Endocrine - Placenta Development: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| (6 intermediate revisions by the same user not shown) | |||
| Line 29: | Line 29: | ||
==Some Recent Findings== | ==Some Recent Findings== | ||
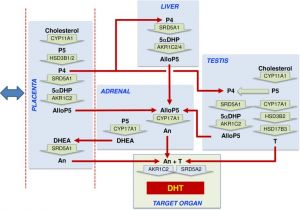
[[File:Model male androsterone synthesis.jpg|alt=Model male second trimester androsterone synthesis|thumb|Model male second trimester androsterone synthesis{{#pmid:30763313|PMID30763313}}]] | |||
{| | {| | ||
|-bgcolor="F5FAFF" | |-bgcolor="F5FAFF" | ||
| | | | ||
* ''' | * '''Expression of steroidogenic enzymes in human placenta according to the gestational age'''{{#pmid:30896833|PMID30896833}} "Female sex steroid hormones, including {{estradiol}} (E2) and {{progesterone}} (P4), serve significant physiological roles in pregnancy. In particular, E2 and P4 influence {{placenta}} formation, maintain pregnancy and stimulate milk production. These hormones are produced by ovaries, adrenal glands and the placenta, of which the latter is a major endocrine organ during pregnancy. However, the mechanism of hormone production during pregnancy remains unclear. In the present study, the regulation of steroid hormones and steroidogenic enzymes was examined in human placenta according to gestational age. In human placental tissues, expression levels of steroidogenic enzymes were determined with reverse transcription‑quantitative polymerase chain reaction and western blotting. The mRNA and protein expression of CYP17A1, HSD17B3 and CYP19A1, which are associated with the synthesis of dehydroepiandrosterone (DHEA) and E2, was elevated at different gestational ages in human placenta. In addition, to evaluate the correlation between serum and placental‑produced hormones, steroid hormone levels, including pregnenolone (PG), DHEA, P4, testosterone (T) and E2, were examined in serum and placenta. Serum and placenta expression of DHEA and E2 increased with gestational age, whereas T and P4 were differently regulated in placenta and serum. To confirm the mechanism of steroidogenesis in vitro, placental BeWo cells were treated with E2 and P4, which are the most important hormones during pregnancy. The mRNA and protein expression of steroidogenic enzymes was significantly altered by E2 in vitro. These results demonstrated that concentration of steroid hormones was differently regulated by steroidogenic enzymes in the placenta depending on the type of the hormones, which may be critical to maintain pregnancy." | ||
* '''Alternative (backdoor) androgen production and masculinization in the human fetus'''{{#pmid:30763313|PMID30763313}} "Masculinization of the external genitalia in humans is dependent on formation of 5α-dihydrotestosterone (DHT) through both the canonical androgenic pathway and an alternative (backdoor) pathway. The fetal testes are essential for canonical androgen production, but little is known about the synthesis of backdoor androgens, despite their known critical role in masculinization. ...Results show that androsterone is the principal backdoor androgen in the male fetal circulation and that DHT is undetectable (<1 ng/mL), while in female fetuses, there are significantly lower levels of androsterone and testosterone. In the male, intermediates in the backdoor pathway are found primarily in the placenta and fetal liver, with significant androsterone levels also in the fetal adrenal. Backdoor intermediates, including androsterone, are only present at very low levels in the fetal testes. This is consistent with transcript levels of enzymes involved in the alternate pathway (steroid 5α-reductase type 1 [SRD5A1], aldo-keto reductase type 1C2 [AKR1C2], aldo-keto reductase type 1C4 [AKR1C4], cytochrome P450 17A1 [CYP17A1]), as measured by quantitative PCR (qPCR). These data identify androsterone as the predominant backdoor androgen in the human fetus and show that circulating levels are sex dependent, but also that there is little de novo synthesis in the testis. Instead, the data indicate that {{placenta}}l progesterone acts as substrate for synthesis of backdoor androgens, which occurs across several tissues. Masculinization of the human fetus depends, therefore, on testosterone and androsterone synthesis by both the fetal testes and nongonadal tissues, leading to DHT formation at the genital tubercle. Our findings also provide a solid basis to explain why placental insufficiency is associated with disorders of sex development in humans." | |||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | {| class="wikitable mw-collapsible mw-collapsed" | ||
| Line 48: | Line 44: | ||
Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Endocrine+Placenta ''Endocrine Placenta''] | Search term: [http://www.ncbi.nlm.nih.gov/pubmed/?term=Endocrine+Placenta ''Endocrine Placenta''] | ||
|} | |} | ||
{| class="wikitable mw-collapsible mw-collapsed" | |||
! Older papers | |||
|- | |||
| {{Older papers}} | |||
* '''Review - The endocrine function of human placenta: an overview'''{{#pmid:26615903|PMID26615903}} "During pregnancy, several tightly coordinated and regulated processes take place to enable proper fetal development and gestational success. The formation and development of the placenta is one of these critical pregnancy events. This organ plays essential roles during gestation, including fetal nourishment, support and protection, gas exchange and production of several hormones and other mediators. Placental hormones are mainly secreted by the syncytiotrophoblast, in a highly and tightly regulated way. These hormones are important for pregnancy establishment and maintenance, exerting autocrine and paracrine effects that regulate decidualization, placental development, angiogenesis, endometrial receptivity, embryo implantation, immunotolerance and fetal development. In addition, because they are released into maternal circulation, the profile of their blood levels throughout pregnancy has been the target of intense research towards finding potential robust and reliable biomarkers to predict and diagnose pregnancy-associated complications." | |||
* '''The feto-placental unit, and potential roles of dehydroepiandrosterone (DHEA) in prenatal and postnatal brain development'''{{#pmid:26485665|PMID26485665}} "Synthesis of dehydroepiandrosterone (DHEA) by the fetal adrenal gland is important for placental oestrogen production, and may also be important for modulating the effects of glucocorticoids on the developing brain. ... Together, the studies outlined in this review indicate that the androgen DHEA is an important hormone of adrenal and Central Nervous System (CNS) origin in the fetal and postnatal spiny mouse. Disturbance of the development of these fetal tissues, and/or of the relationship between the fetal adrenal gland and placenta during pregnancy, may have significant consequences for fetal development, placental function, and maturation of the brain. It is proposed that such disturbances of normal adrenal function could account for some of the neuropathologies that arise in juvenile and adult offspring following illness and stress experienced by the mother during pregnancy." | |||
* '''Human Chorionic Gonadotropin Induces Human Macrophages to Form Intracytoplasmic Vacuoles Mimicking Hofbauer Cells in Human Chorionic Villi'''{{#pmid:23128164|PMID23128164}} The most characteristic morphological feature of macrophages in the stroma of placental villi, known as Hofbauer cells, is their highly vacuolated appearance. They also show positive immunostaining for human chorionic gonadotropin ({{hCG}})." | |||
* '''Serum biomarkers for predicting pregnancy outcome in women undergoing IVF'''{{#pmid:22563548|PMID22563548}} "This study was performed to assess the prognostic value of serum hCG, progesterone, and inhibin A levels measured at 11 days post-ET for predicting pregnancy outcome in women participating in IVF. Between May 2005 and April 2008, sera were obtained from 70 infertile women who underwent IVF-ET at 11 days post-ET and stored. HCG, progesterone, and inhibin A levels were measured by commercial enzyme-linked immunosorbent assay kits. The predictive accuracy of {{hCG}}, progesterone, and inhibin A levels for establishment of intrauterine pregnancy and ongoing pregnancy was calculated by receiver-operating characteristic curve analysis. For the prediction of intrauterine and ongoing pregnancy, serum hCG was better than progesterone and inhibin A. The predictive performance of progesterone and inhibin A was similar. The serum progesterone and inhibin A levels were significantly correlated each other (r=0.915, p=0.010)." | |||
|} | |||
==Human Chorionic Gonadotrophin== | ==Human Chorionic Gonadotrophin== | ||
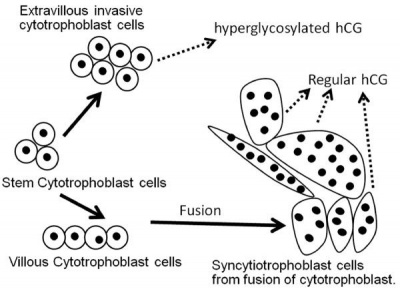
[[File:Trophoblast hCG function.jpg|thumb|Trophoblast hCG function]] | [[File:Trophoblast hCG function.jpg|thumb|Trophoblast hCG function]] | ||
| Line 107: | Line 114: | ||
:'''Links:''' {{implantation}} | :'''Links:''' {{implantation}} | ||
==Other Factors== | |||
* placental tachykinins | |||
* neurokinin B | |||
* endokinin | |||
Latest revision as of 08:44, 21 May 2019
| Embryology - 21 May 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Introduction
For complete notes on placenta development and function see Placenta Development.
Lecture - Placenta Development
| Placenta Links: placenta | Lecture - Placenta | Lecture Movie | Practical - Placenta | implantation | placental villi | trophoblast | maternal decidua | uterus | endocrine placenta | placental cord | placental membranes | placenta abnormalities | ectopic pregnancy | Stage 13 | Stage 22 | placenta histology | placenta vascular | blood vessel | cord stem cells | 2013 Meeting Presentation | Placenta Terms | Category:Placenta | ||
|
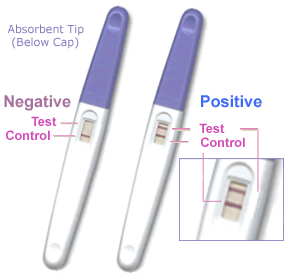
- Human chorionic gonadotrophin (hCG) - like leutenizing hormone, supports corpus luteum in ovary, pregnant state rather than menstrual, maternal urine in some pregnancy testing
- Human chorionic somatommotropin (hCS) - or placental lactogen stimulate (maternal) mammary development
- Human chorionic thyrotropin (hCT)
- Human chorionic corticotrophin (hCACTH)
- progesterone and estrogens - support maternal endometrium
- Progesterone-Associated Endometrial Protein (PAEP, pregnancy protein-14, PP14) - glycoprotein hormone from decidualized endometrium with possible immunoregulatory activities.
- IGF binding protein-1 (IGFBP-1) peptide hormone from decidualized endometrium increases during the first trimester, peaks during the second trimester, falls briefly, and peaks a second time before term.[1]
- Relaxin - corpus luteum then placenta in the first trimester, then declines in the second trimester. Binds to relaxin receptors on both the decidua and cytotrophoblasts.
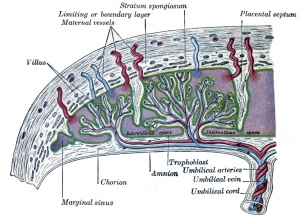
- Placenta - Maternal (decidua) and Fetal (trophoblastic cells, extra-embryonic mesoderm) components
- Endocrine function - maternal and fetal precursors, synthesis and secretion
- Protein Hormones - chorionic gonadotropin (hCG), chorionic somatomammotropin (hCS) or placental lactogen (hPL), chorionic thyrotropin (hCT), chorionic corticotropin (hCACTH)
- hCG - up to 20 weeks, fetal adrenal cortex growth and maintenance
- hCS – rise through pregnancy, stimulates maternal metabolic processes, breast growth
- Steroid Hormones - progesterone (maintains pregnancy), estrogens (fetal adrenal/placenta)
- Protein Hormones - chorionic gonadotropin (hCG), chorionic somatomammotropin (hCS) or placental lactogen (hPL), chorionic thyrotropin (hCT), chorionic corticotropin (hCACTH)
Some Recent Findings
|
| More recent papers |
|---|
|
This table allows an automated computer search of the external PubMed database using the listed "Search term" text link.
More? References | Discussion Page | Journal Searches | 2019 References | 2020 References Search term: Endocrine Placenta
|
| Older papers |
|---|
| These papers originally appeared in the Some Recent Findings table, but as that list grew in length have now been shuffled down to this collapsible table.
See also the Discussion Page for other references listed by year and References on this current page.
|
Human Chorionic Gonadotrophin
Human chorionic gonadotrophin (hCG) like leutenizing hormone, supports corpus luteum in ovary, pregnant state rather than menstrual. Presence in the maternal urine is the basis of some pregnancy testing.
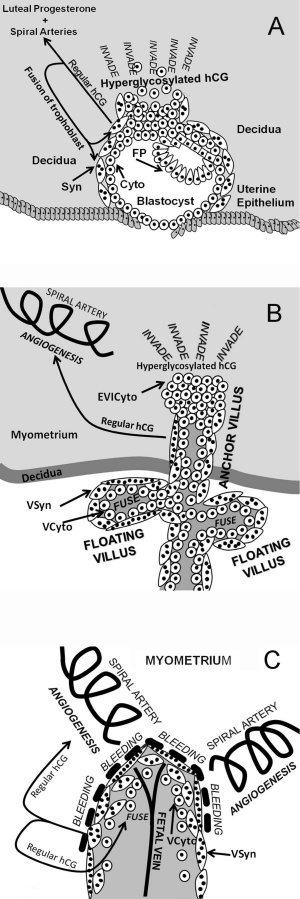
The level of the hyperglycosylated form (HCG-H) is high during the implantation phase and then decreases to the end of the first trimester.[8] In addition to the implantation role it has suggested immune cell modulation and endothelial functions.
Trophoblast cell hCG
- hCG Links: hCG | Trophoblast hCG function | Trophoblast cell hCG | implantation | placenta | NIH - The History of the Pregnancy Test
Placental Estrogen
Maternal late pregnancy shows a large increase in estriol levels.[9]
- fetal adrenal - gland production of dehydroepiandrosterone sulphate (DHEAS)
- fetal liver - conversion to 16α-OH-DHEAS
- placenta - sulphate removal to allow aromatization to estriol.
Placental estrogen, mainly estriol, suppresses gonadotropin secretion from the maternal pituitary gland. Maternal estrogen levels are often a useful indicator of fetal well being.[10][11]
- Uterus - stimulates growth of the myometrium, antagonizes the myometrial-suppressing activity of progesterone.
- Mammary Gland - stimulates mammary gland ductal and alveolar growth.
- Fetal Ovary - stimulates development of female fetal ovary.[12]
A second role for fetal adrenal DHEA-S is possible regulation of the effects of glucocorticoids on the developing brain.[5]
- Links: adrenal
Placental Progesterone
The fetal placenta production of progesterone throughout the second and third trimesters is a key to maintaining the uterus in the non-menstral pregnant state.[13] A second identified immunological role is in modulation of cytokine production.[14]
Human Chorionic Somatommotropin
Human chorionic somatommotropin hormone (hCS, CSH) is also called placental lactogen, due to its role in stimulating maternal mammary development, there may be little growth (somatogen) effects of this hormone. The hormone is structurally similar to both growth hormone (GH) and prolactin (PRL} and binds strongly to PRL receptors but weakly to GH receptors.
- Links: mammary gland
Cortiticotropin Releasing Hormone
The placenta synthesises urocortins (Ucn 1, Ucn 2, Ucn 3), cortiticotropin releasing hormone (CRH) analogues.[15] Appears to be produced by chorio-decidual cells.
Relaxin
The placenta and corpus luteum produce relaxin, a 6 kDa peptide hormone structurally similar to insulin. The hormone stimulates in early pregnancy both uterine growth and vascularization associated with implantation. It is also postulated to have other roles in the menstrual cycle[16]
- Links: implantation
Other Factors
- placental tachykinins
- neurokinin B
- endokinin
References
- ↑ Rutanen EM. (2000). Insulin-like growth factors in obstetrics. Curr. Opin. Obstet. Gynecol. , 12, 163-8. PMID: 10873115
- ↑ 2.0 2.1 O'Shaughnessy PJ, Antignac JP, Le Bizec B, Morvan ML, Svechnikov K, Söder O, Savchuk I, Monteiro A, Soffientini U, Johnston ZC, Bellingham M, Hough D, Walker N, Filis P & Fowler PA. (2019). Alternative (backdoor) androgen production and masculinization in the human fetus. PLoS Biol. , 17, e3000002. PMID: 30763313 DOI.
- ↑ Hong SH, Kim SC, Park MN, Jeong JS, Yang SY, Lee YJ, Bae ON, Yang HS, Seo S, Lee KS & An BS. (2019). Expression of steroidogenic enzymes in human placenta according to the gestational age. Mol Med Rep , 19, 3903-3911. PMID: 30896833 DOI.
- ↑ Costa MA. (2016). The endocrine function of human placenta: an overview. Reprod. Biomed. Online , 32, 14-43. PMID: 26615903 DOI.
- ↑ 5.0 5.1 Quinn TA, Ratnayake U, Dickinson H, Castillo-Melendez M & Walker DW. (2016). The feto-placental unit, and potential roles of dehydroepiandrosterone (DHEA) in prenatal and postnatal brain development: A re-examination using the spiny mouse. J. Steroid Biochem. Mol. Biol. , 160, 204-13. PMID: 26485665 DOI.
- ↑ Yamaguchi M, Ohba T, Tashiro H, Yamada G & Katabuchi H. (2013). Human chorionic gonadotropin induces human macrophages to form intracytoplasmic vacuoles mimicking Hofbauer cells in human chorionic villi. Cells Tissues Organs (Print) , 197, 127-35. PMID: 23128164 DOI.
- ↑ Kim JH, Shin MS, Yi G, Jee BC, Lee JR, Suh CS & Kim SH. (2012). Serum biomarkers for predicting pregnancy outcome in women undergoing IVF: human chorionic gonadotropin, progesterone, and inhibin A level at 11 days post-ET. Clin Exp Reprod Med , 39, 28-32. PMID: 22563548 DOI.
- ↑ Evans J. (2016). Hyperglycosylated hCG: a Unique Human Implantation and Invasion Factor. Am. J. Reprod. Immunol. , 75, 333-40. PMID: 26676718 DOI.
- ↑ Raeside JI. (2017). A Brief Account of the Discovery of the Fetal/Placental Unit for Estrogen Production in Equine and Human Pregnancies: Relation to Human Medicine. Yale J Biol Med , 90, 449-461. PMID: 28955183
- ↑ Rainey WE, Rehman KS & Carr BR. (2004). The human fetal adrenal: making adrenal androgens for placental estrogens. Semin. Reprod. Med. , 22, 327-36. PMID: 15635500 DOI.
- ↑ Parker CR. (1999). Dehydroepiandrosterone and dehydroepiandrosterone sulfate production in the human adrenal during development and aging. Steroids , 64, 640-7. PMID: 10503722
- ↑ Albrecht ED & Pepe GJ. (2010). Estrogen regulation of placental angiogenesis and fetal ovarian development during primate pregnancy. Int. J. Dev. Biol. , 54, 397-408. PMID: 19876841 DOI.
- ↑ Pavlicev M & Norwitz ER. (2018). Human Parturition: Nothing More Than a Delayed Menstruation. Reprod Sci , 25, 166-173. PMID: 28826363 DOI.
- ↑ Raghupathy R & Szekeres-Bartho J. (2016). Dydrogesterone and the immunology of pregnancy. Horm Mol Biol Clin Investig , 27, 63-71. PMID: 26812877 DOI.
- ↑ Fekete EM & Zorrilla EP. (2007). Physiology, pharmacology, and therapeutic relevance of urocortins in mammals: ancient CRF paralogs. Front Neuroendocrinol , 28, 1-27. PMID: 17083971 DOI.
- ↑ Marshall SA, Senadheera SN, Parry LJ & Girling JE. (2017). The Role of Relaxin in Normal and Abnormal Uterine Function During the Menstrual Cycle and Early Pregnancy. Reprod Sci , 24, 342-354. PMID: 27365367 DOI.
Reviews
Costa MA. (2016). The endocrine function of human placenta: an overview. Reprod. Biomed. Online , 32, 14-43. PMID: 26615903 DOI.
De Groot LJ, Chrousos G, Dungan K, Feingold KR, Grossman A, Hershman JM, Koch C, Korbonits M, McLachlan R, New M, Purnell J, Rebar R, Singer F, Vinik A, Tal R, Taylor HS, Burney RO, Mooney SB & Giudice LC. (2000). Endocrinology of Pregnancy. , , . PMID: 25905197
John RM. (2013). Epigenetic regulation of placental endocrine lineages and complications of pregnancy. Biochem. Soc. Trans. , 41, 701-9. PMID: 23697929 DOI.
Evain-Brion D & Malassine A. (2003). Human placenta as an endocrine organ. Growth Horm. IGF Res. , 13 Suppl A, S34-7. PMID: 12914725
Articles
Search PubMed
Search Pubmed: endocrine placenta development
Additional Images
External Links
External Links Notice - The dynamic nature of the internet may mean that some of these listed links may no longer function. If the link no longer works search the web with the link text or name. Links to any external commercial sites are provided for information purposes only and should never be considered an endorsement. UNSW Embryology is provided as an educational resource with no clinical information or commercial affiliation.
- National Institutes of Health (USA) Office of History - The History of the Pregnancy Test | of Pregnancy Testing
Terms
| Endocrine Terms | ||
|---|---|---|
| ||
|
Glossary Links
- Glossary: A | B | C | D | E | F | G | H | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | Numbers | Symbols | Term Link
Cite this page: Hill, M.A. (2024, May 21) Embryology Endocrine - Placenta Development. Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Endocrine_-_Placenta_Development
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G