Integumentary System - Hair Development
| Embryology - 2 Jun 2026 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
Hair formation, hair follicle or pilli development, is an excellent example of two distinct developmental processes: epithelio-mesenchymal interactions and pattern formation. The differentiated hair follicle will eventually contain 20 or more different cell types. Melanocytes, which provide the hair colour, have a neural crest origin, and with ageing their numbers decline leading to whitening (grey) of the hair process.
Hair follicle development in humans begins as an epithelial-mesenchymal interaction at week 9 - 12. This initial lanugo hair is replaced in the late fetal or early neonate by vellus and terminal hairs. A second round of development occurs during puberty under the influence of steroidal hormones.
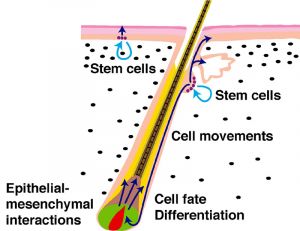
The hair follicle is also a site for stem cells, allowing replacement of the follicle.
- Historic: 1910 Hair
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Hair Embryology | Hair Development | Vellus Hair Development | Terminal Hair Development | Hair Follicle Development |
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Textbooks
- Human Embryology (2nd ed.) Larson Chapter 14 p443-455
- The Developing Human: Clinically Oriented Embryology (6th ed.) Moore and Persaud Chapter 20: P513-529
- Before We Are Born (5th ed.) Moore and Persaud Chapter 21: P481-496
- Essentials of Human Embryology Larson Chapter 14: P303-315
- Human Embryology, Fitzgerald and Fitzgerald
- Color Atlas of Clinical Embryology Moore Persaud and Shiota Chapter 15: p231-236
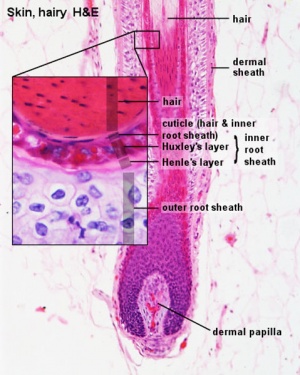
Histology
Adult hair histology.
|
|
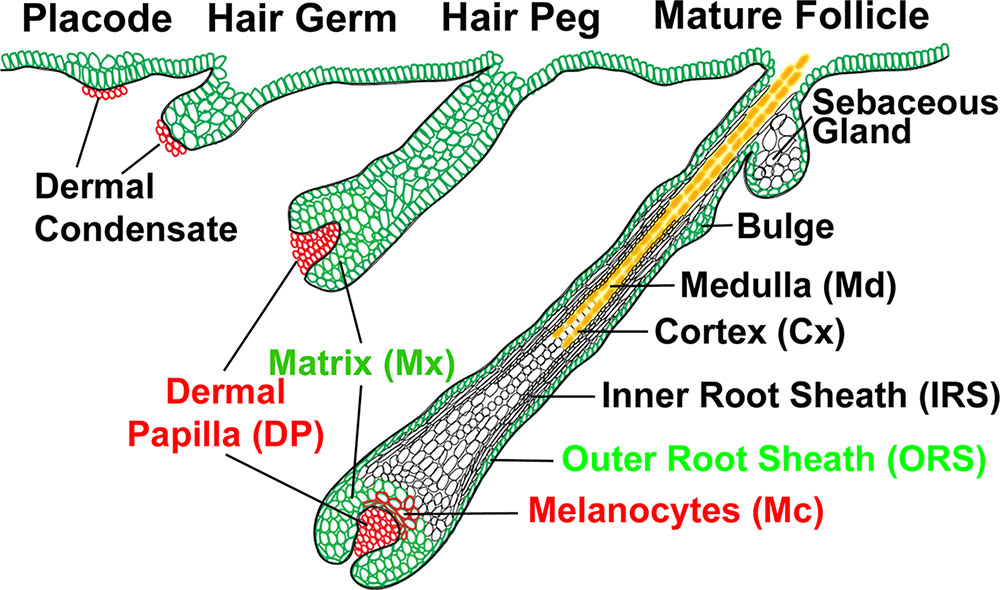
Development Overview
Embryonic
- follicle forms in stratum germinativum of epidermis
- hair bud -> hair bulb
- hair bulb forms hair
- mesenchyme forms hair papilla
- germinal matrix cells become keratinized to form hair shaft
- week 12 - lanugo hair (Latin, lana = wool) - first hair formed replaced postnatally, role in binding vernix to skin
Puberty
Coarse hair in pubis and axilla in both male and female (in males also on face and other body regions chest, etc) (More? Puberty)
Lanugo Hair
Lanugo (Latin, lana = wool) hair is the first hair produced by the fetal hair follicles, appearing at about before 5 months of gestation. The hair is very fine, soft, and usually unpigmented. Normally lost (shed) before birth, though sometimes present at birth. The hair has a role in binding vernix caseosa to skin.
Hair grows over the entire body at the same rate, so the hairs are the same length, and is shed abut 4 weeks before birth. Premature infants can still be covered with these hairs.
Neonatal Hair
Newborn infants have two types of hair:
Vellus Hairs
- short hairs, only a centimetre or two long, and contain little or no pigment
- follicles that produce them do not have sebaceous glands and never produce any other kind of hairs
Terminal Hairs
- long hairs that grow on the head and in many people on the body, arms and legs
- produced by follicles with sebaceous glands
- the hairs in these follicles gradually become thinner and shorter until they look like vellus hairs
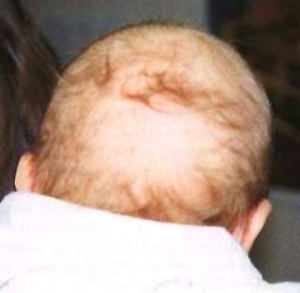
On the head, there are also two periods of hair development in which hair growth begins at the forehead and then extends to the back of the neck.
Then at 2- 3 months old, the first hairs may be shed naturally over an area on the back of the head. This is often mistakenly thought to be due to head rubbing.
Eyelash Hairs
The eyelash hairs function to protect and maintain the health of the lid margin. The adult eyelash hair follicles: produce shorter hair shafts, have no arrector pili muscle, and have a shorter hair cycle.[6] See also a recent review.[7] The eyelid also contains in addition to eyelashes, a number of unique integumentary glands.
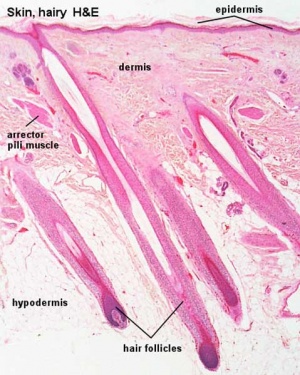
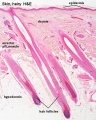
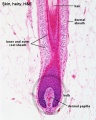
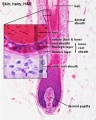
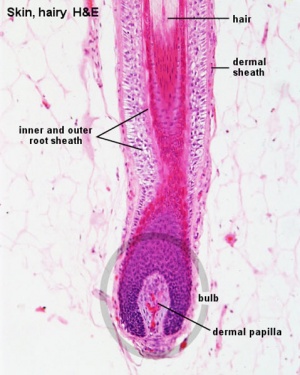
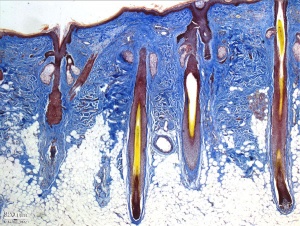
Hair Follicle
This image shows a histological section through the adult skin showing "slices" through 4 hair follicles and their associated glands in different planes. The hair shaft is the yellow structure extending from the bulb within the hair follicle.
From the top down the three major skin layers shown are:
epidermis (red upper region) surface ectoderm in origin, epithelium containing keratinocytes
dermis (dark blue middle region) mesoderm in origin, connective tissue containing many different cell types and collagen fibers.
hypodermis (pale lower region) mesoderm in origin, connective tissue containing many adipose cells.
Hair Follicle Phases
There are several phases of hair follicle growth.
- Anagen Phase - active phase
- Catagen Phase - apoptosis-driven involution, end of active growing phase of the life cycle of the hair, between growing phase (anagen) and resting stage (telogen).
- Telogen Phase - hair follicle resting phase of hair growth cycle.
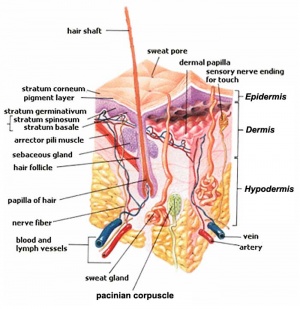
Hair Follicle Stem Cells
Hair follicles contain stem cells located in the bulge[8]
Molecular Hair Development
Signaling pathways in follicle formation:
Shh and its effectors
antagonists for the Wnt (Dkk4)
BMP (Sostdc1)
variant NF-kappaB-signaling cascade, based on lymphotoxin-beta (LTbeta)/RelB.
ctodysplasin regulates the lymphotoxin-{beta} pathway for hair differentiation.[9]
Signaling inhibitors of hair/follicle formation: EGF, FGF5
Wnt signaling
- Initiation and maintenance of hair morphogenesis
- Wnt3a induce hair formation
- Wnt10b promotes hair neogenesis
Sonic Hedgehog Pathway
SHH Knockout mice
- Hair germs comprising epidermal placodes and associated dermal condensates were detected in both control and Shh -/- embryos, but progression through subsequent stages of follicle development was blocked in mutant skin. ...Our findings reveal an essential role for Shh during hair follicle morphogenesis, where it is required for normal advancement beyond the hair germ stage of development. Chin etal Developmental Biology, v 205, n 1, Jan 1999, p 1-9
SHH Activation (inappropriate)
- shh transduction cascade in human epidermis can cause basal cell carcinoma. Here we show that during normal development of avian skin, Shh is first expressed only after the responsiveness to this protein has been suppressed in most of the surrounding ectodermal cells. Forced expression of Shh in avian skin prior to this time causes a disorganized ectodermal proliferation. However, as skin begins to differentiate, the forced expression of Shh causes feather bud formation. Subsequently, expression of Shh in interfollicular epidermis has little or no morphological effect. Restricted responsiveness to Shh in developing skin has functional consequences for morphogenesis and may have important implications for cutaneous pathologies as well
- Morgan etal. Developmental Biology, v 201, n 1, September 1, 1998, p1-12
Pattern Formation Skin
- In birds, the main appendages are the feathers and the foot scales. Their formation results from a series of inductive events between ectoderm (later epidermis) and subectodermal mesoderm (later individualized dermis).
- Morphogenetically, the mesodermal (mesenchymal) component of skin is the predominant tissue, insofar as it controls most morphological and physiological features of developing skin and appendages, notably transformation of ectoderm into epidermis, polarization, proliferation and stratification of epidermal cells, initiation, site, size and distribution pattern of epidermal placodes, species-specific architecture of appendages, regional specification of keratin synthesis.
- The ectodermal (epithelial) component is able to respond to the mesodermal inductive instructions by building feathers and scales in conformity with the specific origin of the dermis. In these epithelial-mesenchymal interactions, extracellular matrix and the microarchitecture of the dermal-epidermal junction appear to play an important role.
Extracellular matrix components
- (primarily collagens, proteoglycans and adhesive glycoproteins) and dermal cell processes close to the epidermal basement membrane become distributed in a microheterogeneous fashion, thus providing a changing substratum for the overlying epidermis. It is assumed that the latter is able to somehow sense the texture and composition of its substratum, and by doing so to appropriately engage in the formation of glabrous, feathered or scaly skin.
- Sengel P Int J Dev Biol 1990 Mar;34(1):33-50
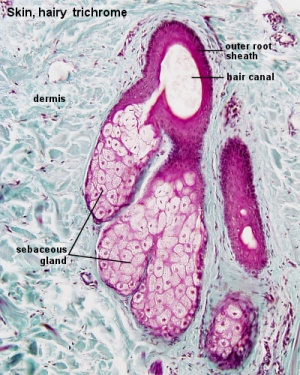
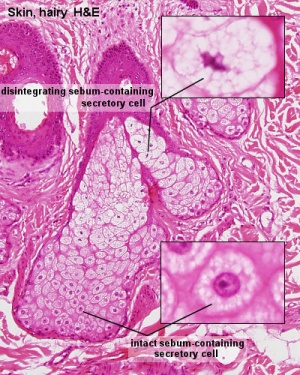
Sebaceous Gland
| Sebaceous Glands are associated with the adult hair follicle and during development secrete vernix caseosa. These glands produce "sebum", that consists of a mixture of lipids, and secret this by a "holocrine" mechanism.
"Holocrine" secretion is where the entire cell is ruptured/lost to release the secreted components. Postnatally these glands are associated with puberty acne, though there are also other abnormalities of these glands.[10] |
Holocrine secretion |
|
Adult skin sebaceous gland histology | |
|---|---|
|
|
- Links: gland
Arrector Pili Muscle
These mesoderm-derived small smooth muscles (hair erector muscle) are associated with the hair (pilosebaceous unit). Their muscle innervation is mainly by noradrenergic fibers and minor cholinergic. Muscle contraction is in response to response to cold or emotional state causes the hair to stand erect ("goosebump").
Muscle Attachments[11]
- proximal end - anchor the bulge areas of hair follicle.
- distal end - situated in the upper dermis.
Puberty Hair Development
The appearance of pubic hair occurs along with ther secondary sexual characteristics (being also staged similarly) and is under endocrine control.
Estrogens- (1 beta-estradiol, E2) involved in skin physiology and are potent hair growth modulators.
Testosterone- Face, trunk and extremities increases hair follicle anagen phase (active) and increases also hair growth rate, thickness, medullation and pigmentation. Effects due to high hormone levels and target organ conversion to 5 alpha-dihydrotestosterone. Pubic hair develops even in absence of 5 alpha-reductase effect.[12]
Tanner Stages
| Stage | Pubic Hair Development |
| 1 | None |
| 2 | Few darker hairs along labia or at base of penis |
| 3 | Curly pigmented hairs across pubes |
| 4 | Small adult configuration |
| 5 | Adult configuration with spread onto inner thighs |
| 6 | Adult configuration with spread to linea alba |
Table based upon the Tanner stages of secondary sexual development.[13]
Histology
Hair Colour
Melanocytes produce melanin which influences hair colour.
Hair Loss
In development the initial hair is lost. In the adult hair loss, or "alopecia", has three distinct forms:
androgenetic alopecia - male- and female-pattern hair loss.
telogen effluvium - alteration of the normal hair cycle, due to many different stress stimuli (severe stress, chemotherapy, childbirth, major surgery, severe chronic illness, rarely occurance in vaccination)
alopecia areata - autoimmune disease, form antibodies against some hair follicles, distinct circular pattern of hair loss.
Abnormalities
References
- ↑ Mok KW, Saxena N, Heitman N, Grisanti L, Srivastava D, Muraro MJ, Jacob T, Sennett R, Wang Z, Su Y, Yang LM, Ma'ayan A, Ornitz DM, Kasper M & Rendl M. (2019). Dermal Condensate Niche Fate Specification Occurs Prior to Formation and Is Placode Progenitor Dependent. Dev. Cell , 48, 32-48.e5. PMID: 30595537 DOI.
- ↑ Peled A, Sarig O, Samuelov L, Bertolini M, Ziv L, Weissglas-Volkov D, Eskin-Schwartz M, Adase CA, Malchin N, Bochner R, Fainberg G, Goldberg I, Sugawara K, Baniel A, Tsuruta D, Luxenburg C, Adir N, Duverger O, Morasso M, Shalev S, Gallo RL, Shomron N, Paus R & Sprecher E. (2016). Mutations in TSPEAR, Encoding a Regulator of Notch Signaling, Affect Tooth and Hair Follicle Morphogenesis. PLoS Genet. , 12, e1006369. PMID: 27736875 DOI.
- ↑ Rudkouskaya A, Welch I & Dagnino L. (2014). ILK modulates epithelial polarity and matrix formation in hair follicles. Mol. Biol. Cell , 25, 620-32. PMID: 24371086 DOI.
- ↑ Amoh Y, Li L, Katsuoka K & Hoffman RM. (2010). Embryonic development of hair follicle pluripotent stem (hfPS) cells. Med Mol Morphol , 43, 123-7. PMID: 20683701 DOI.
- ↑ Shimomura Y & Christiano AM. (2010). Biology and genetics of hair. Annu Rev Genomics Hum Genet , 11, 109-32. PMID: 20590427 DOI.
- ↑ Paus R, Burgoa I, Platt CI, Griffiths T, Poblet E & Izeta A. (2016). Biology of the eyelash hair follicle: an enigma in plain sight. Br. J. Dermatol. , 174, 741-52. PMID: 26452071 DOI.
- ↑ Aumond S & Bitton E. (2018). The eyelash follicle features and anomalies: A review. J Optom , 11, 211-222. PMID: 30017866 DOI.
- ↑ 8.0 8.1 Clayton E, Doupé DP, Klein AM, Winton DJ, Simons BD & Jones PH. (2007). A single type of progenitor cell maintains normal epidermis. Nature , 446, 185-9. PMID: 17330052 DOI.
- ↑ Cui CY, Hashimoto T, Grivennikov SI, Piao Y, Nedospasov SA & Schlessinger D. (2006). Ectodysplasin regulates the lymphotoxin-beta pathway for hair differentiation. Proc. Natl. Acad. Sci. U.S.A. , 103, 9142-7. PMID: 16738056 DOI.
- ↑ Clayton RW, Göbel K, Niessen CM, Paus R, van Steensel MAM & Lim X. (2019). Homeostasis of the sebaceous gland and mechanisms of acne pathogenesis. Br. J. Dermatol. , 181, 677-690. PMID: 31056753 DOI.
- ↑ Narisawa Y, Hashimoto K & Kohda H. (1996). Merkel cells participate in the induction and alignment of epidermal ends of arrector pili muscles of human fetal skin. Br. J. Dermatol. , 134, 494-8. PMID: 8731675
- ↑ <pubmed>3521958</pubmed>
- ↑ Tanner JM. Growth at Adolescence. 2nd ed. Oxford: Blackwell Scientific, 1962.
Journals
Reviews
Grubbs H & Morrison M. (2018). Embryology, Hair. , , . PMID: 30521215
Madaan A, Verma R, Singh AT & Jaggi M. (2018). Review of Hair Follicle Dermal Papilla cells as in vitro screening model for hair growth. Int J Cosmet Sci , 40, 429-450. PMID: 30144361 DOI.
Harland DP. (2018). Introduction to Hair Development. Adv. Exp. Med. Biol. , 1054, 89-96. PMID: 29797270 DOI.
Severin RK, Li X, Qian K, Mueller AC & Petukhova L. (2017). Computational derivation of a molecular framework for hair follicle biology from disease genes. Sci Rep , 7, 16303. PMID: 29176608 DOI.
Shimomura Y. (2016). Journey toward unraveling the molecular basis of hereditary hair disorders. J. Dermatol. Sci. , 84, 232-238. PMID: 27523806 DOI.
Shimomura Y & Christiano AM. (2010). Biology and genetics of hair. Annu Rev Genomics Hum Genet , 11, 109-32. PMID: 20590427 DOI.
López-Bueno A, Villarreal LP & Almendral JM. (2006). Parvovirus variation for disease: a difference with RNA viruses?. Curr. Top. Microbiol. Immunol. , 299, 349-70. PMID: 16568906
Sundberg JP, Peters EM & Paus R. (2005). Analysis of hair follicles in mutant laboratory mice. J. Investig. Dermatol. Symp. Proc. , 10, 264-70. PMID: 16382678 DOI.
Articles
Search PubMed
Search Pubmed: Hair Development | hair follicle development |
Additional Images
Terms
anagen - hair follicle active growth phase, hair follicle progenitors derived from the bulge interact with the mesenchymal dermal papilla cells to generate the lineages of the hair follicle.
catagen - end of active growing phase of the life cycle of the hair, between growing phase (anagen) and resting stage (telogen).
telogen - hair follicle resting phase of hair growth cycle.
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Haziran 2) Embryology Integumentary System - Hair Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Integumentary_System_-_Hair_Development
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G

![Hair follicles contain stem cells located in the bulge[8]](/embryology/images/thumb/3/32/Hair_follicle_development.jpg/400px-Hair_follicle_development.jpg)