Paper - Development and histogenesis of the human pineal organ (1935)
| Embryology - 27 Apr 2024 |
|---|
| Google Translate - select your language from the list shown below (this will open a new external page) |
|
العربية | català | 中文 | 中國傳統的 | français | Deutsche | עִברִית | हिंदी | bahasa Indonesia | italiano | 日本語 | 한국어 | မြန်မာ | Pilipino | Polskie | português | ਪੰਜਾਬੀ ਦੇ | Română | русский | Español | Swahili | Svensk | ไทย | Türkçe | اردو | ייִדיש | Tiếng Việt These external translations are automated and may not be accurate. (More? About Translations) |
Gladstone RJ. and Wakeley CPG. Development and histogenesis of the human pineal organ. (1935) J Anat. 69: 427-454.11. PMID 17104550
| Historic Disclaimer - information about historic embryology pages |
|---|
| Pages where the terms "Historic" (textbooks, papers, people, recommendations) appear on this site, and sections within pages where this disclaimer appears, indicate that the content and scientific understanding are specific to the time of publication. This means that while some scientific descriptions are still accurate, the terminology and interpretation of the developmental mechanisms reflect the understanding at the time of original publication and those of the preceding periods, these terms, interpretations and recommendations may not reflect our current scientific understanding. (More? Embryology History | Historic Embryology Papers) |
Development and Histogenesis of the Human Pineal Organ
By R. J. Gladstone, M.D., F.R.C.S., F.R.S.E. University of London, King’s College
and Cecil P. G. Wakeley, D.Sc., F.R.C.S., F.R.S.E.
Senior Surgeon, King’s College Hospital, and West End Hospital for Diseases of the Nervous System; Hunterian Professor Royal College ‘of Surgeons of England
Introduction
In the past and recently, the study of the pineal system has been approached from widely different standpoints, namely: (1) The morphological. (2) The ontogenetic and biological, with reference to certain problems, such as differentiation, including the association of change of function with correlated changes in form and structure; and questions pertaining to the persistence in living animals of organs which, in some remote palaeozoic epoch, have lost their primary function; also the occurrence of degenerative changes in such structures, and the fate of vestigial organs generally. (3) The clinical, - which is intimately concerned with the physiology of endocrine organs, and the pathological, pharmaceutical and surgical aspects.
Owing to the vestigial condition of the pineal body in Man and Mammals generally, the study of the organ in these has hitherto not received the same minute attention that has been given to it in lower types of Vertebrates. Recently, however, the remarkable progress in diagnosis and treatment of tumours of the pineal gland has done much to focus attention on the structure and exact anatomical relations of the human epiphysis, and the practical application of this work has demonstrated the necessity for further detailed investigation.
A comparison of older with more recent literature on the subject shows that, although the pioneer and classical researches carried out by Dendy, Studnitka and others on the pineal system of Fishes, Amphibia and Reptiles has left little of importance to be added, the later work, especially with regard to the development and histogenesis of the human pineal organ, notwithstanding improved technique in staining of special tissues and the details of cell structure, has left many fundamental points unsettled.
In this communication we shall endeavour to limit ourselves as far as possible to an account of our personal observations on the general development, histogenesis and adult structure of the human pineal gland, and we shall reserve the consideration of the clinical aspect of the subject for a separate publication which will include the description of cases and their treatment, special points in the surgical anatomy of the pineal gland and the neighbouring blood vessels, and a general survey of the results obtained by experimental work on the function of the mammalian pineal gland.
Development of the Human Pineal Organ
An examination of the early human embryos in the Anatomical Department of King’s College, London, indicates that the pineal organ of the human subject arises as a simple diverticulum from the roof of the third ventricle shortly after the 12-mm. stage of development, and that it is preceded by the formation of the posterior commissure. The earliest stage described by Krabbe was observed by him in a 15-mm. human embryo. In front of the pineal diverticulum was a solid mass of cells with spherical nuclei. This mass was directed forward. In addition to the posterior commissure there was a slight indication of the habenular commissure.
In a series of closely graded human embryos belonging to Prof. J. E. Frazer, which he has kindly permitted us to examine, the earliest stage in which a pineal diverticulum could be recognised was in a 15-mm. embryo; it appeared as a single median groove open towards the ventricle and situated in the posterior part of the roof of the thalamencephalon. It extended through fifty-four sections of approximately 10 thickness. Posteriorly the groove ‘widened out, but in part of its extent the margins were in contact, and the appearance suggested that they were about to unite with each other, and in this way cut off a short tube, ending blindly in front and opening posteriorly into the third ventricle. A well-developed posterior commissure lay behind the ~ pineal rudiment.
No pineal diverticulum was recognisable, with certainty, in the stages immediately preceding (14 mm.) and succeeding (16 mm.) the 15-mm. specimen just described. The organ was present, however, in a 17-mm. embryo and in succeeding stages.
Among our own specimens there is a well-developed pineal diverticulum in a 19-mm. embryo and also in a 20-mm. embryo (74 weeks); the latter was sectioned coronally, and thus the plane of the sections was transverse with respect to the brain. In this embryo (text-figs. 1, 2, 3) the pineal organ consists of a posterior hollow diverticulum (text-fig. 1), the lumen of which is in wide communication with the cavity of the third ventricle, and an anterior part, the apex of which is directed forward and which contains a central lumen (textfigs. 2 and 3). This lumen is not actually continuous with that of the posterior part, but there is an intermediate segment in which one sees a radial arrangement of the central cells around a virtual lumen. The general form and position of the organ, with the apex directed forward, closely resembles that which is present in the corresponding stage of development in other Mammals, e.g. the rabbit (text-fig. 4), in Birds (text-fig. 5), in Reptiles (text-fig. 6), in Amphibia (text-figs. 7, 17), and in Fishes.
Text-fig. 1. Pineal evagination, 20 mm. Human embryo. The section passes transversely through the posterior part of the diverticulum, where its lumen is continuous with the cavity of the third ventricle.
Text-fig. 2. Pineal organ of the same embryo as text-fig. 1. The section passes transversely through the anterior part of the organ, and shows the central lumen surrounded by radially arranged ependymal cells.
Text-fig. 3. Median linear reconstruction of the pineal region and posterior commissure of the same embryo, transverse sections of which are shown in text-figs. 1 and 2. The apex of the pineal diverticulum is directed forward.
Text-fig. 4. Median longitudinal section of brain of rabbit embryo (16 days, 11 mm.). The apex of the pineal body is directed forward, and the diverticulum shows a central constriction which partially divides it into an anterior and posterior lobe (compare text-figs. 3, 6 and 7).
Text-fig. 5. Median longitudinal section of the pineal diverticulum of a chick embryo (fourth day). The apex is directed forward.
Text-fig. 6. Semi-diagrammatic representation of a medial longitudinal section of an embryo Sphenodon, showing a constriction marking off the anterior (left) lobe, or “parietal organ”, from the posterior (right) lobe or “‘pineal sac”. The anterior end of the organ is directed forward and is to the left in the figure (after A. Dendy).
Text-fig. 7. Median longitudinal section of the pineal diverticulum of a frog tadpole, showing a constriction in the plane of the cranial capsule, which indicates the future line of separation of the “parietal organ” (Stieda) from the “epiphysis” or ‘“‘ pineal sac”. The apex of the organ is directed forward, and is to the right in the figure.
The wall of the diverticulum consists of all three zones of the neural tube, ependymal, mantle and marginal. The ependymal cells are arranged radially round the lumen of the diverticulum (text-fig. 2), and some of their nuclei show mitotic figures. This layer may therefore be regarded as a proliferating zone of ependymal cells, from which newly formed cells migrate outward into the mantle layer. Externally is a thin reticular or marginal zone. This is in relation with the outer covering of pia mater and mesenchyme, in which a plexus of thin-walled capillary vessels forms a conspicuous feature (textfig. 2).
A median linear reconstruction of the region (text-fig. 3) shows the general appearance of the organ at this stage, and also its relation to the posterior commissure, which is now well developed and extends backward for a considerable distance in the roof of the aqueductus cerebri. The general extent of the commissure at this stage of development is well seen in text-fig. 4, which is drawn from a median section through the brain of a rabbit embryo, and textfig. 5, from a fourth day chick embryo. Plate I A shows the concentration of the fibres of the posterior commissure into a round transverse bundle which commonly takes place at a later stage of development, exemplified in an 8-day chick embryo. A paramedial section (text-fig. 8) of the same rabbit embryo as text-fig. 4 shows the connections and general ventro-caudal direction of the fibres of the posterior commissure as they course backward to the pons, and also fibres passing to the interpeduncular region.
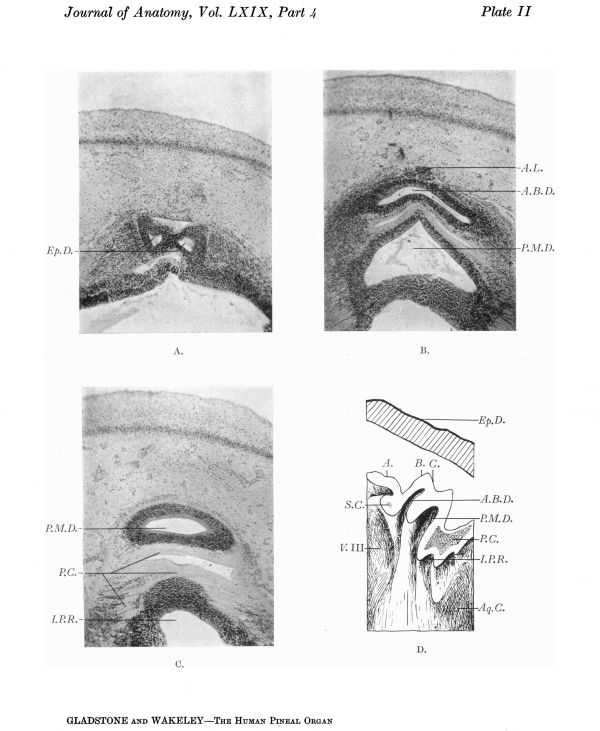
The next stage of development that we shall describe is that seen in a 22-mm. human embryo, obtained at operation by Mr E. McClellan to whom we are also indebted for valuable help in the construction of the wax model, a drawing of which is shown in Plate II D. The model represents a portion of the mid-brain and thalamencephalon magnified 50 diameters. It has been divided into right and left halves by a median sagittal section.
From behind forward the median section of the model (Plate II D) shows a portion of the roof of the aqueductus cerebri, the posterior commissure, and the posterior intercalary plate. The roof of the aqueduct shows a longitudinal median groove and two small pockets produced by thickened bands of the posterior commissure giving rise to a transverse folding of the ependyma. These infrapineal recesses are temporary in nature and will disappear later when the upper part of the intercalary plate is folded backward round the posterior commissure, to form with the commissure a transverse projecting bar between the upper end of the aqueduct and the recessus pinealis. In front of and dorsal to the posterior commissure is a posterior median diverticulum (Plate II B, C, D).
In front of this is an anterior bilobed diverticulum (Plate II B, D). The wall of both the posterior and anterior diverticulum is formed by the whole thickness of the neural tube.
A solid mass of neuro-epithelial cells lies in front of the anterior diverticulum, and appears to have arisen as an outgrowth from its anterior wall. It is partially subdivided into right and left halves by a median fissure, and extends downwards as a flattened band from near the apex to the base of the diverticulum.
Anterior to the two pineal diverticula is an ependymal diverticulum (Plate II A, D), the dorsal or posterior extremity of which is bilobed. The opening of the cavity of this ependymal outgrowth into the third ventricle is to the right of the median plane. Its dorsal wall is directly continuous with the ependymal roof of the third ventricle, and it corresponds to the “dorsal sac”’ of the reptilian brain. In the human foetus it persists as the “‘dorsal”’ or suprapineal diverticulum, which is represented in the adult by the small suprapineal recess.
Text-fig. 8. Paramedian section through the mid-brain of a rabbit embryo (18 days), showing the direction of the fibres of the posterior commissure and their connections with the pons Varolii and interpeduncular region.
It is probable that the anterior pineal diverticulum seen at this stage of development corresponds to the main part of the definitive pineal gland and gives rise to the “posterior lobe” of Krabbe, while the solid mass of neuroepithelial cells in relation to its anterior wall represents the “anterior lobe” of Krabbe. The posterior diverticulum seems to persist for a time as the infrapineal recess which is present in the 6-cm. stage (text-fig. 10).
Text-fig. 9. Transverse section through the pineal organ of a human embryo (6 cm.) showing its relations to the membranes, posterior commissure and subcommissural organ.
Text-fig. 10. A median linear reconstruction of the pineal region of a human embryo (6 cm.) showing the main diverticulum, infrapineal recess, anterior lobe, superior and posterior commissures, the dorsal sac, and an anterior diverticulum possibly representing a vestige of the paraphysis; also the great cerebral vein and opening of the aqueductus cerebri.
If the pineal region be examined at a much later stage of development, e.g. a 6-cm. human embryo, it will be found that the pineal organ has increased considerably in size. Its lumen still communicates with the cavity of the third ventricle, where it opens between the habenular commissure in front and the posterior commissure behind. The long axis of the organ appears to have been rotated backward round a transverse axis passing through its base; so that the apex of the organ, which in the early stages of development was directed forward, now projects upward and backward (text-fig. 10). It lies below the great cerebral vein (text-fig. 10, G.C.V.) and primitive falx cerebri, and above the superior quadrigeminal plate of the mid-brain (text-fig. 10, Q.P.). It is contained in a triangular space bounded below by the roof plate of the neural tube and the layer of pia mater which invests the brain stem, and laterally by right and left membranous laminae, which are attached above to the lower border of the interhemispheric septum or primary falx cerebri. Along the line of junction of the lateral laminae with the lower edge of the interhemispheric septum is a membranous channel which encloses the great cerebral vein (text fig. 11). At a later stage of development when the corpus callosum and fornix grow backward over the thalamencephalon and mid-brain, the thin lower part of the interhemispheric septum disappears (text-fig. 11, S.), while the upper and thicker part persists as the definitive falx cerebri, the inferior sagittal sinus (text-fig. 11, I.8.S.) being developed in its lower border. The septum in the earlier stages of development is much thicker than at the stage described and is formed by a median condensation of the loose mesenchyme which occupies the space between the developing hemispheres. It is not formed by the union of the two layers of a fold, and the same remark applies to the formation of the tentorium and falx cerebelli.
Text-fig. 11. Transverse section through the posterior thalamic region of a human embryo (6 cm.) showing the relation of the cerebral membranes to the great cerebral vein and neural tube at the junction of the lateral plate with the dorsal lamina, and the extreme thinness of the membrane (lower part of interhemispheric septum) which joins the sheath of the great cerebral] vein to the lower margin of the falx cerebri.
Returning to the description of the pineal body itself, there is found at this stage (Plate III A, 4.Z.) an almost solid mass of cells, lying in front of the main or anterior diverticulum and above the habenular commissure. This is the anterior lobe. A recess projecting backward from the posterior wall of the main diverticulum probably corresponds to the posterior pineal diverticulum, described in the preceding specimen (22-mm. embryo). This pocket which has been described as the infrapineal recess disappears at a later stage of development, probably by opening out into the cavity of the third ventricle. The original stalk of the pineal diverticulum thus disappears and the pineal recess of the adult is probably a remnant of the basal part of the central cavity after the distal part of the latter has become closed and separated off from the ventricle. The wall of the apical part of the epiphysis and the anterior lobe is definitely lobulated at this stage, outgrowing finger-shaped processes of the epithelial diverticulum interdigitating with vascular ingrowths from the surrounding pia arachnoid.
Anterior to the pineal diverticulum are two evaginations from the roof of the third ventricle (text-fig. 10). The apices of these outgrowths are directed backward. The more posterior overlaps the base of the anterior lobe of the pineal body, and represents the dorsal sac of Reptiles, while the anterior probably corresponds to the paraphysis of the Reptiles.
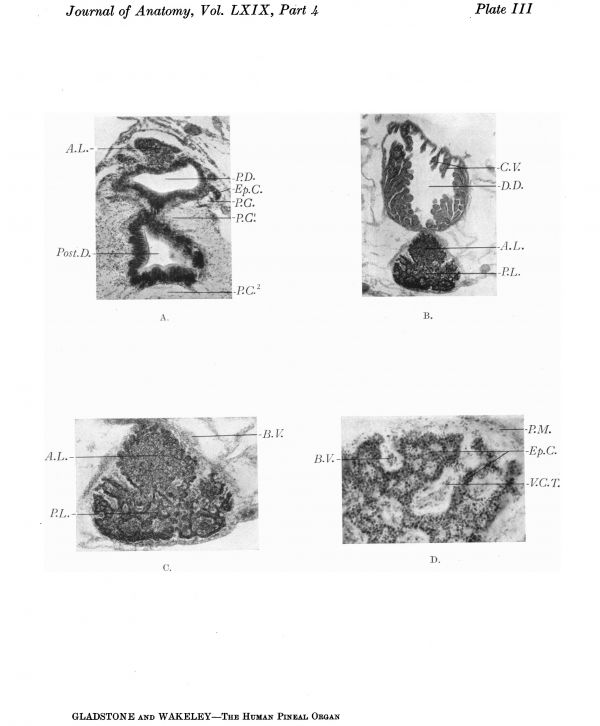
Between the fourth and fifth months of foetal life (text-fig. 12 and Plate III B and C) the distal part of the main posterior lobe and the whole of the anterior lobe of the pineal gland are solid; the stalk and basal part of the posterior lobe are still hollow and in wide communication with the cavity of the third ventricle.
Dorsal to the pineal organ (text-fig. 12, D.D.’, D.D.”’) is a tubular diverticulum from the posterior part of the roof of the third ventricle. This bifurcates behind into two secondary lobes, which extend a short distance beyond the distal end of the pineal organ. The diverticulum is lined internally by a single layer of cubical epithelium, which in many places is invaginated into the lumen of the diverticulum to form a superficial secreting layer of epithelium covering tufts of choroidal villi. If the upper or dorsal wall of the diverticulum is traced forward, it is found to be directly continuous with the choroid plexus of the third ventricle, and since the habenular commissure lies behind the opening of the diverticulum between this and the stalk of the pineal body, it corresponds to the “dorsal sac” of lower Vertebrates.
If the solid “glandular” part of the pineal organ be examined at this stage of development (Plate III B, C, D), it will be seen that both the anterior and posterior lobes consist of a lobulated mass of neuro-epithelial cells, which is penetrated by thin-walled vessels surrounded by loose perivascular sheaths. The epithelial cells are mostly derived from the inner ependymal zone of the diverticulum, but there are also a certain number of cells belonging to the nuclear or mantle zone, which may be distinguished by their position and by being less deeply stained than the ependymal cells, also by their nuclei being vesicular and pale in colour as contrasted with the deeply stained nuclei of the ependymal cells. At the periphery of the gland, branched finger-shaped or club-shaped processes of the epithelial cells grow outward into the surrounding pial tissue, and interdigitate with vascular processes which grow inwards between the epithelial cords (Plate III B, C, D). Further, if cross-sections of the interdigitating processes are observed, it will be seen that the vascular processes of pial tissue appear paler than the surrounding zones of epithelial cells; these vascular mesenchymal areas are limited where they come into contact with the neural epithelium by the external limiting membrane, between which and the zone of deeply stained cells derived from the ependyma are a few sparsely scattered pale cells belonging to the mantle zone. The general arrangement of the epithelial tubes or cords, and their relation to the ingrowing vascular processes, is seen with diagrammatic clearness in Plate IV B and D, photographed from a section of the “pineal sac” of an adult Sphenodon.)
Text-fig. 12. Median linear reconstruction of the pineal region of a 44 months’ human foetus showing anterior opening of aqueductus cerebri; the columnar SC PL. epithelium of the subcommissural organ between this and the posterior PRA commissure, above which is the pineal organ consisting of a main posterior lobe which in the greater part of its extent is solid and a small anterior lobe. Above and in front of the pineal recess : is the superior commissure and above C.Ep.—\ — ay this the bifid dorsal diverticulum. (S.C-O.) | BER 0, p
Should the cross-section of a vascular ingrowth of pial tissue be taken as the centre of an apparent lobule, this will be seen to consist of a core of sinusoidal blood vessels surrounded by a perivascular sheath of pial tissue, which is in contact peripherally with the external limiting membrane of outgrowing neuro-epithelial processes. Proceeding farther outward from the central pale area formed by the vessels and their loose mesenchymatous sheath, there will be found beyond the limiting membrane the reticular and nuclear zones, the latter containing pale cells with vesicular nuclei, and finally a peripheral zone consisting of deeply stained ependymal cells.
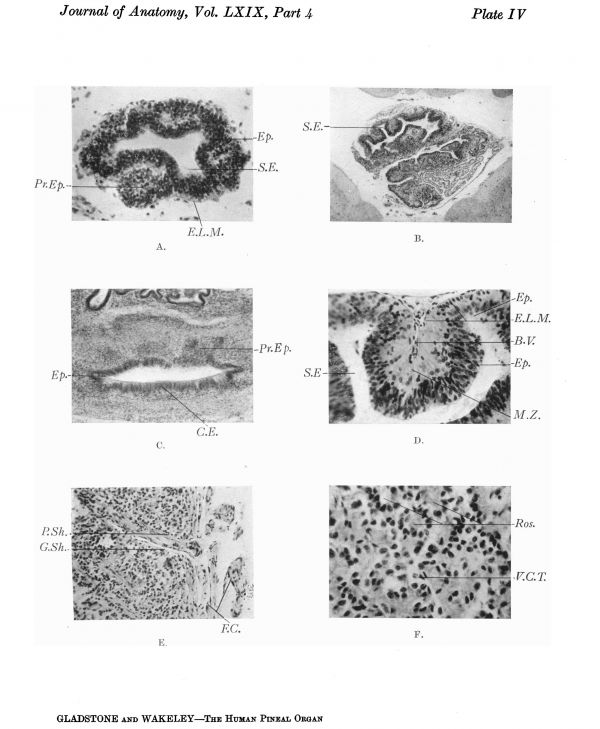
The deeply stained cells surrounding the clear areas have the appearance of epithelial cords cut in various directions. When the section is transverse (Plate IV F), the cords appear as rings of cells, “‘rosettes”’, with deeply stained oval nuclei arranged radially round a palely stained central zone, which is formed by the inner ends of the cells coming into contact in the central axis of the cord. If the section is longitudinal, two parallel rows of nuclei are seen, which are separated by a palely stained axial zone where the inner ends of the cells come into contact in the situation of a virtual lumen. These cords of proliferating ependymal cells are usually grouped in lobules which grow outward between the vascular ingrowths of the pia mater.
There is thus an appearance, produced by cross-sections of the cords, of groups of deeply stained epithelial cells arranged in “ring-like” zones or “rosettes”. These surround the vascular ingrowths, which appear pale. This arrangement of alternating series of branched epithelial and vascular cords is the key to the “mosaic” appearance which is described by Globus and Silbert as characteristic of later stages of foetal life. The “streams” of deeply stained epithelial cells described by these authors are longitudinal sections of the epithelial cords. The central parts of the clear areas of the “mosaic” pattern are transverse or oblique sections through the ingrowing vascular processes, surrounded by pale cells with vesicular nuclei.
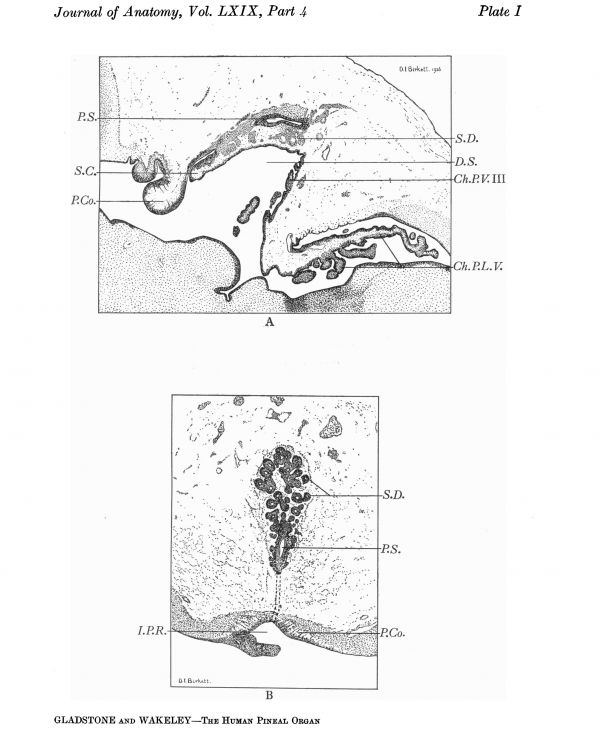
Owing to the radial disposition of the epithelial cells around a central axis which is destitute of nuclei, the general appearance of a group of epithelial cords, cut transversely, is similar to that of an acinar gland, but with the important difference that in the human subject the acini usually have no lumen and no ducts are present. In the early stages of development of the pineal organ in some Birds (Plate I A and B), the main epithelial outgrowths are at first hollow and in communication with a principal duct, which corresponds to the lumen of the primary diverticulum and opens into the cavity of the third ventricle. In these Birds, therefore, the primary condition of the pineal organ shows a complete correspondence to that of an acino-tubular gland; but since in the later stages of development the acini become cut off as hollow vesicles from the duct system, and these vesicles eventually lose their central cavity, it is clear that the difference between the mammalian and avian pineal organ is one of degree rather than of kind, and that the organ in both is built up on essentially the same plan.
- 1 This specimen is from one of the series of microscopical sections illustrating the development of the pineal region of Sphenodon and Geotria which were prepared by the late Prof. Dendy, and we take this opportunity of thanking Prof. D. Mackinnon for permission to make use of this most valuable collection in our present investigation.
Even in the early stages of foetal development in the human subject, the original arrangement of interdigitating epithelial and vascular processes becomes obscured, especially in the centre of the developing organ. This is partly due to degeneration and gradual disappearance of many of the vascular sinuses which form the central structures in the pale areas, and partly to the appearance of cells with pale vesicular nuclei in the tissue between the central core of vascular mesenchyme and the small deeply stained cells. These pale cells are derived from the proliferating deeply stained cords of epithelial cells, and are comparable with the cells of the nuclear or mantle zone of the developing neural tube elsewhere, which are differentiated from the germinal or inner layer of proliferating ependymal cells.
The later post-natal stages of the development of the human pineal organ will be considered in the following section dealing with the structure of the pineal gland.
Structure of the Pineal Gland
A considerable amount of variation exists in the structure of the pineal gland at different age periods and in different individuals. These changes have recently been specially investigated by Globus and Silbert, who have worked on human material ranging from a 54 months’ foetus to old age, and these authors consider that the variability in the structure of tumours arising in the pineal region, either from the pineal body itself or from “rests” in the vicinity of the organ, may be explained on the assumption that the tumour tissue assumes the character in some cases of one age period of its development, and in others of another age period.
Besides variations in structure due to age changes, there are differences in appearance which are brought out by different methods of preparation, and we propose, before dealing with the selective actions produced by the use of special methods of modern technique, to give a short description of the microscopic appearance of a section of the pineal gland of a child of five years of age, stained by the ordinary haematoxylin and eosin method. We shall then endeavour to interpret the appearances seen at this stage of development and in the adult organ by a reference to the earlier stages of embryonic and foetal development, and by the microscopic pictures brought out by the use of differential stains.
Text-fig. 13. Transverse section through the pineal region of a rabbit embryo (18 days) showing the intimate relation of the fundus of the pineal body to the venous sinuses, where the great cerebral vein joins the plexus at the sinus confluens, and the relation of the fibres of the posterior commissure to the subcommissural organ_and ependyma.
Text-fig. 14. Tangential section of the pineal gland of a child, showing the fibrous capsule, lobes and interlobar septa.
The pineal gland at this stage (text-fig. 14) is invested by a fibro-vascular capsule derived from the pia mater, and lined internally by a neuroglial stratum. This capsule sends trabeculae containing blood vessels into the substance of the gland. The trabeculae pass inward between and partially surround the peripheral part of a branched system of lobules which originate from the neuro-epithelium of the primary pineal diverticulum. The lobular areas between the trabeculae communicate with each other in the central part of the ° gland, so that the lobules are not completely enclosed in separate compartments but are continuous with a central mass of parenchymatous tissue which is more uniform in appearance than the lobulated peripheral zone. The lobules consist of a supporting glial tissue which has the character of a fibrillated sponge-work or reticulum enclosing clear intereommunicating spaces. Some of the spaces contain parenchyma cells with vesicular nuclei, while others appear empty. The reticulum is especially noticeable beneath the capsule and in relation with the larger trabeculae. The cell elements of the reticulum have the appearance of being continuous with each other, no intercellular septa ever being visible. The network thus seems to be formed of a plasmodium or spongioplasm within which the nuclei of the cell elements are imbedded. In many of the spaces of the network, however, as stated previously, branched parenchyma cells containing pale vesicular nuclei are present. The processes of these cells may be:
- Continuous with the processes of similar adjacent cells.
- Continuous with the matrix of the general neurospongium.
- They may spread out on the perivascular sheaths of the vessels contained in the trabeculae or fibrous capsule.
The cells with the pale vesicular nuclei belong to the fully developed type of “pineal” or “parenchyma”? cell, and both the nuclei and cytoplasm may contain granules or spherules, which are believed by Dimitrowa and others to be secretory in nature. Many of the parenchyma cells, however, are imbedded in the neurospongium, and in this situation the nucleus is usually smaller and more deeply and uniformly stained than those of the cells just described. Between these two extreme types, both in young and adult specimens, there are numerous intermediate forms (text-fig. 15), both with respect to the type of the nucleus and the position of the cell element, namely, wholly contained within the neurospongium, protruding from this into a space, or completely extruded into the space, and connected with the surrounding structures merely by fine tapering processes.
In adult specimens specifically stained for neuroglia, the distinction between “‘neuroglia cells” and the general plasmodium or syncytial reticulum in which they are imbedded is quite definite, and it is at once evident that the slender neuroglia cells and neuroglial fibres in no sense form the principal constituent of the supporting tissue of the lobules. The former appear as very sparsely scattered branched cells, chiefly of the “‘astrocyte” type, with fine delicate processes lying in a tissue which is principally composed of branched parenchyma cells which form what appears to be a continuous reticulum. This reticulum is permeated by a fine capillary network, and a delicate plexus of thin-walled vessels with their connective tissue sheaths. The fibro-vascular elements are, however, quite inconspicuous, and the branched parenchyma cells form the bulk of the lobular tissue. The proportion that the supporting tissue bears to the fully developed parenchyma cells varies in different specimens. In some there are large tracts of dense finely fibrillated tissue, which either replace the lobular parenchymal tissue in certain parts of the gland, or may penetrate into its substance and thus break it up into small lobules, separated by interlobular septa. This tissue is described as “glial” or “‘fibroglial’’, and contains a varying proportion of neuroglial cells, neuroglial fibres, and fibrous conncctive tissue surrounding blood vessels.
Text-fig. 15. Section of an adult pineal gland stained by van Gieson’s method and eosin, showing the apparent continuity of the reticulum in which the parenchyma cells are imbedded; and also the relation of the parenchyma cells to the spaces of the reticulum and the supporting tissue or neurospongium. An interlobular septum showing capillary blood vessels and nuclei of fibrous connective tissue crosses the upper part of the drawing obliquely. The tissue of the interlobular septum appears to be directly continuous with the neurospongium.
An excessive development of fibroglial tracts and septa occurring in old subjects appears to be an indication of degeneration. The trabecular tissue of the developing organ, in which the vessels with their connective tissue sheaths are imbedded, appears to be largely derived from the primary undifferentiated neurosyncytium, and a balance of power seems to be established between the differentiation and growth of parenchyma cells, and the limiting influence of the fibroglial trabeculae which surround them. The variations in the proportion of parenchyma cells to supporting trabecular tissue, which occurs even in young subjects, may perhaps be explained on the hypothesis that the organ, being vestigial in nature, the power to differentiate parenchyma cells and to maintain their existence is less in some subjects than in others.
The arrangement of the cell elements of the lobules is very irregular, but occasionally an alveolar appearance is present in pineal glands at this period of growth, namely 4-5 years, or even in adult specimens which is similar to that seen in foetal life and at birth (Plate IV F). This affords an indication of the original developmental structure, namely, the outgrowth of proliferating cords of epithelial cells from the germinal or ependymal layer of the primary diverticulum, which alternate with ingrowing vascular processes of the pia mater (see p. 487). .In specimens stained by van Gieson’s method, the interlobular and intralobular fibrous connective tissue, in the substance of the gland, does not show the characteristic pink coloration which is strictly limited to the external fibrous capsule and some of the main trabeculae. It seems, therefore, that the chemical constitution of the intralobular connective tissue is altered in some way by contact with the secretion or tissue fluid contained in the spaces, and does not take on the specific staining reaction that it would do otherwise.
The “parenchymatous” cells with pale vesicular nuclei which occupy the spaces of the reticulum appear to have a position relative to the reticulum which corresponds to the position of nerve cells relative to the supporting glial tissue in the brain and spinal cord.
Both the parenchymatous and the true neuroglial cells of the pineal gland seem to be differentiated from the indifferent cell elements of the primary neurosyncytium, much in the same way as, in the central nervous system, neuroblasts and spongioblasts are developed from undifferentiated parent cells of the primary neurosyncytium of the brain and spinal cord. In the human subject, however, the process of differentiation does not appear to proceed so far as in either the “pineal sac”’ (epiphysis) or in the “‘ retina” of the pineal eye of Reptiles and cyclostomes. In these, definite “‘sensory” and ganglion cells are present which were first described by Studnitka, Dendy, Gisi, Hoffmann, and others, who demonstrated that in the retina of the pineal eye they are normally present and are related to nerve fibres.
The typical parenchyma cells of the mammalian pineal organ seem to be intermediate in character between the “indifferent cell”, which is the parent of the neuroblast, and the fully developed nerve cell. The occasional “‘neuronoid”, “sensory” and true “ganglion” cells, which do not belong to the sympathetic system and which have been observed in the human pineal organ by various authors, might, on this hypothesis, be regarded as cells which under abnormal conditions have completed their full development as nerve cells, whereas the typical parenchyma cells, though undoubtedly arising from neuroblastic tissue, have been transformed during phylogeny into a type of cell which differs from both neuroglial and nerve cells in containing what are apparently secretory granules, and in the absence of an axis cylinder.
Nerve Supply of the Mammalian Pineal Organ
We do not propose in this communication to discuss the general question of the nervous supply of the pineal apparatus, and we shall simply allude to our observations on the nerve supply of the human pineal gland. We have seen in specimens stained by Blair and Davies’ modification of Ranson’s method, plexuses of fine non-medullated fibres and small ganglion cells belonging to the sympathetic system, distributed along the course of the vessels entering and leaving the gland and around the parenchymatous cells. Non-medullated . nerve fibres and cells were also present in the membranes surrounding the pineal gland and at or near its posterior extremity. These correspond to the plexus described by Cajal in the rat, the nervus conari described by Kohlmer, and the retropineal ganglion of Pastori discovered by him in the dog.
We have also seen bundles of medullated nerve fibres in specimens stained, by osmic acid passing between the superior and posterior commissures and the parenchymatous tissue at the base of the gland where they diverge and appear to be distributed between the cell elements; but we have not been able to determine their exact mode of termination, nor have we seen any true ganglionic nerve cells with an axis cylinder process. We have, however, encountered a few large cells of a ‘“‘neuronoid” type, but these resemble aberrant forms of pineal cells rather than true nerve cells.
Our examination of the pineal system of lower Vertebrates and a study of the very extensive literature on the subject has led us to the belief that with the exception of the sympathetic nerves and cells, which are associated with the blood vessels entering and leaving the gland, the nerve supply of the adult mammalian pineal organ, like the organ itself, is tending to become vestigial. Even in lower ‘classes of vertebrate animals, the pineal nerves which were present in early embryonic stages have often completely disappeared in the later stages of embryonic or larval life. This is especially the case when the ‘‘parietal organ” or “terminal vesicle”’, as in the frog tadpole (text-fig. 17), or the “‘ pineal sac” in the mouse embryo becomes separated by a considerable distance from its primary site of origin from the neural tube. The literature bearing upon the nerve supply of the pineal system is voluminous, and the following references are mentioned merely with the object of indicating some of the more important contributions to the subject. Spencer, B., in 1886, first described the parietal nerve in Varanus giganteus: Darkschewitsch, 1886; Béraneck, 1892; Studni¢ka, 1893; Klinckostrém, 1894; Cajal, 1895; Dendy, 1899-1911; Gaskell, 1908, 1910; Krabbe, 1916; Horrax, 1927; Herring, 1927; Pastori, 1928, 1929; Rio Hortega, 1932.
The full complement of nerve fibres and associated ganglia which it may be assumed should be developed in connection with the pineal system, is probably never present in any one type of animal, for in the course of time, the organ has undergone fundamental changes in form, and presumably also of function. These changes have accompanied its evolution in certain lower types of Vertebrates, and its regression or degeneration in many Mammals. The nerve supply of the organ has naturally participated in these changes!
The nerve fibres which one might expect to be present in a fully evolved pineal system, fall into the following categories:
- Somatic afferent, from pineal sensory cells to central nervous system.
- Somatic efferent, from central nervous system to the pineal organ or accessory structures connected with it.
- Visceral afferent, from pineal organ to sympathetic ganglion.
- Visceral efferent, from sympathetic ganglia to pineal organ.
Text-fig. 16. Drawing of a medial longitudinal section through the pineal region of a human subject showing the relations of the pineal organ to the corpus callosum, fornix, great cerebral vein, dorsal diverticulum, and chorioid plexus, superior and posterior commissures, quadrigeminal plate, and the membranes and blood vessels at its posterior pole.
There is abundant evidence in Fishes, Amphibia and Reptiles of nerve fibres, passing between sensory cells of the “retina” of the “pineal eye” and the central nervous system, which it is presumed are afferent to the habenular and other ganglia. In the human subject, medullated nerve fibres connect the anterior and posterior commissures with the parenchyma of the pineal organ, but there seems to be no reliable evidence of the direction of the nerve impulses which traverse these fibres.
It is probable that the sympathetic nerves which accompany the vessels entering and leaving the mammalian pineal gland, and connect it with the retropineal ganglion and plexus on the great cerebral vein, function as sympathetic nerves elsewhere and are both afferent and efferent.
Text-fig. 17. Section through Stieda’s organ of a frog tadpole, at a later stage of development than that shown in text-fig. 7. The terminal vesicle (parietal organ) is completely cut off from the epiphysis, and careful search through sections behind the vesicle failed to reveal any nerve tract or remnant of the original connection between it and the “epiphysis” or brain.
Corpora Arenacea
The presence of corpora arenacea in the pineal glands of young as well as old subjects may be regarded as additional evidence of degenerative changes taking place in a vestigial organ. We do not propose to discuss here their mode of formation further than to mention incidentally that they appear to arise in the first instance by the coalescence of minute droplets of a finely granular material, deposited in pericapillary tissue spaces (text-fig. 18).
Pineal Cysts
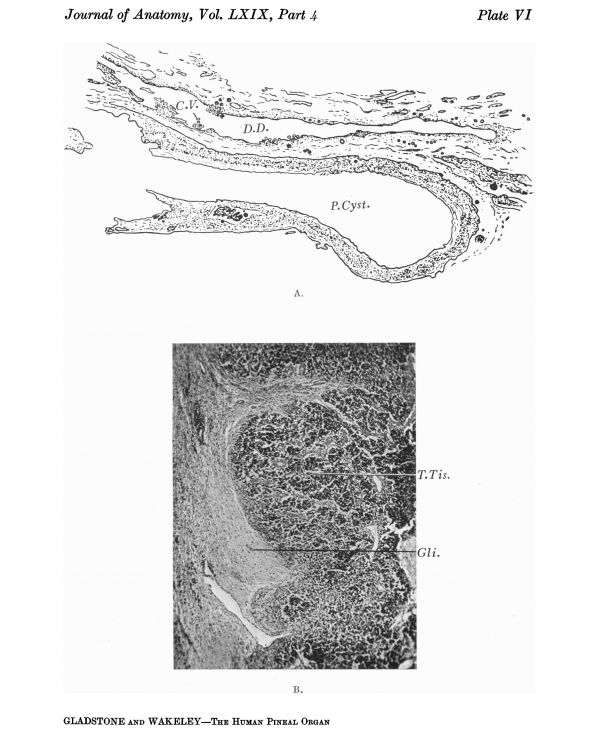
The presence of glial tracts and plaques is of considerable practical importance in connection with the development of cysts. These seem to be most frequently due to the breaking down of the central part of a glial area and the accumulation of tissue fluid in the spaces thus formed. They are often quite irregular in their disposition, and the larger cysts are apparently formed by the coalescence of smaller spaces. A good example of such a cyst is shown in Plates VI A and VC. The specimen was found in a brain in the Anatomy Department, King’s College, London. No history.could be obtained. The wall of the cyst was lined by condensed glial tissue, and no ependymal cells were visible in any of the sections. These were cut serially, and as no epithelial cells were found in the lumen it is probable that their absence could not be accounted for by the epithelium being shed into the cavity of the cyst. Some of the cells forming the immediate lining of typical cysts may be swollen up and rounded, giving a false impression of their nature; these cells must be regarded as pseudo-epithelial, and not in typical cases as being ependymal. The cavum pineale usually disappears early, and though cases have been described in
Text-fig. 18. Section through a perivascular space in an adult human pineal organ showing minute droplets of a finely granular material probably of a colloid nature and indicating an early stage in the formation of a corpus arenaceum.
which remnants of it have persisted in infants and even in adults the majority of cysts met with in the pineal glands of adults appear to be due to the breaking down of the central parts of glial areas, which areas have themselves arisen by a regressive degeneration of localised portions of the parenchyma. If the cysts were due to persistence of portions of the original cavity of the pineal diverticulum one would expect them to be more frequent in the pineal glands of infants and young children than at puberty or in the adult, and they would tend to lie in the central axis of the organ and be regular in their general disposition. They are, however, more frequently found in subjects from the period of puberty onwards, and are often quite irregular in their disposition.
The origin of pineal cysts has been studied by a considerable number of workers, of whom special mention may be made of Krabbe, Marburg and Cooper. These authors have all contributed valuable information on this difficult subject and agree in describing the frequent presence of glial plaques and extreme variability in structure of the pineal gland at different ages. Their interpretation, however, of the appearances found more especially with respect to the lining of the cyst wall by ependymal cells and the formation of the glial plaques differs in certain essential points from what we have described. The evidence of the specimens which we have examined justifies the conclusion that in a large proportion of specimens, with the exception of the pineal recess, the cavity of the primary pineal diverticulum has disappeared completely in young infants and at birth, and also that in one specimen, from a 44 months’ foetus (Plate III B and C, and text-fig. 12) the greater part of the pineal organ was solid, and only the stalk of the diverticulum was hollow.
Cysts lined by ependyma and containing villi are probably derived from the dorsal diverticulum (Plates V E, F and VI A), portions of which may become cut off and persist as closed vesicles. They may be distinguished from the typical pineal cysts by the absence of a peripheral zone of glial tissue outside the pseudo-epithelium which lines the cavity of an ordinary pineal cyst. As a rule only the peduncle of the dorsal diverticulum persists in the adult. It forms the suprapineal recess, which, as is well known, varies considerably in size, but usually has the relations shown in text-fig. 16, where it is seen wedged in between the great cerebral vein above and the pineal gland and superior commissure below.
Dermoid and teratomatous cysts of the pineal region may also be easily distinguished from pineal cysts by the presence of skin and by the contents of the cavity.
Summary and Conclusions
- The pineal organ of Mammals and Birds is derived from a diverticulum, the form and the site of origin of which, between the anterior and posterior commissures, corresponds closely to that of the primary pineal diverticula of Reptiles, Amphibia and Fishes.
- The apex of the primary pineal diverticulum is at first directed forward in all classes of Vertebrates.
- The definitive position of the pineal organ in the human subject with the apex directed backward is largely due to mechanical causes, such as the backward growth of the splenium of the corpus callosum and fornix.
- In the early stages of development of the membranes of the brain, the lower border of the primary interhemispheric septum contains a large vessel which is the precursor of the great cerebral vein and straight sinus. This is continued back into the sinus confluens. At a later stage when the corpus ~ callosum and fornix grow backward above the thalamencephalon and midbrain, this primary vessel is joined by a new vessel, the inferior sagittal sinus. This is developed in the lower border of a thickening of the upper part of the primary septum which becomes the falx cerebri, whereas the thin lower part of the septum in front of the tentorium cerebelli disappears.
- Between the third and fifth months of foetal life there occurs an active proliferation of neuro-epithelial cells derived from the inner or ependymal zone of the pineal diverticulum. These grow outward in the form of cords, the component cells of which are arranged radially round a central axis which is destitute of nuclei. This is accompanied by a simultaneous ingrowth of vascular processes of mesenchyme.
- A special mass of proliferating cells growing from the anterior wall of the main diverticulum gives rise to the solid anterior lobe of Krabbe, whereas the cords growing from the fundus of the diverticulum form the principal solid part of the posterior lobe. The stalk and posterior diverticulum (infrapineal recess) probably open out, and their lumina become incorporated in the cavity of the third ventricle, whereas the cavity of the main or anterior diverticulum, which may be cut off as the “cavum pineale”, usually disappears.
- The neuro-epithelial cells give rise to the glia lining the fibrous capsule and covering the trabeculae, the parenchyma cells and the neuroglial cells (astrocytes). The ingrowing vascular mesenchyme forms the fibrous capsule and the connective tissue basis of the septa and finer trabeculae including the contained vessels. Many of the sinusoidal vessels in the central part of the pale vascular areas seen in the earlier stages of development disappear, leaving only a very fine capillary plexus in the parenchymatous tissue of the lobules.
- The parenchymatous tissue in the adult consists of a reticulum of “branched” pineal cells, among which are a few neuroglial cells chiefly of the “astrocyte” type. The “alveolar” appearance which is sometimes seen in adult specimens is due to the persistence of primary neuro-epithelial cords, cross-sections of which appear as “‘rosettes”’.
- The frequent appearance of glial tracts or plaques containing scattered remnants of degenerating parenchymal cells is an indication of the vestigial condition of the pineal organ in the human subject, and these plaques afford an explanation of the liability of the pineal gland to undergo cystic degeneration, and possibly also the origin in it of certain types of tumour.
- Pineal cysts may result from the breaking down of the central areas of glial plaques. The nuclei of the glial tissue next the lumen of the cyst may give rise to the appearance of a pseudo-epithelium (compare Plate IV D with Plate V D).
- Cells and nerve fibres belonging to the sympathetic system accompany the vessels entering and leaving the pineal organ, and medullated nerve fibres connect the anterior and posterior commissures with the parenchymatous tissue.
True ganglion cells belonging to the central nervous system and having an axis cylinder process, although described by some authors, appear to be very rarely seen in the human pineal gland. Transitional forms exist, which are intermediate between the large cells described as ‘“‘neuronoid” and “‘parenchyma”’ cells,
References
A comprehensive bibliography of the literature on the Pineal System, including its pathological and clinical aspects, will be found in the references marked with an asterisk.
Acutcarro, N. and Sacristayn, J. D. (1912). Trab. Lab. Invest. biol. Univ. Madrid, vol. x.
—— (1913). Trab. Lab. Invest. biol. Univ. Madrid, vol. x1.
BrprForp, T. H. B. (1934). Brain, pt. 1, p. 57.
BERANECK, E. (1892). Anat. Anz. Bd. vi.
BERNARD, H. M. (1887). Nature, Lond., vol. txt, May 21.
(1896). Quart. J. micr. Sct. vol. XXxIx.
Cahal, S. Ramon ¥ (1933). Histology. Translated from the Spanish edition by M. Fernan-Nunez.
CaLvET, J. (1934). L’Ephiphyse. (Gland pineale.) Paris: J. B. Bailliére et Fils.
Cameron, J. (1902-3). Proc. Roy. Soc. Edinb. vol. xxiv. — (1903-4). Proc. Roy. Soc. Edinb. vol. xxv.
— (1903). Anat. Anz. Bd. xxm1. (1904). J. Anat. Physiol. vol. xxv.
CiarKke, J. L. (1860). Proc. Roy. Soc. vol. xt.
—— (1862). Proc. Roy. Soc. vol. x11. Coorzr, Evcenta R. A. (1932). J. Anat., Lond., vol. Lxvu, p. 28.
Cusnine, H. (1914). J. med. Res. vol. xxxI. (1925). Lancet, vol. 11, p. 851.
Curort, G. (1912). Anat. Anz. Bd. xu, S. 657. Dana, C. L. and Berxexey, W. N. (1913). Med. Rec., N.Y., vol. Lxxx1n, p. 835.
Danvy, W. E. (1915). J. exp. Med. vol. xxu1, p. 237. —— (1919). Ann. Surg. vol. Lxx, p. 129.
— (1921). Surg. Gynec. Obstet. ; — (1929). J. Amer. med. Assoc. vol. xc, p. 2012.
Danpy, W. E. and Buackran, K. D. (1914). Amer. J. Dis. Child. vol. vm, p. 406.
DaxRxkScHEwItscu, L. (1886). Neurol. Zbl. Bd. v, S. 29.
Denpy, A. (1899). Quart. J. micr. Sci. vol. xu, N.S. a and 6.
—— (1906). Brit. Assoc. Report (York, 1906).
—— (1907). Quart. J. micr. Sci. vol. Li, N.S.
— (1909). Nature, Lond., vol. txxxxu, Dec. 23.
—— (1910). Proc. Roy. Soc. B, vol. Lxxxm.
—— (1911). Philos. Trans. B, vol. cct, p. 227.
Denpy, A. and Nicuo its, G. E. (1910). Proc. Roy. Soc. B, vol. LxxxxtiI.
Dexter, F. (1902). Amer. J. Anat. vol. 1.
Diitrowa, Mile Z. (1901). Le Nevraze. Thése de Nancy.
EyciesHyMeEr, A. C. (1892). Anat. Anz. Bd. vu, S. 215.
Favanro, C. (1904). Arch. ital. Anat. Embriol. vol. 111, fase. 3.
GasKELL, W. H. (1908). Origin of Vertebrates. London.
—— (1910). Proc. Linn. Soc. Lond. vol. cxxu1.
GuapstonE, R. J. and Duntop, H. A. (1927). J. Anat., Lond., vol. LXx1, p. 366. G
uapstong, R. J. and WaAKELEY, C. P. G. (1920). J. Anat., Lond., vol. Liv, p. 196.
Gtosvs, J. H. (1932). Libman Anniv. Vols. vol. 1, p. 491.
Giosus, J. H. and Suzzert, S. (1931). Arch. Neurol. Psychiat., Lond., vol. xxv, p. 937.
Harris, W. and Cairns, H. (1932). Lancet, vol. 1, p. 3.
Herring, P. T. (1927). Quart. J. exp. Physiol. vol. xvu, p. 125.
Hocusterte|r, F, (1921). Verhandl. d. anat. Ges. a. d. 30. Vers. in Marburg, S. 193-212.
Horrmany, C. K. (1891). Bronn’s Klassen, Bd. v1, Abt. 3, S. 1891.
Horrax, G. (1927). Arch. Neurol. Psychiat., Lond., vol. XXI.
Horrax, G. and Battey, P. (1925). Arch. Neurol. Psychiat., Lond., vol. x11.
KuincKostr6ém, A. DE (1893). Anat. Anz. Bd. vii, 8. 289 u. 561.
Krassg, K. H. (1916). Anat. Hefte, Bd. tiv, S. 191.
—— (1915). (Abstract) Rev. Neurol. Psychiat. vol. x1, p. 300.
Nowikorr, M. (1910). Z. wiss. zool. Bd. xovi, Heft 1.
Pastort, G. (1928). Z. ges. Neurol. Psychiat. Bd. cx1t.
—— (1929). Z. ges. Neurol. Psychiat. Bd. cxxm.
Rio Horeca, P. DEL (1932). Cytology and Cellular Pathology of the Nervous System, vol. u, pp. 635-703.
W. Penfield. RussE x1, D. (1929). Amer. J. Path. vol. v, No. 5, p. 453.
Spencer, B. (1887). Quart. J. Micr. Sci. vol. xvu, p. 165.
Srupniéxa, F. K. (1905). Lehrbuch d. vergl. mikr. Anat. der Wirbelth. Oppel. Fasc. 5.
Sturmer, R. (1913). Histol. Histopath. Arb. Bd. v, Heft 3, S. 417.
Titney, F. and Warren, L. F. (1919). Amer. anat. Mem. vol. 1x, pt. 1, pp. 1-257.
WAKELEY, Crcit P. G. (1934). Med. Pr. Feb. 14, p. 145.
Watrer, K. (1922). Z. ges. Neurol. Psychiat. Bd. Lxx1v.
Warren, J. (1906). Amer. J. Anat. vol. v.
—— (1917). Anat. Rec. No. 11, p. 428.
(1918). J. comp. Neurol. vol. xxvut, pp. 75-135.
Watson, D. M. 8. (1914). Proc. Zool. Soc. Lond. vol. 1, p. 155.
(1914). Proc. Zool. Soc. Lond. vol. 11, p. 749.
Plates
Abbreviations
A.B.D. Anterior bilobed diverticulum. Ant.D. Anterior diverticulum. AL. Anterior lobe. Ag.C. Aqueductus cerebri. B.V. Blood vessel. Cc. Constriction between “pineal sac” and “ pineal eye’’. Ca. Capsule. Cap. Capillary. Cav. Cavity. Col. Cerebellum. CC. Corpus callosum. C.E., C.Ep. Columnar epithelium. C.H. Cerebral hemisphere. Ch.P. Chorioid plexus. Ch.P.V. TIT Chorioid plexus of third ventricle. Ch.P.L.V. Chorioid plexus of lateral ventricle. C.M. Corpus mammillaris. C.N. III Cranial nerve III. C.N.IV Cranial nerve IV. CN.V Cranial nerve V. Cp. Capillary. C.T. Connective tissue. C.T.C. Connective tissue cells. Cr.c. Cranial capsule. Cyst. Cyst containing chorioidal villi. CLV. Chorioidal villi. D.D., D.D.’, D.D.” Dorsal diverticulum and its subdivisions. DS. Dorsal sac. D.M. Dura mater. ELM. External limiting membrane. Ends. Endothelial space. Ep. : Ependyma. Ep.D. Ependymal diverticulum (dorsal sac). Epd. Epidermis. Ep.Z. Ependymal zone. Ep.c. Epithelial column. FC. Vessels in fibrous capsule. FF’. Fornix. GCWV. Great cerebral vein (Galen). Gli., G.Sh. Glial sheath. GLSt. dial stratum (pseudo-epithelium). HG. Habenular ganglion. ISS, Inferior sagittal sinus. ILLS, Interlobar septum. I.Lr.8. Interlobular septum. Inf. Infundibulum. I.P.R. Infrapineal recess. L. Lobule. Le. Lens. LN FPS. Layer of nerve fibres of pineal sac. L.N.F.R. Layer of nerve fibres of retina. Lum. Lumen. M.B. Meynert’s bundle. M.Z. Marginal zone.
N.Z. Nuclear zone.
Op.P.St.
O.T.
P.
Pa.
Pac.
Pa.0.
Par.
P.B.
PC.
P.Co., P.C., P.C4, P.C? P.Cyst.
P.D.
P.E.
P.LP.
P.L.
P.M., P.M, P.M. P.M.D.
P.O.
Post.D.
Opening of pineal stalk. Optic thalamus.
Pulvinar.
? Paraphysis.
Parenchyma cell.
Parietal organ (pineal eye). Parenchyma.
Pineal body.
Pineal cells.
Posterior commissure.
Pineal cyst.
Pineal diverticulum.
Pineal eye (parietal organ). Posterior-intercalary plate. . Posterior lobe.
Pia mater.
Posterior median diverticulum. Pineal organ. : Posterior diverticulum. Peduncle of pineal organ. Pineal recess.
Epithelial process.
Pineal sac.
Pial sheath.
Pineal stalk.
Pons Varolii.
Post-velar arch.
Perivascular space. Quadrigeminal plate.
Retina.
“Rosettes”.
Rathke’s pouch.
Septum interhemisphericum. Superior commissure (habenular commissure). Subcommissural organ.
Sinus confluens.
Superior colliculus. Secondary diverticulum. Secondary evagination. Small darkly stained nucleus. Space.
Splenium.
Sinus transversus.
Stieda’s organ (terminal vesicle). Subependymal layer. Tongue.
Tentorium cerebelli. Thalamus.
Tegmentum.
Thrombus.
Great cerebral vein (V.M. Galeni). Vascular connective tissue. Velar fold.
Ventriculus mesencephalicus. Ventriculus lateralis.
Third ventricle.
Fourth ventricle.
Venous sinuses.
Plate I
A. Medial longitudinal section of the pineal region of a chick embryo (8 days), showing the main pineal diverticulum, and secondary diverticula, which have arisen as hollow outgrowths from the former; also the relation of the superior and posterior commissures to the stalk of the main diverticulum, and the dorsal sac.
B. Oblique coronal section of pineal organ of a chick embryo, of the same age as the preceding, showing the primary and secondary diverticula, and some fibres of the posterior commissure.
Plate II
Transverse sections of pineal region of a 22-mm. human embryo and median aspect of the right half of a model reconstructed from the corresponding series of sections.
A. Section through the posterior bifurcated end of the anterior ependymal diverticulum, in the plane A of the model.
B. Section through the anterior bilobed diverticulum, and posterior median diverticulum, in the plane B of the model.
C. Section through the posterior median diverticulum and posterior commissure in the plane C of the model.
D. Drawing of the median aspect of the right half of the model showing the relations of the superior and posterior commissures to the pineal diverticula.
Plate III
Transverse sections of the pineal region of a 6-cm. human embryo, and of a 44 months’ foetus.
A. Section through the basal part of the main pineal diverticulum of a 6-cm. human embryo showing, in the upper part of the photograph, the solid anterior lobe. Below this is the main pineal diverticulum, the wall of which shows proliferating cords of ependymal cells growing outward, into the surrounding tissue. Below the pineal evagination is a section through the infrapineal recess, the epithelial lining of which is assuming a columnar type. Fibres of the posterior commissure are seen at the sides and below the pineal region.
B. Coronal section through the pineal region of a 44 months’ human foetus showing the relation of the dorsal diverticulum (suprapineal recess) to the pineal gland. Clusters of elongated chorioidal villi project into the lumen of the diverticulum. The pineal gland shows partial subdivision into an anterior and posterior lobe.
C. The pineal gland more highly magnified showing the ingrowth of vascular processes of the pia mater between the outgrowing neuro-epithelial cords.
D. Peripheral portion of the gland x55 D., showing pale areas containing a central core of vascular pia mater, alternating with dark zones, composed of neuro-epithelial cords.
Plate IV
Transverse section through the distal end of the pineal evagination of a 6-cm. human embryo. It shows folding of the wall of the diverticulum and outgrowth of the proliferating ependymal cells into the mantle zone.
Section through the pineal region of an adult Sphenodon (Dendy collection), showing in the upper part of the photograph the pineal evagination, the wall of which is folded in a manner similar to the human, but the extension of the lumen of the diverticulum into the outgrowing processes is much more pronounced. Below and to the right of the photograph are seen sections of portions of the paraphysis and dorsal sac.
Transverse section through the basal part of the pineal evagination of the 44 months’ human foetus, seen in Plate III B, C and D, showing outgrowth of neuro-epithelial cords, more especially from the anterior aspect and sides of the tube; on the posterior aspect (below in photograph) the epithelium is differentiating into the columnar type, characteristic of the subcommissural organ.
Detail of B, x 139 D., showing in the centre a pseudo-lobule, with its central core of vascular pia mater, between two hollow neuro-epithelial outgrowths.
E. Section through the subcapsular part of a pineal gland of an infant (1 year, 4 months), showing the fibrous capsule and the penetration of a blood vessel into the substance of the gland. The vessel is surrounded by a perivascular sheath of fibrous connective tissue, outside which is a glial sheath, the cellular and fibrous components of which are continuous with the neurospongium which forms the supporting tissue of the lobules and contains the parenchyma cells. Portion of the same specimen x 261 D., showing the predominance at this stage of the cells with small dark nuclei, and the arrangement of the cells in cords which when cut in crosssection are seen to be disposed radially round a central core which is destitute of nuclei, giving the acinar appearance sometimes described as “rosettes”.
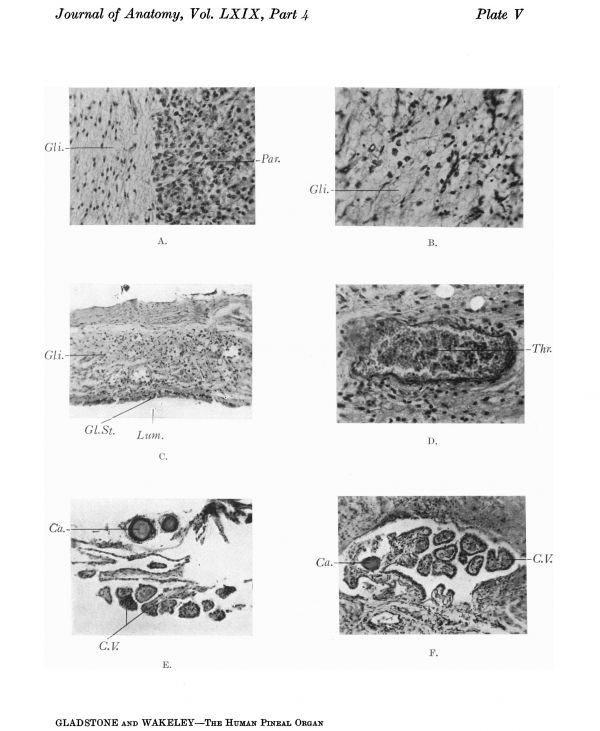
Plate V
Section through an adult human pineal gland showing the parenchymatous tissue on the right and on the left a tract of glial tissue, characterised by sparsely scattered small oval nuclei imbedded in a reticulum of glial fibres.
Section through a glial plaque from the same specimen, x 194 D.
Section through the wall of the pineal cyst shown in A, Plate VI. The lumen of the cyst is below and it is seen to be lined by condensed glial tissue, no ependymal epithelium being visible. Compare with the ependymal epithelium shown in Plate IV C.
The middle zone is formed of a degenerate tissue containing few parenchyma cells, and showing numerous spaces. In the upper part of the section is the fibrous capsule containing blood vessels with thickened walls.
Section through a thrombosed vessel in a pineal tumour. The endothelium lining the vessel is destroyed on the left side, and the vessel is surrounded by a small cell infiltration. The thrombus contains a large proportion of leucocytes.
Portion of the wall of the dorsal diverticulum lying above the pineal cyst shown in Plate VI A, showing chorioidal villi and corpora arenacea.
Section through a cyst lying outside the parenchymatous tissue of a pineal tumour. The cyst is lined by ependyma and chorioidal villi project into its lumen. A corpus arenaceum is imbedded in its wall.
Plate VI
Section of pineal cyst shown in Plate V C and E. The drawing also shows the relation of the dorsal diverticulum to the pineal gland, and chorioidal villi projecting into its lumen. Corpora arenacea are present in the tissue surrounding the pineal cyst and the diverticulum, and are also imbedded in their walls.
Section of pineal tumour showing the lobulated arrangement of the tumour tissue, and tracts of degenerating glial tissue, containing thin-walled blood vessels.
Cite this page: Hill, M.A. (2024, April 27) Embryology Paper - Development and histogenesis of the human pineal organ (1935). Retrieved from https://embryology.med.unsw.edu.au/embryology/index.php/Paper_-_Development_and_histogenesis_of_the_human_pineal_organ_(1935)
- © Dr Mark Hill 2024, UNSW Embryology ISBN: 978 0 7334 2609 4 - UNSW CRICOS Provider Code No. 00098G