Gastrointestinal Tract - Oesophagus Development
| Embryology - 7 Apr 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The oesophagus (British English) or esophagus (American English) or allows the passage of food from the mouth, then pharynx to the stomach by gravity and peristaltic contractions.
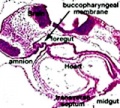
The gastrointestinal tract (GIT) arises initially during the process of gastrulation from the endoderm of the trilaminar embryo (week 3) and extends from the buccopharyngeal membrane to the cloacal membrane. The tract and associated organs later have contributions from all the germ cell layers.
Note that in historic texts the term entoderm is used to describe endoderm and other terminology may also differ from current descriptions.
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Oesophagus Embryology <pubmed limit=5>Oesophagus Embryology</pubmed> |
Textbooks
- Human Embryology Larson Chapter 9 p229-260
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 12 p271-302
- Before We Are Born (5th ed.) Moore and Persaud Chapter 13 p255-287
- Essentials of Human Embryology Larson Chapter 9 p123-146
- Human Embryology Fitzgerald and Fitzgerald Chapter 19,20 p119-123
More? References | Online Textbooks | Historic Textbooks
| UNSW Students | |||||||
|---|---|---|---|---|---|---|---|

|
You have access the following online Embryology resources and textbooks through the UNSW Library. | ||||||

|
Hill, M.A. (2020). UNSW Embryology (20th ed.) Retrieved April 7, 2026, from https://embryology.med.unsw.edu.au
| ||||||

|
Moore, K.L., Persaud, T.V.N. & Torchia, M.G. (2015). The developing human: clinically oriented embryology (10th ed.). Philadelphia: Saunders. (links only function with UNSW connection)
Chapter 11 Alimentary System | ||||||

|
Schoenwolf, G.C., Bleyl, S.B., Brauer, P.R., Francis-West, P.H. & Philippa H. (2015). Larsen's human embryology (5th ed.). New York; Edinburgh: Churchill Livingstone.(links only function with UNSW connection)
Chapter 14 Development of the Gastrointestinal Tract | ||||||
Objectives
- Understanding of germ layer contributions to the early gastrointestinal tract (GIT)
- Understanding of the folding of the GIT
- Understanding of three main GIT embryonic divisions
- Understanding of associated organ development (liver, pancreas, spleen)
- Brief understanding of mechanical changes (rotations) during GIT development
- Brief understanding of gastrointestinal abnormalities
Germ Layer Contributions
- Endoderm - epithelium and associated glands
- Mesoderm (splanchnic) - mesentry, connective tissues, smooth muscle, blood vessels
- Ectoderm (neural crest) - enteric nervous system (neural tube) - extrinsic innervation
Both endoderm and mesoderm will contribute to associated organs.
Gastrointestinal Tract Movies
| Gastrointestinal Tract Movies | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
| |||||||||||||||
|
|
|
|
| |||||||||||||||
|
|
|
|
| |||||||||||||||
| Stage 13 (week 5) | Stage 22 (week 8) | Stage 23 (week 8) | GIT Abnormalities Ultrasound | ||||||||||||||||
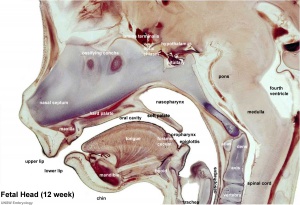
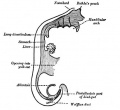
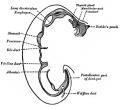
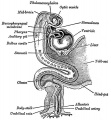
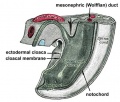
Foregut
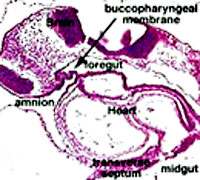
From the oral cavity the next portion of the foregut is initially a single gastrointestinal (oesophagus) and respiratory (trachea) common tube, the pharynx which lies behind the heart. Note that the respiratory tract will form from a ventral bud arising at this level.
- Oral cavity
- Pharynx (oesophagus, trachea)
- Respiratory tract
- Stomach
Development Overview
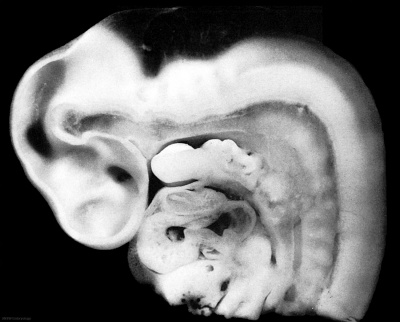
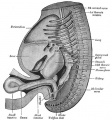
Week 5
(Embryo Carnegie stage 13)
Below is an overview of the sections starting at the level of pharynx compressed dorsoventrally, following the GIT through to the rectum. The most obvious feature is that of a continuous tube initially, attached by dorsoventral mesentery.
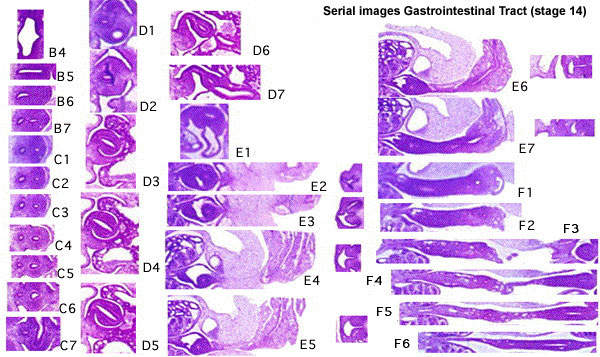
| |||
| Bifurcation of the pharynx into anterior respiratory and posterior oesophagous. | The stomach forming beneath the lung buds and adjacent to the developing liver. | Below the stomach the GIT has a large dorsal mesogastrium and finer ventral mesogastrium. Associated with the tract is the large portal blood vessel derived from the vitelline circulation. | At the bottom curvature of the embryo the mesentry association with the GIT shows extensive vitelline vessels running out through the umbilicus. The hindgut can then be seen, ending at the common urogenital sinus, the cloaca. |
(Embryo Carnegie stage 15)
Later week 5 development showing a sagittal section upper half of embryo.
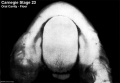
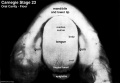
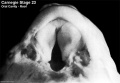
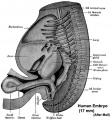
Week 8
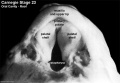
(Embryo Carnegie stage 23)
Images showing both the floor and roof of the embryonic oral cavity in week 8.
- Oral Cavity (stage 23)
Glands
The data below comes from a historic study by Johnson (1910)[4]
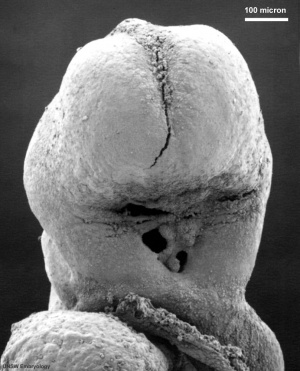
- The oesophagus is at first a simple epithelial tube, the walls of which contain three or four rows of nuclei.
- 20 mm - vaduoles form in the epithelium but the lumen remains pervious throughout. These vacuoles disappear by breaking into the lumen. This causes the epithelium to become thinner and the lumen to increase in size. The cause of the formation of vacuoles has not yet been determined.
- Longitudinal folds of the mucosa are constant structures in the oesophagus. In the upper third of the oesophagus the folds are irregular and variable. In the middle and lower thirds there are four large primary folds. Of these the dorsal and ventral (left and right respectively in the lower part of the oesophagus) develop first; the left and right (ventral and dorsal below) develop soon afterward. Smaller secondary folds, variable in number, appear later at the bases of the primary folds. In the lower part of the oesophagus both primary and secondary folds are twisted through an arc of about 90 degrees in the direction of the hands of a clock. It is probable that this twisting is due to the early rotation of the stomach.
- 55 mm to birth - Areas of ciliated cells are found in the epithelium of the oesophagus. There is both an actual and a relative increase in the amount of surface covered by ciliated cells in embryos up to 187 mm. At birth these areas are relatively smaller. Ciliated cells are absent in the oesophagus of a child of 14 days (seven months premature birth).
- 78 mm - Cardiac glands small areas first seen at this length and will later be found in both the upper and lower ends of the oesophagus. Later these areas evaginate, forming small pockets and grooves. Later, a number of tubular glands grow out from these pockets.
- 240 mm - Oeophageal glands were first observed. They grow out from the epithelium through the muscularis mucosae and lie in the submucosa. In contradistinction to the cardiac glands, their glandular epithelium does not develop until after the excretory ducts are formed. At birth the end pieces of the glands have begun to branch.
Muscles
The oesophagus muscular wall is unique for the gastrointestinal tract in having both smooth and skeletal muscle layers in the muscularis externa upper portion. There is a craniocaudal development with more skeletal muscle in the muscularis externa upper end, a transitional level, and then no skeletal muscle in the lower part. The majority of studies of oesophagus muscle development have used the rodent (mouse, rat) model.
|
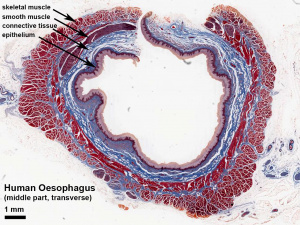
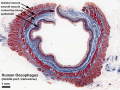
Adult Human Oesophagus (transverse section). (Stain - Masson's Trichrome) |
Smooth Muscle
| The smooth muscle in the oesophagus develops from the splanchnic mesoderm, like the rest of the gastrointestinal tract.
Developmental Sequence: epiblast -> mesoderm -> lateral plate mesoderm -> splanchnic mesoderm -> GIT connective tissue and smooth muscle. |
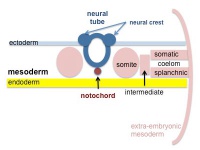
Cartoon showing the splanchnic mesoderm. |
Skeletal Muscle
In animal models (mouse, rat) the oesophagus muscularis externa is initially composed of smooth muscle, and then replaced by skeletal muscle in a craniocaudal progression. This skeletal muscle was historically thought to "transdifferentiate" from smooth muscle.[5] More recent studies suggest a different model with a distinct lineage (skeletal progenitor cell pool) for the skeletal myocytes that form this skeletal muscle.[6][7][8]
Innervation
Neural History
- 1857 Meissner was the first to describe a nerve plexus in the submucosa of the bowel wall.
- 1864 Auerbach described the myenteric plexus between the longitudinal and circular muscle layers.
- 1981 LeDouarin describes neural crest contribution to both plexuses.
Molecular
The endoderm of the developing gastrointestinal tract is a source for patterning signals for both within the tract and also for the surrounding organs and tissues.
- Sox2 - expressed in the anterior part of the primitive gut[9]
- Cdx2 - expressed in the posterior part of the primitive gut[9]
- GDNF - regulate migration of enteric neural crest cells[10]
- endothelin - regulate migration of enteric neural crest cells[10]
Histology
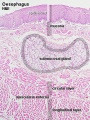
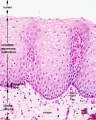
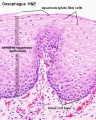
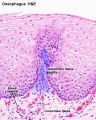
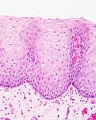
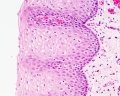
Early embryo esophagus epithelium is simple columnar while the adult oesophagus epithelium is stratified squamous. This also involves a change of keratin expression from K8 to K14.
- Oesophagus Histology: Skeletal and Smooth Muscle | Submucosa Gland | Muscle | Gland-Muscle Animation | Epithelia and Lamina Propria | Labeled Epithelia | Labeled Connective Tissue | Unlabelled | Unlabelled | Oesophagus Development
Abnormalities
The current International Classification of Diseases (ICD-10) code XVII Congenital Malformations Q39 Congenital malformations of oesophagus
- Q39.0 Atresia of oesophagus without fistula Atresia of oesophagus NOS
- Q39.1 Atresia of oesophagus with tracheo-oesophageal fistula Atresia of oesophagus with broncho-oesophageal fistula
- Q39.2 Congenital tracheo-oesophageal fistula without atresia Congenital tracheo-oesophageal fistula NOS
- Q39.3 Congenital stenosis and stricture of oesophagus
- Q39.4 Oesophageal web
- Q39.5 Congenital dilatation of oesophagus
- Q39.6 Diverticulum of oesophagus Oesophageal pouch
- Q39.8 Other congenital malformations of oesophagus Absent Congenital displacement Duplication (of) oesophagus
- Q39.9 Congenital malformation of oesophagus, unspecified
Note ICD-10 is currently being updated to ICD-11 and will have new replacement coding.
Oesophageal Atresia with Tracheo-Oesophageal Fistula
(Q39.1 Atresia of oesophagus with tracheo-oesophageal fistula Atresia of oesophagus with broncho-oesophageal fistula, OA/TOF)
- ICD-11 (beta) LB12.2 Atresia of oesophagus "Oesophageal atresia encompasses a group of congenital anomalies with an interruption in the continuity of the oesophagus, with or without persistent communication with the trachea. In 86% of cases there is a distal tracheooesophageal fistula, in 7% of cases there is no fistulous connection, while in 4% of cases there is a tracheooesophageal fistula without atresia. The remaining cases are made up of patients with OA with proximal, or both proximal and distal, tracheooesophageal fistula."
This abnormality has been shown to be associated with Tbx1 mutations that also include DiGeorge syndrome.[11]
References
- ↑ Chandrashekar J, Hoon MA, Ryba NJ & Zuker CS. (2006). The receptors and cells for mammalian taste. Nature , 444, 288-94. PMID: 17108952 DOI.
- ↑ Perin S, McCann CJ, Borrelli O, De Coppi P & Thapar N. (2017). Update on Foregut Molecular Embryology and Role of Regenerative Medicine Therapies. Front Pediatr , 5, 91. PMID: 28503544 DOI.
- ↑ Ueno S, Yamada S, Uwabe C, Männer J, Shiraki N & Takakuwa T. (2016). The Digestive Tract and Derived Primordia Differentiate by Following a Precise Timeline in Human Embryos Between Carnegie Stages 11 and 13. Anat Rec (Hoboken) , 299, 439-49. PMID: 26995337 DOI.
- ↑ Johnson FP. The development of the mucous membrane of the oesophagus, stomach and small intestine in the human embryo. (1910) Amer. J Anat., 10: 521-559.
- ↑ Patapoutian A, Wold BJ & Wagner RA. (1995). Evidence for developmentally programmed transdifferentiation in mouse esophageal muscle. Science , 270, 1818-21. PMID: 8525375
- ↑ Zhao W & Dhoot GK. (2000). Skeletal muscle precursors in mouse esophagus are determined during early fetal development. Dev. Dyn. , 219, 10-20. PMID: 10974667 <::AID-DVDY1029>3.0.CO;2-2 DOI.
- ↑ Rishniw M, Xin HB, Deng KY & Kotlikoff MI. (2003). Skeletal myogenesis in the mouse esophagus does not occur through transdifferentiation. Genesis , 36, 81-2. PMID: 12820168 DOI.
- ↑ Su PH, Wang TC, Wong ZR, Huang BM & Yang HY. (2011). The expression of nestin delineates skeletal muscle differentiation in the developing rat esophagus. J. Anat. , 218, 311-23. PMID: 21323914 DOI.
- ↑ 9.0 9.1 Raghoebir L, Bakker ER, Mills JC, Swagemakers S, Kempen MB, Munck AB, Driegen S, Meijer D, Grosveld F, Tibboel D, Smits R & Rottier RJ. (2012). SOX2 redirects the developmental fate of the intestinal epithelium toward a premature gastric phenotype. J Mol Cell Biol , 4, 377-85. PMID: 22679103 DOI.
- ↑ 10.0 10.1 Goto A, Sumiyama K, Kamioka Y, Nakasyo E, Ito K, Iwasaki M, Enomoto H & Matsuda M. (2013). GDNF and endothelin 3 regulate migration of enteric neural crest-derived cells via protein kinase A and Rac1. J. Neurosci. , 33, 4901-12. PMID: 23486961 DOI.
- ↑ Mc Laughlin D, Murphy P & Puri P. (2014). Altered Tbx1 gene expression is associated with abnormal oesophageal development in the adriamycin mouse model of oesophageal atresia/tracheo-oesophageal fistula. Pediatr. Surg. Int. , 30, 143-9. PMID: 24356861 DOI.
Online Textbooks
- Developmental Biology (6th ed) Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000. The Digestive Tube and Its Derivatives | Endodermal development of a human embryo
- The Gastrointestinal Circulation Peter R. Kvietys. San Rafael (CA): Morgan & Claypool Publishers; 2010. Table of Contents
- Motor Function of the Pharynx, Esophagus, and its Sphincters. Mittal RK. San Rafael (CA): Morgan & Claypool Life Sciences; 2011. Table of Contents
- Search NLM Online Textbooks "gastrointestinal tract" : Developmental Biology | Endocrinology | Molecular Biology of the Cell | The Cell- A molecular Approach
Historic Textbooks
- The Elements of Embryology by Foster, M., Balfour, F. M., Sedgwick, A., & Heape, W. (1883) The Alimentary Canal and its Appendages
- Text-Book of the Embryology of Man and Mammals by Dr Oscar Hertwig (1892) The Organs of the Inner Germ-Layer The Alimentary Tube with its Appended Organs
- Atlas of the Development of Man Volume 2 by Julius Kollmann (1907) Gastrointestinal
- Text-Book of Embryology by Bailey, F.R. and Miller, A.M. (1921) Alimentary tube and organs
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Reviews
<pubmed></pubmed> <pubmed>28503544</pubmed> <pubmed>27738966</pubmed> <pubmed></pubmed> <pubmed></pubmed>
Articles
<pubmed></pubmed> <pubmed>21950820</pubmed> <pubmed>21595780</pubmed> <pubmed>21323914</pubmed> <pubmed>11683170</pubmed> <pubmed>10906778</pubmed> <pubmed></pubmed>
Search PubMed
Search Pubmed: Oesophagus Development | Oesophagus Development
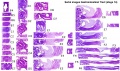
Additional Images
Terms
| Gastrointestinal Tract Terms | ||
|---|---|---|
| ||
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, April 7) Embryology Gastrointestinal Tract - Oesophagus Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Gastrointestinal_Tract_-_Oesophagus_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G