Paper - Chondrification in the hands and feet of staged human embryos
| Embryology - 1 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
O'Rahilly R. Gray DI. and Gardner E. Chondrification in the hands and feet of staged human embryos. (1957) Carnegie Instn. Wash. Publ. 611, Contrib. Embryol., 36:
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Chondrification in the Hands and Feet of Staged Human Embryos
Contributions To Embryology, No. 250
Ronan O'Rahilly, D. I. Gray, and Ernest Gardner
Department; of Anatomy, Wayne State University College of Medicine, Detroit, Michigan, and Stanford University School of Medicine, Stamford, California
With one plate
Introduction
The prenatal development of the skeleton of human limbs has been studied by many investigators. Reports based on their studies have related mainly to ossification, however, as in the recent papers by Noback and Robertson (1951) and O’Rahilly and Meyer (I957). Comparable attention has not been given to the earlier development of the skeletal elements, particularly to those of the hands and feet. Apart from Senior’s (I929) abstract, no adequate account has been published of the onset and sequence of chondrification in a large series of hands and feet.
Investigators generally agree that chondrification begins in the metacarpals before it begins in the carpals, and that the Iunate and pisiform are the last of the carpals to begin to chondrify (Henke and Reyher, 1874; Rosenberg, 1876; Leboucq, I884; Griifenberg, 1905; Martin, I929). A few authors have reported that they could distinguish carpal elements in the blastemal stage (Lewis, 1902; Schmidt-Ehrenberg, 1942; Haines, 1947). Chondrification begins in the proximal phalanges, and extends progressively to the most distal members (Henke and Reyhcr, 1874; Hagen, 1900; Lewis, 1902; Griifenberg, I905; Hesser, 1926). In a 27—mm. embryo, Schulin (1879) found that all the skeletal elements of the hand were ehondrified except the sesamoids.
It is also generally agreed that the digits of the foot, like those of the hand, chondrify in a proximodistal sequence (Henke and Reyhcr, 1874; Schulin, 1879; Schomburg, I900; Bardeen and Lewis, 1901; Leboucq, I904; Bardeen, 1905; Martin, 1929; Trolle, 1948). Several authors have reported chondrification in the foot beginning in embryos of about 14 to 21 mm. in length (Henke and Reyhcr, 1874; liaur, 1886; Bardeen and Lewis, 1901; Bardeen, 1905; Martin, 1929; Hintzsche, 1930; Haines, 1947; Trolle, 1948), but none of these writers has given information regarding the sequence of chendrification in the tarsus. Leboucq (1886) stated that he found the tarsal elements differentiated in a 12-mm. embryo, but it is not clear from his account whether he intended to use “differentiated” as synonymous with “chondrified.” Senior (1929) was the first to report a precise sequence in chondrification in both hand and foot. In an abstract based on a study of 77 embryos, ranging from 12.5 to 24.8 mm. crown-rump length, he reported that with few exceptions the hands and feet fit into one of the stages
- This investigation was supported by research grants A—55o (C2) and 13-532 (C7) from the National Institute of Arthritis and Metabolic Diseases, of the National Institutes of Health, U. S. Public Health Service.
listed in tables I and 2, respectively. Senior pointed out that “chondrification, being cumulative, affords an excellent guide to the relative stage of development of embryonic limbs while the increase in the number of centers lasts.” Streeter (1929) considered using the order of chondrification established by Senior as an aid in staging human embryos. Unfortunately, however, Senior did not publish a complete account of his findings, and attempts to locate his original data have been unsuccessful.
Table 1
| Senior's Stages of Chrondrification in the Hand | |
|---|---|
| Stage | (Numerals refer to digits) |
| 1 | Hamate, capitate, metacarpals 2 to 4 |
| 2 | Metacarpal 5 |
| 3 | Trapezoid |
| 4 | Proximal phalanges 2 to 4 |
| 5 | Triquetrum, trapezium, metacarpal 1, proximal phalanx |
| 6 | Scaphoid, cenlrale |
| 7 | Proximal phalanx |
| 8 | Lunate |
| 9 | Middle pllalanges 2 to 4 |
| 10 | Middle phalanx 5 |
| 11 | Pisiform |
| 12 | Distal phalanx 1 |
| 13 | Distal phalanges 2 to 4 |
| 14 | Distal phalanx 5 |
Table 2
| Senior's Stages of Chrondrification in the Foot | |
|---|---|
| Stage | (Numerals refer to digits) |
| 1 | Metatarsals 2 to 4 |
| 2 | Cuboid, metatarsal 5 |
| 3 | Calcaneus, talus, lateral cuneiform |
| 4 | Intermediate cuneiform |
| 5 | Medial cuneiform, metatarsal 1 |
| 6 | Navicular |
| 7 | Proximal phalanges 2 to 4 |
| 8 | Proximal phalanx 5 |
| 9 | Proximal phalanx 1 |
| 10 | Middle phalanges 2 to 4 |
| 11 | Middle phalanx 5 |
| 12 | Distal phalanx 1 |
| 13 | Distal pbalanges 2 to 4 |
| 14 | Distal phalanx 5 |
Correlation of Senior’s stages of chondrification in both hand and foot with Streeter’s (1942, 1945, 1948, 1949, 1951) developmental horizons seems desirable. For this purpose, a study was made of embryos in the collection of the Department of Embryology, Carnegie Institution of Washington. The writers are indebted to Dr. George W. Corner, former Director, for the opportunity to undertake this work.
Materials and Methods
Serial sections of the limbs of 103 staged human embryos, belonging to horizons xv to xxiii inclusive and ranging from approximately 61/; to 30 mm. in length, were studied. In the case of the upper limb, 102 sectioned embryos were examined; lower limbs of 101 embryos were analyzed.
Since special staining techniques for cartilage matrix had not been used in the preparation of the embryos, it was necessary to establish arbitrary histological criteria for the onset of chondrification. In this study, a blastemal condensation of a skeletal element consists of closely packed cells having indistinct boundaries. When the separation of nuclei in a localized part of a condensation results in a lighter appearance, matrix is presumed to have been deposited in this area, and chondrification is said to have begun.
In the carpals and tarsals, chondrification usually begins near the center, and a more or less rounded ebondrified area surrounded by blastemal tissue is formed. This area then spreads until it reaches the periphery of the blastemal condensation, at which time a perichondrium may be said to exist. Further growth depends on the multiplication of perichondral as well as of cartilage cells, and on the formation of matrix. Chondrification is basically similar in long bones. In the metacarpals and metatarsals, as in other long bones, Streeter’s (1949) five phases in the growth of cartilage can be recognized.
The term “hand” as used throughout this paper includes the carpus, metacarpus, and phalanges. The terminology employed is the Nominu anatomica emandata of 1955, translated into English where appropriate.
Observations
Upper Limb
Horizon xv (6.0 to 11.0 mm.). five embryos examined: Carnegie nos. 2, 721, 650.}, 6506, 8997.
A central, mesenchymal condensation for muscle and bone in the proximal part of the limbs was present in two specimens and absent in three. The central part of this condensation was avascular. A photomicrograph illustrating a section of the upper limb at this stage was reproduced by Streeter (1949, fig. 8, pl. 1). An ectodermal ridge, first visible in the previous horizon, was also found at this stage (O’Rahilly, Gardner, and Gray, 1956).
Horizon xvi (7.0 to 12.2 mm.). Ten embryos examined: Carnegie nos. 163, 617, 6507, 6509, 6510, 6512, 6517, 7115, 8112, 8773.
The humerus, radius, and ulna could be distinguished as mesenchymal condensations in most of these embryos. Early chondrification of the humerus was noted in one. The appearance of the humerus during early chondrification is shown in a photomicrograph by Streeter (1949, fig- 9. 121- 1) Horizon xvii (8.6 to 14.5 mm.). Twenty-one embryos examined: Carnegie nos. 353 (1), 559, 623, 940 (1), 1771, 5642 (1), 5893 (1), 6258, 6519, 6520 (1?), 6521, 6631 (1), 6742, 758, 8101, 8118, 8253, 8789 (1), 8969, 8998, 9100. (figures in parentheses in lists of embryos examined in this and subsequent horizons are Senior’s stages of chondrifieation for the hands.) The humerus and radius were chondrifying in almost all these embryos. Although the ulna was chondrifying in many of them, ehondrification was entirely absent in two embryos and doubtful in two others. The radius and ulna were short and widely separated in embryos in which they were chondrifying (fig. 1). Their distal parts, still composed of condensed mesenchyme, were continuous with a common skeletal blastemal mass from which digital prolongations extended (fig. 1). The blastemal condensation of the thumb was very short or absent; that of the fifth digit was short.
Chondrification had not yet begun in the hands of approximately two-thirds of the embryos. In the remaining third, it was beginning in the 3d and 4th metacarpals (fig. 1); the hands of these embryos, therefore, are in early Senior’s stage 1, a stage i11 which Senior included chondrification of the 2d metacarpal and the hamate and capitate. Aside from three embryos in which chondrification had possibly begun in the hamate and capitate, none was found in the carpus. The ectodermal ridge, present since horizon xiv, disappeared during horizon xvii.
Horizon xuiii (11.7 to 18.0 mm.). Seventeen embryos examined: Carnegie nos. 106 (1), 109 (1), 14,4 (6), 175 (2-3). 492 (2), 4430 (5), 6522 (4-5), 6524 (4). 6525 (4). 6527 (4), 6528 (2), 6533 (4). 7707 (2). 3097 (I), 8172 (4). 8355 (I), 9247 (3) The radius and ulna were widely separated from each other, their distal parts still composed of condensed mesenchyme. In the embryos in which the carpus was still mainly blastemal, localized variations in density were noticed, but individual carpal elements were not identified on the basis of these difierences. In the embryos in which several carpal elements had begun to chondrify, however, mesenchymal condensations of the remaining carpal elements were usually distinguishable. Nonsegmented mesenchymal condensations extended distally from the common carpal blastema into the digits in which chondrification had not yet begun.
On the basis of chondrification in the hand, most embryos ranged from an early Senior’s stage 1 to stage 4. although two were found to be in stage 5 and one in stage 6. Four specimens were classified as early stage I because, although they included chondrification in the 3d and 4th metacarpals, one or more of the other elements (2d metacarpal, capitate, hamate) assigned by Senior to this period had not yet begun to chondrify. In the specimen most advanced in chondrification (Seni0r’s stage 6), the following elements were ehondrifying: all carpals except the lunate and pisiform, all five metacarpals, and the proximal phalanges of the 2d to 5th fingers. figure 2 illustrates chondrification at Senior’s stage 5 i11 a hand of an embryo not included in the seventeen listed above.
Horizon xix (15.5 to 21.0 mm.). Twelve embryos ex amined: Carnegie nos. 17 (11), 43 (7), 409 (9), 432 (7), 576 (3).I324 (7-8).I39o (8-9).2II4 (6).440s (7).s6o9 (99). 6150 (9-10?). 8965 (7-9) Streeter’s cartilage phases 1 to 3 were present in the radius and ulna. The styloid processes of both these elements were chondrifying in some specimens, were mesenchymal in others, and were not recognizable in still others. When the styloid process of the ulna was observed, it was very closely related to the triquetrum.
Although a few of the embryos in this age group exhibited chondrification ranging from Senior’s stages 6 to 11, the chondrification of most members of the group could be classified in stages 7 to 9. (By stage 11, all skeletal elements except the distal phalanges have started to chondrify; the eentrale is chondrified by stage 6.) The ehondrific centers i11 the capitate and hamate were larger than those in the other carpals, whereas relatively small areas of the trapezium and trapezoid were chondrified. N0 close correlation was noted between Streeter's point scores for the entire embryo and Senior’s stages of chondrification for the hand, except that the two embryos most advanced in development had the higher Senior stages.
Horizon xx (18.5 to 25.0 mm.). Eleven embryos examined: Carnegie nos. 431 (12), 460 (10-11), 462 (11), 966 (10), 4148 (9-10), 5537 (10), 6202 (11), 6426 (11), 7274 (9).79o6 (9?).8Is7 (12) The humerus, radius, and ulna of some specimens contained phase 4 cartilage.iThe styloid processes of the radius and ulna were mesenchymal in some embryos, whereas they had begun to chondrify in others. The styloid process of the ulna was closely related to the triquetrum in all embryos of this age group. I11 three specimens, the styloid process of the 3d metacarpal was identified, and in one it had begun to chondrify. Chondrification in the hands ranged from Senior's stages 9 to 12. (By stage 12, all skeletal elements except the distal phalanges of the 2d to 5th digits are chondrifying.) The lunate was consistently less advanced in chondrification than the other carpal elements.
Horizon xxi (19.0 to 26.4 mm.). Fourteen embryos examined: Carnegie nos. 22 (13-14), 455 (14), 903C (I4). I008 (I4): I3s8I’ (I4). 2937 (I4): 4090 (13-14), 4960 (I4),5s96 (I4):653I (I2-14).72s4 (II):7392 (I4), 7864 (I3).8s53 (I4) The humerus had a primary bone collar in at least three specimens, the radius in one. Phase 3 cartilage was generally present in the metacarpals and proximal phalanges. The styloid process of the ulna was hook-shaped and chondrified in most specimens. The hook of the hamate appeared as a cellular condensation. Chondrification in the hands ranged from Senior’s stages 11 to 14, the majority of hands belonging in stage 14. Only one specimen was at stage 11. (All skeletal elements of the hand have begun to chondrify by stage 14.) The lunate and pisiform were less advanced in chondrification than the other carpal elements. The centrale had begun to fuse with the scaphoid in some specimens.
Horizon xxii (23.0 to 27.5 mm.). five embryos examined: Carnegie nos. 1458, 3681, 4339, 4638, 8394.
The humerus of all specimens, and the radius and ulna of most, had a bone collar. By horizon xxii, all skeletal elements were chondrifying. Of the distal phalanges, the 1st was generally the most advanced in chondrification. As in the previous horizon, the eentrale had begun to fuse with the scaphoid in some specimens.
Horizon xxiii (23.0 to 32.2 mm.). Seven embryos examined: Carnegie nos. 75, 86, 1945, 2561, 4525, 5621A, 7425 The bone collars of the humerus, radius, and ulna were thicker than in the previous horizon. Calcification had begun in the cartilage matrix, but no cellular, vascular invasion of the bone collar was observed, except in one specimen in which periosteal bttds were beginning to enter the humerus and radius bilaterally. Although cellular condensations of sesamoids were noted in embryos sectioned so as to show these areas favorably, no chondrification was seen in them. The eentrale was partly fused with the scaphoid in all specimens.
Lower Limb
Horizon xv (6.0 to 11.0 mm.). Six embryos examined: Carnegie nos. 2, 721, 6504, 6506, 8997, 9140.
The mesenchyme of the lower limbs was of uniform density in four embryos. In two other specimens, however, a denser arrangement of the nuclei in the central and proximal parts of the limbs suggested an early blastemal condensation for skeleton and muscles. An eetodermal ridge was present for the first time in the lower limb at this stage (O’Rahilly, Gardner, and Gray, I956) Horizon xvi (7.0 to 12.2 mm.). Ten embryos examined: Carnegie nos. 163, 617, 6507, 6509, 6510, 6512, 6517, 7115, 8112, 8773.
A blastemal condensation, avascular in its central part, was present proximally in at least four of these specimens.
Horizon xvii (8.6 to 14.5 mm.). Twenty-one embryos examined: Carnegie nos. 353, 559, 623, 940, 1771, 5642, 5893, 6258, 6519, 6520, 6521, 6631, 6742, 758, 8101, 8118, 8253, 8789, 8969, 8998, 9100.
In the least-advanced limbs at this stage, no blastemal co11densation was observed. In the m0st—advanced limbs, the entire femur was chondrifying. Centers of chondrification were present in the tibia of some specimens, in the fibula of only two. A common cellular condensation for the tarsus was noted, as well as condensations for the 2d, 3d, 4th, and, occasionally, 5th toes.
Horizon xi/iii (11.7 to 18.0 mm.). Seventeen embryos examined: Carnegie nos. 106, I09, 144 (3), 175, 492 (2), 4430 (2), 6522 (I-2), 652+ 6525» 6527: 6523 (IP): 6533 (1?), 7707, 8097, 8172, 8355, 9247. (figures in parentheses in lists of embryos examined in this and subsequent horizons are Senior's stages of chondrification for the feet.) Chondrification in the tibia extended about halfway to the region of the ankle and included very early Streeter's phase 3 cartilage. The chondrified part of the fibula was very short, and less advanced than that of the tibia. In the majority of embryos, no chondrification was present in the foot. Separate digital prolongations extended distally from the common cellular condensation of the tarsus. A prolongation of the big toe was absent in some specimens and very short i11 others.
In the minority, the chondrifieation of the feet of specimens ranged from 1 to 3 of Senior's stages. (By stage 3, chondrification is present in the cuboid, calcanens, talus, lateral cuneiform, and 2d to 5th metatarsals.) Blastemal condensations extended distally from the cartilage of the metatarsals and, in the 1st digit, distally from the tarsal condensation. The ectodermal ridge, present since horizon xv, disappeared during horizon xviii. figures 3 and 4 illustrate chondrificatio11 in an early Senior’s stage 1 in feet from an embryo not included in the seventeen listed above.
Horizon xix (15.5 to 21.0 mm.). Eleven embryos examined: Carnegie nos. 17 (9), 43 (4 and navicular), 409 (4), 432 (4), 576 (4-5 and mviwlar), 1324 (6), I390 (-1 and navicular), 4405 (3), 5609 (4-5), 6150 (6), 8965 The tibia and fibula were still blastemal in their distal parts. Chondrification in the fibula was less advanced than it was in the tibia. Chondrification in the feet ranged from Senior’s stages 3 to 6, except one specimen at stage 9. (By stage 6, all tarsals and metatarsals have begun to chondrify.) The specimen assigned to Seniors stage 9 on the basis of chondrification in the feet had the maximal Streeter’s point score for horizon xix and an unusually high Senior stage in the hand. In three specimens assigned to stage 4, the navicular was also beginning to chondrify. According to Senior, the navicttlar (stage 6) usually chondrifies after the medial cuneiform a11d 1st metatarsal (stage 5). The center of chondrification in the cuboid was relatively large and well advanced, whereas the centers in the talus and calcanens were relatively small. A cellular condensation of the tuber calcanei appeared in this horizon.
Horizon xx (18.5 to 25.0 mm.). Eleven embryos examined: Carncgie nos. 431 (9), 460 (9), 462 (9), 966 (9), 4148 (4 and navicular), 5537 (7), 6202 (8), 6426 (7-8), 7274 (6), 7966 (7), 8157 (9) The lower ends of the tibia and fibula still consisted of condensed mesenchy1ne and were closely related to each other. Streeter’s phase .4 cartilage was present in the tibia but not in the fibula.
Chondrification iii the feet ranged from Senior’s stages 6 to 9, except one specimen assigned to stage 4, even though it included ehondrification in the navicular. Only one specimen fell into stage 6, whereas about half of the specimens were at stage 9. (By stage 9, all the tarsals, metatarsals, and proximal phalanges have begun to chondrify.) The centers of chondrification in the cuboid and lateral cuneiform were relatively larger than those iii the talus and calcaneus. The cartilage was less advanced in the navicular and medial cuneiform than in other tarsal elements. In the cellular condensations that extended distally from the chondrifying proximal phalanges, there was no segmentation into middle and distal phalanges. figure 5 illustrates chondrification in digits at Senior’s stage 7 in a foot from an embryo not included in the eleven listed.
Horizon x.ri (19.0 to 26.4 mm.). Thirteen embryos examined: Carnegie nos. 22 (10), 455 (11-12), 903C (12), 1008 (12), I358F (12), 2937 (12), 4090 (10-12), 4960 (I0), 5596 (I2), 7254 (9), 7392 (9-10), 7864 (9), 8553 (10).
No bone collars were found in the long bones, although osteoblasts were observed in the perichondrium of the femur and tibia of some embryos. Streeter's phase 4 cartilage was noted in the tibia of all embryos a11d in the fibula of some. The patella was beginning to chondrify in some embryos.
Chonclrification in the feet ranged from Senior‘s stages 9 to 12; about half were at stage 12. (By stage 12, all skeletal elements of the foot are chondrifying except the distal phalanges of the 2d to 5th toes.) The tuber calcanei was beginning to chondrify in some embryos. A cellular condensation of the sustentaculum talare was identifiable in at least one specimen, a11d a condensation of the tuberosity of the 5tl1 metatarsal in another. Streete1"s early phase 3 cartilage was present in the metatarsals a11d proximal phalanges.
Horizon xxii (23.0 to 27.5 mm.). five embryos examined: Carnegie nos. 1458 (12), 3681 (12), 4339 (12), 4638 (12). 8594 (12)A bone collar encircled the shafts of the femur and tibia of most specimens. The fibula contained phase 4 cartilage but lacked a bone collar. The patella was chondrifying in all embryos. All the feet were assigned to Senior’s stage 12. The distal phalanges were present as mesenchymal condensations. The fibula and calcaneus were still closely related to each other.
Horizon xxz'ii (23.0 to 32.2 mm.). Seven embryos examined: Carnegie nos. 75 (14), 86 (I4), 1945 (I3), 2561 (I2). 4525 (I4), 5621A (I3), 7425 (13-14) A definite bone collar did not yet surround the fibula, but a layer of matrix was barely visible in the fibula of one specimen. The bone collar of the tibia of another specimen was being eroded by periosteal buds. The fibula was still closely related to the calcaneus, but usually separated from it by a small part of the talus.
The feet of all embryos were in Senior's stages 13 and 14 except one in Senior’s stage 12. (By stage 14, all the skeletal elements of the foot have begun to chondrify.) No distinction was made between stages 13 and 14 in embryos in which the 2d and 3d phalanges of the 5th digit were present as a single cartilage. Such a specimen was present in the series upon which this report is based.
Discussion
Hand
Although a skeletomuscular condensation was sometimes present in the upper limbs by horizon xv, the humerus, radius, and ulna could not be distinguished as separate blastemal condensations until horizon xvi. Chondrification of these elements began during horizons xvi and xvii, whereas that in the hand, sometimes beginning in horizon xvii, was more commonly under way by horizon xviii.
The sequence of chondrification in the hand agrees with that reported by Senior (1929); all elements except sesamoids were chondrifying by horizon xxi. Thus, chondrification begins in the various elements of the hand during approximately the 6th ovulation week, in embryos ranging from about 11 to 24 mm. in length.
Senior reported that, in his first stage, the hamate, capitate, and metacarpals 2 to 4 chondrify “almost simultaneously.” Data from the present study indicate a slight diilerence in sequence within this first stage, metacarpals 3 and 4 chondrifying first. Senior found no significant deviations from the general sequence for the hand; this conclusion is corroborated in the present investigation. Table 3 lists the horizons that correspond to Senior’s stages of chondrification in the hand.
The centrale, which begins to chondrify at stage 6 (horizons xviii and xix), started to fuse with the scaphoid in some specimens at horizons xxi and xxii, and was partly fused with the scaphoid in all specimens examined in horizon xxiii.
The carpus is first apparent as a common blastemal mass from which digital prolongations extend. Inst before chondrification of a carpal element begins, the blasrema in that area sometimes becomes more densely cellular. According to Haines (1947), in a 13-mm. embryo each carpal element is “represented not by a chondrified centre in the interior of the continuous blastema, but by a separate condensation of the blastema itself, separated from the neighbouring elements by intervening sheets of rarefied tissue. By 14 mm. the carpal . . . elements are chonclrified. . . ." The planes of section of the embryos in the present series were generally unfavorable for distinguishing the individual carpal ele ments during the blastemal stage. When some of the elements had begun to chondrify, however, the adjacent blastemal condensations were more readily identifiable. For example, after the triquetrum had begun to chondrify, the mesenchymal condensation for the pisiform could be more easily delimited.
Nothing in the hand or foot was found that suggested the temporary appearance of structures characteristic of lower vertebrates, such as a prepollex or a postminimus.
Table 3
| Horizons in which a given Senior's Stage of Chrondrification in the Hand has been found | |
|---|---|
| Senior's stage | Horizons |
| 0 | xvii |
| 1 | xvii—xviii |
| 2 | xviii |
| 3 | xviii |
| 4 | xviii |
| 5 | xviii |
| 6 | xviii—xix |
| 7 | xix |
| 8 | xix |
| 9 | xix —xx |
| 10 | xix —xx |
| 11 | xix —xxi |
| 12 | xx —xxi |
| 13 | xxi |
| 14 | xxi on |
The findings reported by Schmidt-Ehrenberg (1942) in the single human embryo (15 mm.) that she examined are not confirmed.
Some upper limbs studied by Bardeen and Lewis (1901), and by Lewis (1902), were included in the present series. Lewis, who examined embryo no. 43 (horizon xix), stated that all proximal phalanges were chondrifying, except that of the thumb; this finding would place the hand in Senior’s stage 6. The present writers, however, found chondrification beginning in the proximal phalanx of the thumb also (Senior’s stage 7). From studies of embryo no. 22 (horizon xxi), Bardeen and Lewis, and Lewis, concluded that of the distal phalanges only the thumb was chondrifying (Senior’s stage 12). The present authors found chondrification in the distal phalanges of the 2d to 4th digits also, but were uncertain about that of the 5th finger (Senior's stages 13 to 14). It should be kept in mind tl1at Bardeen and Lewis may have used different criteria for the onset of chondrification from those employed in the current study.
Foot
A skeletomuscular condensation is ordinarily not present in the lower limb until horizon xvi, and even then not in all specimens. The femur, tibia, and fibula could not be distinguished as separate blastemal condensations until horizon xvii. Chondrification in these elements begins during horizons xvii and xviii, whereas that in the foot, occasionally noted in horizon xvii, is commonly initiated in horizons xviii to xix.
The sequence of chondrification in the foot confirms that reported by Senior, with few variations; all elements except sesamoids were usually chondrifying by horizon xxiii. Thus, chondrification begins in the various elements of the foot between approximately 5‘/§_ and 7 ovulation weeks, in embryos ranging from about 13 to 30 mm. in length.
No clear-cut distinction was found between Senior’s stages I and 2. Although the navicular usually does not ehondrify until stage 6, in several specimens its Chondrification preceded that of the medial cuneiform and Ist metatarsal (stage 5); these feet were assigned to stage 4 plus navicular. Such deviations were minor in the light of the over—all regularity in the sequence of chondrification in both hand and foot.
The foot lags behind the hand in development, and, in any given embryo up to horizon xxii, usually 5 to 1:: fewer chondrifying centers are found in the foot than in the hand (table 5). Table 4 lists the horizons that correspond to Senior’s stages of chondrification in the foot.
Streeter found the degree of development of the humerus to be a valuable aid in staging embryos. The development of other skeletal structures of the limbs likewise follows a definite pattern, and information about them should serve as a further aid in staging embryos. Streeter (1929) had already expressed the hope that the sequence of chondrification worked out by Senior might serve this purpose. Such information about skeletal elements in both upper and lower limbs is summarized in table 6. Table 7 is a more detailed summary of the process of chondrification in the hand and foot.
The tarsus first appears as a common blastemal mass from which digital prolongations extend. As in the hand, it was noted that sometimes the blastema in the area of a tarsal element became more densely cellular just before chondrification began. I-Iaines (1947) reported that in an embryo of I3 mm. each tarsal element was represented by a separate condensation of the blastema, and that by 14 mm. the tarsal elements were chondrified. The planes of section of the embryos in the present series were generally unfavorable for distinguishing the individual tarsal elements during the blastemal phase.
Table 4
| Horizons in which a given Senior's Stage of Chrondrification in the Foot has been found | |
|---|---|
| Senior's stage | Horizons |
| 0 | xviii |
| 1 | xviii |
| 2 | xviii |
| 3 | xviii - xix |
| 4 | xix —xx |
| 5 | xix —xx |
| 6 | xix —xx |
| 7 | xix —xx |
| 8 | xix —xx |
| 9 | xix —xxi |
| 10 | xxi |
| 11 | xxi |
| 12 | xxi - xxiii |
| 13 | xxiii |
| 14 | xxiii on |
Table 5
| The Range of Senior's Stages of the Foot usually found for a Senior's Stage of the Hand up to Horizon xxii | |
|---|---|
| Senior's stages of Hand | Senior's stages of Foot |
| 1 | 0 |
| 2 | 0-2 |
| 3 | |
| 4 | |
| 5 | 1-3 |
| 6 | 3 |
| 7 | 3-6 |
| 8 | |
| 9 | |
| 10 | 4-9 |
| 11 | 7-9 |
| 12 | 9 |
| 13 | 9-12 |
| 14 | |
Note: In terms of the total number of chondrifying centers present, the foot has usually 5 to 12 centers less than the hand in any given embryo up to horizon xxii.
Table 6
| Horizons in which first appearance of first limb elements is usually found | |||
|---|---|---|---|
| Condensed Mesenchyme |
Cartilage | Bone | |
| Humerus | xvi | xvi—xvii | xxi—xxii |
| Radius | xvi | xvii | xxi-xxiii |
| Ulna | xvi | xvii—xviii | xxii-xxiii |
| Hand | xvii | xvii—xxi | In fetus1and after birth |
| Femur | xvii | xvii—xviii | xxii-xxiii |
| Patella | xx | xxi | After birth |
| Tibia | xvii | xvii—xxiii | xxii—xxiii |
| Fibula | xvii | xxii-xviii | In fetus |
| Foot | xvii—xviii | xviii—xxiii or later | in fetus and after birth |
1 Intramembranous ossification at the tips of the distal phalanges of the hand may be in horizon xxiii.
Many investigators have claimed that, during the ontogeny of the limbs, temporary structures of phylogenetic significance are found. In the ease of the human foot, some have reported that structures characteristic of lower vertebrates, such as a prehallux, are present during development (Schmidt-Ehrenberg, I942; Trolle, I948). The present writers have found nothing to substantiate such claims.
Table 7
| Horizons in which first appearance of chondrification in skeletal elements of hand and foot is usually found | ||
|---|---|---|
| Horizons | ||
| Carpus except pisiform | xviii-xix | |
| Pisiform | xix-xxi | |
| Metacarpus | xvii—xxiii | |
| Proximal phalanges | xviii—xix | |
| Middle phalanges | xix-xx | |
| Distal phalanges | xx—xxi | |
| Tarsus | xviii—xix | |
| Metatarsus | xviii—xix | |
| Proximal phalanges | xx | |
| Middle phalanges | xxi | |
| Distal phalanges | xxi—xxiii | |
Some lower limbs studied by Bardeen and Lewis (1901), and by Bardeen (1905), were included in the present series. Bardeen and Lewis examined embryo no. 22 (horizon xxi), and stated that all bones of the leg were in the form of cartilage except “that the terminal phalanges of the 3 outer toes have not yet appeared.” Bardeen, however, reported that embryo no. 22 was similar to embryo no. 17 (horizon xix), in which only the middle phalanx of the 2d digit was chondrifying. His illustration supports this statement; the feet would thus be assigned to Senior’s stages 9 to 10. The present authors found the feet of embryo no. 22 to be in stage I0. Bardeen’s findings would place embryo no. 17 in stages 9 to IO also. The present authors found the feet of this embryo to be in stage 9.
Comparisons based on the investigations of chondrification by Senior and the present authors, and the studies of ossification by Francis and Werle (1939) and Noback and Robertson (1951), indicate that the sequence of chondrification in the hand and foot differs from that of ossification. In the carpus, for example, the hamate and capitate are the first to chondrify and to ossify, but the sequences differ in the other carpal elements. In the tarsus, the cuboid is the first element to chondrify, but the calcancus and talus are the first to ossify. Of the metaearpals and metatarsals, the 2d to 4th begin to chondrify before the 5th, and the 5th before the 1st. The order is usually the same in diaphyseal ossification. The phalanges of both hand and foot begin to chondrify in proximodistal sequence, \vhereas the order is distal, proximal, and middle in diaphyseal ossification. Chondrification is present in the carpus before the tarsus; the converse is true of ossification.
Summary
- Serial sections of the limbs of 103 human embryos, ranging from horizons xv to xxiii inclusive, were examined.
- The skeleton of the hand was blastemal by horizon xvii, and the various elements began to chondrify between horizons xvii and xxi, i.e. approximately during the 6th ovulation week.
- The skeleton of the foot was blastemal by horizons xvii to xviii, and the various elements began to chondrify between horizons xviii and xxiii, or later, i.e. from approximately 5‘/2 to 7 ovulation weeks.
- Chondrification of the various elements of the hand and foot occurred in a definite sequence; Senior’s I4 stages were confirmed.
- Seniors stages of chondrifieation in the hand and foot were correlated with Streeter’s developmental horizons.
Literature Cited
ARDEEN, C. R. 1905. Studies of the development of the human skeleton. Amer. Inur. Anat., vol. 4, pp. 265-302.
and W. H. LEWIS. 1901. The development of the limbs, body—wall and hack in man. Amer. Iour. Anat., vol. I, pp. 1-36.
BAUR, G. 1886. Bemcrkungen iiber den “Astragalus” und das "Intermedium tarsi" der Siiugethiere. Morphnl. Iahrb., vol. 11, pp. 468-483.
Francis, C. C., and P. P. \Vr.m.ta. 1939. The appearance of centers of ossification from birth to 5 years. Amer. Iour. Phys. Anthropol., vol. 24, pp. 273-299.
Gaiirannnae, 1905. Die Entwickelung der Knoehen, Muskeln und Nerven der I-land und der fiir die Bewegungen der I-land bestimmten Muskcln des Unterarms. Anat. I-leftc, vol. 30, pp. 1-154.
I-Lter-.N, W. I900. Die Bildung des Knorpelskeletts beim mensch— lichen Embryo. Arch. f. Anat. u. Physiol., Anat. Abt., pp. 1-40.
Hamias, R. W. 1947. The development of joints. Iour. Anat., vol. 81, pp. 33-55.
HENKE, W., and C. REYIH-'.R. 1874. Studien iiber die Entwickclung der Extremitiiten dcs Menschen, insbesondcre der Gelenkfliichcn. Sitzungsb. tl. k. Akad. d. \Vissensch., Math.nat., vol. 70, pp. 217-273.
Heuser_R, C. 1926. lieitrag zur Kenntnis der Gelenkentwicklung beim Menschen. Morphol. ]ahrb., vol. 55, pp. 489-567. I'IIN'I'lSCl-(E, E. 1930. Beitrag zur Entwieklung des mcnschlichen Fersenbeines. Ztschr. f. mikrosk.-anat. Forsch., vol. 21, pp. 531-551
LEBOUCQ, H. 1884. Rceherches sur la morphologie du carpe chez les mammifx‘.-res. Arch. de l3iol., vol. 5, pp. 35-102. 1886. Sur la morphologie du carpe et du tarse. Anat. Anz., vol. 1, pp. 17-21.
1904. Recherches sur le développement des phalanges terminales des doigts chez l'homme et les mammiferes. Ann. soc. med. de Gand, vol. 84, pp. 227-242.
Lewis, W. H. 1902. The development of the artn in man. Amer. ]our. Anat., vol. I, pp. 145-184.
Martin, P. 1929. Contribution 5 l’étude des articulation (les membres chez l'embryon humain. Thése. 56 pp. Université de Lausanne.
Noback, C. R., and G. G. Robinson. 1951. Sequences of appearance of ossification centers in the human skeleton during the first 5 prenatal months. Amer. Iour. Anat., vol. 89, pp. 1-28.
O'Rahilly, R., I"-.. GartnN1=.R, and D. I. GRAY. 1956. The ectodermal thickening and ridge in the limbs of staged human embryos. Iour. Embryol. and Expcr. Morphol., vol. 4, pp. 254-264.
and D. B. MEYER. 1956. Roentgenographic investigation of the human skeleton during early fetal life. Amer. Iour. Roentgenol., vol. 76, pp. 455-468.
ROSENBERG, E. 1376. Ueber die Entwickelung dcr Wirlielsiiule und das Centrale carpi des Menschen. Morphol. ]ahrb., vol. 1, pp. 83-197.
SCHMIDT-EHRI-‘.I\'l1ERG, E. C. 1942. Die Embryogenese des Extretnitbitenskelettes der Siiugetiere. Iiin Beitrag zur Frage der Entwicklung der Tetrapodengliedmassen. Inauguraldissertation. 131 pp. Geneve.
Set1o:uat;1u:, H. 1900. Untersuchung dcr F.ntwicklung der Muskeln und Knochen des menschlichen Fusses an Sericnschnitten und Rekonstruktionen und unter 7.ul1i.ilfenal1me makroskopischer Priiparation. Dissertation. 61 pp. Giittingen.
SCHULIN, K. 1879. Ueber die Entwickelung und weitere Augbildung dcr Gelenke des menschlichen Kiirpers. Arch. f, Anat. u. Physiol., Anat. Abt., pp. 240-274.
Senior HD. 1929. The chondrification of the human hand and foot skeleton. Anat. Rec., Abstracts, vol. 42, p. 35.
Streeter GL. Chondrification of the hand and foot. (1929) Carnegie Inst. Wash. Year Book No. 28, p. 8.
- 1942. Developmental horizons in human embryos. Description of age group xi, 13 to 20 somites, and age group xii, 221 to 29 somites. Carnegie Inst. Wash. Pub. 541, Contrib. to Embryol., vol. 30, pp. 211-245.
- 1945. Developmental horizons in human embryos. Description of age group xiii, embryos about 4 or 5 millimeters long, and age group xiv, period of indentation of the lens vesicle. Carnegie Inst. Wash. Pub. 557, Contrib. to Embryol,, vol. 31, pp. 27-63.
- 1948. Developmental horizons in human embryos. Description of age groups xv, xvi, xvii, and xviii, being the third issue of a survey of the Carnegie Collection. Carnegie Inst. Wash. Pub. 575, Contrib. to F.mbryol., vol. 32, pp. 133-203.
- 1949. Developmental horizons in human embryos (fourth issue). A review of the histogenesis of cartilage and bone. Carnegie Inst. Wash. Pub. 583, Contrib. to Embryol., Vol. 33, pp. 149-167.
- 1951. Developmental horizons in human embryos. Description of age groups xix, xx, xxi, xxii, and xxiii, being the fifth issue of a survey of the Carnegie Collection. (Prepared for publication by C. II. Heuser and G. W. Corner.) Carnegie Inst. Wash. Pub. 592, Contrib. to Embryol., vol. 34, pp. 165-196.
TROLLE, D. 1948. Accessory bones of the human foot; a radiological, histo-embryological, comparative-anatomical, and genetic study. Translated by E. Aagesen. 272 pp. Copenhagen.
Plate
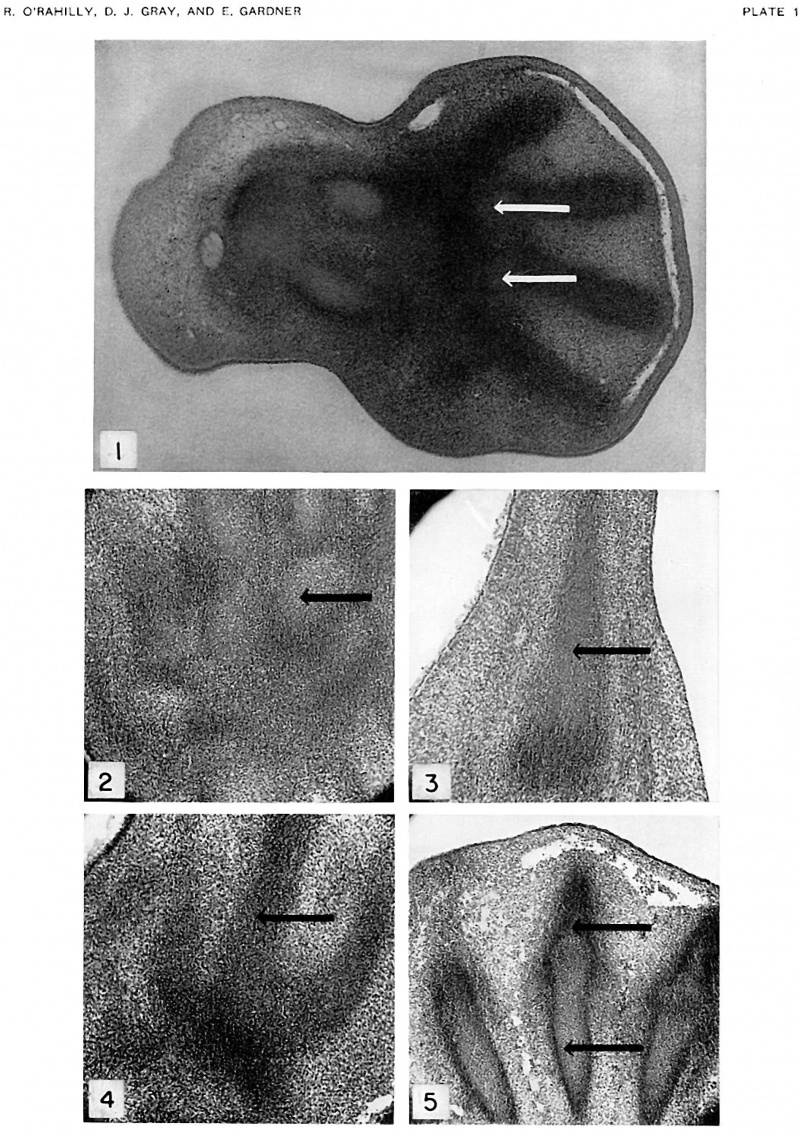
Fig. 1. Frontal section of hand of embryo, Carnegie no. 8789, 11.7 mm., horizon xvii. X56. Senior's stage 1. lilastemal condensations of digits 2 to 5. Radius and ulna are chondrifying. Carpus is blastemal, except very early chondrification in metacarpals 3 and 4, barely suggested it1 this figure by lighter-staining areas (arrows).
Fig. 2. Frontal section of left hand of embryo, Wayne University no. 15, not included in text because the embryo has not been assigned to a Streeter horizon. X75. Senior’s stage 5. Metacarpals 3 to 5 in the upper part of the figure. Hamate indicated by arrow; capitate and trapezoid to its left; triquetrum just below at its right. Radius and ulna in lower part of figure.
Fig. 3. Sagittal section of right foot of embryo, Wayne University no. 15. X75. Senior's stage 1 (early). Early chondrification in metatarsal 4 (arrow). A blastemal condensation for the I cuboid is visible as a more darkly stained region just below the metatarsal.
Fig. 4. Horizontal section of left foot of embryo, Wayne University no. 15. X75. Senior's stage 1 (early). Condensations of metatarsals 2 to 4 in upper part of figure (arrow indicates metatarsal 3). Lighter staining, as compared with tarsus (more darkly stained region just below metatarsals), suggests beginning chondrilication.
Fig. 5. Horizontal section of left foot of embryo, ‘.Vayne University no. 17-2, not included in text because the embryo has not been assigned to a Streeter horizon. X75. Senior's stage 7. Chondrification in metatarsals 2 to 4. Section includes proximal phalanges 3 and 4. Arrows indicate metatarsal 3 and proximal phalanx 3. Part of marginal vein is present above.
Cite this page: Hill, M.A. (2024, May 1) Embryology Paper - Chondrification in the hands and feet of staged human embryos. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Chondrification_in_the_hands_and_feet_of_staged_human_embryos
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G