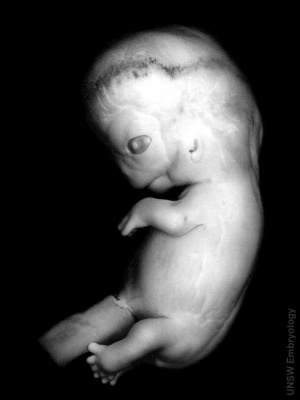
Week 8
We have now reached late embryonic development. Start by looking briefly the process of how the definitive GIT tube is formed and then at the overview of the Carnegie stage 22 embryo GIT from one end to the other. The tract now has a different appearance at different levels; stomach, duodenum, midgut and hindgut.
Then work through the listed specific serial sections of the embryo identifying the GIT features. Alternatively step through the serial sections yourself identifying the tract, its associated mesentries, organs and spaces. Note you should also be comparing the GIT appearance with the earlier embryonic 13 Carnegie stage.
|
<html5media height="500" width="500">File:Stage23 MRI S04.mp4</html5media>
|
Human Embryo MRI at the end of embryonic development (GA week 10).
Observe:
- midgut herniated at the umbilicus, lying outside the ventral body wall, connected by mesentry
- large liver lying directly under the diaphragm and occupying the entire ventral body cavity with organs "embedded" within
- developing pancreas lying in the loop between stomach and duodenum
|
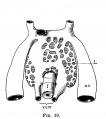
Stage 22 Embryo - Gastrointestinal Tract
| Section
|
Name
|
Description
|
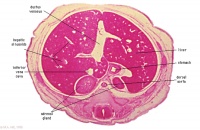
|
E6L
|
Liver. Ductus venosus.
Cardio-oesophageal junction (cf. E5).
Inferior vena cava.
|
|
E7L
|
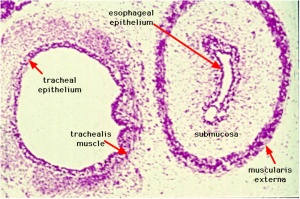
Stomach body, with mucosa, submucosa and muscularis externa.
Lesser sac. Lesser omentum. Pyloroduodenal junction. Folded duodenal mucosa.
Inferior vena cava. Portal vein. Hepatic ducts. Gallbladder.
|
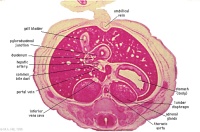
|
F1L
|
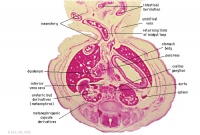
Stomach body. Spleen. Pyloric canal. Duodenum.
Pancreas.
Small intestine loop (jejunum) cut tangentially, ventral to liver.
Portal vein.
|
|
F2L
F3L
|
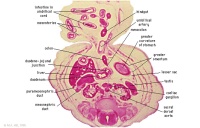
Stomach, spleen. Superior mesenteric artery.
Superior mesenteric vein crossing cranial to body of pancreas.
Tail of pancreas.
Duodenum.
Small intestinal loop herniating from abdominal cavity into the coelom of the umbilical cord (remnant of extra-embryonic coelom).
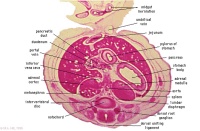
|
|
F4L
|
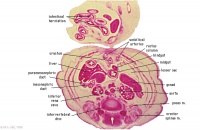
Greater curvature of stomach (tangential section). Lesser sac. Greater omentum. Duodenal/jejunal junction.
Note colon (small lumen, darkly-staining wall) and its mesocolon.
Note the sections of small and large intestine within the umbilical cord coelom and their mesenteries.
Note the thickened jelly to one side of the umbilical cord, containing umbilical vein and R umbilical artery.
|

|
F5L
|
Lesser sac. Greater omentum. Duodenum. Jejunum (cut twice with mesentery in between). Colon and mesocolon.
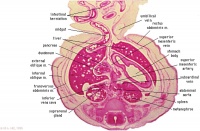
|
|
F6L
|
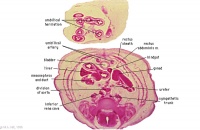
Greater omentum and lesser sac.
Jejunum with mesentery.
Colon with mesocolon.
Three layers of abdominal muscles.
Both umbilical arteries now inside abdominal cavity with urachus between them.
|
|
F7L
|
In abdominal cavity - colon with mesocolon, jejunum. Greater omentum and lesser sac.
Umbilical cord - containing umbilical arteries and small dark allantois. Umbilical cord coelom containing mainly, small intestinal loops with their mesentery.
|
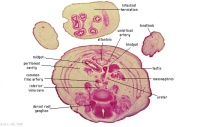
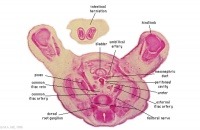
|
G1L
G2L
|
Umbilical cord and coelom containing small intestine loops.
Colon and mesocolon. Jejunum (G1 only).
Bladder with umbilical arteries either side.
Knees.
|
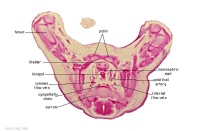
|
G3L
|
Rectum.
Bladder.
Umbilical arteries arising from common iliac arteries.
|
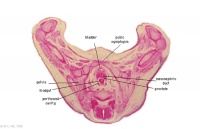
|
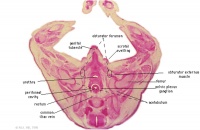
G4L
|
Rectum.
|
|
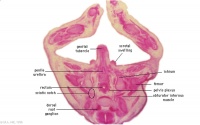
G5L
|
Recto-anal junction with rectovesical pouch of peritoneal cavity.
|
|
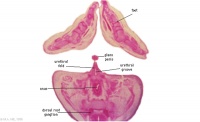
G6L
G7L
|
Anal canal with triangular lumen.
|
Lumen Development
| <html5media height="480" width="255">File:Gastrointestinal tract growth 02.mp4</html5media>
Click Here to play on mobile device
|
This is a simplified animation showing how the gastrointestinal tract wall changes during the late embryonic period.
Week 5 - in this week endoderm in the GIT wall proliferates to the extent of totally blocking (occluding) by week 6, over the next two weeks this tissue degenerates reforming a hollow gut tube. The process is called recanalization (hollow, then solid, then hollow again), abnormalities in this process can lead to duplications or stenosis.
Week 8 - By the end of this week the GIT endoderm tube is a tube once more.
Week 9 - (early fetal) the endoderm of this now hollow tube differentiates into the mucosal epithelium (endoderm).
|
| Page | Play
|
|
- Splanchnic mesoderm will form the submucosa connective tissue and smooth muscle (circular and longitudinal) layers (mesoderm).
- Neural crest cells migrate into this tissue and will form the nerve plexus innervation (ectoderm).
Innervation
Neural Crest colonization occurs in a rostro-caudal sequence forming the 2 associated plexuses.
- week 5 - migrating neural crest cells reach the midgut
- week 7 - neural crest cells have colonized the entire gut
Gastrointestinal Tract Plexuses (enteric nervous system)
| Myenteric plexus
|
Submucosal plexus
|
| Auerbach's plexus
|
Meissner's plexus
|
| Leopold Auerbach (1828–1897) a German anatomist and neuropathologist.
|
Georg Meissner (1829–1905) a German anatomist and physiologist.
|
- first formed plexus
- lies between the outer longitudinal and inner circular smooth muscle layers of muscularis externa
- provides motor innervation to both layers
- secretomotor innervation to the mucosa
- both parasympathetic and sympathetic input
|
- forms 2-3 days after the myenteric plexus
- formed by cells migrating from the myenteric plexus
- innervates smooth muscle of the muscularis mucosae
- only parasympathetic fibers
|
| Links: enteric nervous system | intestine | neural crest | PMID 25428846
|
Interstitial cells of Cajal (ICCs) - may be mesenchymal in origin, develop within the myenteric plexus and are pacemaker cells that control peristaltic contraction waves.[1]
Organs
Cholangiocytes===
Epithelial cells that line the intra- and extrahepatic ducts of the biliary tree. These cells modify the hepatocyte-derived bile, and are regulated by hormones, peptides, nucleotides, neurotransmitters, and other molecules.
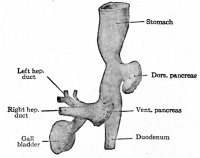
Human (week 4) organ development
|
Note that while the spleen is not a gastrointestinal tract organ, part of the Immune System, it is often described with this system as it develops within the dorsal mesentery. During embryonic development it has a haematopoietic function, with residing blood stem cells, before fetal bone marrow relocation of this population. The adult spleen structure and function was covered in an earlier SH Lecture.
|
Liver
|
Human Embryonic Liver Development
| Week
|
Carnegie Stage
|
Feature
|
Week 4
|
Carnegie stage 11
|
hepatic diverticulum development (ductal plate)
|
|
|
Carnegie stage 12
|
cell differentiation
septum transversum forming liver stroma
hepatic diverticulum forming hepatic trabeculae
|
|
|
Carnegie stage 13
|
epithelial cord proliferation enmeshing stromal capillaries
|
| Week 5
|
Carnegie stage 14
|
hepatic gland and its vascular channels enlarge
hematopoietic function appeared
|
| Week 7
|
Carnegie stage 18
|
obturation due to epithelial proliferation
bile ducts became reorganized (continuity between liver cells and gut)
|
| Week 7 to 8
|
Carnegie stage 18 to Carnegie stage 23
|
biliary ductules developed in periportal connective tissue
produces ductal plates that receive biliary capillaries
|
| Human data[2], see also liver development in the rat embryonic period (Carnegie stages 15-23).[3] (More? Detailed Timeline | Timeline human development)
|
|
| Virtual Slide Features - Stage 22 Liver
|
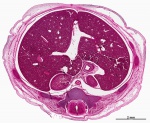
|
Virtual Slide - Stage 22 Liver and Ductus Venosus All Virtual Slides
The links shown in the table below are to specific features shown on the Human embryo (stage 22) Liver and Ductus Venosus virtual slide. See also notes on Liver Development
Clicking the text will open the slide at a detailed view with the structure generally located in the centre of the view. The slide then can also be zoomed out from the set magnification using the controls in the upper left or the mouse.
Use your browser back button to return to this table.
|
You can also make your own selected feature view.
- Set the virtual slide to the region and zoom of interest.
- Click the Permalink (lower righthand corner).
- Then bookmark in your browser, or copy the web address.
See also Permalink help
|
| Cardiovascular
|
Liver
|
Endocrine
|
Musculoskeletal
|
Neural
|
Gastrointestinal
stomach (pylorus)
|
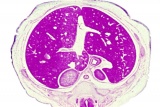
|
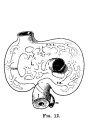
E3 Overview of liver region for selected high power views shown below. Note the position and size of the developing liver spanning the entire abdomen and within the liver the large central ductus venosus.
|
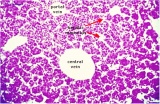
|
E4 Central veins of liver. Radiating appearance of hepatic sinusoids. unlabeled version
|
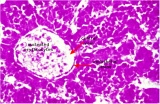
|
E5 Central vein with endothelial lining, containing nucleated erythrocytes, fetal red blood cells. The fetal liver has an important haemopoietic role. unlabeled version
|
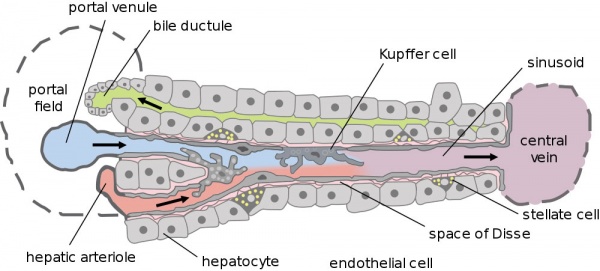
- Hepatic Buds - form hepatocytes, produce bile from week 13 (forms meconium of newborn)
- Vitelline Veins - form sinusoids
- Mesenchyme - form connective tissue and Kupffer cells
|
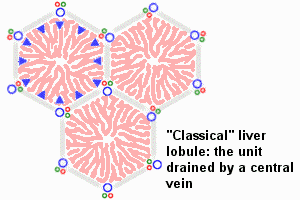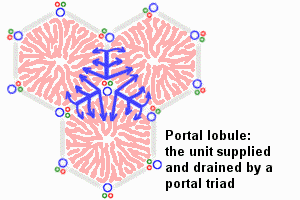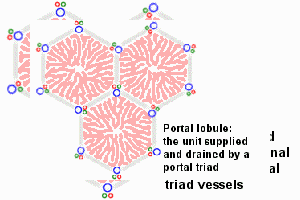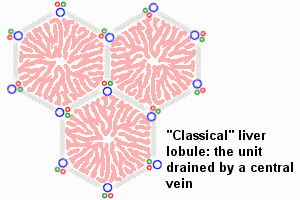
The Adult Liver Lobule
|
- Embryonic Liver and Vasculature
- Links: liver
Pancreas
Exocrine Function - Pancreatic amylase digests starch to maltose. Postnatally, a blood test to detect amylase can be used to diagnose and monitor acute or chronic pancreatitis (pancreas inflammation).
Pancreatic Duct
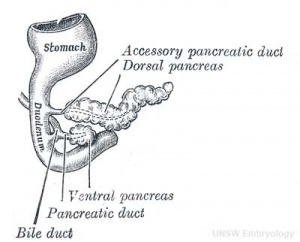
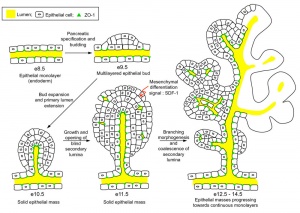
The initial formation of the pancreas as two separate lobes each with their own duct that fuses leads a range of anatomical variations in the adult exocrine pancreatic duct. Pancreatic duct five variation classification: common, ansa pancreatica, branch fusion, looped, and separated. Accessory pancreatic duct (APD, of Santorini) in the embryo is the main drainage duct of the dorsal pancreatic bud emptying into the minor duodenal papilla. In the adult it has been further classified as either long-type (joins main pancreatic duct at pancreas neck portion) and short-type (joins main pancreatic duct near first inferior branch).
- Main Pancreatic Duct (MPD or Wirsung's duct) forms within the dorsal pancreatic bud and is present in the body and tail of the pancreas. Discovered by Johann Georg Wirsung (1589 - 1643) a German physician who worked as a prosector in Padua.
- Accessory Pancreatic Duct (APD or Santorini’s duct) is present mainly in the head of the pancreas. Originally dissected and delineated by Giovanni Domenico Santorini (1681 - 1737) an Italian anatomist.
- Endoscopic Retrograde Cholangiopancreatography (ERCP) is a medical procedure which allows an injected dye to display the duct system on an x ray (pancreatograms).
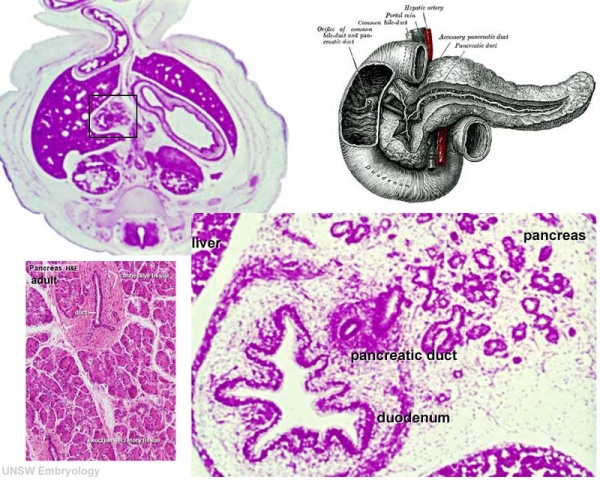
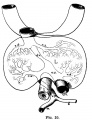
Human (week 8, Stage 22) pancreas
- Functions - exocrine (amylase, alpha-fetoprotein) and endocrine (pancreatic islets)
- Pancreatic buds - endoderm, covered in splanchnic mesoderm
- Pancreatic bud formation - duodenal level endoderm, splanchnic mesoderm forms dorsal and ventral mesentery, dorsal bud (larger, first), ventral bud (smaller, later)
- Duodenum growth/rotation - brings ventral and dorsal buds together, fusion of buds
- Pancreatic duct - ventral bud duct and distal part of dorsal bud, exocrine function
- Islet cells- cords of endodermal cells form ducts, which cells bud off to form islets
- Links: Gastrointestinal Tract - Pancreas Development | Image - Pancreas duct formation
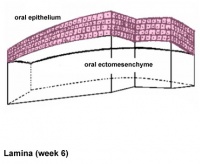
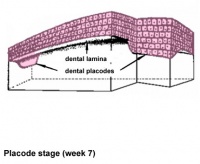
Teeth
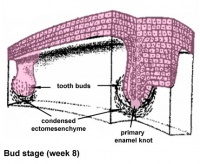
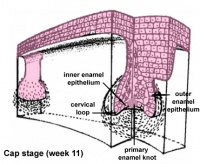
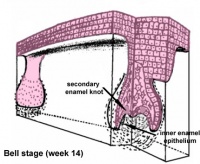
Epitheilal/mesenchymal (ectoderm first pharyngeal arch and neural crest ectomesenchymal cells) interactions in development and has a major contribution from the neural crest. This has been described as 5 stages of development from late embryonic through early fetal period forming the deciduous teeth. Humans have 2 sets of teeth, the deciduous and then the adult teeth that replace them. (More? tooth)
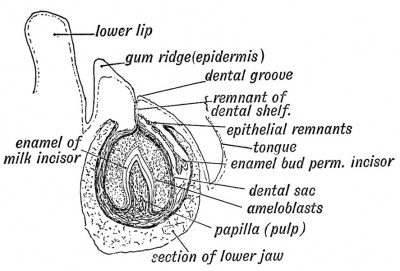
Fetal incisor tooth at 6 months
| Gastrointestinal Tract Terms
|
- allantois - An extraembryonic membrane, endoderm in origin extension from the early hindgut, then cloaca into the connecting stalk of placental animals, connected to the superior end of developing bladder. In reptiles and birds, acts as a reservoir for wastes and mediates gas exchange. In mammals is associated/incorporated with connecting stalk/placental cord fetal-maternal interface.
- amnion - An extra-embryonic membrane, ectoderm and extraembryonic mesoderm in origin, also forms the innermost fetal membrane, that produces amniotic fluid. This fluid-filled sac initially lies above the trilaminar embryonic disc and with embryoic disc folding this sac is drawn ventrally to enclose (cover) the entire embryo, then fetus. The presence of this membrane led to the description of reptiles, bird, and mammals as amniotes.
- amniotic fluid - The fluid that fills amniotic cavity totally encloses and cushions the embryo. Amniotic fluid enters both the gastrointestinal and respiratory tract following rupture of the buccopharyngeal membrane. The late fetus swallows amniotic fluid.
- atresia - is an abnormal interruption of the tube lumen, the abnormality naming is based upon the anatomical location.
- buccal - (Latin, bucca = cheek) A term used to relate to the mouth (oral cavity).
- bile salts - Liver synthesized compounds derived from cholesterol that function postnatally in the small intestine to solubilize and absorb lipids, vitamins, and proteins. These compounds act as water-soluble amphipathic detergents. liver
- buccopharyngeal membrane - (oral membrane) (Latin, bucca = cheek) A membrane which forms the external upper membrane limit (cranial end) of the early gastrointestinal tract. This membrane develops during gastrulation by ectoderm and endoderm without a middle (intervening) layer of mesoderm. The membrane lies at the floor of the ventral depression (stomodeum) where the oral cavity will open and will breakdown to form the initial "oral opening" of the gastrointestinal tract. The equivilent membrane at the lower end of the gastrointestinal tract is the cloacal membrane.
- celiac artery - (celiac trunk) main blood supply to the foregut, excluding the pharynx, lower respiratory tract, and most of the oesophagus.
- cholangiocytes - epithelial cells that line the intra- and extrahepatic ducts of the biliary tree. These cells modify the hepatocyte-derived bile, and are regulated by hormones, peptides, nucleotides, neurotransmitters, and other molecules. liver
- cloaca - (cloacal cavity) The term describing the common cavity into which the intestinal, genital, and urinary tracts open in vertebrates. Located at the caudal end of the embryo it is located on the surface by the cloacal membrane. In many species this common cavity is later divided into a ventral urogenital region (urogenital sinus) and a dorsal gastrointestinal (rectal) region.
- cloacal membrane - Forms the external lower membrane limit (caudal end) of the early gastrointestinal tract (GIT). This membrane is formed during gastrulation by ectoderm and endoderm without a middle (intervening) layer of mesoderm. The membrane breaks down to form the initial "anal opening" of the gastrointestinal tract.
- coelomic cavity - (coelom) Term used to describe a space. There are extra-embryonic and intra-embryonic coeloms that form during vertebrate development. The single intra-embryonic coelom forms the 3 major body cavities: pleural cavity, pericardial cavity and peritoneal cavity.
- crypt of Lieberkühn - (intestinal gland, intestinal crypt) intestinal villi epithelia extend down into the lamina propria where they form crypts that are the source of epithelial stem cells and immune function.
- duplication - is an abnormal incomplete tube recanalization resulting in parallel lumens, this is really a specialized form of stenosis. (More? Image - small intestine duplication)
- esophageal - (oesophageal)
- foregut - first embryonic division of gastrointestinal tract extending from the oral (buccopharyngeal) membrane and contributing oesophagus, stomach, duodenum (to bile duct opening), liver, biliary apparatus (hepatic ducts, gallbladder, and bile duct), and pancreas. The forgut blood supply is the celiac artery (trunk) excluding the pharynx, lower respiratory tract, and most of the oesophagus.
- galactosemia - Metabolic abnormality where the simple sugar galactose (half of lactose, the sugar in milk) cannot be metabolised. People with galactosemia cannot tolerate any form of milk (human or animal). Detected by the Guthrie test.
- gastric transposition - clinical term for postnatal surgery treatment for esophageal atresia involving esophageal replacement. Typically performed on neonates between day 1 to 4. (More? gastrointestinal abnormalities | PMID 28658159
- gastrointestinal divisions - refers to the 3 embryonic divisions contributing the gastrointestinal tract: foregut, Midgut and hindgut.
- gastrula - (Greek, gastrula = little stomach) A stage of an animal embryo in which the three germ layers (endoderm/mesoderm/ectoderm) have just formed. All of these germ layers have contributions to the gastrointestinal tract.
- gastrulation - The process of differentiation forming a gastrula. Term means literally means "to form a gut" but is more in development, as this process converts the bilaminar embryo (epiblast/hypoblast) into the trilaminar embryo (endoderm/mesoderm/ectoderm) establishing the 3 germ layers that will form all the future tissues of the entire embryo. This process also establishes the the initial body axes. (More? gastrulation)
- Guthrie test - (heel prick) A neonatal blood screening test developed by Dr Robert Guthrie (1916-95) for determining a range of metabolic disorders and infections in the neonate. (More? Guthrie test)
- heterotaxia - (Greek heteros = different; taxis = arrangement) is the right/left transposition of thoracic and/or abdominal organs.
- hindgut - final embryonic division of gastrointestinal tract extending to the cloacal membrane and contributing part of the transverse colon (left half to one third), descending colon, sigmoid colon, rectum, part of anal canal (superior), urinary epithelium (bladder and most urethra). The hindgut blood supply is the inferior mesenteric artery.
- inferior mesenteric artery - main blood supply to the hindgut
- intestine - (bowel) part of the gastrointestinal tract (GIT) lying between the stomach and anus where absorption of nutrients and water occur. This region is further divided anatomically and functionally into the small intestine or bowel (duodenum, jejunum and ileum) and large intestine or bowel (cecum and colon).
- intestinal perforation - gastrointestinal abnormality identified in neonates can be due to necrotizing enterocolitis, Hirschsprung’s disease or meconium ileus.
- intraembryonic coelom - The "horseshoe-shaped" space (cavity) that forms initially in the third week of development in the lateral plate mesoderm that will eventually form the 3 main body cavities: pericardial, pleural, peritoneal. The intraembryonic coelom communicates transiently with the extraembryonic coelom.
- meconium ileus intestine obstruction within the ileum due to abnormal meconium properties.
- mesentery - connects gastrointestinal tract to the posterior body wall and is a double layer of visceral peritoneum.
- mesothelium - The mesoderm derived epithelial covering of coelomic organs and also line their cavities.
- Midgut - middle embryonic division of gastrointestinal tract contributing the small intestine (including duodenum distal bile duct opening), cecum, appendix, ascending colon, and part of the transverse colon (right half to two thirds). The midgut blood supply is the superior mesenteric artery.
- neuralation - The general term used to describe the early formation of the nervous system. It is often used to describe the early events of differentiation of the central ectoderm region to form the neural plate, then neural groove, then neural tube. The nervous system includes the central nervous system (brain and spinal cord) from the neural tube and the peripheral nervous system (peripheral sensory and sympathetic ganglia) from neural crest. In humans, early neuralation begins in week 3 and continues through week 4.
- neural crest - region of cells at the edge of the neural plate that migrates throughout the embryo and contributes to many different tissues. In the gastrointestinal tract it contributes mainly the enteric nervous system within the wall of the gut responsible for peristalsis and secretion.
- peritoneal stomata - the main openings forming the pathways for drainage of intra-peritoneal fluid from the peritoneal cavity into the lymphatic system.
- pharynx - uppermost end of gastrointestinal and respiratory tract, in the embryo beginning at the buccopharyngeal membrane and forms a major arched cavity within the phrayngeal arches.
- recanalization - describes the process of a hollow structure becoming solid, then becoming hollow again. For example, this process occurs during GIT, auditory and renal system development.
- retroperitoneal - (retroperitoneum) is the anatomical space (sometimes a potential space) in the abdominal cavity behind (retro) the peritoneum. Developmentally parts of the GIT become secondarily retroperitoneal (part of duodenum, ascending and descending colon, pancreas)
- somitogenesis The process of segmentation of the paraxial mesoderm within the trilaminar embryo body to form pairs of somites, or balls of mesoderm. A somite is added either side of the notochord (axial mesoderm) to form a somite pair. The segmentation does not occur in the head region, and begins cranially (head end) and extends caudally (tailward) adding a somite pair at regular time intervals. The process is sequential and therefore used to stage the age of many different species embryos based upon the number visible somite pairs. In humans, the first somite pair appears at day 20 and adds caudally at 1 somite pair/4 hours (mouse 1 pair/90 min) until on average 44 pairs eventually form.
- splanchnic mesoderm - Gastrointestinal tract (endoderm) associated mesoderm formed by the separation of the lateral plate mesoderm into two separate components by a cavity, the intraembryonic coelom. Splanchnic mesoderm is the embryonic origin of the gastrointestinal tract connective tissue, smooth muscle, blood vessels and contribute to organ development (pancreas, spleen, liver). The intraembryonic coelom will form the three major body cavities including the space surrounding the gut, the peritoneal cavity. The other half of the lateral plate mesoderm (somatic mesoderm) is associated with the ectoderm of the body wall.
- stomodeum - (stomadeum, stomatodeum) A ventral surface depression on the early embryo head surrounding the buccopharyngeal membrane, which lies at the floor of this depression. This surface depression lies between the maxillary and mandibular components of the first pharyngeal arch.
- stenosis - abnormal a narrowing of the tube lumen, the abnormality naming is based upon the anatomical location.
- superior mesenteric artery - main blood supply to the Midgut.
- viscera - the internal organs in the main cavities of the body, especially those in the abdomen, for example the Template:Intestines.
- visceral peritoneum - covers the external surfaces of the intestinal tract and organs within the peritoneum. The other component (parietal peritoneum) lines the abdominal and pelvic cavity walls.
- yolk sac - An extraembryonic membrane which is endoderm origin and covered with extraembryonic mesoderm. Yolk sac lies outside the embryo connected initially by a yolk stalk to the midgut with which it is continuous with. The endodermal lining is continuous with the endoderm of the gastrointestinal tract. The extra-embryonic mesoderm differentiates to form both blood and blood vessels of the vitelline system. In reptiles and birds, the yolk sac has a function associated with nutrition. In mammals the yolk sac acts as a source of primordial germ cells and blood cells. Note that in early development (week 2) a structure called the "primitive yolk sac" forms from hypoblast, this is an entirely different structure.
- yolk stalk - (vitelline duct, omphalomesenteric duct, Latin, vitellus = yolk of an egg) The endodermal connection between the midgut and the yolk sac. See vitelline duct.
|
|
|
Additional Information
| Additional Information - Content shown under this heading is not part of the material covered in this class. It is provided for those students who would like to know about some concepts or current research in topics related to the current class page.
|
Spleen
Embryonic Data (Kyoto Collection)[4]
- Carnegie stage 14 to 17 (week 4-6, GA week 6-8) - appears as a bulge in the dorsal mesogastrium.
- Mesothelium was pseudostratified until stage (16 replaced with high and then low columnar cells.
- Basement membrane was obvious after stage 17.
- Hematopoietic cells detected after stage 18.
- Carnegie stage 20 (week 8, GA week 10) - the spleen is apparent.
- Mesenchymal cells differentiated from cells in dorsal mesogastrium and sinus formation started at stage 20.
- Hilus formation after stage 20.
- Arteries and veins parallel entries at stage 23.
- Intra-splenic folds appear later.
- Rate of increase in spleen length in relation to that of stomach length along the cranial-caudal direction remained constant during 19 - 23.
| Spleen Development Timeline
|
Human Embryonic Spleen Development
| Week
|
Carnegie Stage
|
Feature
|
| Week 5
|
14
|
Carnegie stage 14 to 17
appears as a bulge in the dorsal mesogastrium. Mesothelium pseudostratified.
|
|
|
15
|
|
| Week 6
|
16
|
Mesothelium (pseudostratified} replaced with high columnar cells and then low columnar cells.
|
|
|
17
|
Basement membrane present after this stage.
|
| Week 7
|
18
|
Hematopoietic cells detected.
|
| Week 8
|
20
|
Spleen is now apparent. Mesenchymal cells differentiated from cells in dorsal mesogastrium. Sinus and hilus formation after this stage.
|
|
|
23
|
Arteries and veins parallel entries at this stage.
|
| Human data.[4] Links: spleen | Kyoto Collection | Timeline human development
|
|
Fetal data[5]
- week 15 (GA 17 weeks) - alpha-smooth muscle actin (alpha-SMA)-positive reticulum cells scattered around the arterioles.
- week 18 to 21 (GA 20 - 23 weeks) - alpha-SMA-positive reticulum cells increase in number and began to form a reticular framework. An accumulation of T and B lymphocytes occurred within the framework, and a primitive white pulp was observed around the arterioles.
- week 22 (GA 24 weeks) - antigenic diversity of the reticular framework was observed, and T and B lymphocytes were segregated in the framework. T lymphocytes were sorted into the alpha-SMA-positive reticular framework, and the periarteriolar lymphoid sheath (PALS) was formed around the arteriole. B lymphocytes aggregated in eccentric portions to the PALS and formed the lymph follicle (LF). The reticular framework of the LF was alpha-SMA-negative.
- week 24 (GA 26 weeks) - marginal zone appeared in the alpha-SMA-positive reticular framework around the white pulp.
- Links: spleen
Embryonic Liver Timeline
The table below is a detailed timeline overview of human liver development.
Embryonic Liver Development Timeline
| Carnegie Stage
|
Age (days)
|
CRL (mm)
|
Biliary system
|
Vascular
|
Hepatic parenchyma
|
| 14
|
33
|
7
|
- Bile duct - primordial duct links primitive intestine and liver parenchyma. Thick-walled tube (95 µm diameter) small lumen (22 µm diameter).
- Gall bladder - elongated tube further dilated, thick wall (125 µm diameter) and a narrow lumen (43 µm diameter).
|
- Hepatic sinusoids - intra-hepatic vasculature present
- Three venous tributaries flow into the liver sinusoids - right and left placental vein and a single vitelline vein.
|
- Cords of liver cells fragmented by vascular network of hepatic sinusoids.
- Between pericardial cavity (top) and mesonephros (bottom).
- Upper pole of the liver lies close to the septum transversum and early ventricles.
- Liver occupies the majority of abdominal cavity.
|
| 18
|
46
|
15
|
- Bile duct (future common bile duct), and a common hepatic duct, in contact with liver parenchyma without penetration.
- Primordium of accessory bile tract is an elongated and fusiform gall bladder projecting forward and by a short cystic duct that opens into common bile duct.
- Bile duct empties into second part of duodenum on its posterior side.
|
- Portal system visible - portal vein (100 µm diameter) arises from connection of upper mesenteric vein then at region of hepatic hilum (285 µm) divides into portal branches.
- Left umbilical vein empties into anterior extremity of the left portal branch.
- Ductus venosus (80 µm) connects the initial portion of left portal vein to the inferior vena cava.
- Hepatic venous system 3 branches - left hepatic vein (120 µm in diameter), middle hepatic vein (220 µm in diameter) and right hepatic vein (160 µm in diameter). Flows into the sub-cardinal vein.
|
- Liver parenchyma has two anatomical lobes (right and left lobe), separated by anteroposterior plane formed by placental vein.
|
| 21
|
53
|
22.5
|
Bile duct morphology as earlier stage. Common bile duct empties at the level of the proximal duodenum.
|
- Portal vein arises from joining of splenic vein and superior mesenteric vein. At the level of the hepatic hilum, portal vein divides into two branches, right portal branch (420 µm in diameter) and left portal branch (540 µm in diameter). Right portal branch gives rise to a thin branch to caudate lobe. Ventral branch gives rise to segmental portal veins (VIII and V). Dorsal branch gives rise to the segmental portal veins (VI and VII).
- Ductus venosus connects initial portion of left portal vein to inferior vena cava, just upstream from hepatic vein afferents.
- Hepatic venous system as for previous stage.
|
Hepatic parenchyma a large rounded mass.
|
| 23
|
58
|
27
|
Bile duct morphology as earlier stage.
|
- Portal venous system complete.
- Ductus venosus (40 µm) connects initial portion of portal vein to middle hepatic vein.
- hepatic venous system has changed very little from the previous stage. Three hepatic veins empty into inferior vena cava.
|
- Liver parenchyma roughly oval shape, 2 symmetrical hepatic lobes. The quadrate and caudate lobes are identifiable.
- Upper pole of the liver bounded above by diaphragm.
|
Data from a recent human study[6]
Links: liver | Carnegie stage 14 | 18 | 21 | 23 | simple embryonic timeline | Timeline human development
|
Cholangiocytes
Epithelial cells that line the intra- and extrahepatic ducts of the biliary tree. These cells modify the hepatocyte-derived bile, and are regulated by hormones, peptides, nucleotides, neurotransmitters, and other molecules.
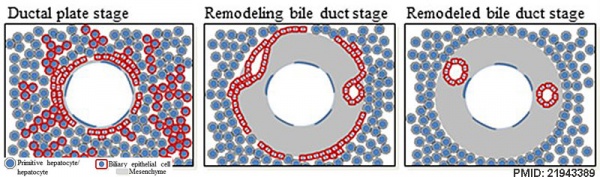
- Three-dimensional reconstructions of intrahepatic bile duct tubulogenesis in human liver[7]
- initial transition of primitive hepatocytes into cholangiocytes shaping the ductal plate
- process of maturation and remodeling where the intrahepatic biliary tree develops through an asymmetrical form of cholangiocyte tubulogenesis.
Teeth Genetics
- Review - PAX9 gene mutations and tooth agenesis[8] "Paired box 9 (PAX9) is one of the best-known transcription factors involved in the development of human dentition. Mutations in PAX9 gene could, therefore, seriously influence the number, position and morphology of the teeth in an affected individual. To date, over 50 mutations in the gene have been reported as associated with various types of dental agenesis (congenitally missing teeth) and other inherited dental defects or variations. The most common consequence of PAX9 gene mutation is the autosomal-dominant isolated (non-syndromic) oligodontia or hypodontia. In the present review, we are summarizing all known PAX9 mutations as well as their nature and precise loci in the DNA sequence of the gene."
- ↑ Radenkovic G, Radenkovic D & Velickov A. (2018). Development of interstitial cells of Cajal in the human digestive tract as the result of reciprocal induction of mesenchymal and neural crest cells. J. Cell. Mol. Med. , 22, 778-785. PMID: 29193736 DOI.
- ↑ Godlewski G, Gaubert-Cristol R, Rouy S & Prudhomme M. (1997). Liver development in the rat and in man during the embryonic period (Carnegie stages 11-23). Microsc. Res. Tech. , 39, 314-27. PMID: 9407542 <314::AID-JEMT2>3.0.CO;2-H DOI.
- ↑ Godlewski G, Gaubert-Cristol R, Rouy S & Prudhomme M. (1997). Liver development in the rat during the embryonic period (Carnegie stages 15-23). Acta Anat (Basel) , 160, 172-8. PMID: 9718390
- ↑ 4.0 4.1 Endo A, Ueno S, Yamada S, Uwabe C & Takakuwa T. (2015). Morphogenesis of the spleen during the human embryonic period. Anat Rec (Hoboken) , 298, 820-6. PMID: 25403423 DOI.
- ↑ Satoh T, Sakurai E, Tada H & Masuda T. (2009). Ontogeny of reticular framework of white pulp and marginal zone in human spleen: immunohistochemical studies of fetal spleens from the 17th to 40th week of gestation. Cell Tissue Res. , 336, 287-97. PMID: 19255788 DOI.
- ↑ Lhuaire M, Tonnelet R, Renard Y, Piardi T, Sommacale D, Duparc F, Braun M & Labrousse M. (2015). Developmental anatomy of the liver from computerized three-dimensional reconstructions of four human embryos (from Carnegie stage 14 to 23). Ann. Anat. , 200, 105-13. PMID: 25866917 DOI.
- ↑ Vestentoft PS, Jelnes P, Hopkinson BM, Vainer B, Møllgård K, Quistorff B & Bisgaard HC. (2011). Three-dimensional reconstructions of intrahepatic bile duct tubulogenesis in human liver. BMC Dev. Biol. , 11, 56. PMID: 21943389 DOI.
- ↑ Bonczek O, Balcar VJ & Šerý O. (2017). PAX9 gene mutations and tooth agenesis: A review. Clin. Genet. , 92, 467-476. PMID: 28155232 DOI.
BGDB: Lecture - Gastrointestinal System | Practical - Gastrointestinal System | Lecture - Face and Ear | Practical - Face and Ear | Lecture - Endocrine | Lecture - Sexual Differentiation | Practical - Sexual Differentiation | Tutorial
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2026, Mayıs 5) Embryology BGDB Gastrointestinal - Late Embryo. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/BGDB_Gastrointestinal_-_Late_Embryo
- What Links Here?
- © Dr Mark Hill 2026, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G