Ectoderm
| Embryology - 22 May 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
The top layer of the early trilaminar embryo germ layers (ectoderm, mesoderm and endoderm) formed by gastrulation. The ectoderm can be though of as having 4 early regions: neural plate, neural crest, surface ectoderm and placodes. Note that there are other pages describing neural (central nervous system; brain and spinal cord) and neural crest (peripheral nervous system; sensory and sympathetic ganglia). Epidermis (integumentary, skin contribution) development will be briefly mentioned due to its ectoderm origin.
The ectoderm contributes to the human embryo:
- nervous system, both central (neural plate) and peripheral (neural crest).
- epidermis of the skin (surface ectoderm) and pigmented cells (neural crest).
- head regions that contribution sensory and endocrine structures (placodes).
- adrenal gland medullary cells (neural crest).
| Ectoderm Links: gastrulation | endoderm | mesoderm | ectoderm | Lecture - Ectoderm Development | Lecture - Neural Development | Lecture - Integumentary Development | neural | neural crest | integumentary | placode | Category:Ectoderm |
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Ectoderm Development | Images | Ectoderm |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
| <html5media width='316' height='500' image="http://php.med.unsw.edu.au/embryology/images/a/a6/Neuralplate_001_icon.jpg">File:Neuralplate_001.mp4</html5media> | This animation shows the embryonic disc from the amniotic cavity side ectoderm (human week 3) onward.
|
|

|
Objectives
- Understanding of events during the third and fourth week of development
- Understanding the process of notochord formation
- Understanding the process of early neural development
- Brief understanding of neural crest formation
- Brief understanding of epidermis formation
- Understanding of the adult components derived from ectoderm
- Brief understanding of early neural abnormalities
Textbook References
- Human Embryology (3rd ed.) Chapter 5 p107-125
- The Developing Human: Clinically Oriented Embryology (6th ed.)
- Moore and Persaud Chapter 18 p451-489
- Essentials of Human Embryology Larson Chapter 5 p69-79
- Before We Are Born (5th ed.) Moore and Persaud Chapter 19 p423-458
Overview
| Template Only - content to be added. Mesoderm has been completed. | |||||||||||||||||||||||||||||||
|
| ||||||||||||||||||||||||||||||
| Overview: Ectoderm | Mesoderm | Endoderm Layers: ectoderm | mesoderm | endoderm | |||||||||||||||||||||||||||||||
| Ectoderm Structures | |||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Notochord
- forms initially as the Axial Process, a hollow tube which extends from the primitive pit , cranially to the oral membrane
- the axial process then allow transient communication between the amnion and the yolk sac through the neuroenteric canal.
- the axial process then merges with the Endodermal layer to form the Notochordal Plate.
- the notochordal plate then rises back into the Mesodermal layer as a solid column of cells which is the Notochord.
Ectoderm
- 2 parts
- midline neural plate
- columnar
- lateral surface ectoderm
- cuboidal
- sensory placodes
- epidermis of skin, hair, glands, anterior pituitary, teeth enamel
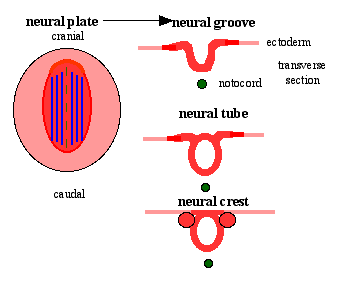
Neural Plate
| <html5media height="500" width="316">File:Neuralplate_001.mp4</html5media> | Development of the neural plate region at the embryonic disc stage.
Dorsal view of the embryonic disc from the amniotic cavity side showing the ectoderm with the central region developing into the neural plate. The neural plate extends from buccopharyngeal membrane to primitive node and forms above the notochord and paraxial mesoderm.The neuroectodermal cells form a broad brain plate and narrower spinal cord region. Three specific regions medial to lateral can also be identified: midline region floor plate, neural plate, edge of neural plate neural crest
|
- extends from buccopharyngeal membrane to primitive node
- forms above notochord and paraxial mesoderm
- neuroectodermal cells
- broad brain plate
- narrower spinal cord
- 3 components form: floor plate, neural plate, neural crest
Neural Determination- neuronal populations are specified before plate folds
- signals from notochord and mesoderm - secrete noggin, chordin,follistatin
- all factors bind BMP-4 an inhibitor of neuralation
- bone morphogenic protein acts through membrane receptor
- lateral inhibition generates at spinal cord level 3 strips of cells
- expression of delta inhibits nearby cells, which express notch receptor, from becoming neurons
- Delta-Notch inetraction- generates Neural strips
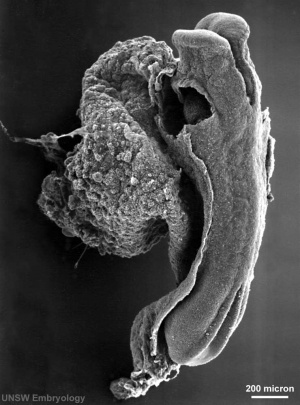
Neural Groove
| <html5media height="480" width="480">File:Neuraltube_001.mp4</html5media> | This animation of early neural development from week 3 onward shows the neural groove fusing to form the neural tube.
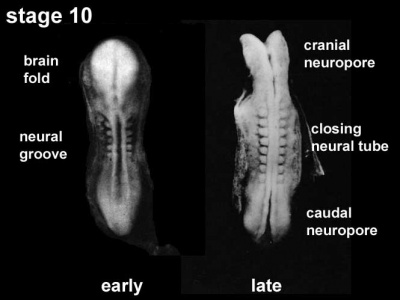
View - Dorsolateral of the whole early embryo and yolk sac. Cranial (head) to top and caudal (tail) to bottom. Yolk sac is shown to the left. Beginning with the neural groove initially fusing at the level of the 4th somite to form the neural tube and closing in both directions to leave 2 openings or neuropores: a cranial neuropore (anterior neuropore) and a caudal neuropore (posterior neuropore). The animation also shows as the embryo grows and folds it increases in size relative to the initial yolk sac. Note also the increasing number of somites over time. |
- forms in the midline of the neural plate (day 18-19)
- either side of which are the neural folds which continues to deepen until about week 4
- neural folds begins to fuse, beginning at 4th somite level
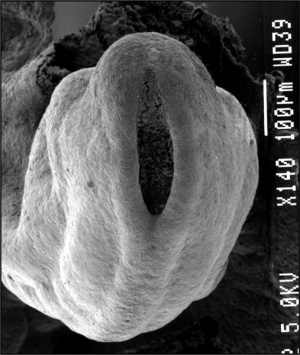
Neural Tube
- the neural tube forms the brain and spinal cord
- fusion of neural groove extends rostrally and caudally
- begins at the level of 4th somite
- closes neural groove "zips up" in some species.
- humans appear to close at multiple points along the tube.
- leaves 2 openings at either end - Neuropores
- cranial neuropore closes before caudal
Failure for the neural tube to close correctly or completely results in a neural tube defect.
Secondary Neuralation
| <html5media height="300" width="320">File:Secondary_neurulation_01.mp4</html5media> | This animation shows the early developmental process often described as secondary neurulation.
Red - site of secondary neurulation | Blue - neural tube
|
Neural Crest
- a population of cells at the edge of the neural plate that lie dorsally when the neural tube fuses
- dorsal to the neural tube, as a pair of streaks
- pluripotential, forms many different types of cells
- cells migrate throughout the embryo
- studied by quail-chick chimeras
- transplanted quail cells have obvious nucleoli compared with chicken
Neural Crest Derivitives
- dorsal root ganglia
- autonomic ganglia
- adrenal medulla
- drg sheath cells, glia
- pia-arachnoid sheath
- skin melanocytes
- connective tissue of cardiac outflow
- thyroid parafollicular cells
- craniofacial skeleton
- teeth odontoblasts
- Links: Neural Crest Development
Ectodermal Placodes
- Specialized ectodermal "patches" in the head region
- Contribute sensory structures - otic placode (otocyst), nasal placode, lens placode
- Contribute teeth
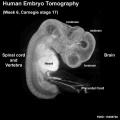
Human Neuralation - Early Stages
The stages below refer to specific Carneigie stages of development.
- Carnegie stage 8 (about 18 postovulatory days) neural groove and folds are first seen
- Carnegie stage 9 the three main divisions of the brain, which are not cerebral vesicles, can be distinguished while the neural groove is still completely open.
- Carnegie stage 10 (two days later) neural folds begin to fuse near the junction between brain and spinal cord, when neural crest cells are arising mainly from the neural ectoderm
- Carnegie stage 11 (about 24 days) the rostral (or cephalic) neuropore closes within a few hours; closure is bidirectional, it takes place from the dorsal and terminal lips and may occur in several areas simultaneously. The two lips, however, behave differently.
- Carnegie stage 12 (about 26 days) The caudal neuropore takes a day to close.
- the level of final closure is approximately at future somitic pair 31
- corresponds to the level of sacral vertebra 2
- Carnegie stage 13 (4 weeks) the neural tube is normally completely closed.
Secondary neurulation begins at stage 12 - is the differentiation of the caudal part of the neural tube from the caudal eminence (or end-bud) without the intermediate phase of a neural plate.
Above text modified from[5]
Movies
| Neural Development | |||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
| ||||||||||||||||||
|
|
|
|
|
| ||||||||||||||||||
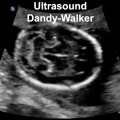
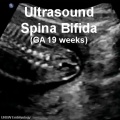
| Abnormalities Ultrasound | |||||||||||||||||||||||
|
|
|
|
| |||||||||||||||||||
References
- ↑ 1.0 1.1 Plouhinec JL, Medina-Ruiz S, Borday C, Bernard E, Vert JP, Eisen MB, Harland RM & Monsoro-Burq AH. (2017). A molecular atlas of the developing ectoderm defines neural, neural crest, placode, and nonneural progenitor identity in vertebrates. PLoS Biol. , 15, e2004045. PMID: 29049289 DOI.
- ↑ 2.0 2.1 Reichert S, Randall RA & Hill CS. (2013). A BMP regulatory network controls ectodermal cell fate decisions at the neural plate border. Development , 140, 4435-44. PMID: 24089471 DOI.
- ↑ Favarolo MB & López SL. (2018). Notch signaling in the division of germ layers in bilaterian embryos. Mech. Dev. , 154, 122-144. PMID: 29940277 DOI.
- ↑ Yaguchi J, Yamazaki A & Yaguchi S. (2018). Meis transcription factor maintains the neurogenic ectoderm and regulates the anterior-posterior patterning in embryos of a sea urchin, Hemicentrotus pulcherrimus. Dev. Biol. , 444, 1-8. PMID: 30266259 DOI.
- ↑ <pubmed>8005032</pubmed>
Textbooks
Online Textbooks
- Developmental Biology by Gilbert, Scott F. Sunderland (MA): Sinauer Associates, Inc.; c2000 Paraxial Mesoderm: The Somites and Their Derivatives
- Molecular Biology of the Cell 4th ed. Alberts, Bruce; Johnson, Alexander; Lewis, Julian; Raff, Martin; Roberts, Keith; Walter, Peter New York and London: Garland Science; c2002 - Figure 21-78. Somite formation in the chick embryo
- Madame Curie Bioscience Database Chapters taken from the Madame Curie Bioscience Database (formerly, Eurekah Bioscience Database) Eurekah.com and Landes Bioscience and Springer Science+Business Media; c2009 Patterning the Vertebrate Neural Plate by Wnt Signaling | Neural Crest Delamination and Migration
Search
- Bookshelf ectoderm | neural plate | neural tube | neural crest
- Pubmed ectoderm | neural plate
Search NLM Online Textbooks: "Ectoderm" : Developmental Biology | The Cell- A molecular Approach | Molecular Biology of the Cell
Reviews
Kutejova E, Briscoe J & Kicheva A. (2009). Temporal dynamics of patterning by morphogen gradients. Curr. Opin. Genet. Dev. , 19, 315-22. PMID: 19596567 DOI.
Charron F & Tessier-Lavigne M. (2007). The Hedgehog, TGF-beta/BMP and Wnt families of morphogens in axon guidance. Adv. Exp. Med. Biol. , 621, 116-33. PMID: 18269215 DOI.
Charron F & Tessier-Lavigne M. (2005). Novel brain wiring functions for classical morphogens: a role as graded positional cues in axon guidance. Development , 132, 2251-62. PMID: 15857918 DOI.
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- Embryo Images Early Cell Populations and Establishment of Body Form | Nervous System Development
- Society for Neuroscience http://web.sfn.org/content/Publications/BrainFacts/index.html Brain Facts
- Anatomy of the Human Body The Neural Groove and Tube
- Environmental Health Perspectives Critical Periods of Vulnerability for the Developing Nervous System: Evidence from Humans and Animal Models | PMC: 1637807 | PMID: 10852851
- Journal Development
Take the Quiz
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 22) Embryology Ectoderm. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Ectoderm
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G

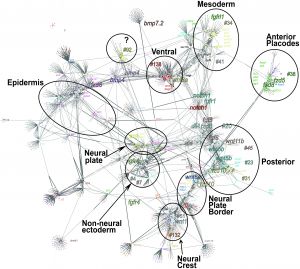
![Zebrafish ectodermal patterning model[2]](/embryology/images/thumb/2/26/Zebrafish_ectodermal_patterning_model.jpg/300px-Zebrafish_ectodermal_patterning_model.jpg)