Transcription/cofactor | Reduced Sry levels, XY sex reversal | a | |
| Sry | Transcription factor | XY sex reversal | XY sex reversal (LOF); XX sex reversal (GOF) | |
| Sox9 | Transcription factor | XY sex reversal | Campomelic dysplasia, XX sex reversal (GOF) | |
| Sox8 | Transcription factor | XY sex reversal in combination with partial loss of Sox9 function | a | |
| Fgf9 | Signaling molecule | XY sex reversal | a | |
| Dax1 | Nuclear receptor | Impaired testis cord formation and spermatogenesis | Hypogonadism | |
| Pod1 | Transcription factor | XY sex reversal | a | |
| Dhh | Signaling molecule | Impaired differentiation of Leydig and PM cells | XY gonadal dysgenesis | |
| Pgdra | Receptor | Reduction in mesonephric cell migration | a | |
| Pgds | Enzyme | No phenotype | a | |
| Arx | Transcription factor | Abnormal testicular differentiation | X-linked lissencephaly with abnormal genitalia | |
| Atrx | Helicase | ND | ATRX syndrome | |
| Insl3 | Signaling factor | Blockage of testicular descent | Cryptorchidism | |
| Lgr8 | Receptor | Blockage of testicular descent | Cryptorchidism | |
| Hoxa10 | Transcription factor | Blockage of testicular descent | Cryptorchidism | |
| Hoxa11 | Transcription factor | Blockage of testicular descent | Cryptorchidism | |
| Amh | Hormone | No Müllerian duct degeneration | Persistent Müllerian duct syndrome | |
| Misrl1 | Receptor | No Müllerian duct degeneration | Persistent Müllerian duct syndrome | |
| Pax2 | Transcription factor | Dysgenesis of mesonephric tubules | a | |
| Lim1 | Transcription factor | Agenesis of Wolffian and Müllerian ducts | a | |
| Dmrt1 | Transcription factor | Loss of Sertoli and germ cells | XY femaleb | |
| Ovary-determining pathway | ||||
| Wnt4 | Signaling molecule | Müllerian duct agenesis, testosterone synthesis, and coelomic vessel formation | XY female (GOF) | |
| FoxL2 | Transcription factor | Premature ovarian failure | BPES | |
| Dax1 | Nuclear receptor | XY sex reversal (GOF) | XY sex reversal (GOF) | |
| RSPO1 | Signaling molecule | XX sex reversal (LOF) | XX sex reversal (LOF) | |
| Table Legend | ||||
|
a No mutations in human sexual disorders identified to date.
b Candidate gene for 9p deletion, XY sex reversal. | |||
| Table data modified[10] | ||||
Orphan Nuclear Receptors
Orphan nuclear receptors are proteins with a nuclear receptor domain structure that lack identified signaling ligands.
Steroidogenic factor-1 (NR5A1, SF-1) and liver receptor homolog-1 (NR5A2, LRH-1) both bind to the same DNA sequences, with different and non-overlapping effects on targets (see review[11]).
- SF-1 is expressed mainly in steroidogenic tissues - adrenal development, sexual differentiation, and Leydig cell function
- LRH-1 in tissues of endodermal origin and gonads - development beyond gastrulation
Human Genital
Studies of human genital development are covered in several early historical studies[12][13][14][15][16][17] and a later 1980's review article based on the Carnegie Collection embryos.[18]
| Structure | Stage Range | Carnegie Stages (Embryo No.) | Study |
|---|---|---|---|
| Cloaca | 11 - 19 | 11 (164), 13 (186}, 14 (80), 15 (2), 16 (221) 19 (43) | Pohlman (1911)[12] |
| Rete | 16 - 23 | 16 (1836), 17 (544), 18 (423; 511; 841), 19 (432), 20 (368; 460), 21 (22; 455; 2937), 23 (75; 782; 1945) | Wilson {1926a}[15] |
| External genitalia | 20, 23 | 20 (2393) 23 (950) | Wilson (1926b)[16] |
| vagina | 17 - 23 | 17 (353), 20 (966), 22 (584A; 4304; 4339; 4638), 23 (4205; 4289; 5725). | Koff (1933)[17] |
| external genital | embryonic to fetal | 16 (792); 18 (492}; to fetal 100mm | Spaulding (1921)[14] |
| pelvis | 15 - 23 | 15, 16, 17, 18, 23 (no embryo numbers available) | Pillet (1967-1971)[19][20][21][22] |
| Some data from[18] | Links: genital | Carnegie Collection |
Animal Models
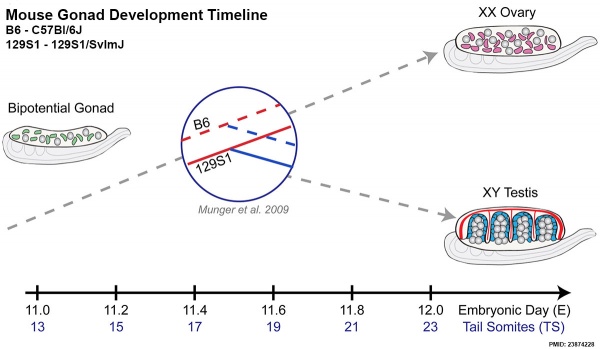
Mouse
Mouse E11.0 to E12.0 shows the critical transition in the gonad from a bipotential to sexually-differentiated state. Based upon transcriptome analysis.[5]
Historic
See also section Historic Embryology Images.
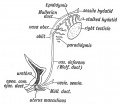
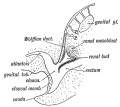
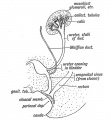
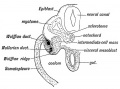
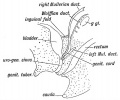
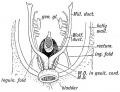
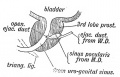
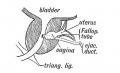
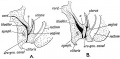
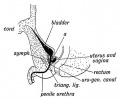
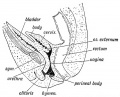
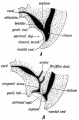
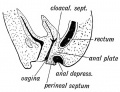
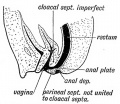
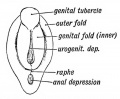
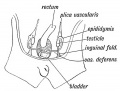
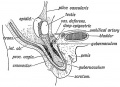
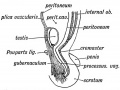
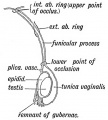
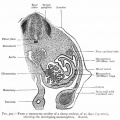
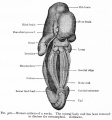
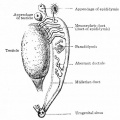
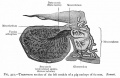
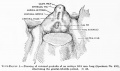
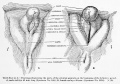
Historic Images of Genital Changes
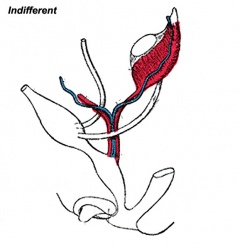
|
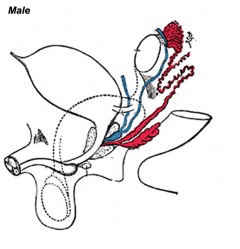
|
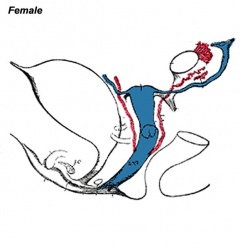
|
| Urogenital indifferent | Urogenital male | Urogenital female |
Additional Images
Historic Embryology Images
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
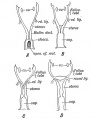
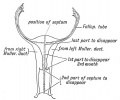
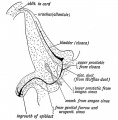
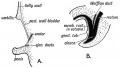
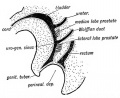
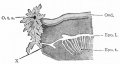
Keith, A. (1902) Human Embryology and Morphology. London: Edward Arnold.
Chapter 9 - The Uro-genital System
- The Uro-genital System: Fig. 79. Wolffian Body | Fig. 80. Wolffian and Genital Ridges | Fig. 81. Female Wolffian Body Remnants | Fig. 82. Male Wolffian Body Remnants |Fig. 83. Renal Bud | Fig. 84. Ureter in the Bladder | Fig. 85. Wolffian and Müllerian Ducts | Fig. 86. Genital Ducts 3rd month | Fig. 87. Müllerian Ducts 3rd month | Fig. 88. Uterus | Fig. 89. Uterus and Vagina | Fig. 90. Prostate remnants of Müllerian Ducts | Fig. 91. Prostate showing an unusual Uterus Masculinus | Fig. 92. Female Uro-genital Sinus | Fig. 93. Male Uro-genital Sinus | Fig. 94. Vagina and Uterus at 7th month | Fig. 95. Division of the Cloaca | Fig. 96. Imperforate Anus | Fig. 97. Cloacal Septum has failed to fuse with Perineal Septum | Fig. 98. The Uro-genital Cleft 2nd month | Fig. 99. Male bladder and urethra at birth | Fig. 100. Ectopia Vesicae | Fig. 101. Prostatic Tubules | Fig. 102. Testis in a foetus of 2£ months | Fig. 103. Testis at the 6th month | Fig. 104. Inguinal Canal and Coverings of the Testis | Fig. 105. Processus Vaginalis | Figures
References
- ↑ Gredler ML, Patterson SE, Seifert AW & Cohn MJ. (2020). Foxa1 and Foxa2 orchestrate development of the urethral tube and division of the embryonic cloaca through an autoregulatory loop with Shh. Dev. Biol. , 465, 23-30. PMID: 32645357 DOI.
- ↑ Kajioka D, Suzuki K, Nakada S, Matsushita S, Miyagawa S, Takeo T, Nakagata N & Yamada G. (2019). Bmp4 is an essential growth factor for the initiation of genital tubercle (GT) outgrowth. Congenit Anom (Kyoto) , , . PMID: 30714224 DOI.
- ↑ León NY, Reyes AP & Harley VR. (2019). Differences of sex development: the road to diagnosis. Lancet Diabetes Endocrinol , , . PMID: 30803928 DOI.
- ↑ Shen J, Isaacson D, Cao M, Sinclair A, Cunha GR & Baskin L. (2018). Immunohistochemical expression analysis of the human fetal lower urogenital tract. Differentiation , 103, 100-119. PMID: 30287094 DOI.
- ↑ 5.0 5.1 Munger SC, Natarajan A, Looger LL, Ohler U & Capel B. (2013). Fine time course expression analysis identifies cascades of activation and repression and maps a putative regulator of mammalian sex determination. PLoS Genet. , 9, e1003630. PMID: 23874228 DOI.
- ↑ Funke S, Flach E, Kiss I, Sándor J, Vida G, Bódis J & Ertl T. (2010). Male reproductive tract abnormalities: more common after assisted reproduction?. Early Hum. Dev. , 86, 547-50. PMID: 20674196 DOI.
- ↑ Lin C, Yin Y, Veith GM, Fisher AV, Long F & Ma L. (2009). Temporal and spatial dissection of Shh signaling in genital tubercle development. Development , 136, 3959-67. PMID: 19906863 DOI.
- ↑ Wu X, Ferrara C, Shapiro E & Grishina I. (2009). Bmp7 expression and null phenotype in the urogenital system suggest a role in re-organization of the urethral epithelium. Gene Expr. Patterns , 9, 224-30. PMID: 19159697 DOI.
- ↑ Lubusky M, Studnickova M, Skrivanek A, Vomackova K & Prochazka M. (2012). Ultrasound evaluation of fetal gender at 12-14 weeks. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub , 156, 324-9. PMID: 22660228 DOI.
- ↑ 10.0 10.1 Wilhelm D, Palmer S & Koopman P. (2007). Sex determination and gonadal development in mammals. Physiol. Rev. , 87, 1-28. PMID: 17237341 DOI.
- ↑ Meinsohn MC, Smith OE, Bertolin K & Murphy BD. (2019). The Orphan Nuclear Receptors Steroidogenic Factor-1 and Liver Receptor Homolog-1: Structure, Regulation, and Essential Roles in Mammalian Reproduction. Physiol. Rev. , 99, 1249-1279. PMID: 30810078 DOI.
- ↑ 12.0 12.1 Pohlman AG. The development of the cloaca in human embryos. (1911) Amer. J Anat. 12: 1-26.
- ↑ Wood-Jones F. The morphology of the external genitalia of the mammala. (1914) Lancet. 1017-1023.
- ↑ 14.0 14.1 Spaulding MH. The development of the external genitalia in the human embryo. (1921) Contrib. Embryol., Carnegie Inst. Wash. Publ. 81, 13: 69 – 88.
- ↑ 15.0 15.1 Wilson KM. Origin and development of the rete ovarii and the rete testis in the human embryo. (1926) Carnegie Instn. Wash. Publ. 362, Contrib. Embryol., Carnegie Inst. Wash., 17:69-88.
- ↑ 16.0 16.1 Wilson KM. Correlation of external genitalia and sex-glands in the human embryo. (1926) Carnegie Instn. Wash. Publ. 363, Contrib. Embryol., Carnegie Inst. Wash. 18: 23-30.
- ↑ 17.0 17.1 Koff A. Development of the vagina in the human fetus. (1933) Contrib. Embryol., Carnegie Inst. Wash. Publ. 443, 24: 59-60.
- ↑ 18.0 18.1 O'Rahilly R. (1983). The timing and sequence of events in the development of the human reproductive system during the embryonic period proper. Anat. Embryol. , 166, 247-61. PMID: 6846859
- ↑ Pillet J. Reconstruction des organes pelviens d’embryons humains de 12,5 et de 25 mm CR. (1967) Ass Anat 51: 819-827.
- ↑ Pillet J. Reconstruction du pelvis d’un embryon humain de 7.5 mm (Stade XVI de Streeter) CR. (1968) Ass Anat 52: 1013-1023.
- ↑ Pillet J (1969) Reconstruction des organes génito«-urinaires et des veines pelviennes d’un embryon de 12,5 mm (Stade XVII de Streeter) CR. Ass Anat 53 : 1817-1824.
- ↑ Pillet J. Reconstruction des organes pelviens d’un embryon de 5 mm (Stade XV de Streeter) CR. (1971) Ass Anat 54:705-715
Reviews
Meinsohn MC, Smith OE, Bertolin K & Murphy BD. (2019). The Orphan Nuclear Receptors Steroidogenic Factor-1 and Liver Receptor Homolog-1: Structure, Regulation, and Essential Roles in Mammalian Reproduction. Physiol. Rev. , 99, 1249-1279. PMID: 30810078 DOI.
Bashamboo A, Eozenou C, Rojo S & McElreavey K. (2017). Anomalies in human sex determination provide unique insights into the complex genetic interactions of early gonad development. Clin. Genet. , 91, 143-156. PMID: 27893151 DOI.
Cohn MJ. (2011). Development of the external genitalia: conserved and divergent mechanisms of appendage patterning. Dev. Dyn. , 240, 1108-15. PMID: 21465625 DOI.
Nakhuda GS. (2008). The role of mullerian inhibiting substance in female reproduction. Curr. Opin. Obstet. Gynecol. , 20, 257-64. PMID: 18460940 DOI.
Articles
Search PubMed
Search Pubmed: Genital System Development | Genital Development
NCBI - Policies and Guidelines | PubMed | Help:Reference Tutorial
Terms
| Genital System Terms (expand to view) |
|---|
Note there are additional glossaries associated with spermatozoa, oocyte renal.
|
| Other Terms Lists |
|---|
| Terms Lists: ART | Birth | Bone | Cardiovascular | Cell Division | Endocrine | Gastrointestinal | Genital | Genetic | Head | Hearing | Heart | Immune | Integumentary | Neonatal | Neural | Oocyte | Palate | Placenta | Radiation | Renal | Respiratory | Spermatozoa | Statistics | Tooth | Ultrasound | Vision | Historic | Drugs | Glossary |
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
| System Links: Introduction | Cardiovascular | Coelomic Cavity | Endocrine | Gastrointestinal Tract | Genital | Head | Immune | Integumentary | Musculoskeletal | Neural | Neural Crest | Placenta | Renal | Respiratory | Sensory | Birth |
Cite this page: Hill, M.A. (2024, June 19) Embryology Genital System Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Genital_System_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G